Abstract
Background
Brain magnetic resonance imaging (MRI) is a crucial tool for clinical evaluation of the brain and neuroscience research. Obtaining successful non-sedated MRI in children who live in resource-limited settings may be an additional challenge.
Objective
To present a feasibility study of a novel, low-cost MRI training protocol used in a clinical research study in a rural/semi-rural region of Colombia and to examine neurodevelopmental factors associated with successful scans.
Materials and methods
Fifty-seven typically developing Colombian children underwent a training protocol and non-sedated brain MRI at age 7. Group training utilized a customized booklet, an MRI toy set, and a simple mock scanner. Children attended MRI visits in small groups of two to three. Resting-state functional and structural images were acquired on a 1.5-Tesla scanner with a protocol duration of 30-40 minutes. MRI success was defined as the completion of all sequences and no more than mild motion artifact. Associations between the Wechsler Preschool and Primary Scale of Intelligence (WPPSI), Movement Assessment Battery for Children (MABC), Behavioral Rating Inventory of Executive Function (BRIEF), Child Behavior Checklist (CBCL), and Adaptive Behavior Assessment System (ABAS) scores and MRI success were analyzed.
Results
Mean (SD) age at first MRI attempt was 7.2 (0.2) years (median 7.2 years, interquartile range 7.1-7.3 years). Twenty-six (45.6%) participants were male. Fifty-one (89.5%) children were successful across two attempts; 44 (77.2%) were successful on their first attempt. Six (10.5%) were unsuccessful due to refusal or excessive motion. Age, sex, and scores across all neurodevelopmental assessments (MABC, TVIP, ABAS, BRIEF, CBCL, NIH Toolbox Flanker, NIH Toolbox Pattern Comparison, WPPSI) were not associated with likelihood of MRI success (P=0.18, 0.19, 0.38, 0.92, 0.84, 0.80, 1.00, 0.16, 0.75, 0.86, respectively).
Conclusion
This cohort of children from a rural/semi-rural region of Colombia demonstrated comparable MRI success rates to other published cohorts after completing a low-cost MRI familiarization training protocol suitable for low-resource settings. Achieving non-sedated MRI success in children in low-resource and international settings is important for the continuing diversification of pediatric research studies.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Brain magnetic resonance imaging (MRI) is a safe and important diagnostic imaging tool for pediatric clinical care and neuroscience research. Structural brain differences, as detected by MRI, are important indicators of the basis for child neurodevelopmental delays and neurologic disabilities [1, 2]. Furthermore, the expansion of MRI as a research technique to a variety of international settings is an important step in diversifying clinical research participant cohorts and increasing the applicability of clinical norms and references to populations around the globe [3].
Brain MRI in pediatric settings is challenging due to the need to have the child free of motion for the duration of the MRI protocol to avoid movement artifacts, which can limit interpretation of the images. In clinical settings, children often require sedation when they are too young to cooperate with staying still for the duration of an MRI, which adds a higher level of complexity to this procedure. In newborns, recent protocols have been developed which are becoming more widely used to obtain successful non-sedated neonatal MRIs during natural sleep [4]. MRIs done for pediatric research often cannot include sedation due to more than minimal risk with anesthesia, which limits its use to understand the developing brain over certain periods of child development [5, 6]. In addition, sedation, though effective in achieving MRI images of high quality, is expensive, time-consuming, and carries certain risks, even under optimal conditions [4, 6, 7]. Prior studies have suggested that there may be long-term effects of anesthesia on brain development [8,9,10]. Furthermore, the need for sedation may result in MRIs being less feasible and economically viable in settings with limited healthcare infrastructure or limited financial resources [3]. Thus, for both clinical and research scans, utilizing techniques to train children to successfully have an MRI without sedation is of great benefit.
Studies have shown that children are more likely to be successful in lying still for awake MRI scans by 6 to 8 years of age [11, 12]. Some children and even adults, however, may still have difficulties completing the MRI due to the nature of the MRI environment, including loud noises and narrow spaces that may induce claustrophobia [4, 11]. It has been shown that young children undergoing non-sedated brain MRI are most successful after they have received comprehensive training and familiarization with the MRI environment [13,14,15,16]. Previous studies have shown success rates ranging from 75 to 100% in non-sedated pediatric MRI following training with a shell mock MRI scanner [5, 6, 17, 18]; however, the definition of MRI “success” also varies across studies, as success from a clinical diagnostic perspective may differ from research study protocols that apply more stringent criteria for head motion, such as a maximum displacement parameter of ≤3 mm reported by de Bie et al. [6]. Barnea-Goraly et al. have demonstrated success using a novel, low-cost non-sedated MRI training protocol in the United States that does not require a commercial mock scanner, reporting 100% success in acquisition of T1-weighted scans and 92.5% success in acquisition of diffusion-weighted scans (defining success as completion of the entire scan and no more than mild motion artifact) [5].
