Abstract
Background
Technetium 99mTc-pertechnetate is the most common and accurate noninvasive method of preoperative investigation for Meckel diverticulum. Despite introducing various methods to increase the sensitivity of the study, there are many case reports of false-negative Meckel scans. A repeat scan is sometimes requested in patients with a high suspicion for Meckel diverticulum and negative or equivocal first Meckel scan.
Objectives
The purpose of this retrospective study is to evaluate the value of repeat scintigraphy for these patients.
Materials and methods
Seven hundred fifty-three Meckel scans were recorded retrospectively. In 33 cases (22 male and 11 female; mean age: 6.8 years), the Meckel scintigraphy was repeated either due to a high clinical suspicion of Meckel diverticulum and a negative study (n = 21) or due to equivocal findings in the first scan (n = 12). The study was interpreted as positive if an abnormal focal activity was identified in the abdomen and pelvis during the procedure. The results were correlated with pathology and clinical symptoms.
Results
Seven out of 12 (58%) equivocal studies were positive on the second study. Six of them were proven to be positive at operation (confirmed by pathology) while one of them was negative on laparoscopy. From 21 negative first scans with a high suspicion for Meckel diverticulum, three (14%) were positive on the second study. All three were proven to be Meckel diverticulum on pathology.
Conclusion
Repeat Meckel scans in patients with equivocal findings on the first study or a negative result with a high clinical suspicion for a Meckel diverticulum are useful especially in cases in which the first study had been done without appropriate preparation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meckel diverticulum is the most common congenital anomaly of the gastrointestinal tract due to persistence of the omphalomesenteric duct [1]. This anomaly represents a true diverticulum and may contain ectopic gastric tissue in about 50–60% of the cases [2, 3]. The secretion from ectopic gastric mucosa may lead to pain, bleeding and/or perforation [2–4]. More than 50% of patients with complications are younger than 2 years of age and bleeding accounts for most cases [2, 5]. In the majority of cases, the patients are asymptomatic and a Meckel diverticulum may often be discovered incidentally. However, even in those cases with complications and symptoms, the diagnosis is often difficult.
Various imaging techniques, including conventional barium study, high-resolution ultrasound and CT scan, have been introduced for detection of Meckel diverticulum [6]. 99mTc-pertechnetate is the most common and accurate noninvasive method of preoperative investigation for Meckel diverticulum containing ectopic gastric mucosa with overall sensitivity of 85%, specificity of 95% and accuracy of 90% in children [5]. Despite introducing various methods to increase the sensitivity of the study, there are still many case reports of false-negative and false-positive Meckel scans [7–10]. A repeat scan is sometimes requested in patients with persistent gastrointestinal bleeding and high suspicion for Meckel diverticulum and negative or equivocal first Meckel scan. The purpose of this retrospective study is to evaluate the value of repeat scintigraphy for these patients.
Materials and methods
Patients
This study was approved by our institutional review board. Seven hundred fifty-three Meckel scans, from 2000 to 2013, were reviewed retrospectively. Out of 753 cases, 696 were interpreted as negative, 41 positive and 16 equivocal. In 33 cases (33/753, 4.3%) (22 male and 11 female; mean age: 6.8 years), the Meckel scintigraphy was repeated either due to a high clinical suspicion of Meckel diverticulum and a negative study (n = 21) or due to equivocal findings in the first scan (n = 12). In almost all cases, the reason for a high clinical suspicion was gastrointestinal bleeding with or without pain with any definite underlying diagnosis.
Imaging
Scintigraphy was performed in all patients after 4 h of fasting prior to radiotracer injection. Ranitidine pretreatment was infused for almost all patients 1 h before radiotracer administration (1 mg/kg diluted with saline to 20 ml total volume and infused over 20 min) to reduce gastric secretion and thus increase the accumulation of activity in the gastric mucosa. Barium study was not done for at least 72 h before the study. Previous perchlorate administration or laxatives were avoided before scintigraphy. For studies performed prior to 2011, the administered activity was calculated based on the standardized pediatric dose scales. Studies performed after 2011 were based on the 2010 North American consensus guidelines (1.85 Mbq/kg) [11].
