Abstract
The effects of using plant ingredients in Senegalese sole (Solea senegalensis) diet on immune competence and intestine morphology and microbial ecology are still controversial. Probiotics or immunostimulants can potentially alter the intestinal microbiota in a way that protects fish against pathogens. The current study aimed to examine the intestine histology and microbiota and humoral innate immune response in juvenile sole fed diets with low (35 %) or high (72 %) content of plant protein (PP) ingredients supplemented with a multispecies probiotic bacteria or autolysed yeast. Fish fed the probiotic diet had lower growth performance. Lysozyme and complement activities were significantly higher in fish fed PP72 diets than in their counterparts fed PP35 diets after 17 and 38 days of feeding. At 2 days of feeding, fish fed unsupplemented PP72 showed larger intestine section area and longer villus than fish fed unsupplemented PP35. At 17 days of feeding, fish fed unsupplemented PP72 showed more goblet cells than the other dietary groups, except the group fed yeast supplemented PP35 diet. High dietary PP level, acutely stimulate fish innate immune defence of the fish after 2 and 17 days of feeding. However, this effect does not occur after 73 days of feeding, suggesting a habituation to dietary treatments and/or immunosuppression, with a reduction in the number of the goblet cells. Fish fed for 38 days with diets supplemented with autolysed yeast showed longer intestinal villus. The predominant bacteria found in sole intestine were Vibrio sp. and dietary probiotic supplementation caused a reduction in Vibrio content, regardless of the PP level.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aquaculture is one of the fastest growing food-producing sectorsin the world (FAO), and during the last years, much effort have been done to improve fish growth by manipulation of dietary formulations. Senegalese sole (Solea senegalensis) is a promising flatfish species in the European aquaculture, due to its high commercial value and nutritional properties. The ability of Senegalese sole to efficiently use novel plant-based diets (Silva et al. 2009) is an important asset towards the intensification and commercialization of this species. Currently, diseases outbreaks remain a primary constraint to aquaculture expansion. One of the newest scientific areas of high interest to improve fish growth efficiency and to prevent and/or control fish diseases is the application of probiotics and microbial feed additives as alternatives to antibiotics (Dimitroglou et al. 2011; Magnadottir 2010). These feed additives or dietary raw materials favourably affect animal performance and welfare, particularly through the modulation of the intestinal microbiota which plays a critical role in maintaining host health (Tuohy et al. 2005) and through the modulation of the immune system, influencing various humoral and cellular defences (Nayak 2010).
Among promising probiotic candidates, the Bacilli class has been widely assayed in fish and numerous studies have demonstrated that its administration enhances immune responses and disease resistance (Aly et al. 2008; Kumar et al. 2008; Newaj-Fyzul et al. 2007; Salinas et al. 2005). On the other hand, the use of some selectively fermented ingredients like yeasts allows specific changes in the composition and/or activity of the gastrointestinal microflora that confers benefits upon the host’s wellbeing and health (Marcel 2008). Natural immunostimulants are valuable for activating the fish immune system and protecting fish against adverse conditions (Anderson 1992; Sakai 1999). Among them, the yeast Saccharomyces cerevisiae has been found to be a good enhancer of the trout immune system (Siwicki et al. 1994) and it can also add nutritional value in fish diets as a possible alternative to commonly used fish meal protein (Rumsey et al. 1992).
The microbial flora present in fish intestine plays an important role on breaking down ingested food or inhibiting the colonization of the intestine by pathogens (Manzano et al. 2012). The composition of bacterial communities in fish can be affected by several factors, including the environment and their interaction with diet. A balanced intestinal microbiota constitutes as an efficient barrier against pathogen colonization, producing metabolic substrates (e.g. vitamins and short-chain fatty acids) and stimulating the immune system (Gaggìa et al. 2010). In this context, probiotics and some immunostimulants may play a significant role to improve the resistance to pathogenic bacteria colonization and enhancement of host mucosa immunity, thus reducing pathogen load and an enhancing health status of the animals (Choct 2009). Bacterial diversity has an important role in the function of ecosystems (Bell et al. 2005). Biodiversity protects ecosystems against a decline in their functionality and allows for adaptation to changing conditions, as the coexistence of many bacterial species provides effective fail-safe measures to help keep the pathogen bacteria from spreading (Yachi and Loreau 1999). The complex microbial ecology of the intestinal tract (GI) provides nutritional benefit and protection against pathogens. Moreover, microbial ecology may play an important role in modulating interactions between environment and the immune system (Balcázar et al. 2007). The potential pathogens present within the GI tract should be reduced in order to avoid mucosal damage and improve absorptive surface area (Merrifield et al. 2010). Rawls et al. (2004) observed that microbiota stimulated intestinal epithelial proliferation and influenced enterocyte morphology.
The aim of the current study was to evaluate the effects of dietary plant protein levels intercropped with a multispecies probiotic (from the Bacilli class) or an autolysed yeast supplementation on growth performance, innate immune response, intestine morphology and microbiota diversity in Senegalese sole (S. senegalensis).
Materials and methods
The feeding experiment took place at the CIIMAR facilities (University of Porto, Portugal). The current study was conducted under the supervision of an accredited expert in laboratory animal science by the Portuguese Veterinary Authority (1005/92, DGV-Portugal, following FELASA category C recommendations) and according to the guidelines on the protection of animals used for scientific purposes from the European directive 2010/63/UE. The current study is an extension of a submitted work by Batista et al. (2016).
Feed and fish
Nowadays, marine fish diets are composed of at least 30 % plant ingredients. Six isoproteic (55 %) and isolipidic (8 %) diets with similar amino acid composition were formulated to contain 35 % (PP35) or 72 % (PP72) of plant ingredients (Table 1) each supplemented with PROB and YEAST (commercial products) or unsupplemented (UN) in a 2 × 3 factorial design. PROB (AquaStar® Growout - Biomin® Holding GmbH, Herzogenburg, Austria) was a multispecies probiotic (Bacillus sp., Pediococcus sp., Enterococcus sp., Lactobacillus sp.) incorporated at 1.34 × 1010 CFU kg−1 diet (CFU—colony-forming unit) in the extruded pellets by means of vacuum coating (Dinnisen Pegasus vacuum mixer, PG-10VCLAB) using fish oil as a carrier. YEAST (Levabon® Aquagrow - Biomin®) was autolysed yeast (S. cerevisiae) supplemented in the mixture at 4 g kg−1 diet. Diets were prepared according to the normatives ISO (6887-1: 1999, 7218: 2007, 6498: 2012) for the microbiological examinations. Thereafter, the isolation and enumeration of bacteria in the diets followed the European standards for Enterococcus spp. (EN 15788: 2009), Lactobacillus spp. (EN 15787: 2009), Pediococcus spp. (EN 15786: 2009) and Bacillus spp. (EN 15784: 2009).