In this study, we developed a low-cost MRI training protocol for use in a rural/semi-rural area of Latin America adapted from our procedures that have been successfully used in several other studies of child neurodevelopment in the United States [12]. Our objective was to assess the feasibility and success rates of our brain MRI training protocol in a typically developing pediatric research cohort of 7-year-old children from a rural/semi-rural region of Colombia. We examined factors within this sample that may affect MRI success, including associations between success in brain MRI scans and neurodevelopmental assessment scores in the domains of cognition, motor abilities, and executive function. The development of low-cost MRI training methods for children that can be used in an international setting is crucial for feasibility of global health pediatric MRI research in low-resource regions.
Materials and methods
Study sample
Fifty-seven healthy 7-year-old Colombian children in a neurodevelopment study [19] underwent MRI training in preparation for non-sedated scans. All children were Spanish speaking and resided in Colombia’s lowest socioeconomic stratum (a Colombian classification system based on residential housing characteristics including public service availability, road type, topography, and building characteristics [20]) in rural and semi-rural towns in the Municipality of Sabanalarga, in the Department of Atlántico, Colombia. These 57 participants were originally selected as non-exposed controls in a study of neurodevelopment following antenatal viral exposure; thus, they are considered healthy and typically developing children of their communities. As such, upon enrollment, all children met the inclusion criteria for the original study: they had no diagnosed medical, psychological, or behavioral conditions; no history of neurologic, hearing, or vision problems not corrected by glasses; no caregiver-reported developmental concerns; no history of physical or occupational therapy for developmental delays; and were not born preterm (≤36 weeks gestation) [19]. Children were also excluded if their mother reported exposure to an infectious disease during pregnancy (e.g., Zika, Dengue, Chikungunya) [19]. The participants were age-matched to cases at the time of evaluation and recruited from the same rural and semi-rural towns as the cases in the neurodevelopmental study via door-to-door recruitment and flyers distributed at the local schools. Upon enrollment, informed written consent was obtained from the parent or legal guardian of all participants. The study was approved by the Institutional Review Board of Children’s National Hospital, Washington, DC and the Institutional Review Committee and the Independent Committee on Research Ethics (CIRCIE), Barranquilla, Colombia. This study is registered on ClinicalTrials.gov, NCT04398901.
MRI training procedures
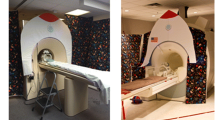
Training occurred in groups of five to ten children at a community site prior to their MRI scans and was conducted by a trained research coordinator. Children were transported by a different study team member to the community site in groups and were able to watch their peers undergo the mock MRI training process (Fig. 1). The research coordinator demonstrated the scanning process using a Playmobil Radiology Playset that includes a miniature model MRI scanner and a toy “patient” and “radiologist” (Brandstätter Group, Zirndorf, Germany). As originally described by Barnea-Goraly et al. and previously used by our group, a colorful collapsible play tunnel simulated the scanning environment [5, 12]. The research coordinator guided each child through practicing lying still in the tunnel for 3 to 5 min as the child listened to typical sounds of an MRI through headphones connected to a phone or computer. A dishware container at the top of the play tunnel modeled a head coil (Fig. 1). During the practice session, the research coordinator provided gentle reminders to keep still if children moved their heads or limbs to help children understand the behavioral requirements for a successful scan. As part of the training, children also listened to a read-aloud of a customized booklet designed for the purpose of the neurodevelopment study which explained the MRI procedure and included photos of a child undergoing an MRI at the same radiology suite in Barranquilla, Colombia, where their imaging would occur (Fig. 2). Participants received a copy of the booklet to take home with them as well as a sticker reward for their cooperation. The total cost of the training supplies for all subjects was less than $100 USD.