Dynamic images were obtained for 15 min after injection of 99mTc-pertechnetate (128*128 matrix, 60 s per frame) followed by sequential static images for 45 to 60 min in anterior, posterior and lateral projections (256*256 matrix, 2–5 min per view). The images were obtained in a supine position with large field dual head equipped with a low-energy high-resolution collimator.
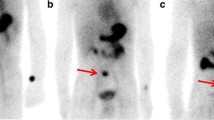
The reports were reviewed in this retrospective study. The study was reported as positive if an abnormal focal activity was identified in the abdomen and pelvis that could not be ascribed to a physiological activity. A typical pattern was defined as a focal abnormal activity, usually in the right lower quadrant (Fig. 1), visualized at the same time as activity in the normal gastric mucosa. However, the activity might be seen later than the stomach and anywhere within the abdomen and pelvic region in atypical forms of Meckel diverticulum scans. The study was reported as equivocal if a focal area of activity was seen in the abdomen and pelvis, but the differentiation between a focal activity due to Meckel diverticulum and physiological activity in the kidneys, ureter, bladder or blood pool activity was not possible even with the aid of lateral views, delayed views and post void images (Fig. 2). The study was reported as negative when no definite focal increased activity was identified in the abdomen and pelvis during the procedure or if the activity was proven to be due to a physiological radiotracer activity.
Meckel scans in a 7-year-old boy with painless rectal bleeding. a The initial scan shows foci of activity were detected in the abdomen likely due to renal activity. On the right lateral view, there was a suspicious focal activity anterior and inferior to the kidney (arrows). This could be due to the activity in the ureter, or bowel. However, an ectopic gastric mucosa could not be completely ruled out. b Repeat study after 1 day revealed a focal activity in the right abdomen below the right kidney (arrows), which was confirmed to be Meckel diverticulum with ectopic gastric mucosa
The positive results were confirmed by pathology. The negative studies were also reviewed to identify any possible false-negative results. Other diagnostic imaging modalities, colonoscopy and upper gastrointestinal findings as well as follow-up clinical symptoms were reviewed in negative Meckel diverticulum scans.
Results
Details of the patients with a repeat scan are shown in Table 1. From 12 equivocal first Meckel diverticulum studies, 7 (58%) were reported as positive, 4 as negative and 1 as equivocal in the repeat scans. From seven positive studies on the second scans, six were proven to be Meckel diverticulum in surgery (confirmed by pathology) while one was negative on laparoscopy. Four out of 12 equivocal cases were negative on repeat study. One was diagnosed with ulcerative colitis and for the other three no definite diagnosis was identified. The last equivocal case was again equivocal on the repeat scan. Laparoscopy did not identify a Meckel diverticulum but showed reactive lymphoid follicles in the abdomen adjacent to the bowels.
From 21 negative first scans with a high suspicion for Meckel diverticulum, 18 (86%) were negative and 3 (14%) were positive on the repeat study. All three were proven to be Meckel diverticulum on pathology. A number of other pathologies, including duodenitis, gastritis, colitis, Crohn disease and juvenile polyp, were identified for the 18 negative/negative scans (Table 1). The exact possible reasons for the negative first study/positive repeat scans in the three cases were not clear. However, in one instance the study had been done without ranitidine preparation, in a second instance washout of the activity might have played a role and in the last instance the Meckel diverticulum was located just behind the bladder.
