Abstract
We studied potential denitrification activity and the underlying denitrifier communities in soils from a semiarid savanna ecosystem of the Kavango region in NE Namibia to help in predicting future changes in N2O emissions due to continuing changes of land use in this region. Soil type and land use (pristine, fallow, and cultivated soils) influenced physicochemical characteristics of the soils that are relevant to denitrification activity and N2O fluxes from soils and affected potential denitrification activity. Potential denitrification activity was assessed by using the denitrifier enzyme activity (DEA) assay as a proxy for denitrification activity in the soil. Soil type and land use influenced C and N contents of the soils. Pristine soils that had never been cultivated had a particularly high C content. Cultivation reduced soil C content and the abundance of denitrifiers and changed the composition of the denitrifier communities. DEA was strongly and positively correlated with soil C content and was higher in pristine than in fallow or recently cultivated soils. Soil type and the composition of both the nirK- and nirS-type denitrifier communities also influenced DEA. In contrast, other soil characteristics like N content, C:N ratio, and pH did not predict DEA. These findings suggest that due to greater availability of soil organic matter, and hence a more effective N cycling, the natural semiarid grasslands emit more N2O than managed lands in Namibia.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Emissions of N2O are a global concern, because the gas contributes to the degradation of ozone in the stratosphere [1]. Moreover, because each N2O molecule traps 300 times more radiant energy per century than a CO2 molecule, N2O contributes considerably to global climate change [2]. In temperate ecosystems, managed soils are the major sources of atmospheric N2O [3–5], because the greater availability of organic substrates in soils under agriculture drives relatively high N transformation rates [6]. Temperate soils and their responses to changes in land use, for instance the conversion of grasslands into crop fields or pastures grazed by animals, are well-studied [4, 7].
In contrast, far less is known about how soils in semiarid ecosystems may respond to changes in land use. This makes predicting how the continuing conversion of pristine savannas into managed lands will affect the contribution of these regions to global greenhouse gas emissions difficult. On a global scale, savannas, and seasonally dry ecosystems in warm climatic regions, cover a land surface of more than 30 × 106 km2. Hence, despite low average daily fluxes of 0.32 mg N2O m−2 [8], these regions represent a significant source of atmospheric N2O (4.4 Tg N2O year−1) and savanna soils contribute 25 % of N2O fluxes from tropical soils [3]. In Africa, savanna ecosystems cover a large part of the continent’s land mass and host the majority of the population. In 2005, Africa contributed only 16 % to total anthropogenic N2O emissions, with agriculture accounting for 42 % (71 % if burning is included) of the African N2O emissions [9]. This low level was attributed to extremely low inorganic fertilizer inputs into most of the sub-Saharan African soils [10]. However, N2O emissions have more than doubled in Africa from 1970 to 2005 and are predicted to double again until 2050 due to the prospected green revolution and the associated changes in land use and land management [9].
To study the effects of land use, climate variations, and other habitat influences on savanna biodiversity and ecosystem functioning and services, the Biota Southern Africa project was launched in 2001. This research project concentrates on studies in Namibia and the western parts of South Africa and aims at providing scientific support for conservation and sustainable use of biodiversity in Namibia and South Africa. Our research focused on soil ecosystems in the Kavango region in northern Namibia and the likely consequences of these systems’ ongoing conversion to agricultural land. In the Kavango region, the cropping area increased by 3.9 % per year because of clearing of natural vegetation from 1943 to 1996 [11]. Previous studies of semiarid ecosystems explored the effects of changes in land use on changes in N2O emissions from soils in response to biotic, abiotic, and seasonal influences [12, 13]. In woodland and managed savannas, similar fluxes of N2O were observed [8], but converting natural systems to agricultural use generally increases greenhouse gas release. For instance, clearing of humid primary forest in Costa Rica increased N2O production, probably due to an increase in N cycling and higher diffusion rates in drying soils [14]. Further, compaction due to trampling by cattle, and fertilizer use dramatically increased N2O release from tropical grasslands [4, 15].
We hypothesized that land use change in the Kavango region would affect the microbial communities involved in denitrification in the soils with feedback effects on N2O fluxes in situ. Denitrifiers and nitrifiers are the main producers of N2O from soils [16]. Even in arid and semiarid soil ecosystems, where bulk soils are generally aerobic, denitrification accounts for a significant fraction of N lost from the soils and drives N2O emissions [17–19]. We used the denitrifier enzyme activity (DEA) assay as a proxy for N2O production by denitrification in the field and linked these data to the abundance and composition of denitrifier communities in different soil types subjected to different types of land use (pristine, currently under cultivation, fallow). Pristine, more C-rich soils showed a higher propensity to produce N2O than nutrient-depleted, managed soils, which was related to the abundance and composition of the underlying denitrifier communities.
Material and Methods
Site Characteristics and Soil Sampling
For this study within the “BIOTA Biodiversity Observatories in Africa” framework [20], the Mile 46 Livestock Development Centre (LDC) in the Kavango region in northeast Namibia was chosen. The Kavango region is dominated by semiarid savannas with ferralic arenosol as the prevalent soil type. Many of these soils are poor, weakly developed with reduced soil horizonation, and generally not very productive due to limited fertilizer inputs [21]. The majority of the Namibian population (70 %) depends on subsistence farming by rain-fed and dryland cropping [22]. Soil samples were collected between March 19 and 24, 2007 in the neighborhood of Mutompo village (18.30 S; 19.25 E; 1180 m NN) in the Kavango region of northeastern Namibia (Fig. S1). The climate is semiarid with a mean temperature of about 22.6 °C and an average rainfall of 527 mm year-1 which occurs primarily from November to March [23]. The soil parent material is formed by degraded dune sands of the Kalahari basin, and soil types are mainly dystric to hypereutric arenosols [24]. The region is characterized by forest savanna with northern Kalahari dry woodlands as the main vegetation type [25]. Common plant species in Mutompo are Combretum species, Terminalia sericea, Burkea africana, Eragrostis echinochloides, and Guibourtia coleosperma [23, 26]. Detailed descriptions of the vegetation of the Kavango region can be found in Strohbach and Petersen [27]. Land use of the area started with the construction of drinking water wells in the 1970s. The communities use the land for rain-fed arable fields, for open-access cattle grazing with little control over stocking rates and grazing times, and for firewood collection [28].