Senegalese sole were obtained from a commercial fish farm (Aquacria S.A., Portugal) and transported to the rearing facilities of CIIMAR. After a 5-week acclimation period, fish were distributed into 18 fibreglass tanks of 50 L each, at a density of 5.8 kg m−2 (31 fish per tank, 33.1 ± 0.2 g each) and fed the experimental diets in triplicate. The tanks were supplied with seawater (32.0 ± 0.1 ppm NaCl, 19.7 ± 0.5 °C, 0.33 ± 0.27 mg L−1 NH3, 0.14 ± 0.07 mg L−1 NO2) at a flow rate of 1.5 L min−1, in a controlled semi-closed recirculation system under a constant photoperiod (12 h light:12 h darkness). Rearing conditions were monitored during the entire trial. The recirculation system was equipped with UV lights and ozone generator to hamper bacteria growth in the rearing water. The trial lasted 73 days and fish were fed to apparent satiety based on visual observation of acceptance and refusal of feed using temporized automatic feeders.
Sampling procedures
Prior to sampling, fish were fasted for 24 h and then sacrificed with an overdose of ethyl 3-aminobenzoate methanesulfonate (MS-222, 200 mg L−1). All fish were individually weighed and measured at the beginning and at the end of the experiment in order to determine growth performance. Ten fish from the initial stock and two fish from each tank at the end of the trial (6 fish per treatment) were sampled and stored at −20 °C for body composition analyses. Total feed consumption was registered during the entire feeding trial. The innate immune response and intestine histology were analysed at days 3, 17, 38 and 73 of the feeding trial.
For the evaluation of humoral innate immune parameters (lysozyme, peroxidase and alternative complement pathway—ACH50), blood was withdrawn from the caudal vein of nine fish per treatment using heparinized syringes and centrifuged at 5000×g for 10 min at 4 °C. Liver and intestine of each fish was sampled, cut and fixed in buffered formalin to histology analysis. To examine intestinal microbiota, the whole intestine of other nine fish per treatment was aseptically removed at 17, 38 and 73 days of feeding the experimental diets.
For microbial ecology determination, the intestinal contents of three non-fasted fish per tank were sampled by stripping and pooled prior homogenized in 1 mL sterile phosphate-buffered saline (PBS, pH 7.2).
Chemical analyses of diets and body composition
All chemical analyses were carried in duplicate according to AOAC (2006). Fish were minced without thawing using a meat mincer, pooled and used to determine their dry matter content (105 °C for 24 h). Fish were then freeze-dried and analysed for dry matter, ash (Nabertherm L9/11/B170; Germany; 550 °C for 6 h), crude protein (N × 6.25, Leco N analyser, Model FP-528, Leco Corporation, St. Joseph, USA), crude lipid (petroleum ether extraction, 40–60 °C, Soxtherm, Gerhardt, Germany) and gross energy (adiabatic bomb calorimeter, Werke C2000, IKA, Germany).
Humoral innate immune parameters
All measurements were done in triplicate on a Power-Wave™ microplate spectrophotometer (BioTek Synergy HT, USA). Plasma lysozyme activity (EU mL−1) was determined using a turbidimetric assay adapted to microtitre, as described by Hutchinson and Manning (1996). One lysozyme enzyme unit (EU) was defined as the amount of lysozyme that caused a decrease in absorbance per minute. Plasma peroxidase activity (EU mL−1) was measured following the procedure adapted to S. senegalensis by Costas et al. (2011), defining that 1 unit of peroxidase produces an absorbance change of 1 OD. Alternative complement pathway (ACH50) activity was based on the lysis of rabbit red blood cells (2.8 × 108 cells mL−1), as reported by Sunyer and Tort (1995). ACH50 units were defined as the concentration of plasma giving 50 % lysis of rabbit red blood cells.
Histological evaluation
The samples (liver and a segment with 1.5 cm length from the proximal intestine) were fixed in phosphate-buffered formalin (4 %, pH 7, VWR, Portugal) for 24 h. They were subsequently dehydrated and embedded in paraffin according to standard histological procedures. Thereafter, 3 micra transversal sections were obtained and stained with haematoxylin and eosin (H&E, Merck, Portugal) and periodic acid-Schiff (PAS, Merck, Portugal) before examination under a light microscope (Olympus BX51, cell^B software, GmbH, Hamburg, Germany).
Three different intestine sections of each animal were used for quantitative measurements. The intestine section area (mm2), villus height (μm), muscular layer thickness (from serosa to submucosa, μm) and goblet cells (number per section) positive to PAS staining of the intestinal wall were measured according to Batista et al. (2014). The villus width (μm) was measured across the base of the villus at the luminal surface of ten selected villi per section.
In the liver, the occurrence of possible pathological damage (presence or absence of defined vacuoles indicator of fat degeneration) was evaluated. For each sampled fish, the cytoplasm vacuolization degree (H&E) and glycogen content (PAS) of the hepatocytes were analysed (Fig. 1). Observations were consistently made using a combination of low magnification (objective of 10×) for noticing the general aspects and then with higher magnification (objective of 40×) for categorizing. Ten randomly sampled fields were analysed per section. Evaluation of the hepatocyte vacuolization degree was made using a semi-quantitative approach, according to the following three grades and criteria: 0 (none)—less than 1/3 of the hepatocyte cytoplasm shows vacuoles, 1 (moderate)—between 1/3 and less than 2/3 of the hepatocyte cytoplasm shows vacuolization, 2 (high)—more than 2/3 of the hepatocyte cytoplasm shows vacuolization. Assessment of hepatocyte carbohydrates (namely glycogen) content also followed a semi-quantitative approach, with three degrees: 0 (none)—when hepatocytes in PAS sections did not stain positively or had a very weak colour, 1 (moderate)—PAS sections presented a median staining, 2 (high)—PAS sections present hepatocytes with a very strong staining.
Intestinal microbiota
Microbial populations were isolated from the intestinal content as described by Merrifield et al. (2009). Intestinal content samples were serially diluted to 10−2 with PBS and 100 μL were spread onto duplicate tryptone soy agar (TSA, 0.9 % NaCl) plates. Plates were incubated at 25 °C for 48 h and colony-forming units (CFU) g−1 was calculated from statistically viable plates (i.e. plates containing 30–300 colonies). One hundred colonies of one of these plates were randomly collected from each dietary treatment assayed and cultured on TSA to obtain pure cultures. Then the colonies were identified by the amplification and sequencing of a fragment of 16S rDNA. Briefly, the colonies isolated were suspended in 100 μL of sterile Milli-Q water. Samples were boiled at 100 °C for 15 min and adjusted to 1 mL. These suspensions were centrifuged at 12,000 rpm for 5 min and 1–5 μL of the supernatant was used to carry out the PCR reactions. The fragment 16S rDNA was amplified using the universal primers SD-Bact-0008-a-S20 (5′ AGAGTTTGATCCTGGCTCAG 3′) and SD-Bact-1492-a-A-19 (5′ GGTTACCTTGTTACGACTT 3′) (Kim and Austin 2006). Polymerase chain reactions were carried out in a 50 μL reaction mixture that included 5 pmol of each primer, 200 μM dNTPs, 1× PCR buffer, 2 mM MgCl2, 1 U BIOTAQ DNA Polymerase (Bioline, London, UK) and 1 μL of a boiled colony suspension. Thermal cycling consisted of an initial step of 2 min at 95 °C and 35 cycles of 30 s at 95 °C, 30 s at 52 °C and 40 s at 72 °C and a final extension step 5 min at 72 °C. Polymerase chain reaction products were electrophoresed on 1 % agarose gel, visualized via ultraviolet transillumination and then they were purified with a QiaQuick PCR purification kit (QIAGEN, Hilden, Germany). The PCR products were sequenced by cycling sequencing using SD-Bact-0008-a-S20 and SD-Bact-1492-a-A-19 by Macrogen (Corea). The resulting sequences (∼500 bp) were compared with the sequences from the National Center for Biotechnology Information (NCBI) or Greengenes DNA sequence database using the BLAST sequence algorithm (Altschul et al. 1990). Chimaeric sequences were identified by using the CHECK CHIMERA program of the Ribosomal Database Project (Maidak et al. 1999), and the sequences reported in this study have been deposited in the GenBank database under the following accession numbers: KU725824 to KU725849.
