Abstract
Weaning stress has serious negative effects on piglets’ health and the swine industry. Probiotics-fermented Chinese herbal medicines are potential feed additives to ameliorate weaning stress. In this study, the effects of probiotics-fermented Massa Medicata Fermentata (MMFP) on intestinal homeostasis were evaluated in weaning piglets. Dietary supplementation with MMFP promoted the development of the intestinal structure and elevated the concentrations of lactic acid and short-chain fatty acids (SCFAs) in the intestinal contents and antioxidant capacities in serum. MMFP reduced the levels of inflammatory factors in the intestinal mucosa. Microbial community analysis demonstrated that MMFP led to the selective and progressive enrichment of lactic acid- and SCFA-producing bacteria along the gastrointestinal tract, in particular, OTUs corresponding to Lactobacillus, Streptococcus, Acetitomaculum, Roseburia, and Eubacterium xylanophilum group, while MMFP reduced the relative abundance of pathogenic bacteria. On the contrary, antibiotics had negative effects on intestinal histology and increased the relative abundance of pro-inflammatory bacterium, such as Marvinbryantia, Peptococcus, Turicibacter, and Blautia. Correlation analysis reflected that the bacteria enriched in MMFP group were positively correlated with enhanced intestinal homeostasis, which suggested that dietary supplementation with MMFP enhanced host intestinal homeostasis by modulating the composition of gut microbiota and the levels of beneficial SCFAs, thus ameliorating weaning stress in piglets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Weaning transition is a crucial period in the piglet’s growth cycle. In the swine industry, compared with the natural environment, shorter suckling period as well as abrupt changes in dietary and social environment frequently lead to anorexia, growth retardation, severe enteric infections, or even death (Gresse et al. 2017; Lalles et al. 2007). In general, antibiotics are widely used to treat bacterial infections and promote animal growth. However, the overuse of antibiotics results in a growing number of drug-resistant pathogens and raises important concerns about animal and human health. Antibiotic-resistant strains of enterobacteria, such as Clostridium difficile, Enterobacteriaceae, Enterococcus spp., and Staphylococcus aureus, pose serious risks to human health (Gross 2013; Tanne 2012). Antibiotics are being gradually banned in various countries. Therefore, new and efficient feed additives to improve weaning stress in piglets are needed.
Intestinal dysfunction, such as stomachache, diarrhea, constipation, and anorexia, is the most common and primary symptom during the animal weaning period. Huge diversity of microorganisms inhabit in the gastrointestinal (GI) tract of mammals and provide many beneficial effects to the host (Buffie and Pamer 2013; Kamada et al. 2013). Commensal bacteria in the intestine can inhibit the expansion of pathogens through colonization resistance and defensive metabolites. Besides, organic acids, produced by the commensal bacteria, contribute to intestinal homeostasis and systemic immunity (Louis et al. 2014). Therefore, manipulation of intestinal microbiota is likely to be one of the keys to ameliorate weaning stress.
Massa Medicata Fermentata (MMF) is a widely clinically used Chinese herbal medicine, mainly for intestinal dysfunction (Zhang et al. 2012). In general, MMF is spontaneously fermented with red bean, Semen Armeniacae Amarum., Artemisia carvifolia, Polygonum hydropiper L., Xanthium sibiricum, wheat bran, and flour. Chinese herbal formulas containing MMF have attenuated ethanol-induced acute gastric injury in rats (Shin et al. 2013) and alleviated neomycin-induced colon injury and disorders in gut microbiota (Bose et al. 2013). However, pathogens and fungal toxins, such as aflatoxin, are usually detected in traditional MMF (Liu et al. 2017). This restricts its clinical application. According to the microbial community dynamics during MMF fermentation (Xu et al. 2013), we modified the fermentation strains with definite probiotics (Bacillus sp., Lactobacillus sp., Streptococcus thermophilus, Enterococcus faecalis, Saccharomyces cerevisiae, Aspergillus oryzae, and Mucor racemosu) instead of random and spontaneous strains (Wang et al., submitted for publication). These probiotics were widely used in food industry and have been proved beneficial to the health of the host (de Almada et al. 2015). Probiotics-fermented Massa Medicata Fermentata (MMFP) combines the advantages of Chinese herbal medicines, digestive enzymes, and probiotics, which has given it great potential value for development and application.
In this study, we aimed to evaluate the effects of MMFP as a novel feed additive on weaning stress in piglets. We systematically analyzed the effects of dietary supplementation with MMFP on intestinal histology, the intestinal immune system, the antioxidant capacities in serum, and the levels of organic acids in intestinal contents. The microbial composition along the GI tract and feces was analyzed through high-throughput sequencing. This is the first report about the ability of probiotics-fermented Chinese medicine to improve weaning stress in piglets. The current study may contribute to the development of new and efficient feed additives. As reviewed by Heinritz et al. (Heinritz et al. 2013), there are strong physiological similarities between humans and pigs, so this study could serve as a reference for the clinical application of probiotics-fermented Chinese medicines.
Materials and methods
Animals, housing, and diets
The experiment was conducted by completely random design. A total of 24 healthy and well-developed piglets (Songliao black swine, initial body weight 15.61 ± 1.52 kg) were used in this study. These pigs were randomly assigned to three treatments (eight pigs per treatment) the day after weaning (day 0). The treatment groups were (a) Con group, in which pigs were fed a basal diet (Supplementary Table S1); (b) Ant group, in which pigs were fed the basal diet supplemented with colistin sulphate (0.3 g/kg feed) and bacitracin zinc (0.5 g/kg feed); and (c) MMFP group, in which pigs were fed the basal diet supplemented with MMFP (2 g/kg feed). MMFP was fermented with Bacillus licheniformis CICC24236, Bacillus subtilis CICC24434, Lactobacillus reuteri CICC6119, Lactobacillus plantarum CICC20261, Streptococcus thermophilus CICC6216, Enterococcus faecalis CGMCC1.10682, Saccharomyces cerevisiae CICC1005, Aspergillus oryzae CICC2013, and Mucor racemosu CICC40491 according to the National Drug Standards (1998) (Wang et al., submitted for publication). The composition of nutritional ingredients was according to the National Research Council (2012). Animals were housed in a controlled environment (26–28 °C, humidity 65–70%) with feed and water ad libitum during a 49-day trial period. The animal experiment was approved by the Institutional Animal Care and Use Committee of Jilin University (IACUC), and the animals were maintained according to the laboratory animal-guideline for ethical review of animal welfare.
Performance measurements and sample collection
Body weight and feed consumption were measured weekly, and average daily gain (ADG) and average daily feed intake (ADFI) were calculated. In this study, ADG was calculated as follows: ADG = (final body weight − initial body weight) / 49. Only survived pigs on day 49 were calculated. The ADFI was calculated as follows: ADFI = (feed consumption each week) / (7 days × n), n = the number of survived pigs. Fecal consistency score (0 = normal feces; 1 = soft and moist feces; 2 = mild diarrhea; 3 = severe, watery diarrhea) was recorded daily (Casey et al. 2007). The diarrhea incidence was evaluated according to a previous study (Sun et al. 2008). Piglets’ morbidity and mortality was recorded throughout the trial period.