Group MRI familiarization and training session. A group MRI training session in a community center in Sabanalarga, Colombia. A 7-year-old child lays in the play tunnel that served as a mock MRI scanner while wearing headphones to simulate the scanning environment. A member of the study team plays MRI sounds as the child lays still for 3-5 minutes. Another 7-year-old child in the training session observes the child practicing while waiting for their turn in the mock scanner
Spanish-language MRI booklet excerpts. a Cover page (translation, My MRI Day); 18 photos included in the booklet were taken at Sabbag Radiólogos, where children underwent brain MRI. (b) Sample page (translation, Now they are going to put this helmet over my head…/…and this warm blanket. I am very comfortable! I could take a good nap./I wear these headphones to hear and talk with the MRI person). Sample page explaining the steps of the MRI scan session. (c) Final customizable page (translation, What a fun day! We’ll see you on your MRI day! / My MRI day / Attach a photo here of yourself on your MRI day). The radiology team was equipped with an instant-print camera to take photos of children in the MRI suite to be added to their booklets. The full text of the booklet can be found in Supplementary Material 1
Non-sedated MRI acquisition
On a separate day, children and their parents were driven to the MRI site (Sabbag Radiólogos) in the city of Barranquilla by a study team member. Travel time was over an hour and often complicated by weather conditions such as heat and flash flooding. Two to three children were scheduled on each appointment day and were transported to the site together. Children were provided lunch and waited in a family-friendly area of the radiology center that was separate from clinical patients. Radiology staff took instant-print photos of the children in the radiology suite to be attached to their MRI booklets as part of a prize for their “MRI day,” in addition to a toy or other incentive for their participation (Fig. 2). All children were escorted by a parent (typically their mother) at all times of the MRI visit and a coordinator from the imaging center remained with the children and families throughout the entire process. Parents were invited to join their child in the scanning room during the scan with their own set of headphones on and were able to have their hand touching their child to ensure the child knew their parent was with them. Hesitant children or those who initially refused the scan were permitted to watch peers successfully complete the MRI before attempting or trying again. If a child was ultimately unsuccessful on their first scan session due to either poor image quality (attributable to excessive motion) or refusal, they had the opportunity to try again during a second scan session on a different date. If a child was hesitant or wanted to prematurely leave the scanner, they were praised for their attempt and the interaction was focused on maintaining and developing trust with the scanning team to leverage for any second attempt. Second attempts were also conducted in groups of two to three children. Mock scanner and other behavioral MRI trainings were not repeated prior to a second scan session, but all other aspects of the day of testing were the same. The length of time between training and MRI acquisition, as well as between first and second MRI attempts, varied due to radiology appointment and family availability; no minimum or maximum times were set.
Scans were acquired on a GE 1.5-T scanner (General Electric, Milwaukee, WI) using an eight-channel head coil. The 30-40-min MRI protocol included resting-state functional and structural images. Sequences included sagittal 3D T1-weighted spoiled gradient recalled (SPGR), sagittal 3D T-2-weighted CUBE, axial resting-state fMRI, axial diffusion tensor imaging (DTI), and axial T-2-weighted fluid-attenuated inversion recovery (FLAIR) images. After 10 min of resting-state functional sequences, children watched a pre-selected movie through MRI-compatible video goggles (Resonance Technology Inc., Los Angeles, CA) during the remaining structural sequences. To help minimize motion, children were wrapped in a blanket and had foam placed inside the head coil to hold their head in place. Sequences with visible motion were repeated during the same scanning session when able. The MRI data were transferred to Children’s National Hospital, Washington, DC, where the data were reviewed for quality and interpreted by a pediatric neuroradiologist (GV).
Neurodevelopmental assessment
As a part of the neurodevelopmental study in which these children were enrolled as controls, children completed a battery of cognitive and motor assessments on the same day as their MRI training [19]. Cognitive skills were assessed using the Wechsler Preschool and Primary Scale of Intelligence (WPPSI), which was administered by trained pediatric neuropsychologists [21]. Full-scale IQ (FSIQ) was calculated according to the Spanish manual norms used as a measure of overall intelligence. The standard score of the Test de Vocabulario en Imágenes Peabody (TVIP, the Spanish adaptation of the Peabody Picture Vocabulary Test) was used to determine child receptive language development. The Movement Assessment Battery for Children 2nd edition (MABC) was used for motor skills evaluation in the domains of manual dexterity, aiming and catching, and balance [22]. We calculated MABC overall scores as per the manual. The TVIP and MABC were administered by trained research coordinators. The Spanish versions of the parent forms Behavioral Rating Inventory of Executive Function (BRIEF), Child Behavior Checklist (CBCL), and Adaptive Behavior Assessment System (ABAS) were used to assess executive functioning, psychosocial functioning, and adaptive behavior [23,24,25]. The BRIEF General Executive Composite (GEC), CBCL Total Problems score, and the ABAS General Adaptive Composite (GAC) were used in analysis. Trained research coordinators assisted parents and caregivers in filling out all parent forms as needed. The age-corrected standard scores for the Flanker Inhibitory Control and Attention Test and the Pattern Comparison Processing Speed Test from the NIH Toolbox were also analyzed following administration by trained pediatric neuropsychologists.
