Abstract
A large body of evidence was compiled in the recent decades showing a noteworthy increase in the detection of pharmaceutical drugs in aquatic ecosystems. Due to its ubiquitous presence, chemical nature, and practical purpose, this type of contaminant can exert toxic effects in nontarget organisms. Exposure to pharmaceutical drugs can result in adaptive alterations, such as changes in tissues, or in key homeostatic mechanisms, such as antioxidant mechanisms, biochemical/physiological pathways, and cellular damage. These alterations can be monitored to determine the impact of these compounds on exposed aquatic organisms. Among pharmaceutical drugs in the environment, antibiotics are particularly important because they include a variety of substances widely used in medical and veterinary practice, livestock production, and aquaculture. This wide use constitutes a decisive factor contributing for their frequent detection in the aquatic environment. Tetracyclines are the individual antibiotic subclass with the second highest frequency of detection in environmental matrices. The characterization of the potential ecotoxicological effects of tetracycline is a much-required task; to attain this objective, the present study assessed the acute toxic effects of tetracycline in the freshwater fish species Gambusia holbrooki by the determination of histological changes in the gills and liver, changes in antioxidant defense [glutathione S-transferase (GST), catalase (CAT), and lipoperoxidative damage] as well as potential neurotoxicity (acetylcholinesterase activity). The obtained results suggest the existence of a cause-and-effect relationship between the exposure to tetracycline and histological alterations (more specifically in gills) and enzymatic activity (particularly the enzyme CAT in liver and GST in gills) indicating that this compound can exert a pro-oxidative activity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
A large amount of chemical agents are released in the aquatic environment as a consequence of direct human use. Among the most frequent sources of pollutants, one can mention hospital and domestic discharges as the major contributors, namely of substances of therapeutic use. This new class of pollutants has recently emerged and includes new or unregulated substances whose environmental presence frequently results from personal use (Verlicchi et al. 2010). This set of particular substances includes drugs, personal care products, and dermocosmetics. Among these compounds, several substances can be significantly challenging to the aquatic ecosystem (Deblonde et al. 2011) even if the compounds are found in low concentrations. The disposal of these compounds, albeit not presently legislated or regularly monitored, requires their characterization in toxicity terms to obtain relevant data that clearly establish the potential toxic effects resulting from their environmental presence, continuous release, low levels of elimination, and interaction with a multiplicity of living organisms in the wild. In addition, the continuous input of such substances into the wild (resulting from an overall inefficacy of sewage-treatment systems, which allowed the broad definition of “pseudo-persistent substances”) causes a chronic exposure of aquatic organisms (Barceló 2003) with the potential onset of immediately unnoticeable, cumulative, and subtle but long-term deleterious effects.
Pharmaceuticals are somewhat peculiar in terms of environmental effects: they are designed to exert biological activity being able to readily cross biological membranes. Drugs are always partially resistant to biotransformation and are daily released in large amounts to the aquatic environment (Halling-Sorensen et al. 1998; Jones et al. 2002). Drugs are also somewhat lipophilic (to favor their absorption through biologic membranes), and the co-occurrence of a large number of distinct metabolites in the same matrix increases the possibility of toxicological interactions (Halling-Sorensen et al. 1998; Cleuvers 2003). The presence of drug residues in the aquatic compartment can be responsible for the exposure of aquatic nontarget organisms resulting in deleterious effects that have in fact been already documented (e.g., Péry et al. 2008). This “cross-talk” can indeed result in a cumulative, irreversible set of deleterious implications that can compromise the population sustainability (Daughton and Ternes 1999).