Twenty-nine plots (Table 1, Table S2) were chosen representing combinations of land use (P: pristine, Fa: fallow for up to 10 years, and Fi: field) and topsoil type (LD = dark and loamy, BR = brown-reddish and slightly loamy, and PS = pale and pure sandy), according to the Munsell notation [29]. Due to the homogenous parent material, the darkness of the soil is an indicator for the nutritional value of the soils [30]. One sample (P/PS-15) was excluded from all statistical analyses, because repeated extractions did not yield PCR-amplifiable DNA. Soil in situ temperature upon sampling was 30–31 °C. After removing mulch from the surface, soil samples from the surface layer (0–10 cm; diameter 5 cm) were taken within a plot of 20 by 20 m. Nine soil cores each (i.e., 18 cores in total) were collected at regular distances along two intersecting, rectangular sampling transects. Samples were pooled per plot, homogenized, and sieved through a 2-mm mesh to remove large roots and pebbles. In the field, each soil homogenate was divided into two fractions (2 g into cryovials and the remainder into sterile plastic bags) and transported to the laboratory at the nearby Mile 46 Livestock Development Centre within a few hours. The cryovials were frozen and transported under liquid nitrogen to the laboratory in Germany, whereas the bags containing the remaining soil were kept at 4 °C and also transported to Germany.
Determination of Soil Water Content, pH, and Nitrate Concentrations
Soil water content (weight %) was determined gravimetrically. Soil (5–10 g) was weighed, dried at 105 °C overnight, and weighed again. To determine pH, an amount of soil equivalent to 1 g dry weight was suspended in 5 ml 0.01 M CaCl2 solution. The soil slurries were incubated for 2 h at room temperature before pH was measured with a pH meter (Sentix 41; WTW, Weilheim, Germany). Soil electrical conductivity was determined in the laboratory in Hamburg (Germany) in a slurry (10 g soil/25 ml water) with a conductivity sensor (WTW).
Nitrate concentrations in soil were determined after extraction from an amount of soil equivalent to 2 g dry weight of soil in 10 ml bi-distilled water and shaking the suspension at 150 rpm for 1 h at 4 °C. Afterwards, the soil suspension was filtered (round filter 2095, Schleicher & Schuell GmbH, Dassel, Germany) and nitrate was measured with an ion chromatograph (IC, Sykam, Fürstenfeldbruck). Data were evaluated with the PeakSimple software (version 3.54, SRI Instruments, Torrence, CA, USA).
Denitrifier Enzyme Activity Assay
The DEA assay [31] measures the abundance of denitrification enzymes and allows comparison of the potential denitrification rates across samples. Aliquots equal to 3 g dry weight of soil were placed in serum bottles to which 3 ml of a substrate solution containing glucose, potassium nitrate (both 10 mM), and chloramphenicol (0.1 g l−1) were added. The serum bottles were capped with butyl stoppers and oxygen was removed by flushing the bottles with N2. Reduction of N2O to N2 was blocked by adding acetylene to 10 % of the gas phase. All experiments were set up in triplicates from the homogenized material and were incubated at 25 °C on a horizontal shaker at 200 rpm. After flushing the syringe with N2 to prevent contamination of the gas phase with oxygen, head-space samples of 0.5 ml were collected every 60 min. Nitrous oxide was analyzed by gas chromatography (GC) with a 63Ni electron capture detector (ECD; Carlo Erba GC 8000; Fisons Instruments, Mainz-Kastel, Germany). N2O production rates per hour were inferred from the period of linear increase in N2O concentration.
DNA Extraction
Total DNA was extracted from 0.5 g frozen soil using the FAST DNA® Spin Kit for Soil Kit according to the manufacturer’s instructions (MP Biomedicals Germany, Heidelberg, Germany). Additional washing (3×) of the DNA with 5.5 M guanidine thiocyanate (pH 7.0) was introduced as additional cleaning to reduce the amount of humic substances in the DNA preparation [32]. Subsequently, the purity and quantity of the DNA were determined by UV spectrophotometry at 260 and 280 nm (Biophotometer, Eppendorf, Hamburg, Germany) and using the Quant-iT™ PicoGreen® dsDNA Assay Kit (Invitrogen GmbH, Karlsruhe, Germany).
T-RFLP Analysis
PCR amplifications of nirS and nirK genes from total environmental DNA extracts were performed in a total volume of 25 μl using the primer pairs cd3aF-R3cd [33, 34] and nirK1F-nirK5R [35] and PCR conditions described previously [34, 36]. Both primer pairs exclude detection of ammonia oxidizer species. The reverse primers R3cd and nirK5R were 5′-end labeled with 6-carboxyfluorescein (Sigma-Aldrich GmbH, Taufkirchen, Germany). Replicate PCRs (5) were combined and analyzed by electrophoresis on 1.5 % [w/v (weight in volume)] agarose gels followed by staining with ethidium bromide (0.5 mg l−1). Bands were visualized by UV excitation. For purification, fragments of proper size were excised from the agarose gel and eluted using the Wizard® SV Gel and PCR Clean-Up System (Promega GmbH, Mannheim, Germany).
