Abstract
The harbor seals (Phoca vitulina) from Helgoland (North Sea) and Anholt (Kattegat, Baltic Sea) are top predators within the marine food web and an indicator species of the environmental contamination. Furthermore, they are a main tourist attraction. Despite these important roles, little is known about the health and pollutant contamination of these seals. The objective of this study was therefore to investigate 18 essential and nonessential/toxic elements (Al, As, Be, Ca, Cr, Cu, Fe, K, Mn, Mo, Ni, P, Pb, Rb, S, Se, Sr, and Zn) in blood samples using inductively coupled plasma mass spectrometry and total X-ray-fluorescence spectrometry. Blood concentrations of mineral nutrients, such as Ca, K, P, and S, were within the reference ranges described for harbor seals. Likewise, for the trace elements, As, Be, Rb, Se, and Sr, no significant differences were observed compared with previous studies. Interestingly, blood concentrations of nine nonessential as well as essential trace metals (Al, Cr, Cu, Fe, Mn, Mo, Ni, Pb, Zn) measured significantly lower in the offshore living seals from Helgoland and Anholt compared with results obtained from animals living close to urbanized areas, such as the Wadden Sea and Elbe estuary. This suggests that industrial emissions, sewage deposition, shipping traffic and dredging tasks might be the cause of increased metal concentrations of inshore harbor seals.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The harbor seal (Phoca vitulina) is the most abundant marine mammal in the North and Baltic Sea. Seals living closed to coastal areas are strongly influenced by anthropogenic activities impacting on their habitat, such as fishery and environmental pollution. For these ecosystems, organic and metal contaminant concentrations have been analyzed in several tissues, e.g., in liver, kidney, or blubber from harbor seals, obtained during postmortem investigations (Boon et al. 2002; Das et al. 2008; Ahrens et al. 2009; Kuenstl et al. 2009; Weijs et al. 2009a, b, c; Agusa et al. 2011; Kakuschke et al. 2012; Dietz et al. 2012). Blood sampling of harbor seals is considered minimally invasive and allows the analyses of pollutants, biomarkers, and health parameters additionally to in vitro cell experiments from living animals. For the North Sea, several investigations have been performed to measure contaminants (Griesel et al. 2006; Das et al. 2008; Griesel et al. 2008; Kakuschke et al. 2009; Weijs et al. 2009a, b, c; Kakuschke et al. 2010b), assess immune cell functions (De Swart 1995; Ross 1995; Kakuschke et al. 2005; Das et al. 2008; Dupont et al. 2013), or analyse biomarkers (Fonfara et al. 2008; Hasselmeier et al. 2008; Kakuschke et al. 2010a; Grebe et al. 2010, 2011, 2012; Kakuschke et al. 2013) in blood samples of harbor seals. Most of these studies focused on animals closed to urbanized areas along the Wadden Sea coast.
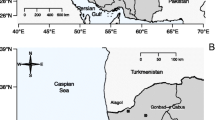
Helgoland, which is located 46 km off the German coastline (54°10′58.9″N 7°53′52.2″E), is characterized by its unique flora and fauna. In particular, the presence of a harbor seal population with up to 300 animals makes Helgoland highly attractive for tourists. Live and dead seals are regularly counted and investigations of behavior and animal movements have been performed in previous studies (Adelung and Müller 2008; Abt 2011a, b; van Neer et al. 2015). However, only a few health and pollution studies have been realized. In three previous studies, we published acute phase protein blood concentrations of haptoglobin, C-reactive protein, and transferrin from harbor seals (Kakuschke et al. 2010a; Grebe et al. 2011; Kakuschke et al. 2013). Additionally, a postmortem study of seals from Helgoland described macroscopic findings, blood parameters as well as metal and organic contaminants of several body fluids and tissues (Kakuschke et al. 2012).
Anholt is located 50 km off the Danish coastline (56°42′50.7″N 11°33′16.9″E) in the Kattegat, Baltic Sea. Up to 1000 harbor seals use the island as a haul-out site (Chudzinska 2009). The seal population of Anholt was the first affected by the phocine distemper virus epidemics (PDV) in 1988 and 2002 (Härkönen et al. 2006; Hall et al. 2006). In 2007, another increase of dead harbor seals was reported (Härkönen et al. 2008). Researchers investigated the population status, movement patterns, diet, human interactions, and diving behaviour of seals in southern Scandinavia including Anholt (Chudzinska 2009; Andersen 2011). However, pollution studies of this area are limited (Olsson et al. 1994; Dietz et al. 2012).