Fresh fecal samples were collected at day 0 and day 49 as follows: nine sampling points were randomly selected in each pen, and then fecal samples from three points were mixed together as a specimen (three fecal samples per pen). At the end of the trial period, carotid artery blood samples from all of the survived pigs were collected by vacuum blood collection tubes and centrifuged at 3000 rpm for 10 min at 4 °C. One animal from each pen was selected randomly and sacrificed. Intestinal tissues were collected from standardized areas of the ileum (100 cm from the ileocecal valve), cecum (central part of the cecum), and colon (central part of the colon). Contents form the cecum, proximal colon (colon 1), medial colon (colon 2), distal colon (colon 3), and rectum were collected with sterile tubes. Mucosa from the jejunum, ileum, cecum, proximal colon (colon 1), medial colon (colon 2), distal colon (colon 3), and rectum was gently scraped with sterile glass slides. All of the samples were stored at − 80 °C before processing.
Histological analysis
Intestinal tissues were flushed with sterile PBS, fixed in 10% neutral-buffered formalin, processed, embedded in paraffin, cut at 5 μm thickness, and stained with hematoxylin and eosin (H&E). More than six complete crypt-villus units were randomly selected from each sample. Measurements of mucosal thickness, villus height, and crypt depth were performed using a Nikon biological microscope (E100) equipped with an ISH500 camera (Tucsen, Fuzhou, China) and ISCapture (version 3.5) morphological analysis software.
Analysis of antioxidant capacities in serum
The levels of total antioxidant capacity (T-AOC), superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), catalase (CAT), and malondialdehyde (MDA) in serum were determined using commercially available assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions.
Analysis of inflammatory factors in serum and intestinal mucosa
The levels of monocyte chemotactic protein 1 (MCP-1), interleukin 1β (IL-1β), interleukin 6 (IL-6), and interleukin 10 (IL-10) in serum and the intestinal mucosa were determined using commercial enzyme-linked immunosorbent assay (ELISA) kits (CUSABIO®, Wuhan, China) according to the manufacturer’s instructions.
Analysis of microbial metabolites in intestinal contents
Concentrations of lactic acid and SCFAs in intestinal contents were analyzed by high-performance liquid chromatography (HPLC). Sample pretreatment and derivatization were according to previous studies (Gao et al. 2017) with slight modifications. Briefly, 1 g of intestinal contents were suspended with 10 mL ddH2O and thoroughly homogenized by vortexing for 1 min. Then, samples were centrifuged at 12,000×g for 5 min. 3-Nitrophenylhydrazine hydrochloride (3-NPH·HCl, Aladdin®, Shanghai, China) was dissolved in methanol at a concentration of 20 mM. 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (1-EDC·HCl, Aladdin®, Shanghai, China) was dissolved in 3% pyridine–methanol solution at a concentration of 0.25 M. Each sample (50 μL) was mixed with 200 μL of 3-NPH·HCl solution and 100 μL of 1-EDC·HCl solution, followed by incubation at 60 °C for 20 min. After cooling, the mixture was filtered through a 0.22 μm filter.
HPLC was performed using an ACCHROM S6000 high-performance liquid chromatograph (ACCHROM, Japan) equipped with a XAqua C18 4.6 mm × 250 mm column (ACCHROM, China). The mobile phase was composed of methanol–water (32:68), with the column temperature of 40 °C, flow rate of 1.2 mL/min, and wavelength of 230 nm.
DNA extraction, sequencing, and data processing
Genomic DNA was extracted from intestinal contents and feces using a TIANamp Stool DNA Kit (Tiangen, Beijing, China) according to the manufacturer’s instructions. The final yield and quality of the extracted DNA were determined spectrophotometrically using a NANODROP 1000TM UV/VIS Spectrophotometer (Thermo, USA). The v3 and v4 regions of the 16S rRNA gene were amplified using forward primers of the sequence 5′-CCTACGGRRBGCASCAGKVRVGAAT-3′ and reverse primers of the sequence 5′-GGACTACNVGGGTWTCTAATCC-3′. PCR was performed in a total of 20 μL volume, containing 1 × PCR Buffer (Mg2+ plus), 200 μM of dNTP, 0.1 μM each of the primer, 1 U of TaKaRa Taq Polymerase (Takara, Dalian, China), and 10 ng of template DNA. The v3–v4 regions were amplified at 94 °C for 3 min, followed by 24 cycles of 94 °C for 5 s, 57 °C for 90 s, and 72 °C for 10 s and a final extension at 72 °C for 5 min. The first round PCR products were used as templates for second round amplicon enrichment PCR. At the same time, indexed adapters were added to the ends of the 16S rDNA amplicons to generate indexed libraries ready for downstream NGS sequencing on Illumina Miseq.
Statistical analysis
The data are expressed as the mean values ± SEM. Statistical analyses were performed in SPSS Statistics 17.0 using the tables and data generated above. A value of p < 0.05 was considered statistically significant. Multiple comparisons were conducted using one-way analysis of variance (ANOVA). Figures were plotted with OriginPro 8 SR0 (v8.0724). The alpha-diversity of microbial communities was calculated by Mothur software (v.1.30). Heatmaps of bacterial abundance were made using R (V.3.2.3). Principal component analysis and correlation analysis were performed using MetaboAnalyst 3.6 (http://www.metaboanalyst.ca/faces/upload/StatUploadView.xhtml).
Sequence data accession number
High-throughput sequencing data have been submitted to the NCBI Sequence Read Archive (SRA) under accession numbers SRP126208 and SRP126217.
Results
MMFP ameliorates weaning stress in piglets
Antibiotic treatment significantly improved the growth rate (Fig. 1, p < 0.01) and ADFI (Table 1) of piglets compared with Con group. Although MMFP lagged behind the Ant group, pigs in the MMFP group showed a higher growth rate (Fig. 1a, p = 0.065) and ADFI (Table 1) compared with the Con group from the 6th week on. At the 7th week, the body weight and ADG of pigs in the MMFP group were significantly higher than those in the Con group (Fig. 1, Table 1, p < 0.05). MMFP decreased diarrhea incidence compared with the Con group (Table 1). Two pigs in the Con group died from diarrhea, and one pig in the Ant group died from acute encephalitis. There was no death in the MMFP group. Thus, dietary supplementation with MMFP improved the growth performance and stress response caused by weaning effectively, and reduced mortality.
