Abstract
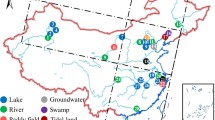
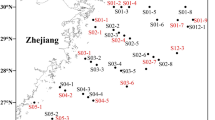
Ammonia-oxidizing archaea (AOA) are ubiquitously found in diverse habitats and play pivotal roles in the nitrogen and carbon cycle, especially in estuarine and coastal environments. Despite the fact that the diversity and distribution of AOA are thought to be tightly linked to habitats, little is known about the relationship that underpins their genomic traits, adaptive potentials, and ecological niches. Here, we have characterized and compared the AOA community in three estuaries of China using metagenomics. AOA were the dominant ammonia oxidizers in the three estuaries. Through phylogenetic analyses, five major AOA groups were identified, including the Nitrosomarinus-like, Nitrosopumilus-like, Aestuariumsis-like, Nitrosarchaeum-like, and Nitrosopelagicus-like groups. Statistical analyses showed that the aquatic and sedimentary AOA communities were mainly influenced by spatial factors (latitude and water depth) and environmental factors (salinity, pH, and dissolved oxygen) in estuaries, respectively. Compared to AOA dwelling in terrestrial and marine habitats, estuarine AOA encoded more genes involved in glucose and amino acid metabolism, transport systems, osmotic control, and cell motility. The low proteome isoelectric points (pI), high content of acidic amino acids, and the presence of potassium ion and mechanosensitive channels suggest a “salt-in” strategy for estuarine AOA to counteract high osmolarity in their surroundings. Our findings have indicated potential adaptation strategies and highlighted their importance in the estuarine nitrogen and carbon cycles.
Key points
• Spatial and environmental factors influence water and sediment AOA respectively.
• Estuarine AOA share low proteome isoelectric value and high acid amino acids content.
• AOA adaptation to estuaries is likely resulted from their unique genomic features.




Similar content being viewed by others
Data availability
The raw reads for samples and reconstructed AOA MAGs from the CRE, the JRE, and the PRE are available in the National Omics Data Encyclopedia (NODE) database (http://www.biosino.org/node) under the Bioproject accession numbers OEP001524, OEP000961, and OEP004282, respectively.
References
Abby SS, Kerou M, Schleper C (2020) Ancestral reconstructions decipher major adaptations of ammonia-oxidizing archaea upon radiation into moderate terrestrial and marine environments. Mbio 11(5):e02371-e2420
Alves RJE, Minh BQ, Urich T, von Haeseler A, Schleper C (2018) Unifying the global phylogeny and environmental distribution of ammonia-oxidising archaea based on amoA genes. Nat Commun 9(1):1–17
Armenteros JA, Tsirigos KD, Sønderby CK, Petersen TN, Winther O, Brunak S, von Heijne G, Nielsen H (2019) SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat Biotechnol 37(4):420–423
Barnett DW, Garrison EK, Quinlan AR, Strömberg MP, Marth GT (2011) BamTools: a C++ API and toolkit for analyzing and managing BAM files. Bioinformatics 27(12):1691–1692
Bernhard AE, Landry ZC, Blevins A, de la Torre JR, Giblin AE, Stahl DA (2010) Abundance of ammonia-oxidizing archaea and bacteria along an estuarine salinity gradient in relation to potential nitrification rates. Appl Environ Microbiol 76(4):1285–1289
