Abstract
The brewing industry is constantly evolving, driven by the quest for novel flavours and fermentation characteristics that cater to evolving consumer preferences. This study explores the genetic and phenotypic diversity of European farmhouse yeasts, traditionally used in rural brewing practices and maintained outside of pure culture industrial yeast selection. We isolated landrace brewing yeast strains from diverse geographical locations across Europe, including Norway, Lithuania, Latvia, and Russia, and also included African farmhouse brewing strains from Ghana. Our genomic analysis using long-read and short-read whole genome sequencing uncovered a genetically distinct group that diverges from industrial brewing yeasts. This group, which is closely related to ale brewing strains, is preliminarily named the ‘European Farmhouse’ group and shows greater predicted admixture from Asian fermentation strains. Through genomic and phenotypic analyses, including flavour metabolite analysis via headspace gas chromatography-mass spectrometry, sugar metabolite analysis via high-performance liquid chromatography, and wort fermentation analysis, we found a broad spectrum of fermentation capabilities, from rapid and efficient fermentation to unique aroma and flavour compound profiles, potentially offering novel traits for brewing applications. This study highlights the importance of preservation of brewing cultural heritage knowledge and resources including yeast cultures.
Key points
• A large set of geographically diverse farmhouse brewing strains were characterized
• Norwegian and Baltic farmhouse brewing strains form a distinct genetic group
• Farmhouse strains show considerable diversity in fermentation and flavour formation
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding the genetic diversity and characteristics of beer yeast is essential for the brewing industry to develop new flavours, aromas, and fermentation traits that meet modern consumer preferences and changing industry conditions. Currently, the beer industry is rapidly evolving, with an increasing number of craft breweries worldwide (Garavaglia and Swinnen 2018). These breweries rely on the diversity of yeasts to create unique beers that set them apart in a competitive market. Therefore, it is vital to study and preserve all available brewing yeast strains. Today, the majority of the commercially used brewing strains tend to cluster into one of two independently domesticated ‘Beer’ groups (Gallone et al. 2016; Gonçalves et al. 2016; Peter et al. 2018; Krogerus et al. 2019). However, non-industrialized brewing strains are still likely to be maintained in traditional brewing systems across the globe (Cubillos et al. 2019).
Farmhouse brewing is the practice where farmers brewed beer for use in their own households from their own grain. Historically, farmhouse brewing was nearly universal among farmers across Europe (Garshol 2020a) and also widespread in other parts of the world. The farmers maintained their own yeast cultures in isolation from those used in modern industrial brewing, and their fermentation practices differed substantially from those in modern brewing. Specifically, the farmers fermented at much higher temperatures (Garshol 2020b) and much faster (Garshol 2022) than modern brewers. Drying the yeast was also very common (Garshol 2020a). These yeasts are known as ‘farmhouse yeasts’. Despite the widespread use of farmhouse yeasts in farmhouse brewing, little is known about their genetic makeup and their potential to offer new flavours and fermentation traits relevant to modern brewing practices. Farmhouse yeasts are potentially unique due to their isolation and adaptation to specific environments (e.g. warm fermentation temperatures and drying) over long periods, sometimes resulting in genetic variations that are not present in industrial brewing yeasts (Preiss et al. 2018; Foster et al. 2022). As such, farmhouse yeasts can offer significant potential for the brewing industry. Beyond commercial applications, the study and preservation of traditional yeasts contribute to the knowledge of cultural heritage in brewing.
In contrast to industrial brewing yeasts which are maintained typically by cryopreservation, traditional farmhouse brewers today use simpler methods such as reusing refrigerated yeast or drying the yeast between batches of beer. These cultures often contain multiple strains of yeast, whereas most commercially produced yeast strains are a monoculture. Because of this, these yeasts may also be referred to as ‘landrace’ yeasts. Traditional farmhouse brewing practices also differ from modern/industrial brewing in several other ways. A wider diversity of beer production methods are used in traditional farmhouse brewing, including techniques such as not boiling the wort (Norway, Denmark, Sweden, Finland, Estonia, Latvia, Lithuania), boiling the mash (Finland), extensive use of juniper and juniper infusions (Norway, Finland, Sweden, Estonia, Latvia, Lithuania), baking the mash (Lithuania, Russia), and using grains such as sorghum and millet (Ghana) (Djameh et al. 2019; Garshol 2020a).
Previous research has explored kveik yeast, a Norwegian type of farmhouse yeast, which has gained popularity due to its beneficial features in the brewery. Opportunities identified in kveik yeast include its ability to ferment at high temperatures, reducing energy consumption in the brewing process, and its unique flavour profiles which can provide new beer styles to meet the growing consumer demand (Preiss et al. 2018; Krogerus et al. 2018b). To date, kveik has been demonstrated as useful in various fermentations including beer (Preiss et al. 2018; Kawa-Rygielska et al. 2021, 2022; Foster et al. 2022; Dippel et al. 2022; Paszkot et al. 2023), Scotch Whiskey (Waymark and Hill 2021), and acid whey beverages (Luo et al. 2021). Recently, it was also shown that kveik yeast has enhanced accumulation of trehalose compared with industrial brewing yeasts, which may explain its enhanced stress resistance (Foster et al. 2022). These traits show a broad range of potential for this Norwegian farmhouse yeast.
We hypothesized that farmhouse yeast strains isolated from a more geographically diverse set of sources may also show beneficial fermentation traits and unique genetics when studied. Here, we isolated and selected a group of 35 landrace brewing yeast strains from Norway, the Baltic region, Russia, and Ghana (Fig. 1). These farmhouse yeasts may be genetically distinct from industrial yeasts due to their adaptation to specific environments or niches over time, resulting in genetic and phenotypic variations unique to each strain or group. Early research on kveik isolates, for example, indicate that they are closely related, but genetically distinct, from ale brewing yeast (Preiss et al. 2018; Dondrup et al. 2023). Furthermore, the characterization of non-industrialized brewing yeast could further uncover the complex origins of brewing yeast (Fay et al. 2019; Abou Saada et al. 2022).
We further hypothesize that landrace yeasts may offer new combinations of flavour and fermentation traits relevant to modern brewing practices such as craft brewing. To test these hypotheses, we conducted both whole genome sequencing (short read and long read) and phenotypic analyses, such as flavour metabolite analysis via headspace gas chromatography-mass spectrometry (HS-GC–MS), sugar metabolite analysis via high-performance liquid chromatography (HPLC), and wort fermentation analysis.
Materials and methods
Yeast strain isolation
Yeast samples in either dry or liquid format were received at the research site in Canada from Norway. Upon receipt, yeast samples were hydrated or inoculated into sterile beer wort (10°P) at a rate of approximately 0.5 g/10 mL. The samples were incubated overnight at 25 °C then aseptically streak plated onto WLN agar (Alpha Biosciences, Baltimore, USA) and incubated for 5 days. Unique colony morphologies and colour patterns on WLN agar were selected and sub-streaked onto fresh WLN agar. The yeasts from Ghana were obtained in a powdered format from dried sediments of a pito brew received from a traditional brewer in Accra, Ghana. The samples were inoculated into 10 mL YPD (1% yeast extract, 2% peptone, 1% glucose) and incubated with shaking at 30 °C for 24 h after which single yeast colonies from the cultures were isolated on YPD agar plates. Pure cultures were cryopreserved at − 80 °C prior to analysis. The colonies were verified as Saccharomyces cerevisiae by performing sequence analysis of the ITS region of the yeast isolates. Table 1 lists the strains selected for analysis. The additional strains included in the genomic analyses are listed in Supplementary Table S1).
Yeast genome sequencing and analysis
Short read Illumina sequencing was performed either as described in Foster et al. (2022) or otherwise DNA extractions were performed using the ZymoBIOMICS™ DNA Miniprep Kit (Zymo Research, Irvine, USA). Short-read sequencing of strains was performed at SeqCenter LLC (Pittsburgh, PA). Illumina sequencing libraries were prepared using the tagmentation-based and PCR-based Illumina DNA Prep kit and custom IDT 10 bp unique dual indices (UDI) with a target insert size of 320 bp. No additional DNA fragmentation or size selection steps were performed. Illumina sequencing was performed on an Illumina NovaSeq 6000 sequencer in one or more multiplexed shared-flow-cell runs, producing 2 × 151 bp paired-end reads. Demultiplexing, quality control, and adapter trimming were performed with bcl-convert (v4.1.5; Illumina, San Diego, USA).
Long-read sequencing experiments were carried out at the University of Guelph, University of Waterloo, and VTT. In the two former locations, high molecular weight yeast genomic DNA was extracted as prescribed by Oxford Nanopore according to Denis et al. (2018). Sequencing was performed with the Oxford Nanopore MinION instrument on a R9.4 flow cell using the SQK-LSK109 ligation sequencing kit (Oxford Nanopore Technology, UK). At VTT, DNA was extracted using a Monarch® HMW DNA Extraction Kit for Tissue (New England Biolabs, Ipswich, USA) and the supplied protocol for yeast. Sequencing was carried out on an Oxford Nanopore MinION MK1C instrument on a R10.3 flow cell using the SQK-LSK110 ligation sequencing kit (Oxford Nanopore Technology, UK). Reads were basecalled using Guppy version 5.0.11 (Oxford Nanopore Technology, UK) using the ‘super high accuracy’ model.