Aliquots of the PCR product (50–100 ng DNA) were cleaved with 3 U of restriction endonuclease (MspI for nirS and HaeIII for nirK; New England Biolabs, Frankfurt/Main, Germany). After cleavage, enzymes and excess salts were removed from the digest with Autoseq G-50 columns (Amersham-Pharmacia Biotech, Freiburg, Germany). Aliquots (2 μl) were mixed with 11 μl of deionized formamide (Applera, Darmstadt, Germany) and 0.3 μM of an internal DNA fragment length standard (X-Rhodamine MapMarker® 30–1000 bp; BioVentures, Murfreesboro, TN). Restriction fragments were separated on an automated DNA sequencer (ABI PRISM® 3130xl Genetic Analyzer, Applied Biosystems, Weiterstadt, Germany), and the lengths of fluorescently labeled terminal restriction fragments (T-RFs) were determined by comparison with the internal standard using GeneMapper software (Applied Biosystems). Peaks >30 bp were analyzed by aligning fragments to the size standard. Reproducibility of patterns was confirmed for repeated terminal restriction fragment length polymorphism (T-RFLP) analysis using the same DNA extracts. A difference of less than 2 bp in estimated length between different profiles was the basis for considering fragments identical. Patterns from different samples were normalized to identical total fluorescence units by an iterative normalization procedure [37].
Quantitative PCR
The abundance of functional marker genes was determined using primer pairs nirK1F-nirK5R [35] and nirS4QF-nirS6QR [38] which also exclude detection of ammonia oxidizers and cycling conditions given in Table S1. Cloning confirmed that despite different primers and cycling conditions used for nirS-based T-RFLP and qPCR analyses, the genetic diversity targeted was essentially the same (data not shown). Gene fragments were amplified in 25-μl reaction mixtures containing 0.5 mM of each primer, 12.5 μl of SYBR green PCR master mix (SYBR® Green JumpStart™ Taq ReadyMix™; Sigma-Aldrich GmbH, Taufkirchen, Germany), 3 μl DNA diluted template corresponding to 15 ng of total DNA, and 500 ng of T4gene32 protein (New England Biolabs). Thermal cycling, fluorescent data collection, and data analysis were done using an iCycler iQ5 (Bio-Rad Laboratories GmbH, Munich, Germany) and the iQ5 Optical System Software 2.0. Standard curves were obtained using serial dilutions of a known amount of plasmid DNA containing the respective fragment of the nirK and nirS gene. Lack of inhibitory effects of co-extracted substances in soil DNA was proven by diluting soil DNA extracts and by quantifying a known amount of plasmid DNA added to soil DNA extracts.
Statistical Analysis
Effects of soil type and land use on soil characteristics (pH, C:N ratio, C and N content, nitrate), denitrification activity (DEA), and denitrification gene abundance (nirK and nirS) were studied by hierarchical analyses of variance. According to the sampling design (Table 1), in a first step, the sites with pale-sandy soils (PS) were compared with the sites characterized by other soils (LD or BR), and then in a factorial submodel, the effects of soil type (BR, LD) and land use (pristine, field, fallow) and their interactions were studied. The effects of land use were further partitioned into two orthogonal contrasts: (1) pristine vs. cultivated (field or fallow) and (2) fields vs. fallows.
To obtain the main gradients in the composition of the nirK- and nirS-type denitrifier communities, correspondence analyses (CA) were carried out with the relative height of the T-RF peaks. λ 1 and λ 2 stands for the eigenvalues of the first and second axes in the ordination diagram. The scores of the sites along the first two axes were then used for further analyses. The effects of soil type and land use on the composition of the denitrifier community expressed as the first two axes of the CA ordination (Ax1 nirK, Ax2 nirK, Ax1 nirS, Ax2 nirS), and the effects of soil type, land use, and denitrifier community composition on DEA were studied by multiple regression analyses. Potential explanatory variables for the composition of the denitrifier communities were PS (yes/no), BR (yes/no), pristine (yes/no), currently cultivated as a field (yes/no), pH, C, and nitrate content. Potential explanatory variables for DEA were the same variables plus the four CA axes, i.e., denitrifier community composition. Soil N content was not included as a potential explanatory variable, because it was strongly intercorrelated with C content (r = 0.92). Regressions with all possible combinations of explanatory variables were calculated and the Bayesian information criterion (BIC) used to measure the strength of evidence for each candidate model. The BIC was preferred over the Akaike information criterion, because it favors models with less parameters [39]. In the results, we present the models with the lowest BIC. Nitrate concentration, nirK and nirS copy numbers, and DEA were log transformed prior to analysis to satisfy the assumptions of homoscedasticity and normally distributed residuals. Most statistical analyses were carried out with IBM-SPSS 20, the correspondence analyses with CANOCO 4.5 [40], and the multiple regression analyses with the leaps package in R 2.15.1.
Results
Effects of Soil Type and Land Use on the Physicochemical Characteristics of the Soils
While conductivity was essentially the same for all soils sampled (21.3 ± 3.7 μS cm−1; P > 0.22), other soil characteristics were influenced by the soil type, land use, and the interaction between soil type and land use. Sites with pale and sandy soils differed strongly from all other sites in terms of pH (5.3 vs. 6.3; Table 2, Fig. 1a) and C:N ratio (15.3 vs. 10.4; Table 2, Fig. 1b). The pH and C:N ratio of all loamy sites were similar, but the type of land use influenced the total C and N content and NO3-N concentration (Table 2). Total soil C content depended on soil type and was influenced by cultivation. C content was higher in pristine soils that had never been cultivated (0.66 ± 0.07 %) than in other soils (0.42 ± 0.02 %). In LD soils, C content at sites currently under cultivation was higher than at fallow sites (0.51 vs. 0.39 %), whereas for BR soils, the opposite (0.37 vs. 0.42 %) was the case (Fig. 1c). Total N concentration was higher in pristine soils than in those that were or had been under cultivation, and these differences were stronger in loamy-dark (0.075 vs. 0.042 %) than in brown-reddish soils (0.052 vs. 0.039 %) (Fig. 1d). Moreover, total N concentration was higher in loamy-dark soils currently under cultivation than in fallows (0.048 vs. 0.036 %), but was lower in brown-reddish field soils than in fallows (0.037 vs. 0.041 %) (Fig. 1e). The concentration of NO3-N was much higher at cultivated than at fallow sites (1.03 vs. 0.14 μg gdw−1 soil) (Fig. 1f).
