Abstract
Production of menaquinone-7 (MK-7) by Bacillus subtilis natto is associated with major drawbacks. To address the current challenges in MK-7 fermentation, studying the effect of magnetic nanoparticles on the bacterial cells can open up a new domain for intensified bioprocesses. This article introduces the new concept of application of iron oxide nanoparticles (IONs) as a pioneer tool for MK-7 process intensification. In this order, IONs with the average size of 11 nm were successfully fabricated and characterized for possible in situ removal of target substances from the fermentation media. The prepared particles were used for decoration and immobilization of B. subtilis natto cells. Presence of iron oxide nanoparticles significantly enhanced the MK-7 specific yield (15 %) as compared to the control samples. In addition, fabricated IONs showed a promising ability for in situ recovery of bacterial cells from the fermentation media with more than 95 % capture efficiency. Based on the results, IONs can be implemented successfully as a novel tool for MK-7 production. This study provides a considerable interest for industrial application of magnetic nanoparticles and their future role in designing an intensified biological process.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Menaquinone-7 (MK-7), a vitamin K homolog, contributes significantly in cardiovascular and bone health and can only be synthesized thorough a fermentation of Bacillus subtilis species mainly Bacillus subtilis natto (Berenjian et al. 2015; Mahanama et al. 2011, 2012). However, this valuable compound is not readily available due to the presence of several tedious and inefficient unit operations which has resulted a current price of US$5 million/kg (Wohlgemuth 2009; Berenjian et al. 2015). Process intensification as a method for making significant changes in the bioprocess plant can be an alternative approach to address the current challenges in MK-7 fermentation. These reductions can come from decreasing the size of individual equipment or from removing the number of involved unit operations (Marques and Fernandes 2011; Vaghari et al. 2015).
A bioreactor has a key role in fermentation process, as it dictates both the product quality and the extent of the downstream requirements. Designing intensified bioreactors that can integrate in situ cell recovery and product formation will permit us to deliver a high-quality product without an extensive downstream purification sequence. Cell immobilization is one of the promising and most applied techniques for intensification of downstream processes. As compared to centrifugation, this approach has significant advantages such as simple equipment requirement and low energy consumption while allowing for the reusability of cells. The common immobilization techniques are based on imbedding the cells in a polymeric matrix like calcium alginate. This matrix acts as a barrier for mass transfer and provides cells with a different microenvironment as compared to fermentation broth.
Iron oxide nanoparticles (IONs) have been used for bacterial cell immobilization and separation (Liu et al. 2004). The surface of bacterial cells can be simply decorated with IONs via nonspecific forces such as hydrogen bonds and electrostatic ore hydrophobic interactions (Li et al. 2009; Chwalibog et al. 2010; Huang et al. 2010). Decorated bacteria show a significant response to magnetic field and easily can be separated by applying a magnetic field (Ansari et al. 2009; Li et al. 2009; Chwalibog et al. 2010; Huang et al. 2010). In addition, immobilization with magnetic nanoparticles would not inhibit the mass transfer around microorganisms and combines the advantages of cell immobilization with those of free cell fermentation (Ansari et al. 2009).
Despite the considerable potential benefits, application of IONs in fermentation technology is associated with considerations. To date, research about the effects of magnetic IONs on a variety of microbial cells have been controversial and sometimes contradictory. Many of the previous studies revealed that IONs can interrupt the integrity of cell membrane and adversely affect normal cell functionalities (Berry et al. 2004; Liu et al. 2004; Can et al. 2009). Contrary, some studies have reported that IONs at certain concentrations show no negative toxic or inhibitory effects on some microbial cells and also there are growth stimulatory effects (Grumezescu et al. 2010; Ficai et al. 2012; Islam et al. 2012; Gholami et al. 2015). By reviewing the literature, it seems that the effect of IONs on the growth and metabolic profile of microorganisms depends on the cell type. Hence, evaluating the potential toxicity and the effect of IONs on specific microbial cells is critical in order to establish their performance for designing an intensified biological process. The present work, therefore, was designed to study the potential effect of magnetic immobilization on Bacillus subtilis natto cells growth, MK-7 production and the possibility of in situ product recovery and cell recycling as compared to untreated and free floating fermentation conditions.