Statistical analysis
Following each set of MRI scan sessions, MRI sequences acquired and reasons for missing sequences, if any, were documented in REDCap [26] for each participant. Neurodevelopmental assessment scores and demographic information were stored in REDCap. Sociodemographic data were summarized as means and standard deviation for continuous variables and as count and percentage for categorical variables. A successful MRI was defined as one in which the child remained in the scanner for the duration of all sequences and any motion artifacts were mild and did not detract from the diagnostic accuracy of the images. We used logistic regression to evaluate the extent to which any given variable predicts whether a subject had an MRI success on their first attempt. We used this approach since it is equivalent to using a negative binomial model for the number of attempts needed for a successful MRI scan that includes censoring for those who do not have a success. Predictor variables included sex, age at MRI, MABC overall score, TVIP standard score, ABAS GAC, BRIEF GEC, CBCL Total Problems, NIH Toolbox Flanker age-corrected standard score, NIH Toolbox Pattern Comparison age-corrected standard score, and WPPSI FSIQ. Separate models were used to evaluate each predictor variable. As a sensitivity analysis, we also examined how much results changed for each predictor variable when the following covariates were added to the model singly: sex, age, and the gap in time between MRI date and training date.
Results
MRI acquisition outcomes
Children were a mean (SD) age of 7.2 (0.2) years (median 7.2 years, interquartile range [IQR] 7.1-7.3) at the time of their first MRI attempt. Twenty-six (45.6%) participants were male. MRI training occurred between 1 and 122 days (median 21 days) before the first MRI attempt. The mean (SD) age at MRI training was 7.1 (0.1) years (median 7.1 years, IQR 7.0-7.1 years). Between November 2022 and June 2023, after a single visit 44 of 57 (77%) of children successfully completed the MRI; success rates improved to 51 of 57 (89%) children after a second visit (Fig. 3). The 13 initial failures were due to either refusal upon arrival at the imaging center (n=10) or excessive motion during image acquisition (n=3) (Fig. 3). All three children with excessive motion in their initial scans were successful in a second attempt, as were four children who had refused the first attempt. Of the remaining six children whose first MRI was unsuccessful due to refusal, two made a second attempt but did not successfully complete the MRI due to refusal, and four children declined a second attempt (Fig. 3). Second scan sessions occurred between 41 and 229 days after initial MRI training.
Factors associated with MRI success
Neither age at MRI (P=0.18) nor child sex (P=0.19) predicted whether participants successfully completed the MRI. Days between MRI preparation and first MRI appointment also did not differ between unsuccessful and successful participants (P=0.90). MRI success was not significantly associated with performance on any of the neurodevelopmental assessments (Table 1). The range of neurodevelopmental assessment scores in this sample can be found in Supplementary Material 2.
Discussion
We developed a low-cost, portable, and engaging Spanish-language brain MRI training protocol and demonstrated its success at preparing 7-year-old children from a low-resource area of Latin America for non-sedated brain MRI, with 89% overall success in usable image acquisition (defined as completion of all sequences with no motion artifacts that detracted from the diagnostic accuracy of the images) in our cohort of 57 children. To our knowledge, this is the first time that pediatric MRI success rates with comprehensive training in an international low-resource setting have been published. While it has been demonstrated that MRI training and familiarization improve pediatric MRI success rates, most studies investigating pediatric MRI training success take place in high-resource settings such as academic medical centers in the United States, the United Kingdom, Canada, and Australia [18]. Many studies also include clinical patient populations who may be more familiar with healthcare settings prior to their MRI than participants in this research cohort. To our knowledge, this is the first Spanish-language protocol that has been published. Nonetheless, the success levels demonstrated in this report are comparable to those obtained from children seen in medical centers such as Stanford University, St. Michael’s Hospital/University of Bristol, and Alberta Children’s Hospital following mock MRI preparation, highlighting the potential for low-resource clinical and research sites to achieve comparable pediatric MRI results to those achieved in a high-resource environment [5, 27, 28]. The ability to successfully harness brain MRI for clinical diagnostic purposes and for research may be particularly important in lower-resource settings with less healthcare infrastructure and where children may be at greater risk of abnormal neurodevelopment from influences such as adverse infectious and environmental exposures, nutritional deficits, and violence-related posttraumatic stress [29,30,31,32,33,34,35].