Reads were trimmed to a minimum length and quality of 1000 bp and Q8 using NanoFilt (De Coster et al. 2018). Genomes were de novo assembled from the long reads using Flye version 2.9 (Kolmogorov et al. 2019). Assemblies were first polished using Medaka version 1.4.3 and then with the short reads using NextPolish version 1.3.1 (Hu et al. 2020). Short reads were trimmed and filtered with fastp using default settings (version 0.20.1; Chen et al. 2018). Trimmed reads were aligned to a S. cerevisiae S288C reference genome (Engel et al. 2014) using BWA-MEM (Li and Durbin 2009), and alignments were sorted and duplicates were marked with sambamba (version 0.7.1; Tarasov et al. 2015). Variants were jointly called in all strains using FreeBayes (version 1.32; Garrison and Marth 2012). Variant calling used the following settings: –min-base-quality 30 –min-mapping-quality 30 –min-alternate-fraction 0.25 – min-repeat-entropy 0.5 –use-best-n-alleles 70 -p 2. The resulting VCF file (variant call format) was filtered to remove variants with a quality score less than 1000 and with a sequencing depth below 10 per sample using BCFtools (Li 2011). Variants were annotated with SnpEff (Cingolani et al. 2012). Sequencing coverage was estimated with mosdepth (version 0.2.6; Pedersen and Quinlan 2018). Chromosome copy numbers were estimated based on distribution of alternate allele frequencies, ploidy as measured by flow cytometry, and sequencing coverage.
For phylogenetic analysis, the variants were filtered to retain only single nucleotide polymorphisms and remove sites with a minor allele frequency less than 5%. The filtered single nucleotide polymorphism (SNP) matrix was converted to PHYLIP format (https://github.com/edgardomortiz/vcf2phylip). A random allele was selected for heterozygous sites. A maximum likelihood phylogenetic tree was generated using IQ-TREE (version 2.0.3; Nguyen et al. 2015) run with the ‘GTR + G4’ model and 1000 bootstrap replicates (Minh et al. 2013). In addition, a neighbour-joining tree based on the genetic distance between all biallelic SNPs (no minor allele frequency filtering) was also produced. Genetic distances were calculated with the ‘matchstates’ algorithm in the ‘pofadinr’ package in R to take into account heterozygous sites (Joly et al. 2015). The tree was built using the ‘bionj’ algorithm in the ‘ape’ package in R (Paradis et al. 2004). Finally, a neighbour-joining trees based on MinHash distances (Ondov et al. 2016) between genome assemblies were generated using mashtree (Katz et al. 2019). Mashtree was used with the long-read assemblies produced here, the short-read assemblies from Peter et al. (2018) and Gallone et al. (2016), and the long-read assemblies from O’Donnell et al. (2023).
Population structure was estimated based on the identified SNPs. The SNP matrix produced above was filtered using PLINK (version 1.9; Purcell et al. 2007) by removing sites in linkage disequilibrium (using a 50 SNP window size, 5 SNP step size, and pairwise threshold of 0.5) and with a minor allele frequency < 5%. The thinned SNP matrix was then input into ADMIXTURE v.1.3.0 (Alexander et al. 2009) and was run for 1 to 20 ancestral populations (K). The number of ancestral populations (K) that best represented this data set was chosen based on the lowest cross-validation error. The analysis was also re-run on a data set produced by merging the SNP matrix available from the 1011 yeast genomes data set (Peter et al. 2018) with SNPs identified in the landrace strains sequenced here using the ‘-@’ option in FreeBayes to force calls at all locations in the 1011 gVCF file. VCF files were merged with BCFtools (Li 2011) and then filtered as described above.
Admixture between populations was further estimated from allele frequency data using both TreeMix (Pickrell and Pritchard 2012) and AdmixtureBayes (Nielsen et al. 2023). The thinned SNP matrix used as input to ADMIXTURE above was used for analysis. First, strains belonging to ‘Mosaic group 3’ were removed, and the VCF was converted to TreeMix format using the script from https://github.com/speciationgenomics/scripts/blob/master/vcf2treemix.sh. TreeMix (version 1.13) was run based on the pipeline available at https://github.com/carolindahms/TreeMix. To find the optimum number of migration events, the pipeline was run using the following options: migration events (m) ranging from 1 to 16, ten replicates per migration event, 500 bootstrap replicates, and China_III as the outgroup. OptM (Fitak 2021) was then used to find the optimum number of migration events for the data set (m = 6). The TreeMix pipeline was then rerun with m = 6 as above, but with 30 replicates, and a consensus tree was produced with BITE (Milanesi et al. 2017). D, f4-ratio and f-branch statistics were calculated with DSuite from all the polymorphic sites detected among the 105 strains (Malinsky et al. 2021). For AdmixtureBayes, which estimates a best-fitting admixture graph without priori information, the same input file as for TreeMix was used. AdmixtureBayes was run with 20 MCMC chains with 75,000,000 iterations (-n 1500000). Convergence was tested by discarding the first 30% of samples as burn-in.
Phasing of short reads was carried out using nPhase version 1.2.0 (Abou Saada et al. 2021) and the workflow outlined in (Abou Saada et al. 2022) for the strains that had also been long-read sequenced. The predicted haplotigs for each strain are visualized in Supplementary Fig. S1. The phased SNPs for each predicted haplotig, as output by nPhase, were then applied to the sequence of the S. cerevisiae S288C reference genome to produce sequences for each of the predicted haplotigs. These sequences were further split into 10 kbp windows. In addition to the phased haplotigs, 53 genomes, representative of the 19 main clades identified in (Peter et al. 2018), were obtained from NCBI. These genomes were aligned to the S. cerevisiae S288C reference genome using minimap2 version 2.17-r941, and variants were called using paftools.js (included within minimap2). The pairwise distances between all the phased haplotigs and the representative genomes within each of the 10kbp windows was calculated using snp-dists (https://github.com/tseemann/snp-dists). Within the resulting distance matrices for all 10 kbp windows, we identified among the haplotigs for each of the phased strains the minimum distance compared with each of the 53 representative genomes.
Ploidy with flow cytometry
Ploidy of selected strains was measured using SYTOX Green staining and flow cytometry as described previously (Krogerus et al. 2017).
Yeast propagation and beer fermentation
To perform lab scale fermentations, yeast starters were grown in 20 mL wort at 25 °C for 24 h. A cell count was performed on the yeast starters using a haemocytometer after which the starters were used to pitch fresh wort with an original gravity of 10.5°P (specific gravity = 1.043 ± 0.0014) at a rate of 1 million cells/mL/°P. The wort used for the fermentation was made from pale barley malt (Rahr) and hopped with 2.4 g/L cascade hops. Three biological replicates of the fermentations were carried out in 500-mL glass bottles fitted with airlocks and incubated without shaking at 25 °C for 7 days. Specific gravity readings of the fermenting wort were measured daily using a DMA35 handheld density meter (Anton Paar GmbH, Graz, Austria). At the end of the fermentation, 20 mL of the fermented wort was taken, filter-sterilized, and stored for HPLC and GC–MS analysis.
Sugar and ethanol analysis by HPLC
The fermented beer samples were prepared for HPLC analysis by sterile filtration using a 0.22-µm syringe filter. Samples were prepared in an HPLC vial containing 400 µL of filtered beer and 50 µL of 6% (v/v) isopropanol as the internal standard. Measurements were performed using HPLC with a refractive index detector (RI). An Aminex HPX-87H column was used, where 5 mM sulphuric acid was the mobile phase. The conditions were flow rate 0.6 mL/min, 620 psi, 60 °C.
Aroma compound analysis by HS-GC–MS
Five millilitres of sterile-filtered beer samples and internal standard mix solution [1% (v/v) 3-octanol and 0.005% (v/v) ethyl methyl sulfide] of 20 µL were pipetted into a 20 mL HS vial sealed by a metal screwcap with a PTFE/silicone septa for quantitation of higher alcohols and esters by Agilent GC 7890B coupled with a PAL autosampler (RSI 85, CTC Analytics, Zwingen, Switzerland) and an MSD 5977B (Agilent, Santa Clara, CA, USA).
Beer samples were incubated at 40 °C for 20 min to reach equilibrium, followed by injection of 1 mL headspace with syringe temperature set at 70 °C, inlet temperature at 200 °C, and a split ratio of 10. Separation of volatile compounds was carried out with a DB-WAX UI capillary column (60 m × 0.25 mm, 0.25 um film thickness, Agilent, Santa Clara, CA, USA) and helium as the carrier gas at a constant flow of 1.2 mL/min. The oven program was stated as below: initial temperature held at 35 °C for 1 min, increased to 120 °C by 15 °C/,in; increased to 180 °C by 5 °C/min, further increased to 250 °C by 20 °C/min, and held at 250 °C for 5 min. The temperature of the transfer line to MS was set at 250 °C. The detection was performed at TIC mode (m/z 25–250) with electron-impact ionization at 70 eV, ion source temperature at 230 °C, and quadrupole temperature at 150 °C.
Ethyl hexanoate, ethyl octanoate, ethyl decanoate, phenylethyl acetate (MilliporeSigma, St. Louis, MO, USA), and pre-made standard mixture containing acetaldehyde, n-propanol, ethyl acetate, isobutanol, isopropyl acetate, ethyl propanoate, active amyl alcohol, isoamyl alcohol, isobutyl acetate, ethyl butanoate, n-butyl acetate, and isoamyl acetate (SPEX CertiPrep Inc., Metuchen, NJ, USA) were used to establish standard calibration curves. 3-Octanol was used as the internal standard for quantitation.
Results
Isolation and genomic analysis of landrace yeasts
A cohort of 35 landrace brewing yeasts was selected on the basis of geographic and cultural diversity from a yeast strain collection containing approximately 250 yeast isolates obtained from original farmhouse brewing cultures (Table 1, Fig. 1). The strains were isolated from the following geographic regions.
-
Norway—West: The fjord region of Norway, west of the central Norwegian mountain chain. All strains described as kveik in earlier studies are derived from this region.
-
Norway—East/north: Collected from the village Ål in the Hallingdal region, and have informally been referred to as ‘gong’, after the local dialect word for yeast.
-
Norway—East/south: From Tinn county in the province of Telemark, and have informally been known as ‘berm’. In this region, the brewers now use malt extract rather than mashing malt.