Biller SJ, Mosier AC, Wells GF, Francis CA (2012) Global biodiversity of aquatic ammonia-oxidizing archaea is partitioned by habitat. Front Microbiol 3:252
Blainey PC, Mosier AC, Potanina A, Francis CA, Quake SR (2011) Genome of a low-salinity ammonia-oxidizing archaeon determined by single-cell and metagenomic analysis. PLoS ONE 6(2):e16626
Bollmann A, Laanbroek HJ (2002) Influence of oxygen partial pressure and salinity on the community composition of ammonia-oxidizing bacteria in the Schelde estuary. Aquat Microb Ecol 28(3):239–247
Cébron A, Berthe T, Garnier J (2003) Nitrification and nitrifying bacteria in the lower Seine River and estuary (France). Appl Environ Microbiol 69(12):7091–7100
Cabello-Yeves PJ, Rodriguez-Valera F (2019) Marine-freshwater prokaryotic transitions require extensive changes in the predicted proteome. Microbiome 7(1):1–12
Cabello-Yeves PJ, Zemskaya TI, Zakharenko AS, Sakirko MV, Ivanov VG, Ghai R, Rodriguez-Valera F (2020) Microbiome of the deep Lake Baikal, a unique oxic bathypelagic habitat. Limnol Oceanogr 65(7):1471–1488
Cao H, Auguet J-C, Gu J-D (2013) Global ecological pattern of ammonia-oxidizing archaea. PLoS ONE 8(2):e52853
Cheung S, Mak W, Xia X, Lu Y, Cheung Y, Liu H (2019) Overlooked genetic diversity of ammonia oxidizing archaea lineages in the global oceans. J Geophys Res-Biogeo 124(7):1799–1811
Dai M, Wang L, Guo X, Zhai W, Li Q, He B, Kao S-J (2008) Nitrification and inorganic nitrogen distribution in a large perturbed river/estuarine system: the Pearl River Estuary China. Biogeosciences 5(5):1227–1244
Daims H, Lebedeva EV, Pjevac P, Han P, Herbold C, Albertsen M, Jehmlich N, Palatinszky M, Vierheilig J, Bulaev A (2015) Complete nitrification by Nitrospira bacteria. Nature 528(7583):504–509
Dang H, Zhang X, Sun J, Li T, Zhang Z, Yang G (2008) Diversity and spatial distribution of sediment ammonia-oxidizing crenarchaeota in response to estuarine and environmental gradients in the Changjiang Estuary and East China Sea. Microbiology 154(7):2084–2095
Eren AM, Esen ÖC, Quince C, Vineis JH, Morrison HG, Sogin ML, Delmont TO (2015) Anvi’o: an advanced analysis and visualization platform for ‘omics data. PeerJ 3:e1319
Francis CA, Roberts KJ, Beman JM, Santoro AE, Oakley BB (2005) Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. Proc Natl Acad Sci 102(41):14683–14688
Gianoulis TA, Raes J, Patel PV, Bjornson R, Korbel JO, Letunic I, Yamada T, Paccanaro A, Jensen LJ, Snyder M (2009) Quantifying environmental adaptation of metabolic pathways in metagenomics. Proc Natl Acad Sci 106(5):1374–1379
Gunde-Cimerman N, Plemenitaš A, Oren A (2018) Strategies of adaptation of microorganisms of the three domains of life to high salt concentrations. FEMS Microbiol Rev 42(3):353–375
Hou L, Xie X, Wan X, Kao S-J, Jiao N, Zhang Y (2018) Niche differentiation of ammonia and nitrite oxidizers along a salinity gradient from the Pearl River estuary to the South China Sea. Biogeosciences 15(16):5169–5187
Hu H-W, Zhang L-M, Yuan C-L, Zheng Y, Wang J-T, Chen D, He J-Z (2015) The large-scale distribution of ammonia oxidizers in paddy soils is driven by soil pH, geographic distance, and climatic factors. Front Microbiol 6:938
Huerta-Cepas J, Forslund K, Coelho LP, Szklarczyk D, Jensen LJ, Von Mering C, Bork P (2017) Fast genome-wide functional annotation through orthology assignment by eggNOG-mapper. Mol Biol Evol 34(8):2115–2122