MMFP promotes the development of intestinal histology
Results from tissue slices of small intestine (Fig. 2a) showed that there were no obvious structural characters of Paneth cells or goblet cell in pigs from the Ant group and no obvious border of intestinal villi. The microvilli in pigs from the Con and MMFP groups were much more integrated than those from the Ant group. In the MMFP group, the mucosal thickness and villus height in small intestine were significantly higher than those in Con group. Villus height in MMFP group was significantly higher than that in the Ant group (Fig. 2d). However, the ratio of villus height to crypt depth in the Ant group was significantly lower than that in the Con and MMFP groups (Fig. 2e). Measurements of cecum and colon (Fig. 2f) showed that the thickness of the mucous layer in the Ant group was lower than that in the Con and MMFP groups. These results suggest that dietary supplementation with MMFP could promote the development and maturation of intestinal histology, while antibiotics might have negative effects.
Effects of MMFP on intestinal histology. On day 49, one pig from each pen was selected randomly and slaughtered. Photomicrographs of small intestine (a), photomicrographs of cecum (b), and photomicrographs of colon (c) were analyzed. Statistical analysis of intestinal histology (d–f). Data represent mean values ± SEM. Student’s t test was used to calculate p values. *p < 0.05; **p < 0.01; ***p < 0.001
MMFP improves antioxidant capacities in serum
The levels of T-AOC, SOD, GSH-Px, CAT, and MDA in serum were analyzed to evaluate the effects of MMFP and antibiotic on antioxidant capacities. The levels of T-AOC, SOD, GSH-Px, and CAT in the MMFP group were the highest among the three groups (Fig. 3a–d). The level of MDA in the Ant group was significantly higher than that in the Con and MMFP groups (Fig. 3e). These results indicate that dietary supplementation with MMFP could enhance the antioxidant capacities and reduce lipid peroxidation in serum.
Effects of MMFP on antioxidant capacities in serum and intestinal immune system. On day 49, one pig from each pen was selected randomly and slaughtered. Antioxidant capacities in serum (a–e) and the levels of inflammatory factors in mucosa from different segments of GI tract (f–i) were analyzed. Data represent mean values ± SEM. Student’s t test was used to calculate p values. *p < 0.05; **p < 0.01; ***p < 0.001
MMFP reduces inflammatory factors in intestinal mucosa
Levels of inflammatory factors in serum and the mucosa along the gastrointestinal tract were analyzed by ELISA to evaluate the effects of MMFP and antibiotic on immunologic homeostasis. Except for the cecum, MMFP reduced the expression levels of MCP-1, IL-1β, IL-6, and IL-10 in the whole of GI tract compared with the Con and Ant groups (Fig. 3f–i). Levels of inflammatory factors in serum were not significantly different between the Con, Ant, and MMFP groups (Fig. S1).
MMFP elevates organic acids in intestinal contents
Concentrations of organic acids in the intestinal contents were analyzed by HPLC to evaluate the effects of MMFP and antibiotics on microbial metabolism. The concentrations of lactate, acetate, propionate, butyrate, isobutyrate, and total SCFAs were significantly increased in all segments of the large intestine in MMFP group compared with the Con and Ant groups (Fig. 4, p < 0.001). These data indicate that dietary supplementation with MMFP enhanced microbial metabolic activities in the intestinal tract.
The concentrations of organic acids in the intestinal contents. On day 49, one pig from each pen was selected randomly and slaughtered. The concentrations of lactate (a), acetate (b), propionate (c), butyrate (d), isobutyrate (e), and total organic acids (f) in intestinal contents from different segments of GI tract were analyzed. Data represent mean values ± SEM. Student’s t test was used to calculate p values. ***p < 0.001
MMFP affects microbial community structure of intestinal contents
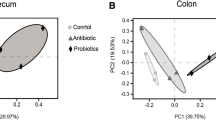
A total of 1,124,498 paired-end reads were obtained from 15 intestinal content samples. After jointing, filtering, and removing chimeras, a total of 909,431 effective sequences were reserved for later analysis (Table S2). Rarefaction curves and Shannon index curves showed that the sequencing depth was enough (Fig. S2a, b). Based on 97% sequence similarity, sequences were assigned to 692 unique OTUs. Phylum-level analysis of the gut microbiome revealed that all regions of the intestinal tract were dominated by Firmicutes (72.81–99.14%), followed by Bacteroidetes (0.45–13.97%). At the genus level, Clostridium sensu stricto 1, Peptoclostridium, Streptococcus, Turicibacter, Parabacteroides, and Intestinibacter were predominant in the intestinal contents (Fig. S2d, e). Principal component analysis (PCA) showed that the microbial compositions of the colonic and rectal contents were more similar to each other than to the cecal contents (Fig. S2c). Alpha diversity analysis (Table S3) revealed that microbial abundance (ACE and Chao1 index) and diversity (Simpson and Shannon index) in cecal contents were lower than those in the colonic and rectal contents.
The heatmap (Fig. 5) revealed that the composition of the microbial community in intestinal contents varied with treatment. In the MMFP group, the relative abundances of Eubacterium xylanophilum group, Clostridium sensu stricto 6, Streptococcus, Acetitomaculum, Roseburia, Coprococcus 1, and Lactobacillus were higher than those in the Con and Ant groups along the GI tract (Fig. 6a). The relative abundance of Ruminococcus was higher in the cecum and proximal colon of the MMFP group compared with the Con and Ant groups (Fig. 6a). The relative abundances of Marvinbryantia, Peptococcus, Turicibacter, and Blautia were increased in the colorectum of the Ant group compared with the Con and MMFP groups (Fig. 6b). However, Candidatus Saccharimonas, Mogibacterium, Solobacterium, Lachnoclostridium, Escherichia-Shigella, and Methanobacteriaceae were more abundant in the colorectum of the Con group than those in the Ant and MMFP groups (Fig. 6c).
Heatmap of microbial community along the gastrointestinal tract. On day 49, one pig from each pen was selected randomly and slaughtered. The microbial community of intestinal contents from different segments of GI tract was analyzed through high-throughput sequencing. Sample ID: Con_Ce, Ant_Ce, and MMFP_Ce: Cecal contents; Con_Co1, Ant_Co1, and MMFP_Co1: contents of proximal colon; Con_Co2, Ant_Con2, and MMFP_Co2: contents of medial colon; Con_Co3, Ant_Con3, and MMFP_Co3: contents of distal colon; Con_Re, Ant_Re, and MMFP_Re: rectal contents
Effects of MMFP on relative abundance of bacteria in different intestinal segments and feces. a Bacteria enriched in intestinal contents of MMFP group. b Bacteria enriched in intestinal contents of Ant group. c Bacteria enriched in intestinal contents of Con group. d Bacteria in fecal samples. Three fecal samples from each pen on day 0 and day 49 respectively were analyzed. On day 49, one pig from each pen was selected randomly and slaughtered. The microbial community of intestinal contents from different segments of GI tract was analyzed. Sample ID: Con_A, Ant_A, and MMFP_A: fecal samples from Con, Ant, and MMFP group respectively on day 0; Con_B, Ant_B, and MMFP_B: fecal samples from Con, Ant, and MMFP group respectively on day 49
Effects of MMFP on microbial community structure of feces
The microbial composition of feces on day 0 and day 49 was analyzed in this study. A total of 1,494,890 paired-end reads were obtained from 18 stool samples. After jointing, filtering, and removing chimeras, a total of 456,963 effective sequences were reserved for later analysis (Table S4). Rarefaction curves and Shannon index curves showed that the sequencing depth was enough (Fig. S3a, b). Based on 97% sequence similarity, sequences were assigned to 1195 unique OTUs. Phylum-level analysis of the feces microbiome revealed that Bacteroidetes (34.09–70.22%) and Firmicutes (27.80–61.52%) were the dominant phyla (Fig. S3d). Alpha-diversity analysis revealed that species richness and diversity increased over time. At the end of the experiment, the number of unique OTUs in the MMFP and Ant group was significantly higher than that in the Con group, and the richness and diversity index in the Ant and MMFP groups showed higher trends than the Con group (Table 2).