-
Lithuania: From the northeastern region known as Aukštaitija, which for decades has been known as a hotspot of farmhouse brewing, with many brewers having started commercial breweries.
-
Latvia: From the Latgale region in eastern Latvia, which culturally and linguistically differs substantially from the rest of Latvia. The strains were collected about 120 km from the Lithuanian border.
-
Russia: All strains were collected in the Chuvash Republic, a region on the south bank of the Volga, 600 km east of Moscow, populated largely by the Chuvash people, who speak a Turkic language. All samples were collected from Chuvash speakers.
-
Ghana: Collected from a pito brewer from Yendi in the northern region of Ghana, approximately 22 km from the northwestern border with Togo. Traditional brewers in this region mainly produce pito (sorghum/millet beer) with backslopped yeast from a previous brew.
Further details for the farmhouse brewing yeasts can be obtained from the Farmhouse Yeast Registry (Garshol 2020c; Registry accessible online at https://www.garshol.priv.no/download/farmhouse/kveik.html). Previously studied and sequenced industrial ale strains as well as kveik strains were also included in the analysis (Preiss et al. 2018; Foster et al. 2022).
To clarify the relatedness and ancestry of the landrace strains, whole genome sequencing of the included strains was performed. Short-read sequence data was previously available for six of the kveik strains included here (Preiss et al. 2018), while the remaining 30 landrace strains were short-read sequenced (average coverage of 125, ranging from 29.5 to 406). The majority of the landrace strains were tetraploid (28 out of 35; Table 1), with many strains (20 out of 35 aneuploid) showing chromosome copy number variations based on read coverage and allele frequency distributions (Supplementary Figs. S2 and S3). Nine of the landrace strains were further long-read sequenced using nanopore sequencing. These nine strains included six of Norwegian origin (from all three sub-regions) and three of Baltic and Russian origin (Table 1). In addition to these landrace strains, long-read sequence data was also obtained for the six brewing and baking strains included as controls (Foster et al. 2022). First, long-read assemblies were produced for these strains. Genome assembly size ranged from 11.8 to 12.3 Mbp, contig number from 16 to 45, and N50 values from 751 to 927 kbp (statistics available in Supplementary Table S2). The metrics suggested high contiguity and completeness of the assemblies.
To better understand how these nine landrace strains, for which long-read assemblies were available, were genetically related to other wild and domesticated S. cerevisiae, an alignment-free neighbour-joining tree was first produced based on minhash distances between the landrace strains and the short-read assemblies from the 1011 yeast genomes data set (Peter et al. 2018) and Gallone et al. (2016). The resulting tree (Fig. 2A and full tree in Supplementary Fig. S4A) grouped seven of the landrace strains as a sister group to the ‘Beer 1/Ale beer’ group. This group included five Norwegian landrace strains, and, surprisingly, two of the Baltic strains (Dras 1 and Rakstins 1). The remaining two landrace strains (Tinn 1 and Marina 1) were placed in the ‘Mixed/Beer-Baking’ group. The six control strains grouped as expected in the ‘Beer 1’ and ‘Mixed’ groups.
Phylogeny of the 35 S. cerevisiae landrace brewing strains sequenced in this study together with larger yeast genome collections. A A neighbour-joining tree generated based on MinHash distances in the assemblies of 1011 yeast genomes (Peter et al. 2018) and long-read assemblies of 9 landrace and 6 control strains generated here. B A neighbour-joining tree generated based on MinHash distances in the 142 assemblies of ScRAP (O’Donnell et al. 2023) and long-read assemblies of 9 landrace and 6 control strains generated here. C Maximum likelihood phylogenetic tree based on SNPs at 310688 sites in 1011 yeast genome strains (Peter et al. 2018), the 35 landrace and 5 brewing control strains sequenced here, and the 5 sequenced NCYC ‘kveik’ strains. D A neighbour-joining tree based on SNPs at 1652813 biallelic sites in 1011 yeast genome strains (Peter et al. 2018), the 35 landrace and 5 brewing control strains sequenced here, and the 5 sequenced NCYC ‘kveik’ strains. The branches containing the ‘Mixed origin’, ‘European Farmhouse’, and ‘Beer 1’/ ‘Ale beer’ populations are highlighted in (A–D). Full trees are available in Supplementary Figs. S4–S5
These results agree with previous work on kveik (Preiss et al. 2018), suggesting that ‘kveik’ strains are closely related to, yet genetically distinct from, domesticated ale yeasts. However, farmhouse strains from outside Norway also clustered with the ‘kveik’ strains, so the group does not seem to be geographically restricted to Norway. Hence, we decided to preliminarily rename this group ‘European Farmhouse’. Interestingly, five strains from the 1011 yeast genomes set, identified as belonging to Mosaic group 3, also grouped in a sister branch to these ‘European Farmhouse’ strains (Supplementary Fig. S4A). This could hint towards an admixed ancestry of these strains, as also hypothesized earlier by Preiss et al. (2018). To ensure that no artefacts were introduced from comparing long-read assemblies with short-read assemblies, a second neighbour-joining tree was produced based on minhash distances between the landrace strains and the recently published long-read assemblies in the ScRAP data set (O’Donnell et al. 2023). The resulting tree again placed the nine landrace and six control strains in the same groups, and the ‘European Farmhouse’ group once again formed a sister group to ‘Beer 1’ (Fig. 2B and full tree in Supplementary Fig. S4B).
Next, we expanded the phylogenetic analysis to the full set of 35 landrace strains. Single nucleotide polymorphisms (SNPs) were first identified from the short-read data. SNPs were also simultaneously called for five control brewing strains, five ‘kveik’ isolates sequenced by NCYC (Bioproject PRJEB42916), and 65 randomly selected S. cerevisiae strains representing 20 of the main S. cerevisiae populations identified by (Peter et al. 2018). A total of 105 strains were included in this initial analysis set. A maximum likelihood phylogenetic tree was then inferred from the resulting SNP matrix consisting of 103,555 variable sites. As with the minhash trees, the majority of the landrace strains (23 of 35, as well as the five ‘kveik’ isolates sequenced by NCYC) grouped together, branching off before the ‘Beer 1’ and ‘Mixed’ strains (Fig. 3A). However, all these strains do not form a single monophyletic group. Seven of the remaining strains grouped within the ‘Mixed’ group, two in ‘Beer 2’, and the two Ghanaian strains among the ‘African Beer’ strains. Of the total of 28 strains in the ‘European Farmhouse’ group, 23 were of Norwegian origin, while the remaining five were of Baltic origin. A sixth strain of Baltic origin grouped together with strains of Mosaic group 3. Like in the minhash tree produced from the long-read assemblies, the Baltic strains formed their own branches in the ‘European Farmhouse’ group. Interestingly, among the Norwegian strains, two sub-groups also form depending on geographical origin. One consists of the strains deriving from Norway—West, the so called ‘kveik’ strains, including those from, e.g. Voss, Stordal Ebbegarden, and Skare, while the other consists of strains isolated in Eastern Norway, in the Norway—East/north and East/south regions, including those from, e.g. Halvorsgard and Skrindo. Of the five landrace strains in the ‘Mixed’ group, four were of Russian origin, while Tinn 1 derives from Norway East/south. In contrast to the assembly-based tree, where the ‘European Farmhouse’ strains grouped as a sister branch between the ‘Beer 1’ and ‘Mixed’ groups, here in the SNP-based tree, the ‘European Farmhouse’ strains branched off closer to the root of the tree after the ‘Mosaic 3’ and ‘Asian fermentation’ strains. This is likely a result of the assembly-based tree not capturing the high levels of heterozygosity found in ‘European Farmhouse’ strains (ranging from 0.34 to 0.55%; Supplementary Table S1), which could indicate an admixed ancestry. A neighbour-joining tree produced from the genetic distance across all 551,621 biallelic sites detected among the strains produced a similar tree structure (Fig. 3B). Here, the ‘European Farmhouse’ strains form a monophyletic group, with sub-groups again forming based on geographical origin.
Phylogeny of the 35 S. cerevisiae landrace brewing strains sequenced in this study. A Maximum likelihood phylogenetic tree based on SNPs at 103555 sites in 105 S. cerevisiae strains. B A neighbour-joining tree based on SNPs at 551621 biallelic sites in 105 S. cerevisiae strains. The set of strains include 35 landrace brewing strains studied here, 5 previously sequenced ‘kveik’ isolates, 5 control brewing strains, and 65 randomly selected strains from Peter et al. (2018) and Gallone et al. (2016) that represent the main S. cerevisiae populations. Information about the strains and their origin is available in Supplementary Table S1. The tree is rooted with the ‘China III’ clade as outgroup. Points in (A–B) are coloured according to geographical origin of the strain
An extended SNP matrix was further produced containing all strains from 1011 yeast genomes (Peter et al. 2018), the 35 landrace and five brewing control strains sequenced here, and the five sequenced NCYC ‘kveik’ strains. This was performed to ensure previous results were not an artefact of the smaller data set overrepresented by the landrace strains. However, because of computational limitations, SNPs could not be called simultaneously in all 1056 strains. Rather, SNPs were recalled in the latter 45 strains based on all the sites in the 1011 yeast genomes gVCF, after which the SNPs in the landrace and brewing strains were merged with those in the 1011 yeast genomes data set. The resulting SNP matrix consisted of 310,688 variable sites after filtering to retain only sites with a minor allele frequency > 1%. As in the assembly-based tree, the maximum likelihood phylogenetic tree produced from this SNP matrix again grouped the ‘European Farmhouse’ strains as a sister branch to the ‘Beer 1’ group (Fig. 2C and full tree available in Supplementary Fig. S5A). A monophyletic clade is formed together with the five previously mentioned strains from Mosaic group 3. Within the ‘European Farmhouse’ clade, the strains of Baltic origin again grouped separately to those of Norwegian origin. Finally, as for the smaller strain set, a neighbour-joining tree was produced from the genetic distance across 1,652,813 biallelic sites detected among the strains. Once again, a similar tree structure was obtained, with the ‘European Farmhouse’ strains forming a geographically subdivided monophyletic sister clade to the ‘Beer 1’ group (Fig. 2D and full tree available in Supplementary Fig. S5B). Together, the results so far highlight that ‘European Farmhouse’ strains are closely related but genetically distinct to ‘Beer 1’/ ‘Ale beer’ brewing strains, and they may have an admixed ancestry. Fascinatingly, this group is not geographically restricted to Norway as indicated by previous preliminary studies restricted to the ‘kveik’ group. Rather, this group consists of farmhouse brewing strains that have been maintained by brewers in Norway and around the Baltic Sea, providing a valuable set of brewing strains genetically distinct from the ‘Beer 1’ strains.