Influence of land use and soil type on a pH; b C:N ratio; c total C content; d, e total N content; and f NO3 content of Namibian savanna soils. Values are means ± 1 SE. For number of replicates, see Table 1
Effects on Denitrifying Enzyme Activity
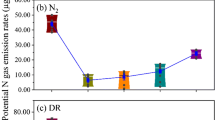
DEA ranged from not detectable in samples generated from cultivated sites to >9 ng N2O-N gdw−1 soil h−1 in samples from pristine plots (Fig. S2). Production of N2O was influenced by soil type and differed between pale and sandy and loamy sites (Table 3) with higher levels for pale and sandy soils (3.10 ng N2O-N gdw−1 soil h−1) than for other soils (0.64 ng N2O-N gdw−1 soil h−1) (Fig. 2a). Land use also influenced DEA with more than threefold higher N2O production rates for the pristine sites (3.73 ng N2O-N gdw−1 soil h−1) than for cultivated soils (0.28 ng N2O-N gdw−1 soil h−1) (Fig. 2b).
Influence of land use and soil type on denitrifier enzyme activity (DEA) in Namibian savanna soils. a Influence of soil type and b influence of land use. Values are means ± 1 SE. For number of replicates, see Table 1
Effects on Denitrifier Abundance
Denitrifiers of the nirK-type were roughly one to two orders of magnitude more abundant than those of the nirS-type (Fig. S3a and b). Denitrifier abundance was determined by soil type, land use, and the interaction of soil type and land use, but community abundance depended on the nitrite reductase gene considered (Table 3). Copy numbers of nirK were similar for pale and sandy and loamy sites, but they were higher at sites with loamy-dark than brown-reddish soils (1.42 × 106 vs. 6.02 × 105 copies gdw−1 soil; Fig. 3a). In contrast, numbers of nirS-type denitrifiers were significantly lower in pale and sandy than in loamy soils (1.05 × 103 vs. 2.67 × 104 copies gdw−1 soil), but similar at all loamy sites (Fig. 3b). Land use influenced the abundance of both nirK-type and nirS-type denitrifiers (Table 3). Denitrifiers of the nirK-type were more abundant at pristine sites than at cultivated sites (2.05 × 106 vs. 7.35 × 105 copies gdw−1 soil; Fig. 3c). This was also the case for the nirS-type denitrifiers (9.9 × 104 vs. 2.27 × 104 copies gdw−1 soil; data not shown). In loamy-dark soils, their numbers differed between sites currently under cultivation and fallows (5.66 × 104 vs. 1.66 × 104 copies gdw−1 soil), but numbers were similar for brown-reddish soils (1.52 × 104 vs. 2.00 × 104 copies gdw−1 soil; Fig. 3d).
Influence of land use and soil type on the abundance of nirK- and nirS-type denitrifiers in Namibian savanna soils. a nirK copy numbers in loamy-dark and brown-reddish soils, b nirS copy numbers in pale-sandy and loamy soils, c nirK copy numbers in pristine and cultivated soils, and d nirS copy numbers in loamy-dark and brown-reddish fields and fallows. Values are means ± 1 SE. For number of replicates, see Table 1. LD loamy-dark, BR brown-reddish soils
Effects on Denitrifier Community Composition
Correspondence analyses revealed strong gradients in the composition of both the nirK-type (λ 1 = 0.43, λ 2 = 0.27) and nirS-type (λ 1 = 0.51, λ 2 = 0.29) denitrifier communities. Overall, denitrifier community composition was best explained by soil type, soil characteristics, and cultivation, but the response of nirK- and nirS-type denitrifiers was community-specific (Table 4). The composition of nirK-type denitrifiers differed between pale-sandy and loamy soils (Fig. S4a) and between brown-reddish and loamy-dark soils, which largely explained differences in community composition along the first ordination axis and between fallow and currently farmed sites. Distinct nirS-type denitrifiers which were prevalent in the pale and sandy soils (Fig. S4b) and soil C content most strongly influenced community differences relative to those in the loamy soils.
Effects of Soil Factors and Denitrifier Communities on Denitrification Activity
Denitrification activity (DEA) increased strongly with soil C content (Fig. 4), but did not correlate significantly with other soil quality metrics. In the best model, land use and the composition of the denitrifier communities, independent of which type of nitrite reductase they had, significantly influenced DEA in addition to soil C (Table 4). Pristine soils sustained higher denitrification activity than managed soils.
Discussion
Influence of Land Use and Soil Type on Soil Characteristics
In this study, we explored soils of the Kavango region in northeast Namibia and the potential activity and communities of denitrifiers, which are a major source of N2O emitted from soils [16]. We observed that land use may alter denitrifier communities and DEA. This suggests the potential for changes in N2O flux, but N cycling in soils also depends strongly upon oxygen availability, soil texture, and soil porosity [41], which were not measured in this study. Intensive agriculture acidifies Namibian soils [24]; the lack of changes in soil pH with conversion of grasslands to agriculture may reflect the low intensity of agricultural practices in the region studied.