The present study reports the first multielement analysis of electrolytes, essential, and toxic trace elements in blood samples from free-ranging, healthy harbor seals from Helgoland and Anholt. Furthermore, results were compared with published data obtained from animals living in different North Sea locations, and from diseased animals.
Materials and Methods
Animals Investigated and Blood Sample Collection
The blood samples analyzed during this study were collected in 2005 and 2006 from 13 free-ranging harbor seals (10 from Helgoland and 3 from Anholt; Table 1; Fig. 1). The seals were captured in individual tube nets and manually restrained for physical examination, to assess sex, length and weight, and to collect blood samples. The maximum time for which the animals were kept for investigations and sampling was 1 h. During this time, the seals were under continuous veterinary observation. All animals appeared healthy on physical examination, showed no clinical signs of disease, and were of normal nutritional status. The seals were classified into two age groups, <1 year and >2 years, based on length and weight.
Blood samples were collected by venipuncture of the epidural vertebral vein using a 1.2 mm × 100 mm needle (TSK-Supra, TSK Laboratory, Japan) and filled into specific lithium heparin (LH) monovettes for metal analysis (Sarstedt AG & Co). The tubes were carefully agitated and kept at ambient temperature during the capture event. Afterwards, the blood samples were transported to the laboratories of the Biologische Anstalt Helgoland, Research and Technology Centre in Büsum, and Helmholtz-Zentrum Geesthacht, Germany, for further processing and storage at −80 °C.
Multielement Analysis
Eighteen elements (Al, As, Be, Ca, Cr, Cu, Fe, K, Mn, Mo, Ni, P, Pb, Rb, S, Se, Sr, and Zn) were analyzed in whole blood according to the procedure described in our previous study (Griesel et al. 2008). For multielement measurements, a microwave digestion system (MarsXpress, CEM GmbH, Kamp-Lintfort, Germany) was used. 500 µL of whole blood was pipetted into perfluoralkoxy (PFA) container; 2 mL sub-boiled nitric acid, 1 mL of hydrogen peroxide, and 50 µL internal standard (1 mg L−1 of yttrium, Merck, Darmstadt, Germany) were added, and the container were heated in a three step program up to 180 °C.
The elements were measured with two different analytical methods. Al, Be, Cr, Mn, Mo, Ni, and Pb were analyzed using inductively coupled plasma mass spectrometry (ICP-MS) equipped with a collision cell (Agilent 7500c ICP-MS, Agilent Technologies, Tokyo, Japan). Measurements of As, Ca, Cu, Fe, K, P, Rb, S, Se, Sr, and Zn were performed by total X-ray-fluorescence spectrometry (TXRF) (Atomika TXRF 8030 C, FEI Company, Oberschleissheim, Germany).
For internal quality control, the reliability of the analytical procedures was confirmed with the human reference material Seronorm™ Trace Elements Whole Blood L-2 (SERO AS, Billingstad, Norway) and/or Clin Check® Whole Blood Control Level II (Recipe, Chemicals + Instruments, Munich, Germany). Additionally, reference material was measured in connection with the participation in the NIST/NOAA 2007 Interlaboratory Comparison Exercise for trace elements in blood of marine mammals (Table 2).
Data Analysis
We used the nonparametric Mann–Whitney U test for comparing element blood concentrations between animals from the island Helgoland with the animals from Rømø and Lorenzenplate (data obtained from Griesel et al. 2008). In case of nondetectable concentrations, the 0.5 LOD-value was used for statistical analyses and graphs. The animals from both studies were within the same age group (>2 years). Due to the fact that no gender-related differences had been found in former studies (Griesel et al. 2008; Dupont et al. 2013), the results for males and females were pooled. The results from Anholt seals were not statistically tested due to the small number and different age of animals. All indicated p values are two-tailed. Statistical significance was designated as p ≤ 0.05.
Results and Discussion
To the authors’ knowledge, our study is the first to report 18 element concentrations, including electrolytes, essential and nonessential/toxic elements measured in blood samples of free-ranging, healthy harbor seals from the islands Helgoland and Anholt (Table 3).
Local Differences of Element Concentrations in Blood Samples from Harbor Seals
Median, minimum, and maximum element concentrations were compared between seals of the islands Anholt (Kattegat, Baltic Sea) and Helgoland (North Sea, Germany) with our previous studies on seals from Rømø (Wadden Sea, Denmark), Lorenzenplate (Wadden Sea, Germany), and Elbe estuary (Neuwerk, Germany) (Table 4; Griesel et al. 2008, Kakuschke et al. 2010a).