Materials and methods
Synthesis of IONs
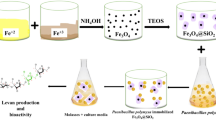
IONs were synthesized by co-precipitation of ferric and ferrous ions with ammonium hydroxide under inert atmosphere. In brief, FeSO4⋅4H2O (0.6 g, 2.2 mmol) and FeCl3⋅6H2O (1.17 g, 3.8 mmol) were dissolved in 50 mL degassed distilled water and the solution was stirred at 70 °C under a nitrogen atmosphere. After 60 min, 5 mL ammonium hydroxide solution (32 %) was quickly added until the target pH (11) was reached. The solution was stirred for another 60 min. Black precipitate was separated by a permanent magnet, washed by hot distilled water several times, and dried overnight in an oven at 50 °C.
Immobilization of bacterial cells with IONs and fermentation
B. subtilis natto (ATCC 6633) were cultured in tryptic soy broth, and cells were harvested and washed with normal saline. The cells were suspended in normal saline and mixed with various concentrations of IONs. The mixtures were incubated in a shaker incubator (150 rpm, 37 °C) for attachment of nanoparticles to the cell surface. After incubation for 15 min, immobilized cells were transferred to fermentation media consisting of 1 % (w/v) yeast extract, 5 % (w/v) glycerol, and 1.5 % (w/v) soy peptone. All the fermentation experiments were conducted at 40 °C for a period of 5 days (Berenjian et al. 2011). A magnetic field (neodymium magnet: 800 gauss) was used for cell separation process studies. Statistical significant was determined by analysis of variance (ANOVA) and was accepted at p < 0.05.
Cell fixation procedures
The bacterial cells suspension was washed with normal saline, and a thin smear was prepared on a glass slide. In order to heat fix the bacterial smear, the air dried slides were passed through the flame of a Bunsen burner four times. The bacterial cells were fixed in 2.5 % glutaraldehyde in normal saline for 45 min. For rinsing, the slides were put in normal saline for 15 min. Cell dehydration was conducted by putting the slides in ethanol using serial concentrations of 30, 50, 70, 80 and 90 % (10 min each). Consequently, prepared samples were left in absolute ethanol for 20 min. The slides were stored at room temperature over night to completely evaporate the ethanol.
MK-7 extraction procedures
MK-7 was extracted from the fermentation media by using a mixture of 2-propanol and n-hexane with the ratios of (1/2, v/v) and 1/4 (liquid/organic, v/v) (Berenjian et al. 2014). In each experiment after the addition of organic solution, the sample was vigorously shaken with a vortex, followed by centrifugation at 3000 rpm for 10 min afterwards. The organic layer was then collected from the aqueous layer to recover the extracted MK-7.
Analytical methods
Transmission electron microscopy (TEM, Philips, CM 10; HT 100 kV), Fourier-transformed infrared spectroscopy (FTIR, Bruker, Vertex 70, FT-IR spectrometer), differential scanning calorimetry (DSC, Thermoanalyser DSC 302), vibrating sample magnetometer and X-ray powder diffraction spectroscopy (Siemens D5000) were used for characterization of prepared nanoparticles. Interactions of IONs with bacterial cell surface were visualized using scanning electron microscopy (SEM, Hitachi S-4700). High-performance liquid chromatography (HPLC, Waters Co., USA) with a photon diode array UV detector was used for the analysis of MK-7 concentration. Samples were separated by C18 Gemini column (5 μm, 250 × 4.6 mm, Phenomenex Co., USA) at 40 °C. The mobile phase consisted of methanol that was used at a flow rate of 1 mL/min. The statistical significant was determined by analysis of variance (ANOVA) test. Statistical significance was accepted at p < 0.05, and data were reported as mean ± standard deviation (SD) of at least three experiments.
Results
Synthesis and characterization of IONs
Synthesis of IONs was conducted in aqueous medium by co-precipitation reaction of Fe (II) and Fe (III). In this method, upon the addition of ammonium hydroxide to the iron salts, a sudden colour change to dark black was observed. This rapid colour change shows the successful formation of iron oxide cores in the solution. Larger magnetite nanoparticles were grown by further continuing the reaction while mixing the solution. Figure 1 shows the TEM micrograph of the prepared IONs. Particles size analyses were conducted using ImageJ software version 1.47v, an image analysis software developed by the NIH (http://imagejnihgov/ij/). The particles size distribution was measured to be from 7 to 20 nm with the mean particle size of 11 nm.