C:N ratios were higher in pale-sandy soils than in loamy soil, and the total C and N content of the soils was influenced by interacting effects of soil type and land use. In Namibia, the N and organic C pools of soils under cultivation are depleted [42]. Similarly, in semiarid grasslands in northeastern Colorado, soil organic matter, potentially mineralizable N, and potentially respirable C were generally lower in soils that had been cultivated than in pristine soils [43]. Our study also found higher N levels in pristine than in cultivated loamy soils. Furthermore, soil N and C contents were higher in loamy-dark soils currently used as fields than in those lying fallow. Land use, i.e., whether a site was field or fallow, also influenced the nitrate content of the soils, with higher nitrate levels in fields than in fallows. Higher nutrient levels in fields than in fallows are in line with the findings of Burke et al. who observed that fallow fields were depleted of nutrients [43]. Burke et al. also found that recovery of active organic matter and nutrient availability requires a fallow period of 50 years [43]. The fact that the fallow sites in this study were abandoned for 10 years at most indicates that time was presumably not sufficient for recovery of at least the loamy-dark soils.
Influence of Land Use and Soil Type on Denitrifier Communities
In soils of the Kavango region, both the nirK- and nirS-type denitrifiers were influenced by soil type and land use, but environmental conditions also resulted in different responses of the two communities, presumably because they occupy distinct ecological niches [44, 45]. While the abundance of nirK-type denitrifiers depended on whether the soil was pristine or under cultivation, the assembly of their communities was strongly determined by the characteristics of the pale-sandy soils and whether it was currently cultivated or lying fallow. However, composition and abundance of denitrifier communities appear to be at least partly intertwined, because they responded to the same environmental stimuli and depended on whether the soil was brown-reddish or loamy-dark. Denitrifiers of the nirS-type were also strongly influenced by characteristics of the pale-sandy soils, which affected both the size and composition of their communities. In addition, their abundance also responded to the type of land use and depended on whether the soil was pristine or cultivated and whether loamy soils were fields or fallows. A further influence on their community composition was soil C content, which agrees with a previous report of the dependence of community composition on dissolved organic C [44]. Denitrifiers are mostly facultative aerobes that prefer a heterotrophic lifestyle. Hence, the availability of C may exert a much stronger selective pressure than availability of N oxides and oxygen.
Influence of Soil Characteristics on Denitrification Activity
Our findings that differences in land use and soil type determined a number of soil characteristics that are relevant for denitrification activity and also influenced the communities mediating the process suggest that denitrification activity and potential N2O emissions of the Namibian savanna soils may indirectly depend on land management and soil type.
Potential N2O production from Namibian savanna soils in our study (range: from non-detectable levels to 9 ng N g−1 soil h−1) was very low compared to that of temperate soils (1–3000 ng N g−1 soil h−1; [45]) and even at the low end of DEA from semiarid grasslands in China (up to 339 ng N2O-N g−1 soil h−1; converted from [46]). Potential N2O emissions estimated by the DEA method are assessed in laboratory studies under optimum conditions (oxygen limitation, optimal temperature, surplus of C and N) and inhibition of N2O reductase by acetylene. Hence, N2O production as estimated by DEA is generally much higher than in field measurements of N2O production, because laboratory-like conditions rarely prevail in the field [47]. DEA reflects the long-term environmental conditions and the pool size of the persisting denitrifying enzymes and hence correlates to denitrification activity in the field [18, 48]. The low potential emissions of N2O from semiarid soils in Namibia are in agreement with the results of a meta-analysis of fluxes from soils of savannas and other seasonally dry ecosystems [8]. At some sites, N2O fluxes were low even during the wet season [49], but their magnitude was apparently influenced by soil organic matter and drainage [50]. Low N2O fluxes were attributed to low soil organic matter, rapid drainage, and low nutrient levels [8]. In the Namibian savanna ecosystem studied here, DEA was higher at pristine and pale-sandy sites than at cultivated and loamy sites and depended primarily on C content and whether the soil was pristine or not. The concurrent availability of C and N and the C:N ratio are critical for denitrification in agricultural soils [51, 52], and the denitrification potential of semiarid grassland soils in China increased after addition of C and/or N [46]. We conclude that Namibian savannas resemble other semiarid grasslands where N and C are limiting denitrification in cultivated rather than in native semiarid grasslands [43]. In these regions, the use of fertilizers is still uncommon and hence harvesting significantly depletes nutrients [10, 43]. Furthermore, changes in the use of Namibian savanna ecosystems such as logging by clear-cutting can alter soil bulk density and organic matter pools [53, 54], which may lead to increased N2O emission from soils [4]. Trampling by grazing livestock also increases N2O emissions from grassland soils [4]. Other management practices like tillage may affect the stability of aggregates, soil C content, and its dynamics [55]. Tillage destroys soil structures that occlude oxygen diffusion to organic materials and hence prevents enzymatic decomposition of C compounds [56, 57]. All this points to a greater availability of soil organic matter and hence a more effective N cycling and a higher potential for N2O emissions in the pristine woodland savannas, which are not disturbed by management and receive higher organic matter input into the soil than the cultivated areas [58].
Influence of Denitrifier Communities on Potential Denitrification Activity
Denitrification activity may depend not only on soil characteristics but also on the size and the composition of the underlying denitrifier communities, although the literature is inconsistent in what matters most (e.g., [44, 59, 60]). In this study, characteristics of the soils determined by land use and soil type influenced both denitrifier abundance as well as community composition. However, DEA of the soils was related only to the composition of the nirK- and nirS-type denitrifier communities while the size of the communities was irrelevant. Although it is currently not possible to correlate differences in denitrification activity to the relative abundance of single terminal restriction fragments or nitrite reductase genotypes, it is known that specific denitrification activity differs among individual strains [61, 62]. This suggests that denitrification activity is determined by the taxonomic makeup of bacterial communities [63], whose development was influenced by the soil characteristics explicitly investigated in this study or by characteristics that were not specified [44] but were covered by the terms land use or soil type.