Calculations of growth performance
Feed conversion ratio (FCR) was calculated as dry feed intake (g)/wet weight gain (g) and the specific growth rate (SGR, % BW day−1) as 100 × (ln W 0 − ln W 1)/days, where W 0 and W 1 are the initial and the final fish mean weights in grammes and days is the trial duration. Voluntary feed intake (VFI, % BW day−1) was calculated as 100 × (dry feed intake (g)/ABW (g)/days), where ABW (average body weight) was calculated as (W 1 + W 0)/2. The net protein utilization (NPU) was calculated as (PBF − PBI)/protein fed, where PBF is the final protein content of fish and PBI is the initial protein content of fish.
Statistical analysis
Statistical analyses (except for intestinal microbiota) were performed with the software SPSS (IBM SPSS STATISTICS, 17.0 package, IBM Corporation, New York, USA). Results are expressed as mean ± standard deviation (SDpooled as weighted average of each group’s standard deviation) and the level of significance used was P ≤ 0.05. Data were analysed for normality (Shapiro-Wilk test) and homogeneity of variance (Levene’s test) and were log-transformed whenever necessary. Data were analysed by a two-way ANOVA with diet and probiotic as main factors. When significant differences were obtained from the ANOVA, Tukey’s post hoc tests were carried out to identify significantly different groups. When data did not meet the ANOVA assumptions, a non-parametric Kruskal-Wallis test was performed. For intestinal microbiota, Kruskal-Wallis test was used to test the significance of the differences among dietary treatments.
Results
Growth performance
Fish grew from 33.1 ± 0.20 to 50.6 ± 1.2 g after feeding the experimental diets for 73 days (Table 2). Growth performance did not differ between PP35 and PP72 groups. PROB groups had significantly lower final body weight (FBW, 45.0 ± 1.9) and daily growth index (SGR, 0.42 ± 0.1) compared to UN groups (50.5 ± 2.0 and 0.58 ± 0.0, respectively). Additionally, UN groups had significantly better feed conversion ratio (FCR, 1.5 ± 0.1) and net protein utilization (NPU, 19.07 ± 2.3) than the PROB (2.2 ± 0.4 and 10.4 ± 1.9, respectively) and YEAST (2.1 ± 0.4 and 13.0 ± 4.6, respectively) supplemented groups. Voluntary feed intake (VFI) was also lower in the UN groups (0.8 ± 0.0) and differed significantly from the YEAST groups (0.9 ± 0.1).
Humoral innate immune parameters
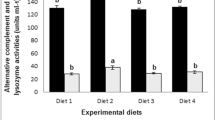
The values of humoral innate parameters analysed are present in Table 3. Peroxidase activity (EU mL−1) was not significantly different (P > 0.05) among dietary groups for any sampling time. Lysozyme activity and ACH50 were not affected by probiotic administration, but were significantly changed by the dietary PP level at 17 and 38 days of feeding. At 17 days, fish from PP72 groups had higher lysozyme (359 ± 201 EU mL−1) and ACH50 (204 ± 55 units mL−1) compared to fish from PP35 groups (177 ± 106 EU mL−1 and 169 ± 19 units mL−1, respectively). At 38 days of feeding, fish from PP72 groups showed higher lysozyme (908 ± 399 EU mL−1) and ACH50 (258 ± 64 units mL−1) activities, compared to fish from PP35 groups (647 ± 457 EU mL−1 and 183 ± 49 units mL−1, respectively).
Histological evaluation
Histological measurements of the intestinal mucosa of fish are present in Table 4 Muscle layer thickness did not vary significantly, regardless of the dietary treatment and sampling time. Intestine section area was significantly different at 2 days of feeding trial, showing the PP72_UN group (2.75 ± 0.66 mm2) with higher area compared to PP35_YEAST (1.93 ± 0.30 mm2) and PP35_UN (1.99 ± 0.42 mm2) groups. Villus was significantly wider (P < 0.01) at 73 days in fish fed PP35_YEAST group (91.01 ± 1.49 μm) than fish fed PP72_YEAST (79.45 ± 7.28 μm) (Fig. 2). Villus length was significantly different (P < 0.05) at 2, 38 and 73 days of feeding the experimental diets (Fig. 2). At 2 days of feeding, fish fed PP72_UN diet had the longest villus (418 ± 30 μm) when compared to fish fed PP35_UN (357 ± 41 μm) and PP72_YEAST (361 ± 13 μm) diets. At 38 days of feeding, fish from YEAST groups had a significant longer villus (413 ± 45 μm) than fish from the UN groups (372 ± 44 μm). The number of goblet cells was significantly different (P < 0.05) at 17 and 73 days of feeding dietary treatments (Fig. 3). At 17 days of feeding, fish fed PP72_UN diet had a significantly (P < 0.05) higher number of goblet cells (379 ± 119) compared to the other dietary treatments, except fish fed PP35_YEAST diet. At 73 days of feeding, both villus length and number of goblet cells were affected by dietary PP level, with fish from PP35 groups having larger villus (455 ± 52 μm) and higher number of goblet cells (363 ± 210) than fish from PP72 groups (395 ± 57 μm and 260 ± 129, respectively).
Histological sections of anterior intestines of Senegalese sole for villus width (VW) and for villus length (VL). VW: (a) PP35_YEAST diet showing higher VW than (b) that corresponds to PP72_YEAST diet (H&E, bar = 100 μm). VL: (a, b) 2 days samples time. (c, d) 73 days sampling time. Note the difference between the villus dimensions in (a) and (c) showing higher villus length when compared with (b) and (d). (a) PP35 diet. (b) PP72_YEAST diet. (c) PP35 diet. (d) PP72_YEAST diet (H&E, bar = 250 μm)
Histological sections of anterior intestines of Senegalese sole for number counting of goblet cells (GC). (a) PP72 diet and (b) PP72_PROB diet, both at 17 days sampling time. (c) PP35 diet and (d) PP72_YEAST diet at 73 days sampling time. Note the difference observed between the (a) and (c) showing higher number of GC, compared with (b) and (d) (PAS stained, bar = 100 μm)
Liver histology revealed different degrees of vacuolization at 2 and 73 days of feeding. Fish from the PP72 groups showed a higher vacuolization (grade 3) compared to fish from PP35 groups (grade 1).