The FTIR spectrum of IONs is presented in Fig. 2 which shows the characteristic peaks of Fe-O at about 644 and 450/cm. In a co-precipitation reaction, the surface of the magnetite nanoparticles is modified by OH groups from aqueous medium. This phenomenon is due to the coordination of unsaturated surface iron atoms with water molecules as well as hydroxyl ions (Ebrahiminezhad et al. 2012). The OH groups absorb infrared waves at 3434/cm (stretching point) and 1629/cm (deforming point) (Ebrahiminezhad et al. 2012, 2013; Islam et al. 2012; Wei Wang et al. 2012).
DSC curve of IONs (Fig. 3) shows endothermic peak at 176.5 °C. This peak was resulted from oxidation and change in crystallinity of iron oxide crystals. Based on saturation magnetisation analysis (Fig. 4), magnetization curve was completely reversible showing the superparamagnetic behaviour of the prepared particles, and the saturation magnetization value was found to be 60 emu/g. This behaviour is so critical for cells separation by applying a magnetic field and again cells dispersion by magnetic field removal. X-ray power diffraction pattern of the produced nanoparticles was shown intensity peaks at 2θ degrees of 30, 35.5, 43, 57 and 63 (Fig. 5). The peaks of the synthesized nanoparticles were compared with the standard data for magnetite crystals. These peaks correspond to (220), (311), (400), (422), (511) and (440) Bragg reflections which are in complete agreement with the standard magnetite confirming the formation of Fe3O4 (Zhao et al. 2006; Ebrahiminezhad et al. 2014, 2015).
Visualization of IONs interactions with bacterial cell surface
The initial step in interaction between bacterial surface and an artificial solid surface is governed mainly by long-range van der Waals and electrostatic interactions between the solid surface and the bacterial cell. While van der Waals forces are generally attractive; the local charges on the bacteria and solid surfaces can lead to electrostatic repulsion or attraction (Jucker et al. 1996). Bacterial cells usually have a strain-dependent net negative charge on the cell wall, and the magnitude of this negative charge varies from strain to strain (Dickson and Koohmaraie 1989). The surface of the bacterial cells contains both positive and negative charges in different magnitudes. Therefore, there would be both attraction and repulsion forces for interaction with a particular surface (Dickson and Koohmaraie 1989).
Islands with various charges in bacterial cell wall provide possibility for interactions with surfaces having even a negative charge. There are some evidence for decoration and immobilization of bacterial cells with IONs with negative zeta potential (Li et al. 2009). However, the efficiency of the magnetic immobilization techniques can be influenced by the rate of bacterial attachment to the applied IONs. The cell surfaces of B. subtilis natto is covered by extracellular polymeric substances (EPS), which contain abundant functional groups such as carboxyl, hydroxyl and phosphate (Soneshein et al. 2002). These functional groups contribute to the robust attachment between IONs and bacteria. Once nanoparticles are mixed with bacterial cells, the IONs could be easily attached onto the surfaces of bacterial cells using hydrogen bonds and nonspecific forces such as hydrophobic or electrostatic interactions (Larsen et al. 2009; Bromberg et al. 2011). Figure 6 is illustrating successful decoration of B. subtilis natto cell surface with IONs as compared to untreated sample. The sizes of produced IONs were about several orders of magnitude smaller than bacterial cells, and the high surface/volume ratio of nanoparticles provided promising surface area for attachment onto the bacterial cell wall and consequently bacterial capture. Due to nonspecific interactions between B. subtilis natto cell wall and IONs, the IONs are randomly attached on to the bacterial surface. Unfortunately, this interaction is not uniform among all the cells, and one can see heavily decorated cells beside some cells with weak decoration (Fig. 6).
Effect of IONs on bacterial growth and MK-7 production
A comparative study on the growth of magnetic immobilized and free cells of B. subtilis natto was conducted. As can be depicted from Fig. 7, bacterial growth was affected by the presence of nanoparticles in a time-dependent manner. Increasing the nanoparticles concentration from 50 to 150 \( \mu \)g/mL resulted in no significant difference on the cell growth (p > 0.05). However, as compared to free-floating bacteria, attachment of the IONs on bacterial cell surface resulted in approximately 5 % growth inhibition by the third day of fermentation. This inhibition was more prefunded by the end of fermentation (p < 0.05). The cell density reached 1.2 × 1011 CFU/mL by the end of fermentation for untreated samples. This value was approximately 10 % higher than the immobilized cells. Moreover, further increase in IONs concentration (more than 150 μg/mL) resulted in sudden agglomeration and sedimentation of bacterial cells and nanoparticles. Therefore, the maximum applicable concentration for IONs was chosen to be 150 μg/mL.