Admixture analysis reveals Asian influence on European Farmhouse genomes
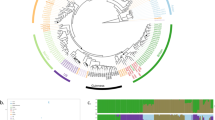
To further investigate the possibility of admixed ancestry among the ‘European Farmhouse’ strains, population structure and recent admixture events were estimated based on the polymorphic sites among the 105 strains in the initial SNP-based analysis set (visualized in Fig. 3A and B) using a range of methods. First, the SNP matrix was filtered to remove sites in linkage disequilibrium and with minor allele frequencies < 5%. Using ADMIXTURE, the number of populations that best represented this data set was eight (Fig. 4A). Here, the ‘European Farmhouse’ strains formed their own population, with a subset of the strains showing admixture with the ‘Beer 1’ and ‘Asian fermentation’ populations. The five ‘Mosaic group 3’ strains from the 1011 yeast genomes set, which in both the minhash- and SNP-based phylogenies grouped close to the ‘European Farmhouse’ strains, showed distinct patterns compared with the latter. Because of the high representation of landrace strains in the set of 105 strains, the analysis was further repeated with the larger data set produced above by merging the 1011 yeast genomes and the landrace and brewing strains sequenced here. After the same filtering as above, ADMIXTURE was rerun, and 16 populations best represented this expanded data set (Supplementary Fig. S6). Now, the ‘European Farmhouse’ and ‘Beer 1’ strains grouped in the same population, with all ‘European Farmhouse’ strains also showing evidence of admixture from strains in the ‘Asian fermentation’ population (Fig. 4B). Among the Norwegian landrace strains, the Norway East/(north and south) strains had a lower proportion of ‘Asian fermentation’ alleles compared with the Western ‘kveik’ strains. The Baltic landrace strains, on the other hand, showed a higher proportion of ‘Asian fermentation’ alleles compared with the Norwegian farmhouse strains, which geographically makes sense.
Population structure of the 35 S. cerevisiae landrace brewing strains and reference strain data sets. A Population structure of the set of 105 S. cerevisiae strains estimated with ADMIXTURE based on SNPs at 47954 sites. Each strain along the x-axis is represented by a vertical bar partitioned into colours based on estimated membership fractions to the resolved populations for K = 7 and 8 assumed ancestral populations. Strains are grouped according to the population they were assigned in Peter et al. (2018), with exception to the ‘European Farmhouse’ strains. B Population structure of a merged set of 35 S. cerevisiae landrace brewing strains and the 1011 yeast genomes strains estimated with ADMIXTURE based on SNPs at 31766 sites. Strains belonging to the ‘Mixed origin’, ‘Ale beer’, ‘Asian fermentation’, and ‘European Farmhouse’ populations are represented along the x-axis by a vertical bar partitioned into colours based on estimated membership fractions to the resolved populations for K = 15 and 16 assumed ancestral populations. Plots covering all strains for K = 8 to 20 assumed ancestral populations are available as Supplementary Fig. S6
To test for migration events, we also ran TreeMix and AdmixtureBayes on the thinned SNP matrix. With TreeMix, the optimal number of migration events that best explained the variation in the set was predicted to be six using the ‘Evanno’ method implemented in OptM (Fitak 2021). Consistent with previous work (Fay et al. 2019; Abou Saada et al. 2022), migration events from the Asian fermentation populations to both the root of the ‘Beer 1’- ‘European Farmhouse’- ‘Mixed origin’ branch (35% migration weight) and the Beer 2 population (14% migration weight) were predicted (Fig. 5A). In addition, a third admixture event from the Asian fermentation populations to the ‘European Farmhouse’ population was observed (20% migration weight), providing additional evidence for the ‘kveik’ and other ‘European Farmhouse’ strains emerging from admixture between a ‘Beer 1’ ancestor and a strain of Asian origin. The ‘European Farmhouse’ strains therefore exhibit a higher proportion of Asian ancestry compared with ‘Beer 1’ strains. These migration events are also supported by D, f4-ratio and f-branch statistics calculated from the full set of polymorphic sites across the 105 strains (Supplementary Fig. S7). The multiple migration events from the Asian fermentation populations into the brewing populations seems to suggest a fitness advantage for the ‘Wine–Asian fermentation’ intraspecific hybrid in a malt fermentation environment.
Phylogenetic networks inferred by A Treemix and B AdmixtureBayes in the set of 105 S. cerevisiae strains. A Using the ‘Evanno’ method implemented in OptM, six migration events between populations inferred by Treemix were selected and are shown with arrows indicating the direction toward the recipient population and coloured according to migration weight (ancestry percentage received from the donor). The scale bar shows ten times the average standard error of the entries in the sample covariance matrix. B The network graph topology with highest posterior probability as derived by AdmixtureBayes. The data set converged to and was best described by 20 admixture events. Red squares indicate admixture events, while blue ovals indicate populations. Percentage numbers on the branches and branch thickness represent admixture proportions
AdmixtureBayes was also used to estimate an admixture graph for the set of 105 strains studied. The tool uses a reversible jump Monte Carlo Markov Chain algorithm to sample high-probability admixture graphs and has been shown to infer graph topologies more accurately than TreeMix (Nielsen et al. 2023). Here, after running 75,000,000 iterations, the population structure was best described by 20 admixture events. The topology with highest posterior probability suggested the ‘European Farmhouse’ population formed from admixture between a ‘Beer 1’ ancestor (77% contribution) and a node derived from an ancestor of the ‘Asian Fermentation’ and ‘Sake’ branch (23% contribution) (Fig. 5B). Likewise, admixture from these Asian fermentation populations was also predicted in the formation of the ‘Mixed origin’- ‘Beer 1’ branch, as well as the ‘Beer 2’ population. Hence, results here further confirm our previous observations. AdmixtureBayes was also rerun on the extended SNP data set (strains of 1011 yeast genomes and the 35 landrace strains studied here). Strains from 1011 yeast genomes that had not been assigned to a clade, or belonged to one of the three mosaic regions, were excluded from the analysis. The resulting top topology also included 20 admixture events and predicted similar admixture events in the forming of the ‘European Farmhouse’ and other brewing and baking populations (Supplementary Fig. S8).
Finally, SNP phasing was used to demonstrate the strong contribution from Asian fermentation strains in the ‘European Farmhouse’ strains. For 17 of the strains where long-read sequences were available (this included nine landrace strains), phased haplotypes were produced using nPhase (Abou Saada et al. 2021). Similar to the approach described by Abou Saada et al. (2022), the obtained haplotypes for each strain were then divided into 10 kbp windows. Within each of the 17 strains, and each 10 kbp window, the sequences of the strain’s haplotypes were compared with those of 53 genomes selected to represent the major populations described in Peter et al. (2018), by calculating the pairwise genetic distance. For each window, the haplotype with the smallest distance was retained. The genome-wide average minimum distance for each of the 17 phased strains compared with the 53 representative genomes was then plotted (Fig. 6). Results reveal that compared with the included control ‘Beer 1’ strains, the ‘European Farmhouse’ strains contain haplotypes with a lower genetic distance to the ‘Asian Fermentation’ and ‘Sake’ strains. Two strains, AEL and BBT, from ‘Mosaic Region 3’ in the 1011 yeast genomes (Peter et al. 2018) and ScRAP data sets (O’Donnell et al. 2023) were also included in the analysis, since they clustered close to the ‘European Farmhouse’ strains in phylogenetic analysis. Here, however, they showed distinct profiles to that of the ‘European Farmhouse’ strains, particularly showing a higher minimum genetic distance to the ‘Beer 1’ strains. Taken together, results here strongly support the hypothesis of kveik and other ‘European Farmhouse’ strains forming from the admixture of a ‘Beer 1’ ancestor and a strain with origins in Asian fermentations.
Minimum genetic distance among the 17 phased haplotypes (columns) compared with 53 S. cerevisiae reference strains representing the major populations found in Peter et al. (2018) (rows). Analysis was carried out on haplotypes that were divided into 10 kbp windows. For each of the 17 strains with phased haplotypes, and each 10 kbp window, the sequences of the strain’s haplotypes were compared with those of the 53 reference genomes by calculating the pairwise genetic distance. For each window, the haplotype with the smallest distance was retained. Heatmap squares are coloured based on the genome-wide average minimum pairwise distance observed between each phased and the reference strain. Hierarchical clustering was performed on the columns
Beer fermentation and metabolite analysis
While genomic analyses revealed that the studied landrace strains were genetically distinct from traditional brewing strains, we also wanted to test whether they were phenotypically different in beer fermentations. To test this, small-scale fermentations in a standard pale ale wort (100% Maris otter pale ale barley malt hopped with Cascade hops) were carried out with a selection of 21 landrace and five brewing strains. Fermentations were conducted at 25 °C which was a compromise between the temperature optima typical of ale yeast fermentation (~ 16–25 °C) and farmhouse yeast fermentation (~ 28–40 °C). Overall, the majority of the tested landrace strains could efficiently ferment the wort, reaching final specific gravities as low as the brewing controls (Fig. 7A; Supplementary Fig. S9). The highest fermentation rates were observed among the landrace strains, with the ‘European Farmhouse’ strains Rakstins 1, Hornindal 1, and Skare 1 among the top performers. Interestingly, a number of ‘European Farmhouse’ strains in the inland Norway subgroup were also among the bottom performers, including Marem 1, Skrindo 2, and Skrindo 5. Overall, no statistical difference (p > 0.05; one-way ANOVA) was observed for fermentation rate between the different groups of strains (Fig. 7B). Six landrace strains, including the three previously mentioned slow ‘European Farmhouse’ strains, the two Ghanaian strains, and Pundurs 1, produced beer with higher final gravity, suggesting inability to use either of the main wort sugars maltose or maltotriose. Indeed, these beers contained either residual maltose or maltotriose (Fig. 7A).