At 2 days of feeding, fish fed diets supplemented with YEAST showed higher hepatocytes glycogen content (more than 2/3 of the liver positively stained) when compared with the fish fed the diets supplemented with PROB. At 17 days of feeding, fish fed PP72 presented hepatocytes with a weak PAS staining, indicating a very low content of carbohydrates (namely glycogen).
Intestinal microbiota
The intestinal bacteria counts varied from 8.2 × 105 to 2.4 × 107 CFU mL−1, as an effect of the diet and sampling time (Table 5). The inclusion of 35 or 72 % of plant ingredients in the diet induced a significant reduction of the microbial counts in fish intestine after 38 or 73 days of feeding. In Table 5, it is possible to observe that the supplementation with the autolysed yeast in PP35 and PP72 diets induced significant differences in the level of intestinal microorganisms in comparison with the microbial recounts obtained in fish fed the non-supplemented diets. On the contrary, these significant differences were not observed when PP35 and PP72 diets were supplemented with the multispecies probiotic bacteria.
In general, the highest relative percentage corresponded to Vibrio sp. (24 to 99 %). Probiotic supplementation greatly reduced the concentration of intestinal Vibrio sp. (Table 5), especially in fish fed low dietary PP level (PP35_PROB). The duration of the dietary treatment had a clear effect on the reduction of intestinal Vibrio sp. concentration in fish fed PP35_PROB. Vibrio harveyi and Vibrio owensii were identified in fish fed PP72_PROB and PP72_YEAST and not identified in fish fed PP72_UN (Table 6). In addition, Staphylococcus saprophyticus was observed only in the intestine of fish fed PROB diets (Tables 6 and 7).
Discussion
Probiotics confer beneficial actions to the host or to their environment through different modes of action. Their application in aquaculture has been regarded as a sustainable and promising strategy not only in the context of disease control but also in nutrition, growth and immunity (Lazado and Caipang 2014).
The capacity of Senegalese sole to cope with diets in which the marine-derived protein was replaced by plant ingredients has been previously reported (Cabral et al. 2011, 2013; Silva et al. 2009). Similarly, to previous studies, the current study demonstrated that growth performance of Senegalese sole was not affected by high content of plant ingredients. The final body weight and FCR were, however, negatively affected by dietary probiotic supplementation (PROB diet). In fact, the effects of probiotics and prebiotics on fish growth performance are often contradictory. Probiotic improved the feed efficiency and growth performance in Senegalese sole (García de la Banda et al. 2012), Japanese flounder (Taoka et al. 2006) and Atlantic cod (Lauzon et al. 2010). Nevertheless, Ferguson et al. (2010) showed no clear effect on growth performance in Nile tilapia fed with Pediococcus acidilactici supplementation, whereas Gunther and Jimenez-Montealegre (2004) observed a growth depression in tilapia fed Bacillus subtilis.
Humoral immune parameters as well as some intestinal morphology parameters showed a high standard deviation, indicating that the data points are spread out over a wider range of values. As readers are generally interested in knowing variability within the sample, descriptive data should be precisely summarized with SD (Barde and Barde 2012). However, for many biological variables the standard deviation can differ significantly across groups in some data sets and the most important is that the data meet the assumptions of the normal distribution model that we used.
All humoral immune parameters analysed, plasma lysozyme, peroxidase and alternative complement (ACH50) activities were not significantly altered by PROB or YEAST. This trend was also observed by Batista et al. (2014) working with sole fed the same multispecies bacteria and Díaz-Rosales et al. (2009) working with sole fed with two different Shewanella sp. However, ACH50 was enhanced when rainbow trout were fed for 4 weeks with diets supplemented with heat-inactivated multispecies bacteria (Choi and Yoon 2008) and gilthead seabream fed for 3 weeks (Salinas et al. 2008). In addition, serum lysozyme activity increased significantly from 438 ± 75 U mL−1 (control) to 1269 ± 134 U mL−1 in rainbow trout fed for 14 days with diets supplemented with a autochthonous intestinal B. subtilis (Newaj-Fyzul et al. 2007). These changes in the innate immune responses were associated to the inhibitory effect of that probiotic against the pathogenic Aeromonas sp.
Previous studies have demonstrated that oral administration of fungal carbohydrates enhances fish innate immune response and protection against infections (Efthimiou 1996; Esteban et al. 2001; Siwicki et al. 1994), although this effect seems to be temporary (reversible) and dependent on its inclusion level and feeding duration (Ortuño et al. 2002). In the current study, innate immune parameters were affected by the increase of dietary plant ingredients content. At 17 and 38 days of feeding trial, fish from PP72 groups had higher values of lysozyme and ACH50 activities than fish from PP35 groups, suggesting a stimulation of the innate immune activity by dietary high PP content. Also, Geay et al. (2011) observed an increase in the ACH50, while the lysozyme was lower, in fish fed a PP diet when compared to fish fed fishmeal-based diet. On the other hand, Sitjà-Bobadilla et al. (2005) reported that high level of fishmeal replacement by plant protein sources had no significant effect on lysozyme activity. Jalili et al. (2013) showed that rainbow trout fed diets with high PP levels (70 and 100 %) resulted in a decrease on the ACH50, as reported by Sitjà-Bobadilla et al. (2005) in gilthead seabream. Hepatocytes provide the major source of complement factors (Abelseth et al. 2003), and the progressive fat degeneration of liver with the PP inclusion could decrease complement proteins synthesis (Sitjà-Bobadilla et al. 2005). Liver from fish fed PP75 diets showed a higher level of vacuolization compared to fish fed PP35 diets. The use of high levels of plant ingredients is recognized to have several disadvantages, particularly related to the amino acid profiles and unsaturated fatty acid imbalances but especially due to their levels of antinutritional factors (Geay et al. 2011). In the current study, high PP level seems to result in an acute stimulation of lysozyme and ACH50, since at 17 and 38 days, fish fed PP72 diets showed higher activities than fish fed PP35 diets. Nevertheless, at 73 days of feeding trial, such effects disappeared. Food legumes and cereals may have adverse effects on some fish species after intake, resulting in morphological problems in liver and intestine that could lead to metabolic failure (Russell et al. 2001). PP72 diet, when compared to PP35, was formulated to contain a low amount of soybean meal, but high content of mixed plant ingredients, including insoluble pea protein, as protein source replacing fishmeal. Thus, the dietary incorporation level of plant protein sources used in the current study may have provided a high level of antinutritional factors, especially after 73 days of feeding.
Intestinal microbiota is confined within a highly specialized barrier defences, composed by the stratified mucous layer, an epithelium and a lamina propria with several innate and adaptive immune cells (Maynard et al. 2012). An increase in intestine section area, villus length and villus width are directly related to an increase of surface area, which may indicate improvement of intestinal absorptive capacity nutrients (Caspary 1992). At 2 days of feeding, fish fed PP72_UN showed larger intestine section area and longer villus than fish fed PP35_UN. At 17 days of feeding, fish fed PP72_UN showed more goblet cells than the other dietary groups, except the group fed PP35_YEAST. At 73 days, however, villus length and width and goblet cells were significantly increased in fish fed low PP diets compared to high PP diets. The latter may indicate that at 2 and 17 days, fish fed the diets formulated with high levels of plant ingredient level had an enhancement on the immune defence and an improvement in the intestinal surface area in contact with nutrients. However, after 73 days of feeding, these effects were reversed, probably due to habituation of fish and/or to a negative effect of chronic ingestion of high PP diets and presence of antinutritional factors. Intestinal epithelium is covered by a layer of mucus continuously produced by goblet cells and being the first line of defence against microbes (Maynard et al. 2012). So, the reduced number of goblet cells, after 73 days of feeding fish with the PP72 diets, causes a decrease in mucous production, thus probably reducing intestinal protection capacity of those fish.