However, magnetic immobilization with IONs showed no negative effect on MK-7 production. As shown in Fig. 8, the presence of different IONs concentrations (50–150 μg/L) did not affect MK-7 production as compared to the control (p > 0.05). MK-7 production was measured to be about 12 ± 0.7 mg/L by the last day of fermentation. Evaluation of MK-7 production under treatment with magnetic nanoparticles revealed 99 % similarity in the total MK-7 production among the immobilized samples.
Further investigation on MK-7 specific yield (Eq. 1), showed a significantly higher specific yield while using IONs as compared to untreated and free-floating samples at the end of the fermentation period (p < 0.05). However, there was no significant specific yield difference among the studied samples on day 3. As can be depicted from Fig. 9, MK-7 specific yield was affected by the presence of nanoparticles in a concentration dependent manner by the end of the fermentation. The highest MK-7 specific yield of 5.14 was reached while using 150 μg/L IONs. Moreover, the presence of 50 μg/L IONs resulted in the lowest MK-7 specific yield (5.03); this specific yield was still 8 % higher as compared to untreated samples.
Cells separation and recovery
The effects of IONs concentration on bacteria capture were examined by using B. subtilis natto. The prepared IONs were successfully attached onto the surfaces of bacterial cell wall. The formed decorated cells were removed from the fermentation media in several minutes with the aid of an external magnetic field (800 gauss). The cell capture efficiencies were increased by enhancing IONs dosages. The capture efficiency for 50 μg/L IONs was only 85 % after 30 min. When the dosage of IONs was increased to 100 μg/L, the capture efficiency was improved to 89 %. However, the highest bacterial removal was achieved in the presence of 150 μg/L over the other examined concentrations. This phenomenon can be justified by the presence of more magnetic particles on the bacterial surface. Furthermore, increasing the separation time from 30 min to 2 h showed no significant difference on the capture efficiency of decorated cells (p > 0.05). The cell capture efficiencies were slightly increased to 87, 90 and 97 %, respectively, in IONs dose dependent manner.
Following the magnetic field separation, the suspended particles were washed once with sterile deionized water and were reused. The particle suspension was continuously pumped through the closed loop at a rate of 1.5 L/min. For all studied IONs concentrations, the bacteria capture efficiencies slightly decreased with increasing the regeneration cycles. The cell capture efficiencies were more than 85 % for all IONs concentrations in the first cycle, whereas the capture efficiencies in the second cycles decreased to 76, 83 and 87 %, respectively. Additionally, MK-7 production were 9.8 ± 0.81, 10.06 ± 0.11 and 10.43 ± 0.21 mg/L for the cells decorated with 50, 100 and 150 μg/mL IONs, respectively. These results point out that although the increasing bacterial capture cycles slightly decreased the capture efficiency, yet, pronounced number of cells could be captured. This observation indicated that IONs decorated B. subtilis natto cells exhibited good reusability and high MK-7 specific yield. Magnetic separation technology is scalable and can be easily integrated in a recycle loop in a bioreactor to achieve a rapid recovery of bacterial clusters. Intensified bioprocess by integrating MK-7 formation and B. subtilis natto recovery can be achieved by the use of magnetized system in order to reduce the number of downstream steps and enhance the efficacy of the process.