Discussion
The sensitivity of the 99mTc-pertechnetate Meckel scan for detection of the Meckel diverticulum containing ectopic gastric mucosa is quite high in children (almost 85–90%). The sensitivity is much lower in adults (about 62%) [12]. Many factors may affect the sensitivity of the scintigraphy (Table 2). Patient preparation, the technique of study and paucity of ectopic gastric mucosa are among the factors that may affect the result. Several techniques have been introduced to increase the sensitivity. Premedication with histamine H2 blockers (cimetidine, ranitidine, famotidine), glucagon and pentagastrin, bladder lavage, and nasogastric suctioning and recently SPECT and SPECT/CT have been reported to increase the sensitivity [4, 12–14]. A repeat Meckel scan is another method to increase the sensitivity and accuracy. Our retrospective study shows that a repeat Meckel scan can be considered in patients with an equivocal finding on the first study or a negative result with a high clinical suspicion for Meckel diverticulum. In almost all cases in our study, the reason for high clinical suspicion was repeated gastrointestinal bleeding without any definite underlying diagnosis in other imaging or non-imaging investigations.
One of the reasons of a false-negative Meckel diverticulum scan is insufficient gastric mucosa. This may happen more frequently in adults since diverticula with enough gastric mucosa are usually diagnosed earlier in life due to the higher risk of complications. Unfortunately, due to the retrospective nature of the study, we could not assess the size of the ectopic gastric mucosa in the pathology. However, at least in one of our cases with a negative first study/positive repeat scan, the size of the ectopic gastric mucosa was very small and located at the tip of Meckel diverticulum (case 14).
Using pentagastrin to increase acid production (thus increasing the activity of the mucus-producing cells), histamine receptor antagonists to reduce secretions from the gastric mucosal cells (thus increasing the accumulations of the radiotracer) and glucagon to suppress the peristalsis and transit of any secreted pertechnetate through the small bowel have been introduced to increase the sensitivity of the scintigraphy [4, 12, 13, 14]. In our cases, one of the patients (case 15) with a negative first scan/positive repeat study did not receive ranitidine before the first scan (Fig. 3). The positive repeat scan can be due to increased accumulation of the radiotracer after ranitidine preparation, which is in line with the result of other studies reporting a higher sensitivity with cimetidine or ranitidine preparation [13, 15].
Meckel scans in a 9-month-old girl. a Anterior view 40–45 min after the radiotracer injection. No focal activity is detected. The early images were also negative (not shown here). b Repeat study after 1 month revealed a focal activity in the left abdomen that was confirmed to be Meckel diverticulum on pathology (arrow)
Saline lavage of the bladder during the procedure is another method to increase the sensitivity by removing the activity that may obscure Meckel diverticulum [16]. Post-void imaging and lateral views are usually helpful to differentiate a Meckel diverticulum activity from urinary activity. In our study, there was one case in which a repeat scan in a negative first scan showed a very faint small activity located just behind the upper portion of the bladder. This was visualized only on the posterior view. The study was reported as a positive study in the repeat scan and was proven to be Meckel diverticulum on pathology. In this case, the size of ectopic gastric mucosa was also very small.
Further evaluation with SPECT or SPECT/CT is another method to increase the sensitivity when the clinical suspicion for Meckel diverticulum is high and the planar images are negative or equivocal [17–19]. This method is especially useful when the ectopic gastric mucosa is located behind the bladder. However, SPECT study is not always feasible especially with a restless patient. Further evaluation with SPECT study is necessary in case of any suspicious activity around the bladder or other hot organs and if the patient cannot void completely and there is a contraindication for catheterization,. SPECT study would have probably detected one of the cases in our series on the first scan with a small-sized ectopic gastric mucosa near to the bladder (case 14).
Dilution of activity is another possibility for a false-negative study, which may happen when a patient has active bleeding [20]. This could be an explanation for one of our cases as illustrated in Fig. 4. None of the three negative first study/positive repeat scan had pain during the first study to suggest intussusception or vascular compromise as the possible causes of false-negative study. However, these possibilities cannot be excluded based on the clinical history. Irrespective of the possible causes of a false-negative study, our study shows that if the clinical suspicion for Meckel diverticulum is high, a repeat study will be useful for a more accurate diagnosis.