PCA analysis (Fig. S3c), histogram of species richness at the genus level (Fig. S3e), and the heatmap (Fig. S4) showed pronounced changes in microbial composition between the start (day 0) and end (day 49) of the experiment. Compared with the start, the relative abundance of Lactobacillus decreased significantly at the end of the experiment. However, Lactobacillus was much more abundant in the MMFP group compared with the Con and Ant groups on day 49 (Fig. 6d). In addition, at the end of the experiment, the relative abundance of Campylobacter in the Con group was significantly higher than that in the Ant and MMFP groups. These results indicate that MMFP could not only ameliorate the reduction of Lactobacillus caused by weaning but also inhibited the proliferation of pathogenic bacteria.
Correlation analysis between metabolic profiles and microorganisms
Correlation analysis (Fig. 7) showed significant correlations between piglet metabolic profile and the relative abundances of bacteria in GI tract. The relative abundances of Clostridium sensu stricto 6, Acetitomaculum, Ruminococcaceae UCG-013, Coprococcus 1, Peptoclostridium, Streptococcus, Roseburia, Eubacterium xylanophilum group, and Lactobacillus had positive correlations with the levels of T-AOC, GSH-Px, and CAT in serum and the concentrations of SCFAs in the intestinal contents, while these bacteria were negatively correlated with levels of MDA in serum and inflammatory factors in intestinal mucosa. In contrast, the relative abundances of Turicibacter, Blautia, Marvinbryantia, Peptococcus, Methanobacteriaceae, Family XIII, and Parabacteroides had negative correlations with the levels of T-AOC, GSH-Px, and CAT in serum and the concentrations of SCFAs in the intestinal contents, and they had positive correlations with the levels of MDA in serum and inflammatory factors in the intestinal mucosa. There was no obvious correlation between the growth performance of piglets and changes in the relative abundances of fecal microbiota (data not shown).
Discussion
Traditional MMF is spontaneously fermented by bacteria, mold, and yeast (Xu et al. 2013). However, the quality of traditional MMF is unstable, and pathogens have been detected in some MMFs (Liu et al. 2017). This restricts its clinical application. Guided by the Chinese Pharmacopeia, we improved the fermentation of MMF with probiotics, i.e., Bacillus sp., Lactobacillus sp., S. thermophilus, E. faecalis, Saccharomyces cerevisiae, Aspergillus oryzae, and Mucor racemosu. Bacterial strains used in MMFP fermentation are safer and more controllable compared with traditional MMF. Therefore, MMFP combines the advantages of Chinese herbal medicine, digestive enzymes, and probiotics. The current study confirmed that dietary supplementation with MMFP effectively alleviated weaning stress in piglets, reduced the levels of inflammatory factors in the intestinal mucosa, and elevated the antioxidant capacities in serum and the concentrations of organic acids in the intestinal contents.
During the weaning period, weaning stress can cause anorexia, growth retardation, severe enteric infection, or even death, which inflicts significant losses on the swine industry. Many kinds of feed additives, such as a live oral vaccine against ETEC F4, ZnO, essential oils, organic acids, prebiotics, and probiotics, have been used to alleviate weaning stress and pathogenic infections in piglets, as reviewed by Gresse et al. (2017). However, there was no systematic report about the beneficial effects of probiotics-fermented Chinese medicines on weaning piglets. Our study confirmed that MMFP treatment increased ADFI and ADG while reducing diarrhea incidence and mortality. On the one hand, MMF contains various digestive enzymes (Xu et al. 2013), and is usually used to promote food digestion and nutrient absorption. Herbal formulas containing MMF promote gastric emptying and intestinal transit (Xu et al. 1998). On the other hand, MMFP is fermented by probiotics. Dietary supplementation with probiotics, such as Bacillus strain and Lactobacillus rhamnosus GG, has reduced diarrhea and promoted growth in pigs (Loh et al. 2014; Mao et al. 2016). Taken together, the data suggest that the efficacy of MMFP is founded on the synergy between MMF and probiotics.
Weaning can induce acute and long-lasting damage to the intestinal physiology, including reduction of villus height and extension of crypt depth (Boudry et al. 2004; McCracken et al. 1999; Montagne et al. 2007). These dramatic alterations can impact the ability of digestion, absorption, and secretion and ultimately intestinal barrier function. Once disrupted, the increased permeability of the barrier will allow toxins, bacteria, or feed-associated antigens to cross the epithelium, which results in inflammation and diarrhea (Pie et al. 2004). The activation of the immune system will reduce the growth rate, feed intake, and lean tissue deposits (Williams et al. 1997). Clearly, to ameliorate weaning stress, it is critical to reduce the degeneration of the intestinal structure and activation of the immune system during the weaning period.
In the current research, villus height and mucosal thickness were increased in MMFP treatment, which were beneficial to intestinal function. The increased villus height raises the amount of contact and the number of active sites between digesta and epithelium, which benefits the absorption of nutrients. In addition, our results reveal that there were no goblet cells or Paneth cells in the intestine of the Ant group, and antibiotic treatment reduced the ratio of villus height to crypt depth and mucosal thickness of the cecum and colon. A previous study reported that dietary supplementation with antibiotics, such as bacitracin zinc, reduced the intestinal tract weight of broiler chicks (Henry et al. 1987). These results suggest that antibiotics may have adverse effects on the development of the GI tract. In contrast, MMFP promotes the development of the GI tract and improves intestinal barrier function.
The over expression of intestinal pro-inflammatory cytokines has adverse effects on the integrity of the intestinal mucosa and allow increased tissue penetration of antigens (Al-Sadi et al. 2013; Liu et al. 2008). The inhibition of cytokines may prevent the propagation of certain pathogens, which contributes to the stability of the intestinal micro-ecosystem (Walsh et al. 2013). In this study, dietary supplementation with MMFP reduced MCP-1, IL-1β, IL-6, and IL-10 in the intestinal mucosa, which might be one of the reasons why MMFP reduced diarrhea incidence and improved the growth rate. Antioxidant capacities in the serum reflect the host’s capacity to respond to endogenous oxidative damage. Improving the antioxidant capacities has positive effects on weaning stress (Fan et al. 2017; Wei et al. 2016). In this study, MMFP increased the levels of T-AOC, SOD, GSH-Px, and CAT and decreased the level of MDA in serum, which suggests that MMFP ameliorates oxidative stress during the weaning period in piglets.