Helgoland seals showed significantly (Mann–Whitney U test) lower blood concentrations for Cr (p < 0.01), Fe (p < 0.01), Mn (p < 0.01), Mo (p < 0.01), Ni (p < 0.01), Pb (p < 0.05), and Zn (p < 0.01) than seals from Rømø and the Lorenzenplate. Furthermore, results for Al (p < 0.01) were lower in comparison to Rømø seals, Cu (p < 0.01) was lower in comparison to seals from the Lorenzenplate (Fig. 2).
Blood concentrations of Al, Cr, Cu, Fe, Mn, Mo, Ni, Pb, and Zn of free-ranging, obviously healthy harbor seals from Anholt and Helgoland (this study) compared with seals from the Wadden Sea—Rømø and Lorenzenplate (Griesel et al. 2008), and the Elbe estuary (Kakuschke et al. 2010b). The width of the boxes are the 25–75 percentile range, the line inside the box is the Median, the open dot the Mean. The whiskers show the Min and Max values. Black dots are the measured values, whereas the 0.5 LOD-value was used for nondetectable levels
For the trace elements As, Be, Rb, Se, and Sr and the mineral nutrients Ca, K, P, and S, no significant differences were observed between animals from Helgoland and the Wadden Sea locations. Measurements for Ca, K, P, and S were within the normal range described for harbor seals (Griesel et al. 2006).
Despite the small number of animals investigated, the element blood concentrations obtained from Anholt seals were comparable to Helgoland seals (Table 4; Fig. 2). Interestingly, seals living in the Elbe estuary showed the highest blood concentrations for most elements in comparison to other locations (Table 4; Fig. 2; Kakuschke et al. 2010b). However, the animals of Anholt and the Elbe estuary were younger, and it is likely that both, age and localization, have an influence on blood element concentrations.
Despite a reduction of contaminants over the recent years, the Elbe River is still the primary contributor to the contamination of its estuary and the German Bight (Loewe et al. 2006). The Elbe water has four times higher Cu concentrations than the Lake Issel (Western Dutch Wadden Sea), the rivers Ems and Weser (Lower Saxony), as well as Eider (Schleswig–Holstein) are in between (Bakker et al. 2009). Similarly, Pb and Zn concentrations are 3–4 times higher in the rivers Elbe and Weser in comparison to Eider and Ems (Bakker et al. 2009). The high blood concentrations of most elements, including Cu, Pb, and Zn, in seals living in the Elbe estuary are therefore not surprising.
The Wadden Sea areas around Lorenzenplate and Rømø are dominated by North Sea coastal water; however, rivers like Eider, Ribe A, or Rejsby A influence the contamination of these areas (Bakker et al. 2009). This is reflected by Wadden Sea seal blood concentrations of most elements, which were lower than in Elbe seals but higher compared with Helgoland seals. Interestingly, Ni concentrations in the sediment of the northeastern Wadden Sea regions of Dithmarschen, North-Frisia, or Sylt-Rømø were higher and showed an increasing trend over the past years compared with southwest regions of the Wadden Sea (Bakker et al. 2009). Seals from Rømø showed besides elevated Ni blood levels, Ni-specific hypersensitivity reactions, potentially as a result of a chronic metal influence (Kakuschke et al. 2005). Similar immunological reactions were found for the metals Al, Cr, Mo, Pb, Sn, and Ti. These elements (Al, Cr, Mo, Ni, and Pb) also were significantly higher in blood samples from the Wadden Sea seals in comparison to Helgoland seals.
The general picture of a higher contamination of near-urban sites also was found for western Wadden Sea areas, and includes next to metal elements, several organic pollutants. Laane et al. (1999) described the distribution of trace metals in the sediment of the Dutch coastal zone with highest concentrations close to the coast. Dietz et al. (2012) investigated perfluorinated chemicals (PFCs) in seal liver tissue from seven locations ranging from the Wadden Sea of the southern North Sea to the Western Baltic Sea and found the highest concentration of perfluorooctanesulfonic acids (PFOS) in animals from the Wadden Sea.
Decreasing concentrations from estuary to coastal waters and the open sea could be explained by the dilution of estuarine with sea water. This is consistent with the reduction of water metal concentrations in the Germany Bay with an increasing distance from the coast line (Laane et al. 2013). Our results reporting different blood concentrations depending on the localization of the seals support these findings. However, as expected, essential elements physiologically regulated, e.g., Ca, K, P, and S were comparable for all seals investigated, independent of their location.