Hyatt D, Chen G-L, LoCascio PF, Land ML, Larimer FW, Hauser LJ (2010) Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinformatics 11(1):1–11
Jain C, Rodriguez-R LM, Phillippy AM, Konstantinidis KT, Aluru S (2018) High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries. Nat Commun 9(1):1–8
Käll L, Krogh A, Sonnhammer EL (2007) Advantages of combined transmembrane topology and signal peptide prediction—the Phobius web server. Nucleic Acids Res 35(suppl_2):W429–W432
Könneke M, Bernhard AE, José R, Walker CB, Waterbury JB, Stahl DA (2005) Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nature 437(7058):543–546
KamandaNgugi D, Blom J, Alam I, Rashid M, Ba-Alawi W, Zhang G, Hikmawan T, Guan Y, Antunes A, Siam R (2015) Comparative genomics reveals adaptations of a halotolerant thaumarchaeon in the interfaces of brine pools in the Red Sea. ISME J 9(2):396–411
Kanehisa M, Sato Y, Morishima K (2016) BlastKOALA and GhostKOALA: KEGG tools for functional characterization of genome and metagenome sequences. J Mol Biol 428(4):726–731
Kang DD, Li F, Kirton E, Thomas A, Egan R, An H, Wang Z (2019) MetaBAT 2: an adaptive binning algorithm for robust and efficient genome reconstruction from metagenome assemblies. PeerJ 7:e7359
Kerou M, Ponce-Toledo RI, Zhao R, Abby SS, Hirai M, Nomaki H, Takaki Y, Nunoura T, Jørgensen SL, Schleper C (2021) Genomes of Thaumarchaeota from deep sea sediments reveal specific adaptations of three independently evolved lineages. ISME J 15(9):2792–2808
Kitzinger K, Padilla CC, Marchant HK, Hach PF, Herbold CW, Kidane AT, Könneke M, Littmann S, Mooshammer M, Niggemann J (2019) Cyanate and urea are substrates for nitrification by Thaumarchaeota in the marine environment. Nat Microbiol 4(2):234–243
Kowalchuk GA, Stephen JR (2001) Ammonia-oxidizing bacteria: a model for molecular microbial ecology. Ann Rev Microbiol 55(1):485–529
Lairson L, Henrissat B, Davies G, Withers S (2008) Glycosyltransferases: structures, functions, and mechanisms. Annu Rev Biochem 77:521–555
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9(4):357–359
Leininger S, Urich T, Schloter M, Schwark L, Qi J, Nicol GW, Prosser JI, Schuster S, Schleper C (2006) Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 442(7104):806–809
Letunic I, Bork P (2021) Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res 49(W1):W293–W296
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25(16):2078–2079
Li M, Cao H, Hong Y, Gu J-D (2011) Spatial distribution and abundances of ammonia-oxidizing archaea (AOA) and ammonia-oxidizing bacteria (AOB) in mangrove sediments. Appl Microbiol Biotechnol 89(4):1243–1254
Li M, Wei G, Shi W, Sun Z, Li H, Wang X, Gao Z (2018) Distinct distribution patterns of ammonia-oxidizing archaea and bacteria in sediment and water column of the Yellow River estuary. Sci Rep 8(1):1–10
Liu Q, Tolar BB, Ross MJ, Cheek JB, Sweeney CM, Wallsgrove NJ, Popp BN, Hollibaugh JT (2018a) Light and temperature control the seasonal distribution of thaumarchaeota in the South Atlantic bight. ISME J 12(6):1473–1485
Liu X, Pan J, Liu Y, Li M, Gu J-D (2018b) Diversity and distribution of Archaea in global estuarine ecosystems. Sci Total Environ 637:349–358
Luo H, Tolar BB, Swan BK, Zhang CL, Stepanauskas R, Moran MA, Hollibaugh JT (2014) Single-cell genomics shedding light on marine Thaumarchaeota diversification. ISME J 8(3):732–736