Discussions
Various microorganisms show different susceptibilities to IONs, but the exact controlling mechanism to the toxicity is not yet understood. Release of free iron from the IONs could catalyse production of reactive oxygen species (ROS) in the Fenton’s reaction (Ebrahiminezhad et al. 2012). ROS disturb the balance between oxidative pressure and antioxidant defence of cells and cause membrane lipid peroxidation, DNA breakage, protein oxidation and give rise to necrosis and apoptosis (Muller et al. 2007; Karlsson et al. 2009; Singh et al. 2010; Ebrahiminezhad et al. 2012). On the other hand, some investigations have purposed that iron ions can be used as a valuable source for growth promotion (Ebrahiminezhad et al. 2015). This phenomenon is a nanoparticle-specific mechanism which is due to stress or stimuli caused by the surface, size and shape of the particles (Brunner et al. 2006). The nonspecific interactions of IONs with cell membrane compounds have been reported to result in disorganization of lipid packing in the microbial membrane. Such an effect may reduce the transport selectivity of membranes (Epand et al. 2008; Ansari et al. 2009). Bacterial cells were also found to be clogged in between the IONs because of the magnetic property of the particles, while trapped cells exhibited a significant change in shape and metabolic activity (Chatterjee et al. 2011). It is apparent that different factors such as size, shape and composition of nanoparticles can lead to different conclusions (Martin et al. 1994).
Some investigations have been reported a growth inhibitory effect of IONs on bacterial strains including Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli and Listeria monocytogenes (Grumezescu et al. 2010; Ramteke et al. 2010; Chatterjee et al. 2011; Ebrahiminezhad et al. 2012). Meanwhile, there is evidence that IONs exhibited a dose-dependent growth promotion effect in case of Klebsiella pneumoniae, Pseudomonas aeruginosa, Enterococcus faecalis and Candida albicans and even on human cell lines (Grumezescu et al. 2010; Ficai, et al. 2012; Islam et al. 2012; Ebrahiminezhad et al. 2015). It seems that the effect of IONs on the growth and metabolic profile of microorganisms depends on the cells’ physiology. The possible explanation of this phenomenon might be the different cell membrane interactions with the nanoparticles (Ebrahiminezhad et al. 2015).
Based on the results of the present study decoration of B. subtilis natto, cells with IONs reduced the bacterial growth rate. Whereas, total MK-7 production yields were significantly higher for magnetic immobilized cells as compared to free B. subtilis natto cells. Based on the results, the highest MK-7 specific yield and separation efficiency were achieved in the presence of 150 μg/L of IONs as compared to the other investigated concentrations. It seems that higher cell growth may not be necessarily required for the optimum MK-7 production. This study shows that the immobilization of B. subtilis natto cells by IONs has a profound effect on MK-7 specific yield and biosynthesis. This improved metabolic activity is mainly resulted by the interaction between nanoparticles and bacterial cell surface which are governed by surface properties of particles such as their chemistry, hydrophilic/hydrophobic characteristics and surface charge. It has been reported that decoration of bacterial cells with IONs makes the cells more metabolically efficient (Ansari et al. 2009). A possible mechanism for this enhancement is that the bounded nanoparticles to the bacterial surface make the cell membranes more permeable to facilitate the mass transfer. The nonspecific interaction of IONs with membrane compounds may result in disorganization of lipid packing and consequently enhance the membrane permeability (Epand et al. 2008; Ansari et al. 2009).
Process integration such as in situ cell and product removal has emerged as a valuable tool to increase the overall process yield and aims at minimizing the process costs. This study describes the possibility of separation of microbial cells from the bioreactor media by the use of IONs in order to minimize the MK-7 production limitations. Magnetic immobilization with IONs showed a positive effect on MK-7 biosynthesis. Meanwhile, specific yield of MK-7 production was significantly improved in the immobilized cells as compared to untreated cells. Moreover, decoration of B. subtilis natto cells with IONs resulted in significant bacterial capture ability with more than 85 % efficiency. High gradient magnetic separation technology is scalable and can be integrated in a recycle loop of bioreactor to achieve rapid cell recovery. In conclusion, the above results demonstrate that IONs can be applied during the MK-7 fermentation for high efficient bacterial cells recovery without any reduction in the production yield. Indeed, this study might be of considerable interest to industrial MK-7 fermentation to address the current production challenges.