This study did not find factors that influence MRI success, which may be due to the overall low number of children that did not achieve MRI success as well as the low variability in age and some of the other variables we examined. Additionally, given that the children in this study were all typically developing controls, lower variability in neurodevelopmental assessment scores may also contribute to the lack of association observed between their scores and MRI success. Some studies that examined relationships between developmental factors and MRI success found associations between neurodevelopmental outcomes and MRI compliance [5, 27, 36, 37]. Higher cognitive assessment scores were associated with MRI success in a study of typically developing children by Thieba et al., but intelligence was not a significant predictor of MRI completion in a neuro-oncology patient population examined by Weiler-Wichtl et al. or in healthy controls or pediatric type I diabetes patients as seen in Barnea-Goraly et al. [5, 36, 37]. Parent-reported hyperactivity was found to be related to MRI success in the study conducted by Weiler-Wichtl et al., but not by Barnea-Goraly et al. or in a study of children cooled for neonatal encephalopathy and age-matched controls by Woodward et al. [5, 27, 36]. Motor ability, specifically balance, was found to be predictive of MRI success by Woodward et al., but Thieba et al. did not observe a relationship between motor scores and MRI completion [27, 37]. Other factors that have been associated with MRI success, such as memory [36], processing speed [27], and language ability [37], have not been widely studied. Furthermore, none of the aforementioned studies was conducted in low- and middle-income countries or a low-resource region outside of the United States. There are significant gaps in the literature as to how children in low-resource international healthcare settings comply with non-sedated brain MRI.
In our study, we did not find that the number of days between MRI training and the MRI was a significant factor for predicting MRI success. We had hypothesized that children whose MRI training was closer to the MRI day would be more successful, based on prior studies, but this was not shown by our data. In other studies that have examined the efficacy of MRI training and familiarization protocols, training occurred immediately prior to the MRI or a few days before [15, 28]. Thus, our observation that the time between training and scanning did not significantly affect likelihood of success is a particularly important finding. In remote or low-resource settings where appointment scheduling and travel to a hospital or imaging center may be more difficult, our findings support flexibility in the timing of training appointments and scan sessions. The variability in timing for our research participants was due to multiple factors including the COVID-19 pandemic, family schedules, MRI and research team transportation availability, and school and holiday schedules. Future studies may assess participant MRI training skills retention over time from MRI training to determine if there is a time in which participants would require MRI re-training. Future researchers interested in implementing a similar MRI training intervention may also consider conducting training on the day of the MRI to mitigate logistical challenges and standardize the consistency of time between training and MRI scan acquisition.
This study has several limitations. First, the MRI trainings and results reported here were a component of our original study designed to evaluate the impact of antenatal viral exposure on child development. This was a standalone case study of the feasibility and success of this MRI training protocol in a very specific pediatric population of typically developing 7-year-old children from rural and semi-rural regions of Colombia. Thus, all children completed the MRI training protocol, and we are unable to provide a baseline MRI success rate in similar children who did not receive the training. The imaging center where these scans were completed had never acquired non-sedated MRI images in children as young as 7 years old prior to this collaboration for research purposes, so while our low-cost training protocol resulted in high success in MRI acquisition, we are unable to compare success rates following this protocol to success rates in clinical scan acquisition in comparable children at this institution. Due to the lack of an appropriate control group for this study, we are unable to say conclusively whether we could have achieved similar rates of MRI success in this population without the use of our MRI training protocol or another preparation method. To our knowledge, there are no published data regarding non-sedated MRI success rates in 7-year-old children in Colombia, or anywhere else in Latin America, which further limits our ability to determine the impact of our training protocol on these high rates of success. We did not collect data on number of repeat sequences or study length, nor did we grade individual sequence quality; all of these would be useful metrics in future studies of non-sedate MRI acquisition. Our criteria in grading motion artifact were purely qualitative; the implementation of more precise criteria when grading image quality would also improve comparability of the results of studies such as this one. Further, we did not collect data on the success rate in this population using a commercial mock MRI scanner, as opposed to a play tunnel, since this was not available, so we cannot compare the success rates of these two approaches to MRI training. However, Fletcher et al. did not find a significant difference in success rates between mock scanners and other familiarization techniques such as play-based training in a sample of 122 children aged 3-7 years undergoing clinical MRI scans [28].
This study only conducted MRI training in a group setting, and children were always brought to the radiology center with peers, so we are unable to independently evaluate the contributions of the peer support environment and the MRI training protocol itself to the success rate of our population. It is possible that the efficacy of the MRI training protocol would have differed had the training been conducted in a one-on-one format with research staff and children, as most other studies have done. Finally, we did not collect information on the amount of time, if any, that parents or caregivers spent reviewing the “My MRI Day” booklet with their child, practicing lying still, or otherwise contributing to their child’s preparation prior to the scan, in addition to the group training conducted by the study team. Parents in our cohort have variable literacy skills [38], which may have also impacted whether children benefitted from additional preparation outside of the training session, as parents with lower literacy skills may have been less likely to review the MRI booklet with their child. We also did not collect information on satisfaction with the MRI booklet or to what extent participants found the booklet engaging. These factors may have made a difference in the success of each child and should be monitored more closely in future studies.