Beer fermentation and metabolite analysis reveals fermentation and flavour production opportunities among landrace yeasts. A A heatmap showing phenotypic diversity in fermentation performance, sugar consumption and flavour metabolite formation among the 26 landrace and control brewing strains included in the phenotypic analysis. The heatmap is coloured based on Z-scores, and rows are sorted by hierarchical clustering. B The average relative fermentation rate among the strains grouped by population. No statistical difference (p > 0.05; one-way ANOVA) was observed between the populations, but the ‘European Farmhouse’ group showed greatest variation. C and D Principal component analysis of the set of 19 phenotypic traits. C Scores are shown to the left (strains coloured based on population), while D loadings are shown to the right (opacity of the phenotypic traits increases with distance from origin)
Similarly to the fermentation rate, considerable variation in volatile aroma production was also observed within the landrace strains, with strains both among the top and bottom producers of higher alcohols and esters (Fig. 7A). Principal component analysis of the fermentation parameters and aroma compound concentrations revealed that the studied landrace strains showed larger diversity for fermentation performance and aroma compound formation compared with the control brewing strains (Fig. 7C and D). In regard to acetate ester production, the top producers included ‘European Farmhouse’ and ‘Mixed’ strains, such as Marem 1, Ishlei 1, Tinn 1, Skare 1, Hornindal 1, and Marina (Supplementary Fig. S10). Marem 1, in particular, produced beers with high levels of flavour-active esters and was a clear outlier among the strains (Fig. 7C). The lowest formation of esters, in contrast, was observed among the ‘Beer 1’ strains Cali Ale and English Ale II. Overall, total ester formation was significantly higher (p < 0.05; one-way ANOVA) among the strains from the ‘Mixed’ and ‘European Farmhouse’ groups and lowest for those from the ‘Beer 1’ and ‘African Beer’ groups (Supplementary Fig. S11).
Linking genotype with phenotype
The small-scale wort fermentations revealed that a number of the landrace strains did not reach as high fermentation yields as the brewing strain controls and that this was due to residual maltose and maltotriose (Fig. 7). Indeed, the beers produced with the brewing controls had significantly lower concentrations of maltotriose than those produced with the landrace strains (p = 0.038; unpaired two-tailed t-test). The decreased use of maltotriose can be attributed mainly to the presence of up to three different loss-of-function mutations in the main maltotriose permease, MAL11, in many of the landrace strains (Fig. 8A). In addition, strains, like Djonno_1, Voss_1, Marina and the two Ghanaian strains, showed lower copy numbers or complete lack of MAL11. Nevertheless, most landrace strains fermented beer wort, and particularly maltose, efficiently, which could be expected based on their history of use. Among the whole set of 105 strains included in the genomics analysis, strains in the ‘Beer 1’, ‘European Farmhouse’ and ‘Mixed origin’ groups showed the highest copy numbers MAL11, MAL31, IMA2, IMA3, and IMA5 (Supplementary Fig. S12).
A The heatmap depicts sugar consumption, copy numbers of genes involved in maltose and maltotriose transport and metabolism, and allele frequencies of inactivating mutations in MAL11 among the phenotyped strains. B The allele frequencies of inactivating mutations in PAD1 and FDC1 among the strains belonging to the ‘Beer 1’, ‘Beer 2’, ‘Mixed origin’ and ‘European Farmhouse’ populations. Strains are ordered as in Fig. 3A
The landrace strains also showed significant variation in volatile aroma compound production, with strains both among the top and bottom producers of higher alcohols and esters. In regard to acetate ester production, the top producers included Marem 1, Ishlei 1, Tinn 1, Skare 1, Hornindal 1, and Marina (Fig. 7C and Supplementary Fig. 11). Of these strains, those belonging to the Mixed population contained a heterozygous frameshift mutation in FAS2 (Lys1857fs). Mutations in FAS2 have previously been linked to altered ester formation (Aritomi et al. 2014; Trindade de Carvalho et al. 2017). Interestingly, the FAS2BTC.1D allele described in Trindade de Carvalho et al. (2017), which was shown to result in enhanced acetate ester formation, was also found as heterozygous in Marem 1. In addition, a homozygous loss of stop codon was observed in Marem 1 for ATF1, coding for the main enzyme involved in acetate ester biosynthesis. The mutation results in an amino acid sequence that extends for an additional 6 amino acids. This same mutation was also heterozygous in three other landrace strains (Rakstins 1, Skare 1, and Voss 1), while homozygous in the Hefeweizen strains Weizen 1 and Uberweizen. The mutation was also present in the three Hefeweizen strains (Beer072, Beer074, and Beer093) included in the study by Gallone et al. (2016), and only in a single ‘Mosaic Region 3’ strain from the 1011 yeast genomes set (Peter et al. 2018). While Weizen 1 and Uberweizen were not included in the small-scale fermentations performed here, Hefeweizen strains are known for high acetate ester formation (Schneiderbanger et al. 2016; Meier-Dörnberg et al. 2017). In the study by Gallone et al. (2016), for example, Beer093 was among the top producers of acetate esters. The effect of this extended ATF1 enzyme on acetate ester formation should therefore be tested in subsequent studies.
The inability to produce 4-vinyl guaiacol from ferulic acid (phenolic off-flavour negative; POF-) is typically considered a domestication marker in S. cerevisiae (Gallone et al. 2016; Gonçalves et al. 2016). This trait is common particularly among strains in the ‘Beer 1’ population, as 4-vinyl guaiacol is undesired in many beer styles. The POF- phenotype is due to loss-of-function mutations in either PAD1 or FDC1. While the 4-vinyl guaiacol concentrations were not here analysed from the fermentations, the majority of the studied landrace strains contained loss-of-function mutations in either of these genes (Fig. 8B). The POF- landrace strains were mainly found in the ‘European Farmhouse’ population. However, not all ‘European Farmhouse’ strains were POF-, with the two Halvorsgard strains and Skrindo_5 lacking inactivating mutations. Among the strains in the Mixed population, inactivating mutations were common, but heterozygous, indicating the strains were not POF-. Outside the European brewing strains, the POF- phenotype was observed among the ‘African Beer’ (AEN) and ‘Mosaic Region 3’ strains (AEE, AEL, ARA, and BTB) as well. However, no inactivating mutations were observed in the two Ghanaian strains GH1 and GH2 isolated here. Marem 1, Stordal Ebbegarden 1, Ivar_Geithung_1, NCYC_Hornindal_2, and Pundurs 2 contained loss-of-function mutations distinct from those in the ‘Beer 1’ and ‘Mixed origin’ populations.
Discussion
Investigating the potential application of diverse yeasts is important for the brewing industry to meet an evolving market and shifting consumer preferences. Our study provides new insights into the genetic diversity and characteristics of landrace yeasts, highlighting their potential to offer new combinations of flavour and fermentation traits relevant to modern brewing practices. Here, we extend prior work on discovering, documenting, and characterizing landrace brewing yeasts and brewing practices from traditional farmhouse brewers.
Of the landrace yeasts studied here, the Norwegian ‘kveik’ strains have received the most interest in the brewing industry, with several commercial yeast suppliers now offering such strains in their selection. When these strains were first phenotypically and genetically characterized, it was revealed that they possess many traits desirable for beer production (e.g. maltose and maltotriose use, flocculation, and the lack of phenolic off-flavour formation), yet they were genetically distinct from other brewing strains (Preiss et al. 2018; Foster et al. 2022). Here, we show that genetically distinct farmhouse brewing yeasts are not restricted only to Norway, but rather, related strains were also isolated from farmhouse breweries in the Baltic region. Hence, we propose to refer to this group of yeast strains as ‘European Farmhouse’ brewing yeast. Inside this group, geographical origin of the strains could also be distinguished based on genome sequence, with strains from western Norway, eastern Norway, and the Baltic region clustering separately based on genome-wide SNPs. The ‘European Farmhouse’ group is closely related to the ‘Beer 1’ group of brewing strains, and these groups have likely branched off and evolved in parallel before the predicted emergence of the ‘Beer 1’ branch in the sixteenth century (Gallone et al. 2019). In comparison with the ‘Beer 1’ strains, the ‘European Farmhouse’ strains have not been used industrially, nor undergone any pure culture isolations to our knowledge.