Organic acids play important roles in regulating body’s growth and homeostasis. Acetate could inhibit pathogen proliferation and invasion of the intestinal epithelium (Asahara et al. 2009; Fukuda et al. 2011) and increase feed intake and obesity (Perry et al. 2016). From this, we infer that increased acetate in the MMFP group might have reduced the risks of pathogen proliferation and infection, and it also contributed to weight gain in weaning piglets. Butyrate is preferentially used as an energy source by gut epithelial cells. Propionate is mainly metabolized in the liver through gluconeogenesis. Butyrate and propionate inhibit the activity of histone deacetylases (HDACs) in colonocytes and immune cells, which leads to the down-regulation of pro-inflammatory cytokines in colonic macrophages (Louis et al. 2014). Dietary supplementation with sodium butyrate decreased the diarrhea incidence of weaned piglets and enhanced immune function (Fang et al. 2014). The present data demonstrate that MMFP increased the concentrations of butyrate and propionate in the intestinal contents, which indicated that MMFP may contribute to the intestinal immune system and improve energy acquisition from food. In the MMFP group, the concentration of lactate in the cecal contents was higher than that in the colonic and rectal contents, whereas acetate, propionate, and butyrate were lower in the cecal contents. This might confirm that lactate is used as a metabolic substrate for SCFA biosynthesis. The lactate-related trophic chain is considered one of the principal metabolic pathways in mammalian gut ecosystems (Valeriano et al. 2017). Lactate could be utilized to form propionate and pyruvate, and then pyruvate can be used for acetate and butyrate biosynthesis (Flint et al. 2015; Louis et al. 2014). Consequently, in the MMFP group, the increased lactate might have facilitated SCFA biosynthesis. In summary, increased concentrations of organic acids in the MMFP group might have contributed to intestinal homeostasis.
Dietary supplementation with MMFP altered the microbial composition of the intestinal contents, as shown in Fig. 5 and Fig. S2c. The relative abundances of Lactobacillus and Streptococcus were increased in the MMFP group compared with the Con and Ant groups. This had beneficial effects on the health of the host. Lactobacillus is one of “core microbiota” in the GI tract of mammals (Katouli and Wallgren 2005), which can prevent the diarrhea and intestinal infections caused by bacteria and viruses (Konstantinov et al. 2006; Li et al. 2012; Mao et al. 2016). Bacteria of the Lactobacillus group are often decreased during the weaning transition (Gresse et al. 2017), so supplementation of Lactobacillus spp. may decrease the risks of disease. Streptococcus thermophilus has been widely used as probiotic to enhance the immune system and inhibit the proliferation of pathogens (Feng et al. 2017). Acetitomaculum is an acetate-producing bacterium (Greening and Leedle 1989). Roseburia (Duncan et al. 2002; Louis and Flint 2009), Coprococcus (Van den Abbeele et al. 2013), Eubacterium xylanophilum group (Van Gylswyck and van der Toorn 1985), Clostridium sensu stricto (Varel and Pond 1992), and Ruminococcus (Claesson et al. 2012) can produce butyrate. The increased relative abundances of above bacteria in the MMFP group explained the increase of organic acids in the intestinal contents. Correlation analysis showed that the relative abundances of Clostridium sensu stricto 6, Acetitomaculum, Coprococcus 1, Streptococcus, Roseburia, Eubacterium xylanophilum group, and Lactobacillus were positively correlated with the antioxidant activities in serum and negatively correlated with the levels of MDA and inflammatory cytokines, which meant these bacteria have positive effects on host health. Dietary supplementation of MMFP increased the relative abundance of cellulolytic and SCFA-producing bacteria, indicating a shift toward a more adult pig-like intestinal environment associated with increased functional ability for carbohydrate degradation.
The long-term use of subtherapeutic doses of antibiotics, or taking antibiotics for bacterial infections will increase the risks of pathogen proliferation and infection (Fouhse et al. 2016; Mu et al. 2017). A similar pattern appeared in the Ant group in this study. Peptococcus has been significantly enriched in oral squamous cell carcinoma samples (Zhao et al. 2017). Turicibacter has been isolated from a blood culture of a febrile patient with acute appendicitis (Bosshard 2002). Blautia was notably increased in the fecal samples of hyperlipidemic patients compared with the samples from normal volunteers (Wang et al. 2017). The relative abundances of Turicibacter, Blautia, Marvinbryantia, and Peptococcus were increased in the colonic and rectal contents of Ant group (Fig. 6b). Furthermore, the abundance of these bacteria was positively correlated with the levels of MDA in serum and inflammatory factors in intestinal mucosa, and negatively correlated with the antioxidant capacities in serum and the concentrations of organic acids in intestinal contents (Fig. 7). These results suggest that antibiotics may increase the risks of infections and oxidative damage.
The pro-inflammatory bacterium Mogibacterium is more abundant in feces of patients with adenomatous polyps (Hale et al. 2017) and intestinal mucosa of colorectal cancer patients (Candela et al. 2014). Solobacterium moorei can cause bacteremia (Lau et al. 2006), and pathogenic Escherichia-Shigella can cause piglet diarrhea (Gresse et al. 2017). The decreased relative abundances of Mogibacterium, Solobacterium, and Escherichia-Shigella in the intestinal contents of the Ant and MMFP groups reflected that a reduced diarrhea incidence in Ant and MMFP group might occur partially through the inhibition of certain pathogenic bacteria. Methane is a major greenhouse gas. Some evidences show that methanogenesis is associated with slower intestinal and colonic transit (Pimentel et al. 2006; Soares et al. 2005), which may result in constipation (Chatterjee et al. 2007) and dyspepsia. A previous study showed that acidification of the gut lumen caused by high levels of SCFAs inhibited methanogenic activity (Ong et al. 2010). In this study, high levels of SCFAs in the intestinal contents of MMFP group might have inhibited methanogenesis, which had positive effects on food digestion and environment.
It is worth noting that the microbial composition of feces was barely affected by Ant or MMFP treatment at the end of the experiment. The most remarkable changes were that MMFP diminished the reduction of Lactobacillus caused by weaning and reduced the relative abundance of Campylobacter. Campylobacter is a typical enteric pathogen. Campylobacter is abundant in the gut of pigs with dysentery (Burrough et al. 2017), and humans may be infected with Campylobacter through various animal sources (Rosner et al. 2017). The reduction of Campylobacter in the feces of the Ant and MMFP groups suggested that antibiotics and MMFP may decrease the risks of dysentery and diffusion of pathogens. The microbial composition of feces had no correlation with the growth performance or metabolic profiles in our study. These results reveal that the microbiota in the GI tract, instead of in the feces, was closely related to the homeostasis of host, which is in accordance with a previous study (Zhao et al. 2015).