Future researchers can use this MRI training protocol in other low-resource settings to provide effective, easy-to-implement familiarization prior to an MRI. It is important to note that numerous studies have found age to be an important predictive feature of MRI scan success [15, 28, 39], so this protocol may need to be adapted for other age groups given that it was exclusively piloted with 7-year-olds. We did not study the efficacy of this protocol on a diverse age range of children. It is important to note that this case study presents data from a highly homogeneous, typically developing pediatric population recruited specifically to match the sociodemographic characteristics of cases in a separate study of child neurodevelopment. This protocol may require adaptation for clinical usage, as well as for research conducted in populations with different cultural, socioeconomic, or geographical characteristics, or to improve compatibility with distinct healthcare infrastructures.
Conclusion
In this case study of MRI training feasibility, children from a rural/semi-rural setting in Colombia demonstrated similar or greater success in non-sedated MRIs after a low-cost group training as compared to similar-aged children seen in high-resource urban medical centers, with 89% overall scan success. The MRI familiarization methods described in this report may help to maximize non-sedated MRI success among children in low-resource and international settings and the group format that allows for peer modeling may be an effective method for increasing MRI success across pediatric studies. Future research with a well-matched control group that does not receive MRI training, or receives a different type of training, prior to scan acquisition is needed to accurately determine the impact of this familiarization protocol on non-sedated MRI success.
Data, materials, and/or code availability
The data presented in this study are available upon reasonable request from the corresponding author. The data are not publicly available due to privacy considerations for participating children and families. A template version of the MRI training booklet described in the article is available as supplementary material to this article.
References
Momen AA, Jelodar G, Dehdashti H (2011) Brain magnetic resonance imaging findings in developmentally delayed children. Int J Pediatr 2011:386984. https://doi.org/10.1155/2011/386984
Battaglia A, Carey JC (2003) Diagnostic evaluation of developmental delay/mental retardation: an overview. Am J Med Genet C Semin Med Genet 117C:3–14. https://doi.org/10.1002/ajmg.c.10015
Shen FX, Wolf SM, Bhavnani S et al (2021) Emerging ethical issues raised by highly portable MRI research in remote and resource-limited international settings. NeuroImage 238:118210. https://doi.org/10.1016/j.neuroimage.2021.118210
Dong S-Z, Zhu M, Bulas D (2019) Techniques for minimizing sedation in pediatric MRI. J Magn Reson Imaging 50:1047–1054. https://doi.org/10.1002/jmri.26703
Barnea-Goraly N, Weinzimer SA, Ruedy KJ et al (2014) High success rates of sedation-free brain MRI scanning in young children using simple subject preparation protocols with and without a commercial mock scanner–the Diabetes Research in Children Network (DirecNet) experience. Pediatr Radiol 44:181–186. https://doi.org/10.1007/s00247-013-2798-7
de Bie HMA, Boersma M, Wattjes MP et al (2010) Preparing children with a mock scanner training protocol results in high quality structural and functional MRI scans. Eur J Pediatr 169:1079–1085. https://doi.org/10.1007/s00431-010-1181-z
Vanderby SA, Babyn PS, Carter MW et al (2010) Effect of anesthesia and sedation on pediatric MR imaging patient flow. Radiology 256:229–237. https://doi.org/10.1148/radiol.10091124
Davidson AJ, Sun LS (2018) Clinical evidence for any effect of anesthesia on the developing brain. Anesthesiology 128:840–853. https://doi.org/10.1097/ALN.0000000000001972
Jevtovic-Todorovic V (2018) Exposure of developing brain to general anesthesia: what is the animal evidence? Anesthesiology 128:832–839. https://doi.org/10.1097/ALN.0000000000002047
US Food and Drug Administration (2017) FDA Drug Safety Communication: FDA review results in new warnings about using general anesthetics and sedation drugs in young children and pregnant women. 2017-02-05. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-approves-label-changes-use-general-anesthetic-and-sedation-drugs