Martens-Habbena W, Berube P, Urakawa H, de La Torre J, Stahl D (2009) Ammonia oxidation kinetics determine niche separation of nitrifying archaea and and bacteria. Nature 461(7266):976–979
McLusky DS, Elliott M (2004) The estuarine ecosystem: ecology, threats and management. Oxford University Press, Oxford
Merbt SN, Stahl DA, Casamayor EO, Martí E, Nicol GW, Prosser JI (2012) Differential photoinhibition of bacterial and archaeal ammonia oxidation. FEMS Microbiol Lett 327(1):41–46
Milliman JD, Huang-Ting S, Zuo-Sheng Y, Mead RH (1985) Transport and deposition of river sediment in the Changjiang estuary and adjacent continental shelf. Cont Shelf Res 4(1–2):37–45
Mosier AC, Francis CA (2008) Relative abundance and diversity of ammonia-oxidizing archaea and bacteria in the San Francisco Bay estuary. Environ Microbiol 10(11):3002–3016
Mosier AC, Lund MB, Francis CA (2012) Ecophysiology of an ammonia-oxidizing archaeon adapted to low-salinity habitats. Microb Ecol 64(4):955–963
Ngugi DK, Blom J, Alam I, Rashid M, Ba-Alawi W, Zhang G, Hikmawan T, Guan Y, Antunes A, Siam R (2015) Comparative genomics reveals adaptations of a halotolerant thaumarchaeon in the interfaces of brine pools in the Red Sea. ISME J 9(2):396–411
Nicol GW, Leininger S, Schleper C, Prosser JI (2008) The influence of soil pH on the diversity, abundance and transcriptional activity of ammonia oxidizing archaea and bacteria. Environ Microbiol 10(11):2966–2978
Parks DH, Imelfort M, Skennerton CT, Hugenholtz P, Tyson GW (2015) CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res 25(7):1043–1055
Paul S, Bag SK, Das S, Harvill ET, Dutta C (2008) Molecular signature of hypersaline adaptation: insights from genome and proteome composition of halophilic prokaryotes. Genome Biol 9(4):1–19
Peng Y, Leung HC, Yiu S-M, Chin FY (2012) IDBA-UD: a de novo assembler for single-cell and metagenomic sequencing data with highly uneven depth. Bioinformatics 28(11):1420–1428
Pester M, Rattei T, Flechl S, Gröngröft A, Richter A, Overmann J, Reinhold-Hurek B, Loy A, Wagner M (2012) amoA-based consensus phylogeny of ammonia-oxidizing archaea and deep sequencing of amoA genes from soils of four different geographic regions. Environ Microbiol 14(2):525–539
Pester M, Schleper C, Wagner M (2011) The Thaumarchaeota: an emerging view of their phylogeny and ecophysiology. Curr Opin Microbiol 14(3):300–306
Qin W, Zheng Y, Zhao F, Wang Y, Urakawa H, Martens-Habbena W, Liu H, Huang X, Zhang X, Nakagawa T (2020) Alternative strategies of nutrient acquisition and energy conservation map to the biogeography of marine ammonia-oxidizing archaea. ISME J 14(10):2595–2609
Ren M, Feng X, Huang Y, Wang H, Hu Z, Clingenpeel S, Swan BK, Fonseca MM, Posada D, Stepanauskas R (2019) Phylogenomics suggests oxygen availability as a driving force in Thaumarchaeota evolution. ISME J 13(9):2150–2161
Rice P, Longden I, Bleasby A (2000) EMBOSS: the European molecular biology open software suite. Trends Genet 16(6):276–277
Rodriguez-Brito B, Rohwer F, Edwards RA (2006) An application of statistics to comparative metagenomics. BMC Bioinformatics 7(1):1–11
Santos JP, Mendes D, Monteiro M, Ribeiro H, Baptista MS, Borges MT, Magalhães C (2018) Salinity impact on ammonia oxidizers activity and amoA expression in estuarine sediments. Estuar Coast Shelf Sci 211:177–187