Differences of Blood Element Concentrations Comparing Healthy and Diseased Harbor Seals
For most elements no differences were detected between Helgoland seals, which were suspected to be healthy, and four diseased animals (Table 5; Kakuschke et al. 2012).
However, Pb blood concentrations of the diseased harbor seals were higher than the maximum value measured in healthy seals. Pb is known to be toxic and a health risk for marine mammals. A case study (Shlosberg et al. 1997) described progressive liver damage and finally death of a bottlenose dolphin resulting from Pb intoxication. Frouin et al. (2010) reported a decreased lymphoblastic proliferation of Pb incubated lymphocytes obtained from blood and lymph node samples. However, in the present study a correlation of health condition and Pb blood concentrations could not conclude unambiguous. First, the blood concentrations of diseased seals from Helgoland were comparable to those of healthy seals living in the Wadden Sea area and Elbe estuary (Griesel et al. 2008; Kakuschke et al. 2010b). Second, the liver, kidney, and muscle Pb concentrations of these diseased animals were mostly below detection limit (Kakuschke et al. 2012).
Conclusions
Although harbor seals are an indicator species of the ecosystem and a tourist attraction, little is known about the seals from the islands Helgoland and Anholt. Our multielement investigation provides a data set of essential and nonessential/toxic elements in blood samples from these animals. For several metals, our findings support other studies showing higher levels of pollutants in animals living in estuary and inshore habitats, which are influenced by industrial emissions and sewage, shipping traffic and dredging tasks, in comparison to seals living further away from the mainland.
References
Abt K (2011a) Meeressäugerfunde an den Küsten Schleswig-Holsteins 2010. Bericht an Nationalparkverwaltung im Landesbetrieb für Küstenschutzm, Nationalpark und Meeresschutz Schleswig-Holstein, Kiel
Abt K (2011b) Robbenzählungen im schleswig-holsteinischen Wattenmeer 2010. Bericht an Nationalparkverwaltung im Landesbetrieb für Küstenschutz, Nationalpark und Meeresschutz Schleswig-Holstein, Kiel
Adelung D, Müller G (2008) Forschungsverbund MINOSplus – Weiterführende Arbeiten an Seevögeln und Meeressäugern zur Bewertung von Offshore – Windkraftanlagen, Teilvorhaben TP6 – „Seehunde in See“ – Untersuchungen zur räumlichen und zeitlichen Nutzung der Nordsee durch Seehunde im Zusammenhang mit der Entwicklung von Offshore-Windenergieanlagen. Ministeriumfür Umwelt, Naturschutz und Reaktorsicherheit, Universität Kiel, Germany
Agusa T, Yasugi S, Lida A, Ikemoto T, Anan Y, Kuiken T, Osterhaus A, Tanabe S, Iwata H (2011) Accumulation features of trace elements in mass-stranded harbor seals (Phoca vitulina) in the North Sea coast in 2002: the body distribution and association with growth and nutrition status. Mar Pollut Bull 62:963–975
Ahrens L, Siebert U, Ebinghaus R (2009) Temporal trends of polyfluoroalkyl compounds in harbor seals (Phoca vitulina) from the German Bight, 1999-2008. Chemosphere 76:151–158
Andersen SM (2011) Harbour seals and human interactions in Danish waters. Dissertation. University of Southern Denmark and Aarhus University
Bakker J, Lüerßen G, Marencic H, Jung K (2009) Hazardous Substances. Thematic Report No. 5.1. In: Marencic H, de Vlas J (eds) Quality status report 2009. Wadden Sea Ecosystem No. 25. Common Wadden Sea Secretariat, Trilateral Monitoring and Assessment Group, Wilhelmshaven, Germany
Boon JP, Lewis WE, Tjoen-A-Choy MR, Allchin CR, Law RJ, de Boer J, ten Hallers-Tjabbes CC, Zegers BN (2002) Levels of polybrominated diphenyl ether (PBDE) flame retardants in animals representing different trophic levels of the North Sea food web. Environ Sci Technol 36:4025–4032
Chudzinska M (2009) Diving behaviour of harbour seals (Phoca vitulina) from the Kattegat. Masterthesis. University of Aarhus, Denmark