Meckel diverticulum scintigraphy in a 2-year-old boy with gastrointestinal bleeding. Dynamic images as well as delayed static views at 15, 30 and 45 min and lateral and posterior views were acquired (not shown here). Only images at 10, 15, 30 and 45 min are shown. a First study didn’t show any definite focal increased activity in the abdomen and pelvis to suggest an ectopic gastric mucosa. b Repeat study after 2 months revealed a focal activity visualized early at the same time with the gastric activity in the right lower quadrant (arrows). This activity was not seen on delayed images at 30 or 45 min likely due to the washout of the activity (bleeding or bowel movement). Meckel diverticulum was confirmed by pathology
A number of other pathologies were detected in patients with negative first scans and negative repeat studies. These include gastritis, duodenitis, ileitis, colitis, polyps, Crohn disease and anal fissure. Although laparoscopy or surgery was not done in the majority of patients in this group, none of them showed a Meckel diverticulum on follow-up studies or on surgical pathology.
On the other hand, in those cases with equivocal findings on Meckel diverticulum scintigraphy, a repeat scan may be considered to differentiate between a true-positive vs. a false-positive finding. In our study, 58% of the repeat scans (7/12) from the first equivocal scans were positive. Meckel diverticulum was proven in 6 out of 7 (85%). Repeat scan may be helpful to exclude the false-positive results and to avoid unnecessary further invasive investigation or treatment in some of the patients with equivocal findings. Many reasons have been reported for a false-positive Meckel diverticulum scan (Table 3) [2, 5, 10, 12]. In addition to contamination, physiological excretion of the activity into the bowel, any activity related to the urinary system (dilated pelvis, ureter, ureterocele, vesicoureteral reflux, etc.) and other possibilities such as bowel vascular anomalies, bowel ulcerations, inflammation and obstruction are reported as false-positive findings on Meckel diverticulum scan (Table 3).
In our study, the repeat scan correctly identified 6 Meckel diverticulum cases out of 12 equivocal Meckel diverticulum results (50%). In one case, a repeat scan was positive but laparoscopy was negative for Meckel diverticulum. No definite diagnosis was identified in this case. A number of pathologies were detected for the other equivocal first studies with no positive repeat scan including duodenitis, gastritis, multiple angiodysplasia, reactive lymphoid follicles and ulcerative colitis. Thus, a repeat scan for the studies with equivocal results may be considered especially if the clinical suspicion for Meckel diverticulum is high.
It should be emphasized that in order to avoid a repeat scan and to reduce the radiation dose to patients it is necessary to strictly follow the recommended protocols for patient preparation and image acquisition including post voiding images, catheterization and SPECT study, if possible. The effective dose for a 5-year-old child in a Meckel scan is about 1.6 mSv assuming that the body weight is about 20 kg and the dose is 0.05 mCi/Kg (1.85 Mbq/kg) [21].
Our study had some limitations due to the retrospective nature of the study. The exact size of the ectopic gastric mucosa, type of cells (parietal and chief cells) and the location of ectopic gastric mucosa in Meckel diverticulum were not exactly described in all pathology reports. Only in one case was it reported that the size of ectopic gastric mucosa was very small and located in the tip of the Meckel diverticulum. Thus, we could not make any conclusion in terms of the relationship between the size and visualization of ectopic gastric mucosa. SPECT study was not done in our patient population. This is another limitation that may affect the sensitivity. This study is unique in that it evaluates the usefulness of a repeat study when the first study is equivocal or when the first study is negative but clinical suspicion is high and other investigations are not diagnostic.
Conclusion
Repeat Meckel scan in patients with equivocal findings on the first study or a negative result with a high clinical suspicion for Meckel diverticulum may detect Meckel diverticulum containing ectopic gastric mucosa especially in cases in which the first study had been done without appropriate preparation or without SPECT acquisition.