In conclusion, dietary supplementation with MMFP improved the growth rate and average daily feed intake and reduced the diarrhea incidence and mortality of piglets. These results confirmed that MMFP can effectively alleviate weaning stress in piglets. Analysis of morphology and ELISA showed that MMFP promoted the development of intestinal histology and decreased the levels of inflammatory factors in the intestinal mucosa. In addition, MMFP elevated the antioxidant capacities of serum and the concentrations of organic acids in the intestinal contents. Correspondingly, the relative abundances of lactic acid and SCFA-producing bacteria were dramatically increased in the intestinal contents of the MMFP group. Dietary supplementation with MMFP reduced the relative abundances of pathogens in the intestinal contents and feces. These results demonstrate that MMFP enhanced host intestinal homeostasis by modulating the composition of the gut microbiota, thereby ameliorating weaning stress in piglets (Fig. 8). Obviously, the growth-promoting mechanism of MMFP is different from that of antibiotics. Furthermore, correlation analysis between the relative abundances of microbiota and host metabolic profile showed that the microbiota in the GI tract, not feces, modulates host metabolism. More investigations are still needed to explore the effects and mechanisms of probiotics-fermented Chinese medicine. We hope that this report could contribute to the development of new and efficient feed additives, and serve as a reference and a case for discussion.
References
Al-Sadi R, Boivin M, Ma T (2013) Mechanism of cytokine modulation of epithelial tight junction barrier. Front Biosci (Landmark Ed) 14:2765–2778
Asahara T, Shimizu K, Nomoto K, Watanuki M, Tanaka R (2009) Antibacterial effect of fermented milk containing Bifidobacterium breve, Bifidobacterium bifidum and Lactobacillus acidophilus against indigenous Escherichia coli infection in mice. Microb Ecol Health Dis 13(1):16–24. https://doi.org/10.1080/089106001750071663
Bose S, Han KW, Lee MJ, Kim H (2013) Intestinal protective effects of herbal-based formulations in rats against neomycin insult. Evid Based Complement Alternat Med 2013:161278–161213. https://doi.org/10.1155/2013/161278
Bosshard PP (2002) Turicibacter sanguinis gen. Nov., sp. nov., a novel anaerobic, Gram-positive bacterium. Int J Syst Evol Microbiol 52(4):1263–1266. https://doi.org/10.1099/ijs.0.02056-0
Boudry G, Peron V, Le Huerou-Luron I, Lalles JP, Seve B (2004) Weaning induces both transient and long-lasting modifications of absorptive, secretory, and barrier properties of piglet intestine. J Nutr 134(9):2256–2262. https://doi.org/10.1093/jn/134.9.2256
Buffie CG, Pamer EG (2013) Microbiota-mediated colonization resistance against intestinal pathogens. Nat Rev Immunol 13(11):790–801. https://doi.org/10.1038/nri3535
Burrough ER, Arruda BL, Plummer PJ (2017) Comparison of the luminal and mucosa-associated microbiota in the colon of pigs with and without swine dysentery. Front Vet Sci 4:139. https://doi.org/10.3389/fvets.2017.00139
Candela M, Turroni S, Biagi E, Carbonero F, Rampelli S, Fiorentini C, Brigidi P (2014) Inflammation and colorectal cancer, when microbiota-host mutualism breaks. World J Gastroenterol 20(4):908–922. https://doi.org/10.3748/wjg.v20.i4.908
Casey PG, Gardiner GE, Casey G, Bradshaw B, Lawlor PG, Lynch PB, Leonard FC, Stanton C, Ross RP, Fitzgerald GF, Hill C (2007) A five-strain probiotic combination reduces pathogen shedding and alleviates disease signs in pigs challenged with Salmonella enterica Serovar typhimurium. Appl Environ Microbiol 73(6):1858–1863. https://doi.org/10.1128/AEM.01840-06
Chatterjee S, Park S, Low K, Kong Y, Pimentel M (2007) The degree of breath methane production in IBS correlates with the severity of constipation. Am J Gastroenterol 102(4):837–841. https://doi.org/10.1111/j.1572-0241.2007.01072.x
Claesson MJ, Jeffery IB, Conde S, Power SE, O'Connor EM, Cusack S, Harris HMB, Coakley M, Lakshminarayanan B, O'Sullivan O, Fitzgerald GF, Deane J, O'Connor M, Harnedy N, O'Connor K, O'Mahony D, van Sinderen D, Wallace M, Brennan L, Stanton C, Marchesi JR, Fitzgerald AP, Shanahan F, Hill C, Ross RP, O'Toole PW (2012) Gut microbiota composition correlates with diet and health in the elderly. Nature 488(7410):178–184. https://doi.org/10.1038/nature11319
de Almada CN, Nunes de Almada C, Martinez RC, Sant'Ana Ade S (2015) Characterization of the intestinal microbiota and its interaction with probiotics and health impacts. Appl Microbiol Biotechnol 99(10):4175–4199. https://doi.org/10.1007/s00253-015-6582-5
Duncan SH, Hold GL, Barcenilla A, Stewart CS, Flint HJ (2002) Roseburia intestinalis sp nov., a novel saccharolytic, butyrate-producing bacterium from human faeces. Int J Syst Evol Microbiol 52:1615–1620. https://doi.org/10.1099/ijs.0.02143-0
Fan P, Tan Y, Jin K, Lin C, Xia S, Han B, Zhang F, Wu L, Ma X (2017) Supplemental lipoic acid relieves post-weaning diarrhoea by decreasing intestinal permeability in rats. J Anim Physiol Anim Nutr (Berl) 101(1):136–146. https://doi.org/10.1111/jpn.12427
Fang CL, Sun H, Wu J, Niu HH, Feng J (2014) Effects of sodium butyrate on growth performance, haematological and immunological characteristics of weanling piglets. J Anim Physiol Anim Nutr (Berl) 98(4):680–685. https://doi.org/10.1111/jpn.12122
Feng JR, Wang F, Qiu X, McFarland LV, Chen PF, Zhou R, Liu J, Zhao Q, Li J (2017) Efficacy and safety of probiotic-supplemented triple therapy for eradication of Helicobacter pylori in children: a systematic review and network meta-analysis. Eur J Clin Pharmacol 73(10):1199–1208. https://doi.org/10.1007/s00228-017-2291-6
Flint HJ, Duncan SH, Scott KP, Louis P (2015) Links between diet, gut microbiota composition and gut metabolism. Proc Nutr Soc 74(1):13–22. https://doi.org/10.1017/S0029665114001463