While the ‘European Farmhouse’ strains are closely related to ‘Beer 1’ strains, they also show more signs of admixture. In early work, it was hypothesized that the ‘kveik’ strains have an admixed ancestry between a’Beer 1’ strain and one possibly of Asian origin. Here, we provide further evidence to support this hypothesis. A recent study, however, argues that the ‘Kveik’ population, rather than being closely related to the ‘Beer 1’ population, forms a sister group to other domesticated populations closer to the root of the S. cerevisiae tree (Dondrup et al. 2023). In our study, we observed a similar phenomenon of the ‘kveik’ strains grouping closer to ‘Mosaic Region 3’ and Asian fermentation strains when constructing a SNP-based tree (Fig. 3A). However, we believe this is an artefact of the relatively high levels of heterozygosity in, and admixed history of, these strains, which places the ‘European Farmhouse’ branch intermediate of the ‘Mixed origin’- ‘Beer 1’ and ‘Asian Fermentation’- ‘Sake’ branches (Kopelman et al. 2012; Meirmans et al. 2018; Huang et al. 2022). Firstly, in the assembly-based trees (Fig. 2A–B and Supplementary Fig. S4A–B) based on the haploid major allele sequence, the ‘European Farmhouse’ strains form a sister group to the ‘Beer 1’ strains. Secondly, using different algorithms, admixture and migration events from the Asian fermentation strains into a ‘Beer 1’ ancestor was predicted. Thirdly, long-read phasing of the SNPs suggests that the studied ‘European Farmhouse’ strains show lowest genetic distance to the ‘Beer 1’ and ‘Mixed origin’ strains out of the major populations identified in Peter et al. (2018). But, compared with the ‘Beer 1’ and ‘Mixed origin’ strains, the ‘European Farmhouse’ strains also show lower distance to the Asian fermentation strains supporting admixture with the latter. Recent studies on ale yeast have revealed that the ‘Beer 1’ ancestor in itself was also derived from admixture between close relatives of European wine and Asian fermentations strains (Fay et al. 2019; Abou Saada et al. 2022). The ‘European Farmhouse’ strains, in comparison, show an even higher contribution of Asian fermentation alleles. It is unclear why gene flow from the Asian fermentation strains is beneficial in the beer wort environment; however, many domesticated Asian strains show duplications of genes related to maltose metabolism (Duan et al. 2018) and carry the RTM1 cluster which confers resistance to molasses toxicity (Borneman et al. 2016; Pontes et al. 2020). High copy number of the MAL genes is also characteristic of the ‘Beer 1’ and ‘European Farmhouse’ populations, while the RTM1 cluster is also present in the ‘Beer 1’, ‘Mixed origin’, and ‘European Farmhouse’ strains (Supplementary Figs. S13 and S14).
These results led us to wonder how the repeated admixture of Asian fermentation genetics into brewing strain groups have happened historically. While purely speculative, there has been influence of beer brewing in the Middle East and Georgia, eliminated by Islam some time in the twelfth century (Haider 2013). Brewing continued in Georgia, and it is possible that yeast may have spread north of the Caucasus into Russia and beyond either before or shortly after 700 CE. Known viking activity in present-day Russia and Ukraine between 700 CE until at least 1100 CE (Roesdahl 2016; Jarman 2021) could have influenced Scandinavian brewing culture, methods, and beer yeast. Additionally, we note that the Rakstins_1 and Dras_1 strains appear to be genetically ‘European Farmhouse’ yeasts, despite being isolated from sources in the Baltic region. Similarly, the Norway East strains appear to form their own sub-group within the ‘European Farmhouse’ population that appears to span both the north and south regions. The two yeast types referred to as ‘gong’ and ‘berm’ thus appear to be a single group. Unsurprisingly, the Norway East strains clustered next to the kveik strains from Norway West, suggesting that the strains from Norway East form an outgroup of kveik. Further investigation is required to analyse more yeast sources and isolates from traditional brewers in the Baltic region in order to understand whether the occurrence of yeasts from the ‘European Farmhouse’ group is common among Baltic farmhouse brewing yeasts. It is also likely that similar strains have in the past been used for farmhouse brewing in Finland, Sweden, and Denmark. However, in Finland, for example, the farmhouse beer sahti is still commonly brewed, but brewers transitioned to commercial baker’s yeast during the first half of the twentieth century (Räsänen 1975). The same transition took place in the brewing of the Swedish farmhouse ale gotlandsdricke (Salomonsson 1979).
Limited work has so far been carried out on the phenotypic diversity of farmhouse yeasts. Prior work on a relatively small subset of ‘kveik’ isolates has revealed that they tend to ferment wort rapidly, have high thermotolerance, and can produce beers ranging from clean and neutral to strongly aromatic (Preiss et al. 2018; Foster et al. 2022; Kawa-Rygielska et al. 2022; Habschied et al. 2022; Dippel et al. 2022). Here, we reveal that farmhouse yeast, including kveik, show significant phenotypic diversity, both in regard to fermentation dynamics and flavour formation. In our screen of farmhouse strains and industrial controls, we observed both the highest and lowest maximum fermentation rates from strains belonging to the ‘European Farmhouse’ population. So, while kveik yeast are generally considered rapid fermenters, we here reveal that this is not always the case. Many of the landrace strains belonging to the ‘Mixed’ population also showed rapid fermentation. This is anticipated, as the ‘Mixed’ population consists of strains isolated from breweries, bakeries, and distilleries, and tend to have the ability to consume maltotriose (Gallone et al. 2016; Peter et al. 2018). The landrace strains showing poor fermentation were unable to completely use the maltose and maltotriose in the wort naturally limiting ethanol formation, and this appeared linked to lower copy numbers of or non-functional α-glucoside permeases. While the brewing industry has traditionally been interested in fast-fermenting strains with high ethanol yields, there is also an increasing interest towards strains with limited maltose and maltotriose use, as the consumer demand for beer with low and no alcohol content continues to rise (Bellut and Arendt 2019). Landrace strains therefore show potential for many different applications within the brewing industry.
In addition to showing variation in fermentation dynamics, the studied landrace strains showed considerable variation in aroma formation. Farmhouse ales can also have very different flavour profiles, with traditional European farmhouse ales usually being ester-rich and sometimes phenolic, while some kveik strains are known for their ability to produce ‘clean’ flavour profiles even at high fermentation temperatures. Similarly to the fermentation dynamics, we here show that strains from the ‘Kveik’ population were both among the top and bottom producers of higher alcohols and esters, meaning they have potential for producing both cleaner ‘lager-style’ and fruitier ‘pale ale-style’ beers. In contrast with previous work (Preiss et al. 2018), we here also reveal that not all strains in the ‘Kveik’ population are POF- and that the non-kveik landrace strains tended to be POF+ . The vast majority of the POF- strains in the ‘Beer 1’, ‘Mixed origin’, and ‘European Farmhouse’ populations carry the same three inactivating mutations in PAD1 and FDC1, as also highlighted in other recent studies (Gallone et al. 2016; Gonçalves et al. 2016; Abou Saada et al. 2022). This supports that these mutations have been acquired by an early ancestor of the ‘Mixed origin’- ‘European Farmhouse’- ‘Beer 1’ branch. Interestingly, some ‘European Farmhouse’ strains carry a premature stop codon in FDC1 (Lys78*) that is not found in any other ‘Beer 1’ or ‘Mixed origin’ strains, supporting a potential independent domestication for a sub-group of the European farmhouse strains. The unique inactivating mutations in the POF- ‘African Beer’ strains similarly imply an independent domestication for the phenotype in these strains (Abou Saada et al. 2022).
In addition to the direct applied value of landrace brewing strains, they are also valuable from a strain development perspective. Compared with ‘Beer 1’ strains, which tend to be sterile (Gallone et al. 2016; De Chiara et al. 2022), the majority of the kveik strains that have been tested so far have been fertile (Preiss et al. 2018; Dippel et al. 2022). This means that kveik strains can be quite readily used for breeding. The fertility of the non-kveik landrace strains has not been studied, but they are also likely to be fertile, as strains from the ‘Mixed origin’, ‘Mosaic beer’, and ‘African beer’ populations typically are (De Chiara et al. 2022). Landrace strains also tend to be polyploid (here the majority were tetraploid), which makes them good candidates for strain improvement through adaptive evolution. Previous studies have revealed that polyploid strains tend to adapt faster and undergo larger genomic changes during adaptive evolution compared with diploid strains (Selmecki et al. 2015; Lu et al. 2016). Indeed, the technique has been used to enhance ethanol tolerance and sugar utilization of brewing yeast (Brickwedde et al. 2017; Krogerus et al. 2018a). Similar strategies could be applied to further improve the landrace strains; while many of them showed rapid fermentation, some showed incomplete maltose or maltotriose use as a result of mutations in MAL11. Finally, from a strain development perspective, the genomes of the landrace strains can provide new insights and targets for strain engineering. Several kveik strains, for example, have been shown to have enhanced thermotolerance compared with traditional brewing strains, which in turn appears to be linked to enhanced intracellular trehalose accumulation (Foster et al. 2022). The molecular mechanisms responsible for this phenotype have not yet been identified, but several potential causative single nucleotide polymorphisms in genes related to trehalose metabolism have been identified in kveik strains that could be tested. Similarly, targets for other brewing-relevant phenotypes, such as altered aroma formation, could be identified from the genomes of the landrace strains.
In summary, we found genetically distinct and phenotypically useful yeast strains among various landrace brewing yeast cultures used to ferment traditional farmhouse beers. This study helps understand the genetic history of kveik and related landrace yeasts by suggesting that ‘European Farmhouse’ is a sister group to ‘Beer 1’. We further found evidence for increased Asian admixture in the ‘European Farmhouse’ yeast lineage. It remains unknown exactly how modern beer yeasts as well as landrace yeasts such as kveik have evolved over time. Further studies may explore a larger sample set of landrace yeast isolates or populations from the > 60 farmhouse yeast cultures already identified (Garshol 2020c). The unique genetics of such yeasts may yield new technological benefits to the brewing, baking, or biofuel industries including traits such as stress resistance, alcohol tolerance, and flavour/metabolite production. Newly developing and growing industries such as synthetic biology and cellular agriculture may also benefit from the knowledge to develop more robust and metabolically diverse yeasts.
Data availability
Illumina and basecalled ONT sequencing reads have been deposited in NCBI-SRA under BioProject number PRJNA1018716 (https://www.ncbi.nlm.nih.gov/bioproject/). The 15 long-read genome assemblies generated here are available at https://zenodo.org/records/12604786.