Scarlett K, Denman S, Clark DR, Forster J, Vanguelova E, Brown N, Whitby C (2021) Relationships between nitrogen cycling microbial community abundance and composition reveal the indirect effect of soil pH on oak decline. ISME J 15(3):623–635
Schleper C, Nicol GW (2010) Ammonia-oxidising archaea–physiology, ecology and evolution. Adv Microb Physiol 57:1–41
Sieber CM, Probst AJ, Sharrar A, Thomas BC, Hess M, Tringe SG, Banfield JF (2018) Recovery of genomes from metagenomes via a dereplication, aggregation and scoring strategy. Nat Microbiol 3(7):836–843
Sintes E, Bergauer K, De Corte D, Yokokawa T, Herndl GJ (2013) Archaeal amoA gene diversity points to distinct biogeography of ammonia-oxidizing Crenarchaeota in the ocean. Environ Microbiol 15(5):1647–1658
Soppa J (2006) From genomes to function: haloarchaea as model organisms. Microbiology 152(3):585–590
Stahl DA, de la Torre JR (2012) Physiology and diversity of ammonia-oxidizing archaea. Annu Rev Microbiol 66:83–101
Stieglmeier M, Alves RJE, Schleper C (2014) The phylum thaumarchaeota. In: The prokaryotes: other major lineages of bacteria and the archaea. Springer, Berlin
Stocker R, Seymour JR, Samadani A, Hunt DE, Polz MF (2008) Rapid chemotactic response enables marine bacteria to exploit ephemeral microscale nutrient patches. Proc Natl Acad Sci 105(11):4209–4214
Sun X, Zhao J, Zhou X, Bei Q, Xia W, Zhao B, Zhang J, Jia Z (2022) Salt tolerance-based niche differentiation of soil ammonia oxidizers. ISME J 16(2):412–422
Van Kessel MA, Speth DR, Albertsen M, Nielsen PH, den Camp HJO, Kartal B, Jetten MS, Lücker S (2015) Complete nitrification by a single microorganism. Nature 528(7583):555–559
Van Melderen L (2010) Toxin–antitoxin systems: why so many, what for? Curr Opin Microbiol 13(6):781–785
Wang B, Qin W, Ren Y, Zhou X, Jung M-Y, Han P, Eloe-Fadrosh EA, Li M, Zheng Y, Lu L (2019) Expansion of Thaumarchaeota habitat range is correlated with horizontal transfer of ATPase operons. ISME J 13(12):3067–3079
Wang W, Yu Z, Wu Z, Song S, Song X, Yuan Y, Cao X (2018) Rates of nitrification and nitrate assimilation in the Changjiang River estuary and adjacent waters based on the nitrogen isotope dilution method. Cont Shelf Res 163:35–43
Wankel SD, Mosier AC, Hansel CM, Paytan A, Francis CA (2011) Spatial variability in nitrification rates and ammonia-oxidizing microbial communities in the agriculturally impacted Elkhorn Slough estuary California. Appl Environ Microbiol 77(1):269–280
Widderich N, Czech L, Elling FJ, Könneke M, Stöveken N, Pittelkow M, Riclea R, Dickschat JS, Heider J, Bremer E (2016) Strangers in the archaeal world: osmostress-responsive biosynthesis of ectoine and hydroxyectoine by the marine thaumarchaeon Nitrosopumilus maritimus. Environ Microbiol 18(4):1227–1248
Xie W, Luo H, Murugapiran SK, Dodsworth JA, Chen S, Sun Y, Hedlund BP, Wang P, Fang H, Deng M (2018) Localized high abundance of Marine Group II archaea in the subtropical Pearl River Estuary: implications for their niche adaptation. Environ Microbiol 20(2):734–754
Xie Y-G, Luo Z-H, Fang B-Z, Jiao J-Y, Xie Q-J, Cao X-R, Qu Y-N, Qi Y-L, Rao Y-Z, Li Y-X (2022) Functional differentiation determines the molecular basis of the symbiotic lifestyle of Ca. Nanohaloarchaeota Microbiome 10(1):1–13
Yang Y, Zhang C, Lenton TM, Yan X, Zhu M, Zhou M, Tao J, Phelps TJ, Cao Z (2021) The evolution pathway of ammonia-oxidizing archaea shaped by major geological events. Mol Biol Evol 38(9):3637–3648