Fouhse JM, Zijlstra RT, Willing BP (2016) The role of gut microbiota in the health and disease of pigs. Animal Frontiers 6(3):30–36. https://doi.org/10.2527/af.2016-0031
Fukuda S, Toh H, Hase K, Oshima K, Nakanishi Y, Yoshimura K, Tobe T, Clarke JM, Topping DL, Suzuki T, Taylor TD, Itoh K, Kikuchi J, Morita H, Hattori M, Ohno H (2011) Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469(7331):543–549. https://doi.org/10.1038/nature09646
Gao X, Xie Q, Liu L, Kong P, Sheng J, Xiang H (2017) Metabolic adaptation to the aqueous leaf extract of Moringa oleifera lam.-supplemented diet is related to the modulation of gut microbiota in mice. Appl Microbiol Biotechnol 101(12):5115–5130. https://doi.org/10.1007/s00253-017-8233-5
Greening RC, Leedle JA (1989) Enrichment and isolation of Acetitomaculum ruminis, gen. nov., sp. nov.: acetogenic bacteria from the bovine rumen. Arch Microbiol 151(5):399–406
Gresse R, Chaucheyras-Durand F, Fleury MA, Van de Wiele T, Forano E, Blanquet-Diot S (2017) Gut microbiota dysbiosis in postweaning piglets: understanding the keys to health. Trends Microbiol 25(10):851–873. https://doi.org/10.1016/j.tim.2017.05.004
Gross M (2013) Antibiotics in crisis. Curr Biol 23(24):R1063–R1065. https://doi.org/10.1016/j.cub.2013.11.057
Hale VL, Chen J, Johnson S, Harrington SC, Yab TC, Smyrk TC, Nelson H, Boardman LA, Druliner BR, Levin TR, Rex DK, Ahnen DJ, Lance P, Ahlquist DA, Chia N (2017) Shifts in the fecal microbiota associated with adenomatous polyps. Cancer Epidemiol Biomark Prev 26(1):85–94. https://doi.org/10.1158/1055-9965.EPI-16-0337
Heinritz SN, Mosenthin R, Weiss E (2013) Use of pigs as a potential model for research into dietary modulation of the human gut microbiota. Nutr Res Rev 26(2):191–209. https://doi.org/10.1017/S0954422413000152
Henry PR, Ammerman CB, Campbell DR, Miles RD (1987) Effect of antibiotics on tissue trace mineral concentration and intestinal tract weight of broiler chicks. Poult Sci 66(6):1014–1018. https://doi.org/10.3382/ps.0661014
Kamada N, Seo SU, Chen GY, Nunez G (2013) Role of the gut microbiota in immunity and inflammatory disease. Nat Rev Immunol 13(5):321–335. https://doi.org/10.1038/nri3430
Katouli M, Wallgren P (2005) Metabolism and population dynamics of the intestinal microflora in the growing pig. In: Holzapfel WH, Naughton PJ, Pierzynowski SG, Zabielski R, Salek E (eds) Biology of growing animals. vol 2. Elsevier, Amsterdam, pp 21–53
Konstantinov SR, Awati AA, Williams BA, Miller BG, Jones P, Stokes CR, Akkermans AD, Smidt H, de Vos WM (2006) Post-natal development of the porcine microbiota composition and activities. Environ Microbiol 8(7):1191–1199. https://doi.org/10.1111/j.1462-2920.2006.01009.x
Lalles JP, Bosi P, Smidt H, Stokes CR (2007) Nutritional management of gut health in pigs around weaning. Proc Nutr Soc 66(2):260–268. https://doi.org/10.1017/S0029665107005484
Lau SK, Teng JL, Leung KW, Li NK, Ng KH, Chau KY, Que TL, Woo PC, Yuen KY (2006) Bacteremia caused by Solobacterium moorei in a patient with acute proctitis and carcinoma of the cervix. J Clin Microbiol 44(8):3031–3034. https://doi.org/10.1128/JCM.00555-06
Li XQ, Zhu YH, Zhang HF, Yue Y, Cai ZX, Lu QP, Zhang L, Weng XG, Zhang FJ, Zhou D, Yang JC, Wang JF (2012) Risks associated with high-dose Lactobacillus rhamnosus in an Escherichia coli model of piglet diarrhoea: intestinal microbiota and immune imbalances. PLoS One 7(7):e40666. https://doi.org/10.1371/journal.pone.0040666
Liu Y, Huang J, Hou Y, Zhu H, Zhao S, Ding B, Yin Y, Yi G, Shi J, Fan W (2008) Dietary arginine supplementation alleviates intestinal mucosal disruption induced by Escherichia coli lipopolysaccharide in weaned pigs. Br J Nutr 100(3):552–560. https://doi.org/10.1017/S0007114508911612
Liu T, Jia T, Chen J, Liu X, Zhao M, Liu P (2017) Analysis of microbial diversity in Shenqu with different fermentation times by PCR-DGGE. Braz J Microbiol 48(2):246–250. https://doi.org/10.1016/j.bjm.2017.01.002
Loh G, Prieto ML, O'Sullivan L, Tan SP, McLoughlin P, Hughes H, O'Donovan O, Rea MC, Kent RM, Cassidy JP, Gardiner GE, Lawlor PG (2014) Evaluation of the efficacy and safety of a marine-derived Bacillus strain for use as an in-feed probiotic for newly weaned pigs. PLoS One 9(2):e88599. https://doi.org/10.1371/journal.pone.0088599
Louis P, Flint HJ (2009) Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol Lett 294(1):1–8. https://doi.org/10.1111/j.1574-6968.2009.01514.x
Louis P, Hold GL, Flint HJ (2014) The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol 12(10):661–672. https://doi.org/10.1038/nrmicro3344
Mao X, Gu C, Hu H, Tang J, Chen D, Yu B, He J, Yu J, Luo J, Tian G (2016) Dietary Lactobacillus rhamnosus GG supplementation improves the mucosal barrier function in the intestine of weaned piglets challenged by porcine rotavirus. PLoS One 11(1):e0146312. https://doi.org/10.1371/journal.pone.0146312
McCracken BA, Spurlock ME, Roos MA, Zuckermann FA, Gaskins HR (1999) Weaning anorexia may contribute to local inflammation in the piglet small intestine. J Nutr 129(3):613–619. https://doi.org/10.1093/jn/129.3.613
Montagne L, Boudry G, Favier C, Le Huerou-Luron I, Lalles JP, Seve B (2007) Main intestinal markers associated with the changes in gut architecture and function in piglets after weaning. Br J Nutr 97(1):45–57. https://doi.org/10.1017/S000711450720580X
Mu C, Yang Y, Su Y, Zoetendal EG, Zhu W (2017) Differences in microbiota membership along the gastrointestinal tract of piglets and their differential alterations following an early-life antibiotic intervention. Front Microbiol 8:797. https://doi.org/10.3389/fmicb.2017.00797
National Drug Standards (1998) Chinese medicine prescription. China, pp Z19-36. WS3-B-3555-98
National Research Council (2012) Nutrient requirements of swine: eleventh revised edition. The National Academies Press, Washington, DC. https://doi.org/10.17226/13298