References
Abou Saada O, Tsouris A, Eberlein C, Friedrich A, Schacherer J (2021) nPhase: an accurate and contiguous phasing method for polyploids. Genome Biol 22:126. https://doi.org/10.1186/s13059-021-02342-x
Abou Saada O, Tsouris A, Large C, Friedrich A, Dunham MJ, Schacherer J (2022) Phased polyploid genomes provide deeper insight into the multiple origins of domesticated Saccharomyces cerevisiae beer yeasts. Curr Biol 32:1350-1361.e3. https://doi.org/10.1016/j.cub.2022.01.068
Alexander DH, Novembre J, Lange K (2009) Fast model-based estimation of ancestry in unrelated individuals. Genome Res 19:1655–1664. https://doi.org/10.1101/gr.094052.109
Aritomi K, Hirosawa I, Hoshida H, Shiigi M, Nishizawa Y, Kashiwagi S, Akada R (2014) Self-cloning yeast strains containing novel FAS2 mutations produce a higher amount of ethyl caproate in Japanese sake. Biosci Biotechnol Biochem 68:206–214. https://doi.org/10.1271/bbb.68.206
Bellut K, Arendt EK (2019) Chance and challenge: non-Saccharomyces yeasts in nonalcoholic and low alcohol beer brewing–a review. J Am Soc Brew Chem 77:77–91. https://doi.org/10.1080/03610470.2019.1569452
Borneman AR, Forgan AH, Kolouchova R, Fraser JA, Schmidt SA (2016) Whole genome comparison reveals high levels of inbreeding and strain redundancy across the spectrum of commercial wine strains of Saccharomyces cerevisiae. G3 Genes Genomes Genet 6:957–971. https://doi.org/10.1534/g3.115.025692
Brickwedde A, van den Broek M, Geertman J-MA, Magalhães F, Kuijpers NGA, Gibson B, Pronk JT, Daran J-MG (2017) Evolutionary engineering in chemostat cultures for improved maltotriose fermentation kinetics in Saccharomyces pastorianus lager brewing yeast. Front Microbiol 8:1690. https://doi.org/10.3389/fmicb.2017.01690
Chen S, Zhou Y, Chen Y, Gu J (2018) fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34:i884–i890. https://doi.org/10.1093/bioinformatics/bty560
Cingolani P, Platts A, Wang LL, Coon M, Nguyen T, Wang L, Land SJ, Lu X, Ruden DM (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin) 6:80–92. https://doi.org/10.4161/fly.19695
Cubillos FA, Gibson B, Grijalva-Vallejos N, Krogerus K, Nikulin J (2019) Bioprospecting for brewers: exploiting natural diversity for naturally diverse beers. Yeast 36:383–398. https://doi.org/10.1002/yea.3380
De Chiara M, Barré BP, Persson K, Irizar A, Vischioni C, Khaiwal S, Stenberg S, Amadi OC, Žun G, Doberšek K, Taccioli C, Schacherer J, Petrovič U, Warringer J, Liti G (2022) Domestication reprogrammed the budding yeast life cycle. Nat Ecol Evol 6:448–460. https://doi.org/10.1038/s41559-022-01671-9
De Coster W, D’Hert S, Schultz DT, Cruts M, Van Broeckhoven C (2018) NanoPack: visualizing and processing long-read sequencing data. Bioinformatics 34:2666–2669. https://doi.org/10.1093/bioinformatics/bty149
Denis E, Sanchez S, Mairey B, Beluche O, Cruaud C, Lemainque A, Wincker P, Barbe V (2018) Extracting high molecular weight genomic DNA from Saccharomyces cerevisiae. Protoc Exch. https://doi.org/10.1038/protex.2018.076
Dippel K, Matti K, Muno-Bender J, Michling F, Brezina S, Semmler H, Rauhut D, Wendland J (2022) Co-fermentations of kveik with non-conventional yeasts for targeted aroma modulation. Microorganisms 10:1922. https://doi.org/10.3390/microorganisms10101922
Djameh C, Ellis WO, Oduro I, Saalia FK, Haslbeck K, Komlaga GA (2019) West African sorghum beer fermented with Lactobacillus delbrueckii and Saccharomyces cerevisiae : fermentation by-products. J Inst Brew 125:326–332. https://doi.org/10.1002/jib.562
Dondrup M, Eiken HG, Martinussen AO, Haugland LK, Holdhus R, Dolan D, Grellscheid S, Hagen S, Elameen A, Myking T (2023) Traditional Norwegian kveik yeast: an ancient sister group to domesticated Saccharomyces cerevisiae. https://doi.org/10.1101/2023.07.03.547515
Duan S-F, Han P-J, Wang Q-M, Liu W-Q, Shi J-Y, Li K, Zhang X-L, Bai F-Y (2018) The origin and adaptive evolution of domesticated populations of yeast from Far East Asia. Nat Commun 9:2690. https://doi.org/10.1038/s41467-018-05106-7
Engel SR, Dietrich FS, Fisk DG, Binkley G, Balakrishnan R, Costanzo MC, Dwight SS, Hitz BC, Karra K, Nash RS, Weng S, Wong ED, Lloyd P, Skrzypek MS, Miyasato SR, Simison M, Cherry JM (2014) The reference genome sequence of Saccharomyces cerevisiae: then and now. G3 (Bethesda) 4:389–398. https://doi.org/10.1534/g3.113.008995
Fay JC, Liu P, Ong GT, Dunham MJ, Cromie GA, Jeffery EW, Ludlow CL, Dudley AM (2019) A polyploid admixed origin of beer yeasts derived from European and Asian wine populations. PLOS Biol 17:e3000147. https://doi.org/10.1371/journal.pbio.3000147
Fitak RR (2021) OptM : estimating the optimal number of migration edges on population trees using Treemix. Biol Methods Protoc 6:bpab017. https://doi.org/10.1093/biomethods/bpab017
Foster B, Tyrawa C, Ozsahin E, Lubberts M, Krogerus K, Preiss R, van der Merwe G (2022) Kveik brewing yeasts demonstrate wide flexibility in beer fermentation temperature tolerance and exhibit enhanced trehalose accumulation. Front Microbiol 13:747546. https://doi.org/10.3389/fmicb.2022.747546
Gallone B, Steensels J, Prahl T, Soriaga L, Saels V, Herrera-Malaver B, Merlevede A, Roncoroni M, Voordeckers K, Miraglia L, Teiling C, Steffy B, Taylor M, Schwartz A, Richardson T, White C, Baele G, Maere S, Verstrepen KJ (2016) Domestication and divergence of Saccharomyces cerevisiae beer yeasts. Cell 166:1397-1410.e16. https://doi.org/10.1016/j.cell.2016.08.020
Gallone B, Steensels J, Mertens S, Dzialo MC, Gordon JL, Wauters R, Theßeling FA, Bellinazzo F, Saels V, Herrera-Malaver B, Prahl T, White C, Hutzler M, Meußdoerffer F, Malcorps P, Souffriau B, Daenen L, Baele G, Maere S, Verstrepen KJ (2019) Interspecific hybridization facilitates niche adaptation in beer yeast. Nat Ecol Evol 3:1562–1575. https://doi.org/10.1038/s41559-019-0997-9
Garavaglia C, Swinnen J (2018) Economic perspectives on craft beer. Springer International Publishing, Cham
Garrison E, Marth G (2012) Haplotype-based variant detection from short-read sequencing. https://doi.org/10.48550/arXiv.1207.3907
Garshol LM (2020a) Historical brewing techniques: the lost art of farmhouse brewing. Brewers Publications, Boulder, CO, USA
Garshol LM (2020b) Pitch temperatures in traditional farmhouse brewing. J Am Soc Brew Chem 79:181–186
Garshol LM (2020c) The farmhouse yeast registry. Master Brew Assoc Am Tech Q 57:123–128
Garshol LM (2022) Fermentation times in traditional farmhouse brewing. J Am Soc Brew Chem 80:279–285
Gonçalves M, Pontes A, Almeida P, Barbosa R, Serra M, Libkind D, Hutzler M, Gonçalves P, Sampaio JP (2016) Distinct domestication trajectories in top-fermenting beer yeasts and wine yeasts. Curr Biol 26:2750–2761. https://doi.org/10.1016/j.cub.2016.08.040
Habschied K, Krstanović V, Šarić G, Ćosić I, Mastanjević K (2022) Pseudo-Lager—brewing with Lutra® kveik yeast. Fermentation 8:410. https://doi.org/10.3390/fermentation8080410
Haider N (2013) Contesting intoxication. Islam Law Soc 20:48–89. https://doi.org/10.1163/15685195-0002A0002
Hu J, Fan J, Sun Z, Liu S (2020) NextPolish: a fast and efficient genome polishing tool for long-read assembly. Bioinformatics 36:2253–2255. https://doi.org/10.1093/bioinformatics/btz891
Huang J, Bennett J, Flouri T, Leaché AD, Yang Z (2022) Phase resolution of heterozygous sites in diploid genomes is important to phylogenomic analysis under the multispecies coalescent model. Syst Biol 71:334–352. https://doi.org/10.1093/sysbio/syab047
Jarman C (2021) River kings: a new history of the Vikings from Scandinavia to the Silk Roads. William Collins Books, London
Joly S, Bryant D, Lockhart PJ (2015) Flexible methods for estimating genetic distances from single nucleotide polymorphisms. Methods Ecol Evol 6:938–948. https://doi.org/10.1111/2041-210X.12343
Katz L, Griswold T, Morrison S, Caravas J, Zhang S, Bakker H, Deng X, Carleton H (2019) Mashtree: a rapid comparison of whole genome sequence files. J Open Source Softw 4:1762. https://doi.org/10.21105/joss.01762
Kawa-Rygielska J, Adamenko K, Pietrzak W, Paszkot J, Głowacki A, Gasiński A, Leszczyński P (2021) The potential of traditional Norwegian kveik yeast for brewing novel beer on the example of Foreign Extra Stout. Biomolecules 11:1778. https://doi.org/10.3390/biom11121778
Kawa-Rygielska J, Adamenko K, Pietrzak W, Paszkot J, Głowacki A, Gasiński A (2022) Characteristics of New England India Pale Ale Beer produced with the use of Norwegian kveik yeast. Molecules 27:2291. https://doi.org/10.3390/molecules27072291