Das K, Siebert U, Gillet A, Dupont A, Di-Poi C, Fonfara S, Mazzucchelli G, De Pauw E, De Pauw-Gillet MC (2008) Mercury immune toxicity in harbour seals: links to in vitro toxicity. Environ Health 7:52
De Swart (1995) Impaired immunity in seals exposed to bioaccumulated environmental contaminants. Dissertation. Erasmus University Rotterdam, The Netherlands
Dietz R, Riget FF, Galatius A, Sonne C, Teilmann J, Bossi R (2012) Spatial trends of perfluorochemicals in harbor seals (Phoca vitulina) from Danish waters. Sci Total Environ 414:732–737
Dupont A, Siebert U, Covaci A, Weijs L, Eppe G, Debier C, De Pauw-Gillet MC, Das K (2013) Relationships between in vitro lymphoproliferative responses and levels of contaminants in blood of free-ranging adult harbour seals (Phoca vitulina) from the North Sea. Aquat Toxicol 142(143):210–220
Fonfara S, Kakuschke A, Rosenberger T, Siebert U, Prange A (2008) Cytokine and acute phase protein expression in blood samples of harbour seal pups. Mar Biol 3:337–345
Frouin H, Menard L, Measures L, Brousseau P, Fournier M (2010) T Lymphocyte-proliferative responses of a grey seal (Halichoerus grypus) exposed to heavy metals and PCBs in vitro. Aquat Mamm 36:365–371
Grebe M, Proefrock D, Kakuschke A, Broekaert JAC, Prange A (2010) Metallomics approach for the identification of the iron transport protein transferrin in the blood of harbour seals (Phoca vitulina). Metallomics 2:683–693
Grebe M, Proefrock D, Kakuschke A, Broekaert JAC, Prange A (2011) Absolute quantification of transferrin in blood samples of harbour seals using HPLC-ICP-MS. Metallomics 3:176–185
Grebe M, Proefrock D, Kakuschke A, del Castillo Busto ME, Montes-Bayon M, Sanz-Medel A, Broekaert JAC, Prange A (2012) Comparison of different methods for the absolute quantification of harbour seal transferrin glycoforms using HPLC-ICP-MS. J Anal Atom Spec 27:440–448
Griesel S, Mundry R, Kakuschke A, Fonfara S, Siebert U, Prange A (2006) Mineral elements and essential trace elements in blood of seals of the North Sea measured by total-reflection X-ray fluorescence analysis. Spectrochim Acta B 61:1158–1165
Griesel S, Kakuschke A, Siebert U, Prange A (2008) Trace element concentrations in blood of harbor seals (Phoca vitulina) from the Wadden Sea. Sci Total Environ 392:313–323
Hall AJ, Jepson PD, Goodman SJ, Harkonen T (2006) Phocine distemper virus in the North and European Seas: data and models, nature and nurture. Biol Conserv 131:221–229
Härkönen L, Dietz R, Reijnders P, Teilmann J, Harding K, Hall A, Brasseur S, Siebert U, Goodman SJ, Jepson PD, Rasmussen TD, Thompson P (2006) A review of the 1988 and 2002 phocine distemper virus epidemics in European harbour seals. Dis Aquat Organ 68:115–130
Härkönen T, Backlin BM, Barrett T, Bergman A, Corteyn M, Dietz R, Harding KC, Malmsten J, Roos A, Teilmann J (2008) Mass mortality in harbour seals and harbour porpoises caused by an unknown pathogen. Vet Rec 162:555–556
Hasselmeier I, Fonfara S, Driver J, Siebert U (2008) Differential hematology profiles of free-ranging, rehabilitated, and captive harbor seals (Phoca vitulina) of the German North Sea. Aquat Mamm 34:149–156
Kakuschke A, Valentine-Thon E, Griesel S, Fonfara S, Siebert U, Prange A (2005) Immunological impact of metals in harbor seals (Phoca vitulina) of the North Sea. Environ Sci Technol 39:7568–7575
Kakuschke A, Griesel S, Fonfara S, Rosenberger T, Prange A (2009) Concentrations of selected essential and non-essential elements in blood of harbor seal (Phoca vitulina) pups of the German North Sea. Biol Trace Elem Res 127:28–36
Kakuschke A, Erbsloeh HB, Griesel S, Prange A (2010a) Acute phase protein haptoglobin in blood plasma samples of harbour seals (Phoca vitulina) of the Wadden Sea and of the isle Helgoland. Comp Biochem Physiol B Biochem Mol Biol 155:67–71