Ong DK, Mitchell SB, Barrett JS, Shepherd SJ, Irving PM, Biesiekierski JR, Smith S, Gibson PR, Muir JG (2010) Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J Gastroenterol Hepatol 25(8):1366–1373. https://doi.org/10.1111/j.1440-1746.2010.06370.x
Perry RJ, Peng L, Barry NA, Cline GW, Zhang D, Cardone RL, Petersen KF, Kibbey RG, Goodman AL, Shulman GI (2016) Acetate mediates a microbiome-brain-beta-cell axis to promote metabolic syndrome. Nature 534(7606):213–217. https://doi.org/10.1038/nature18309
Pie S, Lalles JP, Blazy F, Laffitte J, Seve B, Oswald IP (2004) Weaning is associated with an upregulation of expression of inflammatory cytokines in the intestine of piglets. J Nutr 134(3):641–647
Pimentel M, Lin HC, Enayati P, van den Burg B, Lee HR, Chen JH, Park S, Kong Y, Conklin J (2006) Methane, a gas produced by enteric bacteria, slows intestinal transit and augments small intestinal contractile activity. Am J Physiol Gastrointest Liver Physiol 290(6):G1089–G1095. https://doi.org/10.1152/ajpgi.00574.2004
Rosner BM, Schielke A, Didelot X, Kops F, Breidenbach J, Willrich N, Golz G, Alter T, Stingl K, Josenhans C, Suerbaum S, Stark K (2017) A combined case-control and molecular source attribution study of human Campylobacter infections in Germany, 2011-2014. Sci Rep 7(1):5139. https://doi.org/10.1038/S41598-017-05227-X
Shin IS, Jeon WY, Shin HK, Cha SW, Lee MY (2013) Banhabaekchulchunma-tang, a traditional herbal formula attenuates absolute ethanol-induced gastric injury by enhancing the antioxidant status. BMC Complement Altern Med 13:170. https://doi.org/10.1186/1472-6882-13-170
Soares AC, Lederman HM, Fagundes-Neto U, de Morais MB (2005) Breath methane associated with slow colonic transit time in children with chronic constipation. J Clin Gastroenterol 39(6):512–515. https://doi.org/10.1097/01.mcg.0000165665.94777.bd
Sun P, Li D, Li Z, Dong B, Wang F (2008) Effects of glycinin on IgE-mediated increase of mast cell numbers and histamine release in the small intestine. J Nutr Biochem 19(9):627–633. https://doi.org/10.1016/j.jnutbio.2007.08.007
Tanne JH (2012) Resistance of enterobacteria to carbapenem antibiotics is a global crisis. BMJ 344:e1646. https://doi.org/10.1136/bmj.e1646
Valeriano VD, Balolong MP, Kang DK (2017) Probiotic roles of Lactobacillus sp. in swine: insights from gut microbiota. J Appl Microbiol 122(3):554–567. https://doi.org/10.1111/jam.13364
Van den Abbeele P, Belzer C, Goossens M, Kleerebezem M, De Vos WM, Thas O, De Weirdt R, Kerckhof FM, Van de Wiele T (2013) Butyrate-producing Clostridium cluster XIVa species specifically colonize mucins in an in vitro gut model. ISME J 7(5):949–961. https://doi.org/10.1038/ismej.2012.158
Van Gylswyck NO, van der Toorn JJTK (1985) Eubacterium uniforme sp. nov. and Eubacterium xylanophilum sp. nov., fiber-digesting bacteria from the rumina of sheep fed corn stover. Int J Syst Bacteriol 35(3):323–326. https://doi.org/10.1099/00207713-35-3-323
Varel VH, Pond WG (1992) Characteristics of a new cellulolytic Clostridium sp. isolated from pig intestinal tract. Appl Environ Microbiol 58(5):1645–1649
Walsh AM, Sweeney T, O'Shea CJ, Doyle DN, O'Doherty JV (2013) Effect of dietary laminarin and fucoidan on selected microbiota, intestinal morphology and immune status of the newly weaned pig. Br J Nutr 110(9):1630–1638. https://doi.org/10.1017/S0007114513000834
Wang Y, Tong Q, Shou JW, Zhao ZX, Li XY, Zhang XF, Ma SR, He CY, Lin Y, Wen BY, Guo F, Fu J, Jiang JD (2017) Gut microbiota-mediated personalized treatment of hyperlipidemia using berberine. Theranostics 7(9):2443–2451. https://doi.org/10.7150/thno.18290
Wei B, Nie S, Meng Q, Qu Z, Shan A, Chen Z (2016) Effects of l-carnitine and/or maize distillers dried grains with solubles in diets of gestating and lactating sows on the intestinal barrier functions of their offspring. Br J Nutr 116(3):459–469. https://doi.org/10.1017/S0007114516001951
Williams NH, Stahly TS, Zimmerman DR (1997) Effect of level of chronic immune system activation on the growth and dietary lysine needs of pigs fed from 6 to 112 kg. J Anim Sci 75:2481–2496
Xu C-T, Ma J-Y, Pan B-R, Ma L-S (1998) Strengthen international academic cooperation and exchanges: prospects in the 21st century: summary of the first world Chinese congress of digestion. World J Gastroenterol 4(6):475–482. https://doi.org/10.3748/wjg.v4.i6.475
Xu Y, Xie YB, Zhang XR, Chen C, Xiang H, Xie Q (2013) Monitoring of the bacterial and fungal biodiversity and dynamics during Massa Medicata Fermentata fermentation. Appl Microbiol Biotechnol 97(22):9647–9655. https://doi.org/10.1007/s00253-013-5187-0
Zhang LX, Gao WY, Wang HY (2012) Review of traditional Chinese medicine processed by fermentation. Zhongguo Zhong Yao Za Zhi 37(24):3695–3700
Zhao W, Wang Y, Liu S, Huang J, Zhai Z, He C, Ding J, Wang J, Wang H, Fan W, Zhao J, Meng H (2015) The dynamic distribution of porcine microbiota across different ages and gastrointestinal tract segments. PLoS One 10(2):e0117441. https://doi.org/10.1371/journal.pone.0117441
Zhao H, Chu M, Huang Z, Yang X, Ran S, Hu B, Zhang C, Liang J (2017) Variations in oral microbiota associated with oral cancer. Sci Rep 7(1):11773. https://doi.org/10.1038/s41598-017-11779-9
Funding
This study was funded by Jilin Province Science and Technology Institute of China (No. 20180201078YY) and Jilin Province Development and Reform Commission of China (No. 2015Y051).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The animal experiment was approved by the Institutional Animal Care and Use Committee of Jilin University (IACUC). All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 1074 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Xie, Q., Sun, S. et al. Probiotics-fermented Massa Medicata Fermentata ameliorates weaning stress in piglets related to improving intestinal homeostasis. Appl Microbiol Biotechnol 102, 10713–10727 (2018). https://doi.org/10.1007/s00253-018-9438-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9438-y