After 38 days of feeding the experimental diets, villus length was significantly increased in YEAST-supplemented groups, compared to unsupplemented (UN) groups. Yeast cells have been reported to be a source of nucleotides, which contribute for the intestinal maintenance in aquatic animals by improving mucosal flora and mucosal surfaces with relative elongation of the intestinal tract (Li et al. 2007). Also, (Abu-Elala et al. 2013) observed that fish treated with S. cerevisiae showed yeast colonization in the intestine, accompanied by an increase in the length and density of the intestinal villus. Such morphological alterations are often associated with an improvement in food digestion and absorption. After 2 days feeding the YEAST diets, fish had a higher hepatocyte glycogen content compared with fish fed diets supplemented with PROB, showing a stimulation of the glycogen liver storage, the first line of energy source when blood glucose concentration falls during stress.
Fish intestinal microbiota changes with the diet, and this effect is of particular interest considering that the effects of high fishmeal replacement by plant ingredients are still poorly understood, especially in flatfish. Molecular techniques have facilitated culture-independent studies, becoming a valid support to traditional techniques (Ercolini 2004). In the current study, from all bacteria isolated from sole intestine, approximately 20 % were not identified. The intestinal tract of fish harbours a high density of non-culturable bacteria and the composition of which has not yet been reported, leading to differences between viable and total microbial counts (Shiina et al. 2006). In this study, it has been possible to demonstrate the ability to modulate the intestinal microbiota of Senegalese sole by feeding diets supplemented either with probiotic or autolysed S. cerevisiae. The results obtained are in accordance with those reported by Standen et al. (2015), who observed in tilapia (Oreochromis niloticus) the modulation of the intestinal microbiota and morphology caused by feeding the fish with the same multispecies probiotic used in the current study. The modulation exerted on the intestinal microbiota by viable and inactivated cells of S. cerevisiae has been reported in tilapia and sturgeon (Huso huso) (Hoseinifar et al. 2011; Waché et al. 2006). In addition, the modulation of intestinal microbiota has also been demonstrated in fish treated with S. cerevisiae fermentation products (He et al. 2011) and glucans obtained from yeast (Kuhlwein et al. 2013). In the current study, the predominant bacteria found in S. senegalensis intestine were Vibrio sp., which is in agreement with other studies carried out with the same flatfish cultured under extensive, semi-extensive or intensive production systems (Martin-Antonio et al. 2007). This trend was also detected in farmed Senegalese sole fed fresh or lyophilized Shewanella putrefaciens (Tapia-Paniagua et al. 2010, 2015).
Fish fed dietary probiotic supplementation (PP35_PROB and PP72_PROB) had reduced intestinal bacteria related to Vibrio species. Previous studies observed that strains of probiotic Bacillus (Liu et al. 2015; Luis-Villasenor et al. 2015; Wu et al. 2014), Enterococcus (Lin et al. 2013) and Pediococcus and Lactobacillus (Munoz-Atienza et al. 2013) showed an antibacterial activity against fish pathogens, including Vibrio.
V. harveyi was detected in sole fed probiotic supplemented and especially in fish fed yeast-supplemented diets, and not detected in fish fed unsupplemented diets. These Vibrio species are included in the Harveyi clade and are well-known as pathogenic for marine-farmed organisms (Austin and Zhang 2006; Cano-Gomez et al. 2011; Gomez-Gil et al. 2004), including S. senegalensis in the case of V. harveyi (Arijo et al. 2005; Zorrilla et al. 2003). However, it is frequent in the presence of virulent and non-virulent strains of V. harveyi (Rico et al. 2008; Zorrilla et al. 2003), and for this reason, more studies are necessary to evaluate the potential virulence of the strains of these species and how diet formulation may modulate bacterial presence in the intestine. On the other hand, the dietary probiotic supplementation in PP35 diet (PP35_PROB) induced the presence of S. saprophyticus. S. saprophyticus is associated to microorganisms that play a critical role in fish fermentation and is known to be resistant to different antibiotics (Sergelidis et al. 2014; Zhang et al. 2015). In the current study, S. saprophyticus were detected solely in fish fed probiotic diets, especially in low PP groups. Although there are no reports that S. saprophyticus caused diseases in fish, Sun et al. (2011) speculate that it may be a potential harmful bacterium as it is often implicated in human urinary tract infections (Kuroda et al. 2005). Its presence in the intestinal microbiota of fish must be considered as a route for the transmission of antibiotic resistance (Chajecka-Wierzchowska et al. 2015).
In conclusion, Senegalese sole was able to cope with high plant ingredient content in replacement of marine-derived ingredients. After 2 and 17 days, diets with high PP content seem to acutely stimulate the innate immune defence of the fish, as suggested by lysozyme and ACH50 activities. However, after 73 days of feeding, fish fed PP72 showed reduced the number of the goblet cells and low hepatic glycogen content, indicating a lower capacity to overcome stress situations.
Our data demonstrates that it is possible to modulate the intestinal microbiota of Senegalese sole by dietary supplementation with probiotic or autolysed S. cerevisiae, even if Vibrio sp. are the predominant bacteria present in sole intestine. The diets with selected probiotics or autolysed yeast were able to reduce Vibrio load, but the appearance of V. harveyi and S. saprophyticus still has associated a potential health risk.
Dietary inclusion of the multispecies probiotic bacteria or autolysed yeast used in this work should be regarded with caution for Senegalese sole, since there was a negative impact on fish growth performance and no clear health benefits were observed at the end of the trial. More studies should be performed to evaluate if the selected probiotics’ capacity to modulate gut microbiota results in clear health benefits for the fish.