Kakuschke A, Valentine-Thon E, Griesel S, Gandrass J, Luzardo OP, Boada LD, Pena MZ, Gonzalez MA, Grebe M, Profrock D, Erbsloeh HB, Kramer K, Fonfara S, Prange A (2010b) First health and pollution study on harbor seals (Phoca vitulina) living in the German Elbe estuary. Mar Pollut Bull 60:2079–2086
Kakuschke A, Gandrass J, Luzardo OP, Boada LD, Zaccaroni A, Griesel S, Grebe M, Profrock D, Erbsloeh HB, Valentine-Thon E, Prange A, Kramer K (2012) Postmortem health and pollution investigations on harbor seals (Phoca vitulina) of the islands Helgoland and Sylt. ISRN Zoology. Article ID 106259
Kakuschke A, Proefrock D, Prange A (2013) C-reactive protein in blood plasma and serum samples of harbour seals (Phoca vitulina). Mar Mamm Sci 29:E183–E192
Kuenstl L, Griesel S, Prange A, Goessler W (2009) Arsenic speciation in bodily fluids of harbor seals (Phoca vitulina) and harbor porpoises (Phocoena phocoena). Environ Chem 6:319–327
Laane R, Sonneveldt HLA, Van der Weyden AJ, Loch JPG, Groeneveld G (1999) Trends in the spatial and temporal distribution of metals (Cd, Cu, Zn and Pb) and organic compounds (PCBs and PAHs) in Dutch coastal zone sediments from 1981 to 1996: a model case study for Cd and PCBs. J Sea Res 41:1–17
Laane R, Vethaak AD, Gandrass J, Vorkamp K, Köhler A, Larsen MM, Strand J (2013) Chemical contaminants in the Wadden Sea: sources, transport, fate and effects. J Sea Res 82:10–53
Loewe P, Becker G, Brockmann U, Dick S, Frohse A, Herrmann J, Klein B, Klein H, Nies H, Schmolke S, Schrader D, Schulz A, Theobald N, Weigelt S (2006) Nordseezustand 2004. Berichte des Bundesamtes für Seeschifffahrt und Hydrographie. Nr.40. Bundesamt für Seeschifffahrt und Hydrographie (BSH): Hamburg, Rostock
Olsson M, Karlsson B, Ahnland E (1994) Diseases and environmental contaminants in seals from the Baltic and the Swedish West-Coast. Sci Total Environ 154:217–227
Ross PS (1995) Seals, pollution and disease: environmental contaminant-induced immunosuppression. Dissertation, University Utrecht, The Netherlands
Shlosberg A, Bellaiche M, Regev S, Gal R, Brizzi M, Hanji V, Zaidel L, Nyska A (1997) Lead toxicosis in a captive bottlenose dolphin (Tursiops truncatus) consequent to ingestion of air gun pellets. J Wildl Dis 33:135–139
Van Neer A, Jensen LF, Siebert U (2015) Grey seal (Halichoerus grypus) predation on harbour seals (Phoca vitulina) on the island of Helgoland, Germany. J Sea Res 97:1–4
Weijs L, Das K, Siebert U, van Elk N, Jauniaux T, Neels H, Blust R, Covaci A (2009a) Concentrations of chlorinated and brominated contaminants and their metabolites in serum of harbour seals and harbour porpoises. Environ Int 35:842–850
Weijs L, Dirtu AC, Das K, Gheorghe A, Reijnders PJH, Neels H, Blust R, Covaci A (2009b) Inter-species differences for polychlorinated biphenyls and polybrominated diphenyl ethers in marine top predators from the Southern North Sea: Part 1. Accumulation patterns in harbour seals and harbour porpoises. Environ Pollut 157:437–444
Weijs L, Dirtu AC, Das K, Gheorghe A, Reijnders PJH, Neels H, Blust R, Covaci A (2009c) Inter-species differences for polychlorinated biphenyls and polybrominated diphenyl ethers in marine top predators from the Southern North Sea: Part 2. Biomagnification in harbour seals and harbour porpoises. Environ Pollut 157:445–451
Acknowledgments
The authors thank all colleagues at the Helmholtz-Zentrum Geesthacht (HZG) and at the Research and Technology Centre in Büsum, the Seal Station Friedrichskoog, the colleagues in Demark, and all participants of the seal catches.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kakuschke, A., Griesel, S. Essential and Toxic Elements in Blood Samples of Harbor Seals (Phoca vitulina) from the Islands Helgoland (North Sea) and Anholt (Baltic Sea): A Comparison Study with Urbanized Areas. Arch Environ Contam Toxicol 70, 67–74 (2016). https://doi.org/10.1007/s00244-015-0205-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-015-0205-0