References
Satya R, O’Malley JP (2005) Case 86: Meckel diverticulum with massive bleeding. Radiology 236:836–840
Park JJ, Wolff BG, Tollefson MK et al (2005) Meckel diverticulum: the Mayo Clinic experience with 1476 patients (1950–2002). Ann Surg 241:529–533
Matsagas MI, Fatouros M, Koulouras B et al (1995) Incidence, complications, and management of Meckel’s diverticulum. Arch Surg 130:143–146
Sfakianakis GN, Conway JJ (1981) Detection of ectopic gastric mucosa in Meckel’s diverticulum and in other aberrations by scintigraphy: ii. indications and methods–a 10-year experience. J Nucl Med 22:732–738
Bemelman WA, Hugenholtz E, Heij HA et al (1995) Meckel’s diverticulum in Amsterdam: experience in 136 patients. World J Surg 19:734–736
Elsayes KM, Menias CO, Harvin HJ et al (2007) Imaging manifestations of Meckel’s diverticulum. AJR Am J Roentgenol 189:81–88
Wilton G, Froelich JW (1982) The “false-negative” Meckel’s scan. Clin Nucl Med 7:441–443
Ariga M, Suga K, Matsunaga N et al (2001) Failure to detect a huge Meckel’s diverticulum with abundant ectopic gastric mucosa on gastric mucosal scintigraphy with Tc-99 m pertechnetate. Clin Nucl Med 26:470–471
Sharma RK, Jain VK (2008) Emergency surgery for Meckel’s diverticulum. World J Emerg Surg 3:27
Tulchinsky M (2006) Meckel’s scan: pitfall in patients with active small bowel bleeding. Clin Nucl Med 31:814–816
Gelfand MJ, Parisi MT, Treves ST et al (2011) Pediatric radiopharmaceutical administered doses: 2010 North American consensus guidelines. J Nucl Med 52:318–322
Schwartz MJ, Lewis JH (1984) Meckel’s diverticulum: pitfalls in scintigraphic detection in the adult. Am J Gastroenterol 79:611–618
Petrokubi RJ, Baum S, Rohrer GV (1978) Cimetidine administration resulting in improved pertechnetate imaging of Meckel’s diverticulum. Clin Nucl Med 3:385–388
Singh PR, Russell CD, Dubovsky EV et al (1978) Technique of scanning for Meckel’s diverticulum. Clin Nucl Med 3:188–192
Rerksuppaphol S, Hutson JM, Oliver MR (2004) Ranitidine-enhanced 99mtechnetium pertechnetate imaging in children improves the sensitivity of identifying heterotopic gastric mucosa in Meckel’s diverticulum. Pediatr Surg Int 20:323–325
Datz FL, Taylor A Jr (1985) Utility of bladder lavage in Meckel’s scanning. Clin Nucl Med 10:804–806
Dillman JR, Wong KK, Brown RK et al (2009) Utility of SPECT/CT with Meckel’s scintigraphy. Ann Nucl Med 23:813–815
Schneider P, Duren C, Reiners C (2010) SPECT-CT image fusion could enhance Meckel scan. World J Pediatr 6:281
Papathanassiou D, Liehn JC, Meneroux B et al (2007) SPECT-CT of Meckel diverticulum. Clin Nucl Med 32:218–220
Sagar J, Kumar V, Shah DK (2006) Meckel’s diverticulum: a systematic review. J R Soc Med 99:501–505
Spottswood SE, Pfluger T, Bartold SP et al (2014) SNMMI and EANM practice guideline for meckel diverticulum scintigraphy 2.0. J Nucl Med Technol 42:163–169
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vali, R., Daneman, A., McQuattie, S. et al. The value of repeat scintigraphy in patients with a high clinical suspicion for Meckel diverticulum after a negative or equivocal first Meckel scan. Pediatr Radiol 45, 1506–1514 (2015). https://doi.org/10.1007/s00247-015-3340-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-015-3340-x