References
Abelseth TK, Stensvag K, Espelid S, Nygaard R, Ellingsen T, Bogwald J, Dalmo RA (2003) The spotted wolffish (Anarhichas minor Olafsen) complement component C3: isolation, characterisation and tissue distribution. Fish Shellfish Immunol 15:13–27
Abu-Elala N, Marzouk M, Moustafa M (2013) Use of different Saccharomyces cerevisiae biotic forms as immune-modulator and growth promoter for Oreochromis niloticus challenged with some fish pathogens. Int J Vet Sci Med 1:21–29
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Aly SM, Abdel-Galil Ahmed Y, Abdel-Aziz Ghareeb A, Mohamed MF (2008) Studies on Bacillus subtilis and Lactobacillus acidophilus, as potential probiotics, on the immune response and resistance of Tilapia nilotica (Oreochromis niloticus) to challenge infections. Fish Shellfish Immunol 25:128–136
Anderson DP (1992) Immunostimulants, adjuvants, and vaccine carriers in fish: applications to aquaculture. Annu Rev Fish Dis 2:281–307
AOAC (2006) Official methods of analysis of AOAC International, 18 edn. Maryland, USA
Arijo S, Chabrillón M, Díaz-Rosales P, Rico RM, Martínez-Manzanares E, Balebona MC, Toranzo AE, Moriñigo MA (2005) Bacteria isolated from outbreaks affecting cultured sole, Solea senegalensis (Kaup). Bull Eur Assoc Fish Pathol 25:148–154
Austin B, Zhang XH (2006) Vibrio harveyi: a significant pathogen of marine vertebrates and invertebrates. Lett Appl Microbiol 43:119–124
Balcázar JL, Blas I, Ruiz-Zarzuela I, Vendrell D, Calvo AC, Márquez I, Gironés O, Muzquiz JL (2007) Changes in intestinal microbiota and humoral immune response following probiotic administration in brown trout (Salmo trutta). Br J Nutr 97:522–527
Barde MP, Barde PJ (2012) What to use to express the variability of data: standard deviation or standard error of mean? Perspect Clin Res 3:113–116
Batista S, Ozório R, Kollias S, Dhanasiri A, Lokesh J, Kiron V, LMP V, Fernandes J (2016) Changes in intestinal microbiota, immune- and stress-related transcript levels in Senegalese sole (Solea senegalensis) fed plant ingredients diets intercropped with probiotics or imunostimulants. Aquaculture 458:149–157
Batista S, Ramos MA, Cunha S, Barros R, Cristóvão B, Rema P, Pires MA, Valente LMP, Ozório ROA (2014) Immune responses and gut morphology of Senegalese sole (Solea senegalensis, Kaup 1858) fed monospecies and multispecies probiotics. Aquac Nutr 21:625–634
Bell T, Newman JA, Silverman BW, Turner SL, Lilley AK (2005) The contribution of species richness and composition to bacterial services. Nature 436:1157–1160
BS EN 15784:2009 (2009) Animal feeding stuffs. Isolation and enumeration of presumptive Bacillus spp., p 18
BS EN 15786:2009 (2009) Animal feeding stuffs. Isolation and enumeration of Pediococcus spp., p 20
BS EN 15787:2009 (2009) Animal feeding stuffs. Isolation and enumeration of Lactobacillus spp., p 20
BS EN 15788:2009 (2009) Animal feeding stuffs—isolation and enumeration of Enterococcus (E. faecium) spp., p 18
Cabral EM, Bacelar M, Batista S, Castro-Cunha M, Ozório ROA, Valente LMP (2011) Replacement of fishmeal by increasing levels of plant protein blends in diets for Senegalese sole (Solea senegalensis) juveniles. Aquaculture 322-323:74–81
Cabral EM, Fernandes TJR, Campos SD, Castro-Cunha M, Oliveira MBPP, Cunha LM, Valente LMP (2013) Replacement of fish meal by plant protein sources up to 75% induces good growth performance without affecting flesh quality in ongrowing Senegalese sole. Aquaculture 380-383:130–138
Cano-Gomez A, Hoj L, Owens L, Andreakis N (2011) Multilocus sequence analysis provides basis for fast and reliable identification of Vibrio harveyi-related species and reveals previous misidentification of important marine pathogens. Syst Appl Microbiol 34:561–565
Caspary WF (1992) Physiology and pathophysiology of intestinal absorption. Am J Clin Nutr 55:299S–308S
Chajecka-Wierzchowska W, Zadernowska A, Nalepa B, Sierpinska M, Laniewska-Trokenheim L (2015) Coagulase-negative staphylococci (CoNS) isolated from ready-to-eat food of animal origin–phenotypic and genotypic antibiotic resistance. Food Microbiol 46:222–226
Choct M (2009) Managing gut health through nutrition. Br Poult Sci 50:9–15
Choi S-H, Yoon T-J (2008) Non-specific immune response of rainbow trout (Oncorhynchus mykiss) by dietary heat-inactivated potential probiotics. Immune Netw 8:67–74
Costas B, Conceição LEC, Dias J, Novoa B, Figueras A, Afonso A (2011) Dietary arginine and repeated handling increase disease resistance and modulate innate immune mechanisms of Senegalese sole (Solea senegalensis Kaup, 1858). Fish Shellfish Immunol 31:838–847
Díaz-Rosales P, Arijo S, Chabrillón M, Alarcón FJ, Tapia-Paniagua ST, Martínez-Manzanares E, Balebona MC, Moriñigo MA (2009) Effects of two closely related probiotics on respiratory burst activity of Senegalese sole (Solea senegalensis, Kaup) phagocytes, and protection against Photobacterium damselae subsp. piscicida. Aquaculture 293:16–21
Dimitroglou A, Merrifield DL, Carnevali O, Picchietti S, Avella M, Daniels C, Güroy D, Davies SJ (2011) Microbial manipulations to improve fish health and production—a Mediterranean perspective. Fish Shellfish Immunol 30:1–16
Efthimiou S (1996) Dietary intake of β-1,3/1,6 glucans in juvenile dentex (Dentex dentex), Sparidae: effects on growth performance, mortalities and non-specific defense mechanisms. J Appl Ichthyol 12:1–7
Ercolini D (2004) PCR-DGGE fingerprinting: novel strategies for detection of microbes in food. J Microbiol Methods 56:297–314
Esteban MA, Cuesta A, Ortuño J, Meseguer J (2001) Immunomodulatory effects of dietary intake of chitin on gilthead seabream (Sparus aurata L.) innate immune system. Fish Shellfish Immunol 11:303–315
Ferguson RMW, Merrifield DL, Harper GM, Rawling MD, Mustafa S, Picchietti S, Balcázar JL, Davies SJ (2010) The effect of Pediococcus acidilactici on the gut microbiota and immune status of on-growing red tilapia (Oreochromis niloticus). J Appl Microbiol 109:851–862
Gaggìa F, Mattarelli P, Biavati B (2010) Probiotics and prebiotics in animal feeding for safe food production. Int J Food Microbiol 141(Supplement 1):S15–S28
García de la Banda I, Lobo C, Chabrillón M, León-Rubio JM, Arijo S, Pazos G, María Lucas L, Moriñigo MÁ (2012) Influence of dietary administration of a probiotic strain Shewanella putrefaciens on Senegalese sole (Solea senegalensis, Kaup 1858) growth, body composition and resistance to Photobacterium damselae subsp piscicida. Aquac Res 43:662–669