Conclusions
Overall, our results suggest that N2O emissions in southern Africa may not increase by the conversion of pristine savanna to cultivated soils as long as fertilizers are not applied on a large scale. In pristine soils, N is conserved by microorganisms and plants via net primary production, thus limiting the availability of N for nitrification and denitrification [64, 65]. However, anthropogenic influences like the continuing conversion of native to agricultural lands together with the predicted inputs of N into African savanna soils by the use of fertilizers have the potential to disrupt the closed N cycle [65]. This may result in increased N2O emissions from agricultural land [66–68], but denitrification activity and hence the size of the N2O fluxes may be limited by the availability of soil C.
References
Crutzen PJ (1970) The influence of nitrogen oxides on the atmospheric ozone content. Q J R Meteorol Soc 96:320–325
IPCC (2001) Climate change 2001: a scientific basis. Intergovernmental panel on climate change. Cambridge University Press, Cambridge
EPA (2010) Methane and nitrous oxide emissions from natural sources, Inventory of U.S. greenhouse gas emissions and sinks: 1990–2008. U.S. Environmental Protection Agency, Washington, pp 1–194
Bhandral R, Saggar S, Bolan NS, Hedley MJ (2007) Transformation of nitrogen and nitrous oxide emission from grassland soils as affected by compaction. Soil Tillage Res 94:482–492. doi:10.1016/j.still.2006.10.006
Gärdenäs AI, Ågren GI, Bird JA, Clarholm M, Hallin S, Ineson P et al (2011) Knowledge gaps in soil carbon and nitrogen interactions—from molecular to global scale. Soil Biol Biochem 43:702–717. doi:10.1016/j.soilbio.2010.04.006
Burger M, Jackson LE (2003) Microbial immobilization of ammonium and nitrate in relation to ammonification and nitrification rates in organic and conventional cropping systems. Soil Biol Biochem 35:29–36. doi:10.1016/S0038-0717(02)00233-X
Rafique R, Hennessy D, Kiely G (2011) Nitrous oxide emission from grazed grassland under different management systems. Ecosystems 14:563–582. doi:10.1007/s10021-011-9434-x9
Castaldi S, Ermice A, Strumia S (2006) Fluxes of N2O and CH4 from soils of savannas and seasonally-dry ecosystems. J Biogeogr 33:401–415. doi:10.1111/j.1365-2699.2005.01447.x
Hickman JE, Havlikova M, Kroeze C, Palm CA (2011) Current and future nitrous oxide emissions from African agriculture. Curr Opin Environ Sustain 3:370–378. doi:10.1016/j.cosust.2011.08.001
Vitousek PM, Naylor R, Crews T, David MB, Drinkwater LE, Holland E et al (2009) Nutrient imbalances in agricultural development. Science 324:1519–1520. doi:10.1126/science.1170261
Mendelsohn J, El Obeid S (2003) Sand and water. A profile of the Kavango region. Struik Publishers, Cape Town
Du R, Lu DR, Wang GC (2006) Diurnal, seasonal, and inter-annual variations of N2O fluxes from native semi-arid grassland soils of Inner Mongolia. Soil Biol Biochem 38:3474–3482. doi:10.1016/j.soilbio.2006.06.012
Xu YQ, Wan SQ, Cheng WX, Li LH (2008) Impacts of grazing intensity on denitrification and N2O production in a semi-arid grassland ecosystem. Biogeochemistry 88:103–115. doi:10.1007/s10533-008-9197-4
Weitz AM, Veldkamp E, Keller M, Neff J, Crill PM (1998) Nitrous oxide, nitric oxide, and methane fluxes from soils following clearing and burning of tropical secondary forest. J Geophys Res D: Atmos 103:28047–28058. doi:10.1029/98JD02144
Stehfest E, Bouwman L (2006) N2O and NO emission from agricultural fields and soils under natural vegetation: summarizing available measurement data and modeling of global annual emissions. Nutr Cycl Agroecosyst 74:207–228. doi:10.1007/s10705-006-9000-7
Conrad R (1996) Soil microorganisms as controllers of atmospheric trace gases (H2, CO, CH4, OCS, N2O, and NO). Microbiol Rev 60:609–640
McLain JET, Martens DA (2006) N2O production by heterotrophic N transformations in a semiarid soil. Appl Soil Ecol 32:253–263. doi:10.1016/j.apsoil.2005.06.005
Peterjohn WT (1991) Denitrification: enzyme content and activity in desert soils. Soil Biol Biochem 23:845–855. doi:10.1016/0038-0717(91)90096-3
Peterjohn WT, Schlesinger WH (1990) Nitrogen loss from deserts in the Southwestern United States. Biogeochemistry 10:67–79. doi:10.1007/BF00000893
Jürgens N, Schmiedel U, Haarmeyer DH, Dengler J, Finckh M, Goetze D et al (2012) The BIOTA Biodiversity Observatories in Africa—a standardized framework for large-scale environmental monitoring. Environ Monit Assess 184:655–678. doi:10.1007/s10661-011-1993-y
Hartemink AE, Huting J (2008) Land cover, extent, and properties of arenosols in Southern Africa. Arid Land Res Manag 22:134–147. doi:10.1080/15324980801957689
Klintenberg P, Seely M (2004) Land degradation monitoring in Namibia: a first approximation. Environ Monit Assess 99:5–21. doi:10.1007/s10661-004-3994-6
Haarmeyer DH, Luther-Mosebach J, Dengler J, Schmiedel U, Finckh M, Berger K et al. (2010) The BIOTA observatories. In: Jürgens N, Haarmeyer DH, Luther-Mosebach J, Dengler J, Finckh M, Schmiedel U (eds.): Biodiversity in southern Africa. Volume 1: patterns at local scale—the BIOTA Observatories, pp. 6–801, Klaus Hess Publishers, Göttingen & Windhoek.