References
Ansari F, Grigoriev P, Libor S, Tothill IE, Ramsden JJ (2009) DBT degradation enhancement by decorating Rhodococcus erythropolis IGST8 with magnetic Fe3O4 nanoparticles. Biotechnol Bioeng 102:1505–1512
Berenjian A, Mahanama R, Talbot A, Biffin R, Regtop H, Valtchev P, Kavanagh J, Dehghani F (2011) Efficient media for high menaquinone-7 production: response surface methodology approach. N Biotechnol 28:665--672
Berenjian A, Mahanama R, Talbot A, Regtop H, Kavanagh J, Dehghani F (2014) Designing of an intensification process for biosynthesis and recovery of menaquinone-7. Appl Biochem Biotechnol 172:1347–1357
Berenjian A, Mahanama R, Kavanagh J, Dehghani F (2015) Vitamin K series: current status and future prospects. Crit Rev Biotechnol 35:199–208
Berry CC, Wells S, Charles S, Aitchison G, Curtis ASG (2004) Cell response to dextran-derivatised iron oxide nanoparticles post internalisation. Biomaterials 25:5405–5413
Bromberg L, Chang EP, Hatton TA, Concheiro A, Magariños B, Alvarez-Lorenzo C (2011) Bactericidal core-shell paramagnetic nanoparticles functionalized with poly(hexamethylene biguanide). Langmuir 27:420–429
Brunner TJ, Wick P, Manser P, Spohn P, Grass RN, Limbach LK, Bruinink A, Stark WJ (2006) In vitro cytotoxicity of oxide nanoparticles: comparison to asbestos, silica, and the effect of particle solubility. Environ Sci Technol 40:4374–4381
Can K, Ozmen M, Ersoz M (2009) Immobilization of albumin on aminosilane modified superparamagnetic magnetite nanoparticles and its characterization. Colloids Surf B Biointerfaces 71:154–159
Chatterjee S, Bandyopadhyay A, Sarkar K (2011) Effect of iron oxide and gold nanoparticles on bacterial growth leading towards biological application. J Nanobiotechnology 9:34
Chwalibog A, Sawosz E, Hotowy A, Szeliga J, Mitura S, Mitura K, Grodzik M, Orlowski P, Sokolowska A (2010) Visualization of interaction between inorganic nanoparticles and bacteria or fungi. Int J Nanomedicine 5:1085–1094
Dickson JS, Koohmaraie M (1989) Cell surface charge characteristics and their relationship to bacterial attachment to meat surfaces. Appl Environ Microbiol 55:832–836
Ebrahiminezhad A, Davaran S, Rasoul-Amini S, Barar J, Moghadam M, Ghasemi Y (2012) Synthesis, characterization and anti-listeria monocytogenes effect of amino acid coated magnetite nanoparticles. Curr Nanosci 8:868–874
Ebrahiminezhad A, Ghasemi Y, Rasoul-Amini S, Barar J, Davaran S (2013) Preparation of novel magnetic fluorescent nanoparticles using amino acids. Colloids Surf B Biointerfaces 102:534–539
Ebrahiminezhad A, Rasoul-Amini S, Davaran S, Barar J, Ghasemi Y (2014) Impacts of iron oxide nanoparticles on the invasion power of Listeria monocytogenes. Curr Nanosci 10:382–388
Ebrahiminezhad A, Rasoul-Amini S, Kouhpayeh A, Davaran S, Barar J, Ghasemi Y (2015) Impacts of amine functionalized iron oxide nanoparticles on HepG2 cell line. Curr Nanosci 11:113–119
Epand RM, Epand RF, Savage PB (2008) Ceragenins (cationic steroid compounds), a novel class of antimicrobial agents. Drug News Perspect 21:307–311
Ficai D, Andronescu E, Ficai A, Voicu G, Vasile B, Ionita V, Guran C (2012) Synthesis and characterization of mesoporous magnetite based nanoparticles. Curr Nanosci 8:875–879
Gholami A, Rasoul-Amini S, Ebrahiminezhad A, Seradj SH, Ghasemi Y (2015) Lipoamino acid coated superparamagnetic iron oxide nanoparticles concentration and time dependently enhanced growth of human hepatocarcinoma cell line (Hep-G2). J Nanomater 45:1–9
Grumezescu AM, Mihaiescu DE, Mogosanu DE, Chifiriuc MC, Lazar V, Calugarescu I, Traistaru V (2010) In vitro assay of the antimicrobial activity of Fe3O4 and CoFe2O4/oleic acid—core/shell on clinical isolates of bacterial and fungal strains. Optoelectron Adv Mat 4:1798–1801
Huang YF, Wang YF, Yan XP (2010) Amine-functionalized magnetic nanoparticles for rapid capture and removal of bacterial pathogens. Environ Sci Technol 44:7908–7913