Geay F, Ferraresso S, Zambonino-Infante J, Bargelloni L, Quentel C, Vandeputte M, Kaushik S, Cahu C, Mazurais D (2011) Effects of the total replacement of fish-based diet with plant-based diet on the hepatic transcriptome of two European sea bass (Dicentrarchus labrax) half-sibfamilies showing different growth rates with the plant-based diet. BMC Genomics 12:1–18
Gomez-Gil B, Soto-Rodriguez S, Garcia-Gasca A, Roque A, Vazquez-Juarez R, Thompson FL, Swings J (2004) Molecular identification of Vibrio harveyi-related isolates associated with diseased aquatic organisms. Microbiology 150:1769–1777
Gunther J, Jimenez-Montealegre R (2004) Effect of the probiotic Bacillus subtilis on the growth and food utilization of tilapia (Oreochromis niloticus) and prawn (Macrobrachium rosenbergii) under laboratory conditions. Rev Biol Trop 52:937–943
He S, Zhou Z, Meng K, Zhao H, Yao B, Ringo E, Yoon I (2011) Effects of dietary antibiotic growth promoter and Saccharomyces cerevisiae fermentation product on production, intestinal bacterial community, and nonspecific immunity of hybrid tilapia (Oreochromis niloticus female x Oreochromis aureus male). J Anim Sci 89:84–92
Hoseinifar SH, Mirvaghefi A, Merrifield DL (2011) The effects of dietary inactive brewer’s yeast Saccharomyces cerevisiae var. ellipsoideus on the growth, physiological responses and gut microbiota of juvenile beluga (Huso huso). Aquaculture 318:90–94
Hutchinson TH, Manning MJ (1996) Seasonal trends in serum lysozyme activity and total protein concentration in dab (Limanda limanda L.) sampled from Lyme Bay, U.K. Fish Shellfish Immunol 6:473–482
ISO 6498:2012 (2012) Animal feeding stuffs - guidelines for sample preparation, p 46
ISO 6887-1:1999 (1999) Microbiology of food and animal feeding stuffs - preparation of test samples, initial suspension and decimal dilutions for microbiological examination - part 1: general rules for the preparation of the initial suspension and decimal dilutions, p 5
ISO-7218:2007 (2007) Microbiology of food and animal feeding stuffs - general requirements and guidance for microbiological examinations, p 66
Jalili R, Tukmechi A, Agh N, Noori F, Ghasemi A (2013) Replacement of dietary fish meal with plant sources in rainbow trout (Oncorhynchus mykiss) effect on growth performance, immune responses, blood indices and disease resistance. Iran J Fish Sci 12:577–591
Kim D-H, Austin B (2006) Innate immune responses in rainbow trout (Oncorhynchus mykiss, Walbaum) induced by probiotics. Fish Shellfish Immunol 21:513–524
Kuhlwein H, Emery MJ, Rawling MD, Harper GM, Merrifield DL, Davies SJ (2013) Effects of a dietary beta-(1,3)(1,6)-D-glucan supplementation on intestinal microbial communities and intestinal ultrastructure of mirror carp (Cyprinus carpio L.). J Appl Microbiol 115:1091–1106
Kumar R, Mukherjee SC, Ranjan R, Nayak SK (2008) Enhanced innate immune parameters in Labeo rohita (Ham.) following oral administration of Bacillus subtilis. Fish Shellfish Immunol 24:168–172
Kuroda M, Yamashita A, Hirakawa H, Kumano M, Morikawa K, Higashide M, Maruyama A, Inose Y, Matoba K, Toh H, Kuhara S, Hattori M, Ohta T (2005) Whole genome sequence of Staphylococcus saprophyticus reveals the pathogenesis of uncomplicated urinary tract infection. Proc Natl Acad Sci USA 102:13272–13277
Lauzon HL, Gudmundsdottir S, Steinarsson A, Oddgeirsson M, Martinsdottir E, Gudmundsdottir BK (2010) Impact of probiotic intervention on microbial load and performance of Atlantic cod (Gadus morhua L.) juveniles. Aquaculture 310:139–144
Lazado CC, Caipang CMA (2014) Atlantic cod in the dynamic probiotics research in aquaculture. Aquaculture 424-425:53–62
Li P, Lawrence AL, Castille FL, Gatlin DM (2007) Preliminary evaluation of a purified nucleotide mixture as a dietary supplement for Pacific white shrimp Litopenaeus vannamei (Boone). Aquac Res 38:887–890
Lin YH, Chen YS, Wu HC, Pan SF, Yu B, Chiang CM, Chiu CM, Yanagida F (2013) Screening and characterization of LAB-produced bacteriocin-like substances from the intestine of grey mullet (Mugil cephalus L.) as potential biocontrol agents in aquaculture. J Appl Microbiol 114:299–307
Liu XF, Li Y, Li JR, Cai LY, Li XX, Chen JR, Lyu SX (2015) Isolation and characterisation of Bacillus spp. antagonistic to Vibrio parahaemolyticus for use as probiotics in aquaculture. World J Microbiol Biotechnol 31:795–803
Luis-Villasenor I, Voltolina D, Gomez-Gil B, Ascencio F, Campa-Cordova A, Audelo-Naranjo J, Zamudio-Armenta O (2015) Probiotic modulation of the gut bacterial community of juvenile Litopenaeus vannamei challenged with Vibrio parahaemolyticus CAIM 170. Lat Am J Aquat Res 43:766–775
Magnadottir B (2010) Immunological control of fish diseases. Mar Biotechnol 12:361–379
Maidak BL, Cole JR, Parker CT Jr, Garrity GM, Larsen N, Li B, Lilburn TG, McCaughey MJ, Olsen GJ, Overbeek R, Pramanik S, Schmidt TM, Tiedje JM, Woese CR (1999) A new version of the RDP (ribosomal database project). Nucleic Acids Res 27:171–173
Manzano M, Iacumin L, Giusto C, Cecchini F, Patthey C, Fontanillas R, Comi G (2012) Utilization of denaturing gradient gel electrophoresis (DGGE) to evaluate the intestinal microbiota of brown trout Salmo trutta fario. J Vet Sci Med Diagn 1:1–6
Marcel BR (2008) Prebiotics: Concept, Definition, Criteria, Methodologies, and Products. In: Handbook of prebiotics. CRC Press, Boca Raton, New York, pp. 39–68
Martin-Antonio B, Manchado M, Infante C, Zerolo R, Labella A, Alonso C, Borrego JJ (2007) Intestinal microbiota variation in Senegalese sole (Solea senegalensis) under different feeding regimes. Aquac Res 38:1213–1222
Maynard CL, Elson CO, Hatton RD, Weaver CT (2012) Reciprocal interactions of the intestinal microbiota and immune system. Nature 489:231–241
Merrifield DL, Burnard D, Bradley G, Davies SJ, Baker RTM (2009) Microbial community diversity associated with the intestinal mucosa of farmed rainbow trout (Oncoryhnchus mykiss Walbaum). Aquac Res 40:1064–1072
Merrifield DL, Dimitroglou A, Foey A, Davies SJ, Baker RTM, Bøgwald J, Castex M, Ringø E (2010) The current status and future focus of probiotic and prebiotic applications for salmonids. Aquaculture 302:1–18
Munoz-Atienza E, Gomez-Sala B, Araujo C, Campanero C, del Campo R, Hernandez P, Herranz C, Cintas L (2013) Antimicrobial activity, antibiotic susceptibility and virulence factors of lactic acid bacteria of aquatic origin intended for use as probiotics in aquaculture. BMC Microbiol 13:15
Nayak SK (2010) Probiotics and immunity: a fish perspective. Fish Shellfish Immunol 29:2–14