Gröngröft A, Herpel N, Petersen A, Mills A (2006) Indication of soil degradation processes in selected drylands of Southern Africa. Proceedings of the international conference: soil and desertification—integrated research for the sustainable management of soils in drylands. Hamburg, Germany
Barnard P (1998) Biological diversity in Namibia—a country study. Namibian National Biodiversity Task Force, Windhoek
Uhlmann E, Görke C, Petersen A, Oberwinkler F (2004) Arbuscular mycorrhizae from semiarid regions of Namibia. Can J Bot 82:645–653. doi:10.1139/b04-039
Strohbach BJ, Petersen A (2007) Vegetation of the central Kavango woodlands in Namibia: an example from the Mile 46 Livestock Development Centre. S Afr J Bot 73:391–401. doi:10.1016/j.sajb.2007.03.002
Pröpper M, Gröngroft A, Falk T, Eschenbach A, Fox T, Gessner U et al. (2010) Causes and perspectives of land-cover change through expanding cultivation in Kavango. In: Biodiversity in southern Africa. Volume 3: implications for land use and management, pp. 1–31. Klaus Hess Publishers, Göttingen & Windhoek
USDA (2002) Soil Survey Technical Note No. 2
Wisch U, Petersen A, Gröngröft A, Eschenbach A (2009) Dryland farming in the Kavango region: effect of land use on soil properties. Zentralbl Geol Palaeontol Teil 1 2008:189–211
Smith MS, Tiedje JM (1979) Phases of denitrification following oxygen depletion in soil. Soil Biol Biochem 11:261–267. doi:10.1016/0038-0717(79)90071-3
Yeates C, Gillings MR (1998) Rapid purification of DNA from soil for molecular biodiversity analysis. Lett Appl Microbiol 27:49–53. doi:10.1046/j.1472-765X.1998.00383.x
Michotey V, Mejean V, Bonin P (2000) Comparison of methods for quantification of cytochrome cd 1-denitrifying bacteria in environmental marine samples. Appl Environ Microbiol 66:1564–1571. doi:10.1128/AEM.66.4.1564-1571.2000
Throbäck IN, Enwall K, Jarvis Å, Hallin S (2004) Reassessing PCR primers targeting nirS, nirK and nosZ genes for community surveys of denitrifying bacteria with DGGE. FEMS Microbiol Ecol 49:401–417. doi:10.1016/j.femsec.2004.04.011
Braker G, Fesefeldt A, Witzel KP (1998) Development of PCR primer systems for amplification of nitrite reductase genes (nirK and nirS) to detect denitrifying bacteria in environmental samples. Appl Environ Microbiol 64:3769–3775
Braker G, Ayala-del-Rio HL, Devol AH, Fesefeldt A, Tiedje JM (2001) Community structure of denitrifiers, bacteria, and archaea along redox gradients in Pacific Northwest marine sediments by terminal restriction fragment length polymorphism analysis of amplified nitrite reductase (nirS) and 16S rRNA genes. Appl Environ Microbiol 67:1893–1901. doi:10.1128/AEM.67.4.1893-1901.2001
Dunbar J, Ticknor LO, Kuske CR (2001) Phylogenetic specificity and reproducibility and new method for analysis of terminal restriction fragment profiles of 16S rRNA genes from bacterial communities. Appl Environ Microbiol 67:190–197. doi:10.1128/AEM.67.1.190-197.2001
Kandeler E, Deiglmayr K, Tscherko D, Bru D, Philippot L (2006) Abundance of narG, nirS, nirK, and nosZ genes of denitrifying bacteria during primary successions of a glacier foreland. Appl Environ Microbiol 72:5957–5962. doi:10.1128/AEM.00439-06
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd ed., XXVI, 488 p.