Kolmogorov M, Yuan J, Lin Y, Pevzner PA (2019) Assembly of long, error-prone reads using repeat graphs. Nat Biotechnol 37:540–546. https://doi.org/10.1038/s41587-019-0072-8
Kopelman NM, Stone L, Gascuel O, Rosenberg NA (2012) The behavior of admixed populations in neighbor-joining inference of population trees. In: Altman R, Dunker K, Hunter L, Murray T, Klein T (eds) Biocomputing 2013. World Scientific, Singapore, pp 273–284
Krogerus K, Seppänen-Laakso T, Castillo S, Gibson B (2017) Inheritance of brewing-relevant phenotypes in constructed Saccharomyces cerevisiae x Saccharomyces eubayanus hybrids. Microb Cell Fact 16:66. https://doi.org/10.1186/s12934-017-0679-8
Krogerus K, Holmström S, Gibson B (2018a) Enhanced wort fermentation with de novo lager hybrids adapted to high-ethanol environments. Appl Environ Microbiol 84:e02302-e2317. https://doi.org/10.1128/AEM.02302-17
Krogerus K, Preiss R, Gibson B (2018b) A unique Saccharomyces cerevisiae × Saccharomyces uvarum hybrid isolated from Norwegian farmhouse beer: characterization and reconstruction. Front Microbiol 9:2253. https://doi.org/10.3389/fmicb.2018.02253
Krogerus K, Magalhães F, Kuivanen J, Gibson B (2019) A deletion in the STA1 promoter determines maltotriose and starch utilization in STA1+ Saccharomyces cerevisiae strains. Appl Microbiol Biotechnol 103:7597–7615. https://doi.org/10.1007/s00253-019-10021-y
Li H (2011) A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinformatics 27:2987–2993. https://doi.org/10.1093/bioinformatics/btr509
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25:1754–1760. https://doi.org/10.1093/bioinformatics/btp324
Lu Y-J, Swamy KBS, Leu J-Y (2016) Experimental evolution reveals interplay between Sch9 and polyploid stability in yeast. PLOS Genet 12:e1006409. https://doi.org/10.1371/journal.pgen.1006409
Luo S (Rossie), DeMarsh TA, DeRiancho D, Stelick A, Alcaine SD (2021) Characterization of the fermentation and sensory profiles of novel yeast-fermented acid whey beverages. Foods 10:1204. https://doi.org/10.3390/foods10061204
Malinsky M, Matschiner M, Svardal H (2021) Dsuite - Fast D -statistics and related admixture evidence from VCF files. Mol Ecol Resour 21:584–595. https://doi.org/10.1111/1755-0998.13265
Meier-Dörnberg T, Hutzler M, Michel M, Methner F-J, Jacob F (2017) The importance of a comparative characterization of Saccharomyces cerevisiae and Saccharomyces pastorianus strains for brewing. Fermentation 3:41. https://doi.org/10.3390/fermentation3030041
Meirmans PG, Liu S, van Tienderen PH (2018) The analysis of polyploid genetic data. J Hered 109:283–296. https://doi.org/10.1093/jhered/esy006
Milanesi M, Capomaccio S, Vajana E, Bomba L, Garcia JF, Ajmone-Marsan P, Colli L (2017) BITE: an R package for biodiversity analyses. https://doi.org/10.1101/181610
Minh BQ, Nguyen MAT, Von Haeseler A (2013) Ultrafast approximation for phylogenetic bootstrap. Mol Biol Evol 30:1188–1195. https://doi.org/10.1093/molbev/mst024
Nguyen LT, Schmidt HA, Von Haeseler A, Minh BQ (2015) IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32:268–274. https://doi.org/10.1093/molbev/msu300
Nielsen SV, Vaughn AH, Leppälä K, Landis MJ, Mailund T, Nielsen R (2023) Bayesian inference of admixture graphs on Native American and Arctic populations. PLOS Genet 19:e1010410. https://doi.org/10.1371/journal.pgen.1010410
O’Donnell S, Yue J-X, Saada OA, Agier N, Caradec C, Cokelaer T, De Chiara M, Delmas S, Dutreux F, Fournier T, Friedrich A, Kornobis E, Li J, Miao Z, Tattini L, Schacherer J, Liti G, Fischer G (2023) Telomere-to-telomere assemblies of 142 strains characterize the genome structural landscape in Saccharomyces cerevisiae. Nat Genet 55:1390–1399. https://doi.org/10.1038/s41588-023-01459-y
Ondov BD, Treangen TJ, Melsted P, Mallonee AB, Bergman NH, Koren S, Phillippy AM (2016) Mash: fast genome and metagenome distance estimation using MinHash. Genome Biol 17:132. https://doi.org/10.1186/s13059-016-0997-x
Paradis E, Claude J, Strimmer K (2004) APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20:289–290. https://doi.org/10.1093/bioinformatics/btg412
Paszkot J, Gasiński A, Kawa-Rygielska J (2023) Evaluation of volatile compound profiles and sensory properties of dark and pale beers fermented by different strains of brewing yeast. Sci Rep 13:6725. https://doi.org/10.1038/s41598-023-33246-4
Pedersen BS, Quinlan AR (2018) Mosdepth: quick coverage calculation for genomes and exomes. Bioinformatics 34:867–868. https://doi.org/10.1093/bioinformatics/btx699
Peter J, De Chiara M, Friedrich A, Yue J-X, Pflieger D, Bergström A, Sigwalt A, Barre B, Freel K, Llored A, Cruaud C, Labadie K, Aury J-M, Istace B, Lebrigand K, Barbry P, Engelen S, Lemainque A, Wincker P, Liti G, Schacherer J (2018) Genome evolution across 1,011 Saccharomyces cerevisiae isolates. Nature 556:339–344. https://doi.org/10.1038/s41586-018-0030-5
Pickrell JK, Pritchard JK (2012) Inference of population splits and mixtures from genome-wide allele frequency data. PLoS Genet 8:e1002967. https://doi.org/10.1371/journal.pgen.1002967
Pontes A, Hutzler M, Brito PH, Sampaio JP (2020) Revisiting the taxonomic synonyms and populations of Saccharomyces cerevisiae—phylogeny, phenotypes, ecology and domestication. Microorganisms 8:903. https://doi.org/10.3390/microorganisms8060903
Preiss R, Tyrawa C, Krogerus K, Garshol LM, van der Merwe G (2018) Traditional Norwegian Kveik are a genetically distinct group of domesticated Saccharomyces cerevisiae brewing yeasts. Front Microbiol 9:2137. https://doi.org/10.3389/fmicb.2018.02137
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MAR, Bender D, Maller J, Sklar P, de Bakker PIW, Daly MJ, Sham PC (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81:559–575. https://doi.org/10.1086/519795
Räsänen M (1975) Vom Halm zum Fass. Suomen muinaismuistoyhdistys, Helsinki
Roesdahl E (2016) The Vikings. Penguin Random House, London
Salomonsson A (1979) Gotlandsdricka. Karlstad Press, Visby
Schneiderbanger H, Koob J, Poltinger S, Jacob F, Hutzler M (2016) Gene expression in wheat beer yeast strains and the synthesis of acetate esters. J Inst Brew 122:403–411. https://doi.org/10.1002/jib.337
Selmecki AM, Maruvka YE, Richmond PA, Guillet M, Shoresh N, Sorenson AL, De S, Kishony R, Michor F, Dowell R, Pellman D (2015) Polyploidy can drive rapid adaptation in yeast. Nature 519:349–352. https://doi.org/10.1038/nature14187
Tarasov A, Vilella AJ, Cuppen E, Nijman IJ, Prins P (2015) Sambamba: fast processing of NGS alignment formats. Bioinformatics 31:2032–2034. https://doi.org/10.1093/bioinformatics/btv098
Trindade de Carvalho B, Holt S, Souffriau B, Lopes Brandão R, Foulquié-Moreno MR, Thevelein JM (2017) Identification of novel alleles conferring superior production of rose flavor phenylethyl acetate using polygenic analysis in yeast. mBio 8. https://doi.org/10.1128/mbio.01173-17
Waymark C, Hill AE (2021) The influence of yeast strain on whisky new make spirit aroma. Fermentation 7:311. https://doi.org/10.3390/fermentation7040311
Funding
Open Access funding provided by Technical Research Centre of Finland. Research at University of Guelph was funded by Natural Sciences and Engineering Research Council of Canada Discovery (NSERC #264792–400922), Alliance-GLAAIR Product Development (UG-GLPD-2021–101214), and Ontario Regional Priorities Partnership program (ON-RP3 06) grants. Research at VTT was funded by the Research Council of Finland (Academy Research Fellow 355120).
Author information
Authors and Affiliations
Contributions
RP conceived the study, designed experiments, and wrote the manuscript.
EF conceived the study, designed experiments, and performed experiments.
LG provided yeast strains and wrote the manuscript.
BF performed data analysis.
EO performed data analysis.
ML performed data analysis.
GM designed experiments and edited the manuscript.
KK conceived the study, designed experiments, performed experiments, performed data analysis, and wrote the manuscript.
All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
Richard Preiss and Eugene Fletcher were employed by Escarpment Laboratories Inc. Kristoffer Krogerus was employed by VTT Technical Research Centre of Finland Ltd. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Preiss, R., Fletcher, E., Garshol, L.M. et al. European farmhouse brewing yeasts form a distinct genetic group. Appl Microbiol Biotechnol 108, 430 (2024). https://doi.org/10.1007/s00253-024-13267-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00253-024-13267-3