Newaj-Fyzul A, Adesiyun AA, Mutani A, Ramsubhag A, Brunt J, Austin B (2007) Bacillus subtilis AB1 controls Aeromonas infection in rainbow trout (Oncorhynchus mykiss, Walbaum). J Appl Microbiol 103:1699–1706
Ortuño J, Cuesta A, Rodrı́guez A, Esteban MA, Meseguer J (2002) Oral administration of yeast, Saccharomyces cerevisiae, enhances the cellular innate immune response of gilthead seabream (Sparus aurata L.). Vet Immunol Immunopathol 85:41–50
Rawls JF, Samuel BS, Gordon JI (2004) Gnotobiotic zebrafish reveal evolutionarily conserved responses to the gut microbiota. Proc Natl Acad Sci U S A 101:4596–4601
Rico RM, Tapia-Paniagua S, Martínez-Manzanares E, Balebona MC, Moriñigo MA (2008) Characterization of Vibrio harveyi strains recovered from diseased farmed Senegalese sole (Solea senegalensis). J Appl Microbiol 105:752–760
Rumsey GL, Winfree RA, Hughes SG (1992) Nutritional value of dietary nucleic acids and purine bases to rainbow trout (Oncorhynchus mykiss). Aquaculture 108:97–110
Russell PM, Davies SJ, Gouveia A, Tekinay AA (2001) Influence of dietary starch source on liver morphology in juvenile cultured European sea bass (Dicentrarchus labrax L.). Aquac Res 32:306–314
Sakai M (1999) Current research status of fish immunostimulants. Aquaculture 172:63–92
Salinas I, Abelli L, Bertoni F, Picchietti S, Roque A, Furones D, Cuesta A, Meseguer J, Esteban MA (2008) Monospecies and multispecies probiotic formulations produce different systemic and local immunostimulatory effects in the gilthead seabream (Sparus aurata L.). Fish Shellfish Immunol 25:114–123
Salinas I, Cuesta A, Esteban MA, Meseguer J (2005) Dietary administration of Lactobacillus delbrueckii and Bacillus subtilis, single or combined, on gilthead seabream cellular innate immune responses. Fish Shellfish Immunol 19:67–77
Sergelidis D, Abrahim A, Papadopoulos T, Soultos N, Martziou E, Koulourida V, Govaris A, Pexara A, Zdragas A, Papa A (2014) Isolation of methicillin-resistant Staphylococcus spp. from ready-to-eat fish products. Lett Appl Microbiol 59:500–506
Shiina A, Itoi S, Washio S, Sugita H (2006) Molecular identification of intestinal microflora in Takifugu niphobles. Comput Biochem Phys D 1:128–132
Silva JMG, Espe M, Conceição LEC, Dias J, Valente LMP (2009) Senegalese sole juveniles (Solea senegalensis Kaup, 1858) grow equally well on diets devoid of fish meal provided the dietary amino acids are balanced. Aquaculture 296:309–317
Sitjà-Bobadilla A, Peña-Llopis S, Gómez-Requeni P, Médale F, Kaushik S, Pérez-Sánchez J (2005) Effect of fish meal replacement by plant protein sources on non-specific defence mechanisms and oxidative stress in gilthead sea bream (Sparus aurata). Aquaculture 249:387–400
Siwicki AK, Anderson DP, Rumsey GL (1994) Dietary intake of immunostimulants by rainbow trout affects non-specific immunity and protection against furunculosis. Vet Immunol Immunopathol 41:125–139
Standen BT, Rodiles A, Peggs DL, Davies SJ, Santos GA, Merrifield DL (2015) Modulation of the intestinal microbiota and morphology of tilapia, Oreochromis niloticus, following the application of a multi-species probiotic. Appl Microbiol Biotechnol 99:8403–8417
Sun YZ, Yang HL, Ma RL, Song K, Lin WY (2011) Molecular analysis of autochthonous microbiota along the digestive tract of juvenile grouper Epinephelus coioides following probiotic Bacillus pumilus administration. J Appl Microbiol 110:1093–1103
Sunyer J, Tort L (1995) Natural hemolytic and bactericidal activities of sea bream Sparus aurata serum are effected by the alternative complement pathway. Vet Immunol Immunopathol 45:333–345
Taoka Y, Maeda H, Jo J-Y, Jeon M-J, Bai SC, Lee W-J, Yuge K, Koshio S (2006) Growth, stress tolerance and non-specific immune response of Japanese flounder Paralichthys olivaceus to probiotics in a closed recirculating system. Fish Sci 72:310–321
Tapia-Paniagua S, Chabrillón M, Díaz-Rosales P, Banda IG, Lobo C, Balebona MC, Moriñigo MA (2010) Intestinal microbiota diversity of the flat fish Solea senegalensis (Kaup, 1858) following probiotic administration. Microb Ecol 60:310–319
Tapia-Paniagua ST, Vidal S, Lobo C, García de la Banda I, Esteban MA, Balebona MC, Moriñigo MA (2015) Dietary administration of the probiotic SpPdp11: effects on the intestinal microbiota and immune-related gene expression of farmed Solea senegalensis treated with oxytetracycline. Fish Shellfish Immunol 46:449–458
Tuohy KM, Rouzaud GC, Bruck WM, Gibson GR (2005) Modulation of the human gut microflora towards improved health using prebiotics-assessment of efficacy. Curr Pharm Des 11:75–90
Waché Y, Auffray F, Gatesoupe F-J, Zambonino J, Gayet V, Labbé L, Quentel C (2006) Cross effects of the strain of dietary Saccharomyces cerevisiae and rearing conditions on the onset of intestinal microbiota and digestive enzymes in rainbow trout, Onchorhynchus mykiss, fry. Aquaculture 258:470–478
Wu HJ, Sun LB, Li CB, Li ZZ, Zhang Z, Wen XB, Hu Z, Zhang YL, Li SK (2014) Enhancement of the immune response and protection against Vibrio parahaemolyticus by indigenous probiotic Bacillus strains in mud crab (Scylla paramamosain). Fish Shellfish Immunol 41:156–162
Yachi S, Loreau M (1999) Biodiversity and ecosystem productivity in a fluctuating environment: the insurance hypothesis. Proc Natl Acad Sci 96:1463–1468
Zhang H, Li Y, Xu K, Wu J, Dai Z (2015) Microbiological changes and biodiversity of cultivable indigenous bacteria in Sanbao larger yellow croaker (Pseudosciaena crocea), a Chinese salted and fermented seafood. J Food Sci 80:M776–M781
Zorrilla I, Arijo S, Chabrillon M, Diaz P, Martinez-Manzanares E, Balebona MC, Moriñigo MA (2003) Vibrio species isolated from diseased farmed sole, Solea senegalensis (Kaup), and evaluation of the potential virulence role of their extracellular products. J Fish Dis 26:103–108
Acknowledgments
We would like to thank to CIIMAR/ICBAS (UP) and FBA (University of Nordland) for the use of the facilities and equipment and for technical support and Mrs. Ligia Lourenço (UTAD) for her histology technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
S. M. G. Batista was supported by FCT – SFRH/BD/76668/2011. This work was also supported by the FCT Projects PEst-OE/AGR/UI0772/2011 and PEst-OE/AGR/UI0772/2014.
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Batista, S., Medina, A., Pires, M.A. et al. Innate immune response, intestinal morphology and microbiota changes in Senegalese sole fed plant protein diets with probiotics or autolysed yeast. Appl Microbiol Biotechnol 100, 7223–7238 (2016). https://doi.org/10.1007/s00253-016-7592-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7592-7