Islam S, Kusumoto Y, Abdulla-Al-Mamun M, Manaka H, Horie Y (2012) Synthesis, characterization and application of Dumbbell-shaped magnetic (Fe3O4 and-Fe2O3) nanoparticles against HeLa (cancer) cells. Curr Nanosci 8:811–818
Jucker BA, Harms H, Zehnder A (1996) Adhesion of the positively charged bacterium Stenotrophomonas (Xanthomonas) maltophilia 70401 to glass and Teflon. J Bacteriology 178:5472–5479
Karlsson HL, Gustafsson J, Cronholm P, Möller L (2009) Size-dependent toxicity of metal oxide particles—a comparison between nano- and micrometer size. Toxicol Lett 188:112–118
Larsen MU, Seward M, Tripathi A, Shapley NC (2009) Biocompatible nanoparticles trigger rapid bacteria clustering. Biotechnol Prog 25:1094–1102
Li YG, Gao HS, Li WL, Xing JM, Liu HZ (2009) In situ magnetic separation and immobilization of dibenzothiophene-desulfurizing bacteria. Bioresour Technol 100:5092–5096
Liu X, Guan Y, Yang Y, Ma Z, Wu X, Liu H (2004) Preparation of superparamagnetic immunomicrospheres and application for antibody purification. J Appl Polym Sci 94:2205–2211
Mahanama R, Berenjian A, Valtchev P, Talbot A, Biffin R, Regtop H, Dehghani F, Kavanagh JM (2011) Enhanced production of menaquinone 7 via solid substrate fermentation from Bacillus subtilis. Int J Food Eng 7:5
Mahanama R, Berenjian A, Regtop H, Talbot A, Dehghani F, Kavanagh JM (2012) Modeling menaquinone 7 production in tray type solid state fermenter. ANZIAM J 53:354–372
Marques MPC, Fernandes P (2011) Microfluidic devices: useful tools for bioprocess intensification. Molecules 16:8368–8401
Martin ST, Morrison CL, Hoffmann MR (1994) Photochemical mechanism of size-quantized vanadium-doped TiO2 particles. J Phys Chem 98:13695–13704
Muller K, Skepper JN, Posfai M, Trivedi R, Howarth S, Corot C, Lancelot E, Thompson PW, Brown AP, Gillard JH (2007) Effect of ultrasmall superparamagnetic iron oxide nanoparticles (ferumoxtran-10) on human monocyte-macrophages in vitro. Biomaterials 28:1629–1642
Ramteke C, Sarangi BK, Chakrabarti T, Mudliar S, Satpute D, Pandey RA (2010) Synthesis and broad spectrum antibacterial activity of magnetite ferrofluid. Curr Nanosci 6:587–591
Singh N, Jenkins GJ, Asadi R, Doak SH (2010) Potential toxicity of superparamagnetic iron oxide nanoparticles (SPION). Nano Rev 1:5358
Soneshein AL, Hoch JA, Losick R (2002) Bacillus subtilis and its closest relatives. ASM Press, Washington DC
Vaghari H, Eskandari M, Sobhani V, Berenjian A, Song Y, Jafarizadeh-Malmiri H (2015) Process intensification for production and recovery of biological products. Am J Biochem Biotech 11:37–43
Wei Wang X, Cheng ZH, Li JS, Yuan ZH (2012) Controllable synthesis and magnetic properties of ferromagnetic nanowires and nanotubes. Curr Nanosci 8:801–809
Wohlgemuth R (2009) The locks and keys to industrial biotechnology. N Biotechnol 25(4):204–213
Zhao SY, Don KL, Chang WK, Hyun GC, Young HK, Young SK (2006) Synthesis of magnetic nanoparticles of Fe3O4 and CoFe2O4 and their surface modification by surfactant adsorption. Bull Korean Chem Soc 27:237–242
Acknowledgments
This investigation was financially supported by Iran National Science Foundation and The University of Waikato, New Zealand.
Compliance with ethical standards
ᅟ
Conflict of interest
The authors declare that they have no competing interests.
Ethics
The article is original and has not been formally published in any other peer-reviewed journal and does not infringe any existing copyright and any other third party rights.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ebrahiminezhad, A., Varma, V., Yang, S. et al. Magnetic immobilization of Bacillus subtilis natto cells for menaquinone-7 fermentation. Appl Microbiol Biotechnol 100, 173–180 (2016). https://doi.org/10.1007/s00253-015-6977-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6977-3