Barkovich MJ, Xu D, Desikan RS et al (2018) Pediatric neuro MRI: tricks to minimize sedation. Pediatr Radiol 48:50–55. https://doi.org/10.1007/s00247-017-3785-1
Yerys BE, Jankowski KF, Shook D et al (2009) The fMRI success rate of children and adolescents: typical development, epilepsy, attention deficit/hyperactivity disorder, and autism spectrum disorders. Hum Brain Mapp 30:3426–3435. https://doi.org/10.1002/hbm.20767
Artunduaga M, Liu CA, Morin CE et al (2021) Safety challenges related to the use of sedation and general anesthesia in pediatric patients undergoing magnetic resonance imaging examinations. Pediatr Radiol 51:724–735. https://doi.org/10.1007/s00247-021-05044-5
De Amorim e Silva C, Mackenzie A, Hallowell L et al (2006) Practice MRI: reducing the need for sedation and general anaesthesia in children undergoing MRI. Australas Radiol 50:319–323. https://doi.org/10.1111/j.1440-1673.2006.01590.x
Dégeilh F, Lacombe-Barrios J, Tuerk C et al (2023) Behavioral-play familiarization for non-sedated magnetic resonance imaging in young children with mild traumatic brain injury. Pediatr Radiol 53:1153–1162. https://doi.org/10.1007/s00247-023-05592-y
Bharti B, Malhi P, Khandelwal N (2016) MRI customized play therapy in children reduces the need for sedation - a randomized controlled trial. Indian J Pediatr 83:209–213. https://doi.org/10.1007/s12098-015-1917-x
Carter AJ, Greer M-LC, Gray SE, Ware RS (2010) Mock MRI: reducing the need for anaesthesia in children. Pediatr Radiol 40:1368–1374. https://doi.org/10.1007/s00247-010-1554-5
Suzuki A, Yamaguchi R, Kim L et al (2023) Effectiveness of mock scanners and preparation programs for successful magnetic resonance imaging: a systematic review and meta-analysis. Pediatr Radiol 53:142–158. https://doi.org/10.1007/s00247-022-05394-8
Mulkey SB, Peyton C, Ansusinha E et al (2023) Preschool neurodevelopment in Zika virus-exposed children without congenital Zika syndrome. Pediatr Res 94:178–184. https://doi.org/10.1038/s41390-022-02373-5
Chica-Olmo J, Sánchez A, Sepúlveda-Murillo FH (2020) Assessing Colombia’s policy of socio-economic stratification: an intra-city study of self-reported quality of life. Cities 97:102560. https://doi.org/10.1016/j.cities.2019.102560
Wechsler D (2012) Wechsler preschool and primary scale of intelligence—fourth edition. Psychol Corp San Antonio TX
Henderson SE, Sugden D, Barnett AL (1992) Movement assessment battery for children-2. Res Dev Disabil
Gioia GA, Isquith PK, Guy SC, Kenworthy L (2000) Behavior rating inventory of executive function: BRIEF. Psychological Assessment Resources Odessa, FL
Achenbach TM (2001) Manual for ASEBA school-age forms & profiles. Univ Vt Res Cent Child Youth Fam
Harrison PL, Oakland T (2000) Adaptive behavior assessment system. Psychological Corporation San Antonio, TX
Harris PA, Taylor R, Thielke R et al (2009) Research electronic data capture (REDCap)--a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inf 42:377–381. https://doi.org/10.1016/j.jbi.2008.08.010
Woodward K, Spencer APC, Jary S, Chakkarapani E (2023) Factors associated with MRI success in children cooled for neonatal encephalopathy and controls. Pediatr Res 93:1017–1023. https://doi.org/10.1038/s41390-022-02180-y
Fletcher S, Lardner D, Bagshawe M et al (2023) Effectiveness of training before unsedated MRI scans in young children: a randomized control trial. Pediatr Radiol 53:1476–1484. https://doi.org/10.1007/s00247-023-05647-0
Bitta M, Kariuki SM, Abubakar A, Newton CRJC (2017) Burden of neurodevelopmental disorders in low and middle-income countries: a systematic review and meta-analysis. Wellcome Open Res 2:121. https://doi.org/10.12688/wellcomeopenres.13540.3
Dix-Cooper L, Eskenazi B, Romero C et al (2012) Neurodevelopmental performance among school age children in rural Guatemala is associated with prenatal and postnatal exposure to carbon monoxide, a marker for exposure to woodsmoke. Neurotoxicology 33:246–254. https://doi.org/10.1016/j.neuro.2011.09.004
Dórea JG (2021) Exposure to environmental neurotoxic substances and neurodevelopment in children from Latin America and the Caribbean. Environ Res 192:110199. https://doi.org/10.1016/j.envres.2020.110199
Flores-Mendoza C, Ardila R, Gallegos M, Reategui-Colareta N (2021) General intelligence and socioeconomic status as strong predictors of student performance in Latin American schools: evidence from PISA items. Front Educ 6:632289. https://doi.org/10.3389/feduc.2021.632289