Ter Braak CJF, Smilauer P (2002) Canoco reference manual and CanoDraw for Windows users guide: software for canonical community ordination [4.5]. Microcomputer Power, Ithaka
Nömmik H (1956) Investigations on denitrification in soil. Acta Agric Scand 6:195–228
Zeidler J, Hanrahan S, Scholes M (2002) Land-use intensity affects range condition in arid to semi-arid Namibia. J Arid Environ 52:389–403. doi:10.1006/jare.2002.0990
Burke IC, Lauenroth WK, Coffin DP (1995) Soil organic matter recovery in semiarid grasslands: implications for the conservation reserve program. Ecol Appl 5:793–801. doi:10.2307/1941987
Enwall K, Throbäck IN, Stenberg M, Soderstrom M, Hallin S (2010) Soil resources influence spatial patterns of denitrifying communities at scales compatible with land management. Appl Environ Microbiol 76:2243–2250. doi:10.1128/AEM.02197-09
Rich JJ, Heichen RS, Bottomley PJ, Cromack K, Myrold DD (2003) Community composition and functioning of denitrifying bacteria from adjacent meadow and forest soils. Appl Environ Microbiol 69:5974–5982. doi:10.1128/AEM.69.10.5974-5982.2003
Wu X, Liu G, Butterbach-Bahl K, Fu B, Zheng X, Brüggemann N (2013) Effects of land cover and soil properties on denitrification potential in soils of two semi-arid grasslands in Inner Mongolia, China. J Arid Environ 92:98–101
Herrera-Arreola G, Vasquez-Murrieta MS, Cruz-Mondragon C, Van Cleemput O, Dendooven L (2008) Nitrous oxide emissions from soils of the semi-arid highlands of Durango, Mexico: a laboratory study. Arid Land Res Manag 22:179–194
Frank DA, Groffman PM (1998) Denitrification in a semi-arid grazing ecosystem. Oecologia 117:564–569
Scholes MC, Martin R, Scholes RJ, Parsons D, Winstead E (1997) NO and N2O emissions from savanna soils following the first simulated rains of the season. Nutr Cycl Agroecosyst 48:115–122. doi:10.1007/s004420050693
Rees RM, Wuta M, Furley PA, Li C (2006) Nitrous oxide fluxes from savanna (miombo) woodlands in Zimbabwe. J Biogeogr 33:424–437. doi:10.1111/j.1365-2699.2005.01423.x
Philippot L, Hallin S, Schloter M (2007) Ecology of denitrifying prokaryotes in agricultural soil. Adv Agron 96:249–305. doi:10.1016/S0065-2113(07)96003-4
Tiedje JM (1988) Ecology of denitrification and of dissimilatory nitrate reduction to ammonium. In: Zehnder AJB (ed) Biology of anaerobic microorganisms. John Wiley and Sons, Inc., New York, pp 179–244
Bossio DA, Girvan MS, Verchot L, Bullimore J, Borelli T, Albrecht A et al (2005) Soil microbial community response to land use change in an agricultural landscape of western Kenya. Microb Ecol 49:50–62. doi:10.1007/s00248-003-0209-6
Johnson CE, Johnson AH, Huntington TG, Siccama TG (1991) Whole-tree clear-cutting effects on soil horizons and organic-matter pools. Soil Sci Soc Am J 55:497–502. doi:10.2136/sssaj1991.03615995005500020034x
Huygens D, Boeckx P, Van Cleemput O, Oyarzún C, Godoy R (2005) Aggregate and soil organic carbon dynamics in south Chilean andisols. Biogeosciences 2:159–174. doi:10.5194/bg-2-159-2005
Bayer C, Martin-Neto L, Mielniczuk J, Pavinato A, Dieckow J (2006) Carbon sequestration in two Brazilian Cerrado soils under no-till. Soil Tillage Res 86:237–245. doi:10.1016/j.still.2005.02.023
Christensen BT (2001) Physical fractionation of soil and structural and functional complexity in organic matter turnover. Eur J Soil Sci 52:345–353. doi:10.1046/j.1365-2389.2001.00417.x
Abbadie L, Lensi R (1990) Carbon and nitrogen mineralization and denitrification in a humid savanna of West Africa (Côte d’Ivoire). Acta Oecol 11:717–728. doi:10.1029/95JD01923
Bremer C, Braker G, Matthies D, Beierkuhnlein C, Conrad R (2009) Plant presence and species combination, but not diversity, influence denitrifier activity and the composition of nirK-type denitrifier communities in grassland soil. FEMS Microbiol Ecol 70:377–387. doi:10.1111/j.1574-6941.2009.00732.x
Morales SE, Cosart T, Holben WE (2010) Bacterial gene abundances as indicators of greenhouse gas emission in soils. ISME J 4:799–808. doi:10.1038/ismej.2010.8
Cavigelli MA, Robertson GP (2000) The functional significance of denitrifier community composition in a terrestrial ecosystem. Ecology 81:1402–1414. doi:10.1890/0012-9658(2000)081%5B1402:TFSODC%5D2.0.CO;2
Ka JO, Urbance J, Ye RW, Ahn TY, Tiedje JM (1997) Diversity of oxygen and N-oxide regulation of nitrite reductases in denitrifying bacteria. FEMS Microbiol Lett 156:55–60. doi:10.1111/j.1574-6968.1997.tb12705.x
Braker G, Dörsch P, Bakken LR (2012) Genetic characterization of denitrifier communities with contrasting intrinsic functional traits. FEMS Microbiol Ecol 79:542–554. doi:10.1111/j.1574-6941.2011.01237.x
Dries H, Pascal B, Pamela T, Leandro P, Oswald VC, Carlos ON et al (2008) Mechanisms for retention of bioavailable nitrogen in volcanic rainforest soils. Nat Geosci 1:543–548. doi:10.1038/ngeo252
Muñoz C, Paulino L, Monreal C, Zagal E (2010) Greenhouse gas (CO2 and N2O) emissions from soils: a review. Chil J Agr Res 70:485–497
Brümmer C, Brüggemann N, Butterbach-Bahl K, Falk U, Szarzynski J, Vielhauer K et al (2008) Soil-atmosphere exchange of N2O and NO in near-natural savanna and agricultural land in Burkina Faso (W. Africa). Biogeosciences 2008:582–600. doi:10.1007/s10021-008-9144-1
Castaldi S, Ariangelo DPR, John G, Nina N, Ruben M, José SJ (2004) Nitrous oxide and methane fluxes from soils of the Orinoco savanna under different land uses. Glob Chang Biol 10:1947–1960. doi:10.1111/j.1365-2486.2004.00871.x
Dick J, Kaya B, Soutoura M, Skiba U, Smith R, Niang A, Tabo R (2008) The contribution of agricultural practices to nitrous oxide emissions in semi-arid Mali. Soil Use Manag 24:292–301. doi:10.1111/j.1475-2743.2008.00163.x
Acknowledgments
This work was funded by the BIOTA South program of the German Federal Ministry of Education and Research (Project 01LC0621E/TP3) and the Max Planck Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Braker, G., Matthies, D., Hannig, M. et al. Impact of Land Use Management and Soil Properties on Denitrifier Communities of Namibian Savannas. Microb Ecol 70, 981–992 (2015). https://doi.org/10.1007/s00248-015-0623-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-015-0623-6