Yin Y, Mao X, Yang J, Chen X, Mao F, Xu Y (2012) dbCAN: a web resource for automated carbohydrate-active enzyme annotation. Nucleic Acids Res 40(W1):W445–W451
Yu D, Chen N, Krom MD, Lin J, Cheng P, Yu F, Guo W, Hong H, Gao X (2019) Understanding how estuarine hydrology controls ammonium and other inorganic nitrogen concentrations and fluxes through the subtropical Jiulong River Estuary, SE China under baseflow and flood-affected conditions. Biogeochemistry 142(3):443–466
Zhang Y, Chen L, Dai T, Tian J, Wen D (2015) The influence of salinity on the abundance, transcriptional activity, and diversity of AOA and AOB in an estuarine sediment: a microcosm study. Appl Microbiol Biotechnol 99:9825–9833
Zhou Z, Zhang G-X, Xu Y-B, Gu J-D (2018) Successive transitory distribution of Thaumarchaeota and partitioned distribution of Bathyarchaeota from the Pearl River estuary to the northern South China Sea. Appl Microbiol Biotechnol 102(18):8035–8048
Zou D, Li H, Du P, Wang B, Lin H, Liu H, Chen J, Li M (2022) Distinct features of sedimentary archaeal communities in hypoxia and non-hypoxia regions off the Changjiang River Estuary. Microbiol Spectr 10(5):e01947-e2022
Zou D, Li Y, Kao SJ, Liu H, Li M (2019) Genomic adaptation to eutrophication of ammonia-oxidizing archaea in the Pearl River estuary. Environ Microbiol 21(7):2320–2332
Zou D, Wan R, Han L, Xu MN, Liu Y, Liu H, Kao S-J, Li M (2020) Genomic characteristics of a novel species of ammonia-oxidizing Archaea from the Jiulong River Estuary. Appl Environ Microbiol 86(18):e00736-e820
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 32225003, 31970105, 992251306, 42141003); the Shenzhen Science and Technology Program (grant number JCYJ20200109105010363); the Innovation Team Project of Universities in Guangdong Province (grant number 2020KCXTD023); Shenzhen University 2035 Program for Excellent Research (2022B002); the China Postdoctoral Science Foundation (grant number. 2022M722175); Long Term Observation and Research Plan in the Changjiang Estuary and the Adjacent East China Sea Project (LORCE) (grant number 14282); and the National Program on Global Change and Air-Sea Interaction (Phase II)—Hypoxia and Acidification Monitoring and Warning Project in the Changjiang Estuary. CZ also acknowledges supports from the Department of Science and Technology of Guangdong Province (grant number 2021B151512008), the Shenzhen Key Laboratory of Marine Archaea Geo-Omics, Southern University of Science and Technology (grant number ZDSYS201802081843490), and the Southern Marine Science and Engineering Guangdong Laboratory (Guangzhou) (grant number K19313901). HL acknowledges the support by the Hong Kong Branch of Southern Marine Science and Engineering Guangdong Laboratory (Guangzhou) (grant number SMSEGL20SC01).
Author information
Authors and Affiliations
Contributions
DZ and ML conceptualized the study. JC, CZ, SK, and HL provided the samples. DZ performed laboratory work, collected the data, performed the analyses and wrote the manuscript with the help from all co-authors.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zou, D., Chen, J., Zhang, C. et al. Diversity and salinity adaptations of ammonia oxidizing archaea in three estuaries of China. Appl Microbiol Biotechnol 107, 6897–6909 (2023). https://doi.org/10.1007/s00253-023-12761-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12761-4