Manrique-Millones DL, Pineda-Marin CP, Millones-Rivalles RB, Dimitrova R (2021) The 7Cs of positive youth development in Colombia and Peru: a promising model for reduction of risky behaviors among youth and emerging adults. In: Dimitrova R, Wiium N (eds) Handbook of positive youth development: advancing research, policy, and practice in global contexts. Springer International Publishing, Cham, pp 35–48
Schady N (2006) Early childhood development in Latin America and the Caribbean. Policy Research Working Paper; No. 3869. World Bank, Washington, DC
van Velez A, Talero-Gutierrez C, Gonzalez-Reyes R (2007) Prevalence of delayed neurodevelopment in children from Bogotá, Colombia, South America. Neuroepidemiology 29:74–77. https://doi.org/10.1159/000109499
Weiler-Wichtl LJ, Fries J, Fohn-Erhold V et al (2023) Initial evidence for positive effects of a psychological preparation program for MRI iMReady in children with neurofibromatosis type I and brain tumors—how to meet the patients’ needs best. J Clin Med 12:. https://doi.org/10.3390/jcm12051902
Thieba C, Frayne A, Walton M et al (2018) Factors associated with successful MRI scanning in unsedated young children. Front Pediatr 6:146. https://doi.org/10.3389/fped.2018.00146
Mulkey SB, Corn E, Williams ME et al (2024) Neurodevelopmental Outcomes of Normocephalic Colombian Children with AntenatalZika Virus Exposure at School Entry. Pathogens 13:170. https://doi.org/10.3390/pathogens13020170
Simhal AK, Filho JOA, Segura P et al (2021) Predicting multiscan MRI outcomes in children with neurodevelopmental conditions following MRI simulator training. Dev Cogn Neurosci 52:101009. https://doi.org/10.1016/j.dcn.2021.101009
Acknowledgements
The authors thank the families who participated in the study. They would like to thank Cinthia Rojas, Maria Paulina Quintero, and Eleonora Mozo for their help in facilitating the MRI research collaboration at Sabbag Radiólogos. They would also like to thank Ana Cecilia Perez, research coordinator at BIOMELab, for implementing the MRI training protocol and scheduling MRI appointments, Samir Rodríguez Charris for transporting participants to the MRI site, and Adalberto Nieto and Leonardo Escalante for acquiring the MRI sequences at Sabbag Radiólogos.
Funding
Research reported in this publication was supported by the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the National Institutes of Health under Award Numbers R01HD102445 and the District of Columbia Intellectual and Developmental Disabilities Research Center (DC-IDDRC) Award P50HD105328 by NICHD (PI, T. Haydar).
Author information
Authors and Affiliations
Contributions
Conceptualization: Sarah Mulkey, Elizabeth Corn, Carlos Cure; methodology: Elizabeth Corn, Sarah Mulkey, Raul Tarud, Gilbert Vezina, Catherine Limperopoulos, Madison Berl; validation: Elizabeth Corn, Regan Andringa-Seed; formal analysis and investigation: Elizabeth Corn, Regan Andringa-Seed, Robert Podolsky; data curation: Elizabeth Corn, Regan Andringa-Seed, Robert Podolsky; writing - original draft preparation: Elizabeth Corn, Regan Andringa-Seed, Meagan Williams, Sarah Mulkey; writing - review and editing: Elizabeth Corn, Regan Andringa-Seed, Meagan Williams, Margarita Arroyave-Wessel, Raul Tarud, Gilbert Vezina, Robert Podolsky, Kushal Kapse, Catherine Limperopoulos, Madison Berl, Carlos Cure, Sarah Mulkey; visualization: Elizabeth Corn, Regan Andringa-Seed, Meagan Williams, Robert Podolsky; supervision: Sarah Mulkey, Carlos Cure; project administration: Elizabeth Corn, Sarah Mulkey, Margarita-Arroyave-Wessel, Carlos Cure; funding acquisition: Sarah Mulkey; resources: Raul Tarud, Carlos Cure
Corresponding author
Ethics declarations
Ethics approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Children’s National Hospital and the Institutional Review Committee and the Independent Committee on Research Ethics (CIRCIE), Barranquilla, Colombia (IRB Pro00012759, most recent approval 04/28/2023). This study is registered on ClinicalTrials.gov, NCT04398901.
Consent
Informed consent was obtained from all subjects involved in the study. Additional informed consent was obtained from all individual participants for whom identifying images are included in this article.
Conflicts of interest
None
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Corn, E., Andringa-Seed, R., Williams, M.E. et al. Feasibility and success of a non-sedated brain MRI training protocol in 7-year-old children from rural and semi-rural Colombia. Pediatr Radiol 54, 1513–1522 (2024). https://doi.org/10.1007/s00247-024-05964-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-024-05964-y