Antibiotic compounds are used in large amounts (estimated between 100,000 and 200,000 tons/year) both in medical and veterinary practice (Kummerer 2009). However, the use of antibiotics is not restricted to humans and/or animals; in agriculture, antibiotics can also be used to promote fruit growth. Other uses include apiculture, cattle production, and aquaculture as growth promoters (by acting against the development of infections and favoring the increase of advantageous bacterial populations in the gut of animals), prophylaxis, and treatment of bacterial infections (van den Booggard and Stobberingh 1999; Cabello 2006). The excretion of drugs by way of urine and feces is responsible for the release of antibiotics residues into sewage-treatment plants (STPs) where antibiotics in general are not fully degraded and reach the water bodies when effluents are released (Hirsch et al. 1999; Castiglioni et al. 2005). Alternatively, antibiotics can also be dispersed by liquid manure when excreted by medicated farmed animals; aquaculture practices are also responsible for the presence of high amounts of antibiotic residues in the aquatic compartments (e.g., Díaz-Cruz et al. 2003). The risk posed by antibiotics cannot be neglected considering the levels in which they have been found (from ng/L to µg/L) (Halling-Sorensen et al. 1998; Fatta-Kassinos et al. 2011) and due to their continuous release in high amounts by STPs (Michael et al. 2013). Among antibiotic drugs, the tetracycline group is of particular relevance because it is frequently detected in the environment (Gu and Karthikeyan 2005). Tetracyclines are wide-spectrum antibiotics, are used in large amounts in human and veterinary clinical practice, and, in some countries, are formulated as part of animal feed to act as growth promoters by acting against harmful bacteria (Halling-Sorensen et al. 1998). Generally, tetracyclines are excreted by urine, mostly in the unchanged form, at approximately 60 % (Chambers 2001). Degradation-product formation depends on environmental conditions (mainly as a function of the water pH; Chen and Huang 2009) with loss of activity (Halling-Sorensen et al. 2002). However, degradation products can be reverted back to the original compounds (e.g., Sarmah et al. 2006); thus, it is highly important to study the parent compound rather than only focusing on the metabolites or degradation products (e.g., Wang and Yates 2008). Tetracyclines have been involved in distinct toxic phenomena including oxidative stress (Dong et al. 2012) and neurotoxicity (Snavely and Hodges 1984; Thomas 1994) in animal species such as earthworms and mammals.
The present work aimed to assess the ecotoxicological deleterious acute effects of tetracycline in the freshwater fish Gambusia hoolbrooki in terms of oxidative stress and damage, neurotoxicity, phase II metabolism (conjugation capacity), and histological alterations, in specific organs such as liver and gills.
Material and Methods
Capture of Test Organisms
G. holbrooki is a widely disseminated poeciliidae native to the United States of America but found in all hydrographic basins of the Iberian Peninsula (Nico and Fuller 2013) due to its invasive nature and high adaptability to adverse conditions (Cabral and Marques 1999). Its use as an animal model is favored by its small body size and adaptability to controlled laboratory conditions (Nunes et al. 2008). Individuals of G. holbrooki were captured in Pateira de Fermentelos, a natural freshwater lagoon in the central region of Portugal; the main anthropogenic inputs to this lagoon are effluents from agriculture and livestock production resulting in contamination of organic origin (Cerqueira et al. 2005; Ribeiro 2012). After their capture using a hand net, fish were transported in plastic boxes with continuous aeration to the laboratory. Only males and sexually immature females, with sizes comprised between 2.0 and 2.5 cm, were selected. All other individuals were discarded immediately after capture. Before exposure, individuals were subjected to a period of quarantine, acclimation, and depuration (15 days) under laboratory-controlled conditions: dechlorinated tap water, temperature 20 ± 1 °C, 16:8-h light-to-dark photoperiod, and continuous aeration. Animals were fed ad libitum with commercial fish food (Sera Vipan flakes). Inspections were daily performed discarding dead and diseased animals.
Fish Quarantine, Exposure and Sacrifice
After the quarantine period, 40 organisms were selected to develop the ecotoxicological assays. This was performed according to the general recommendations for acute tests (96-h) according to the testing guideline Organisation for Economic Co-operation and Development (OECD) no. 203 (OECD 1992). During exposure, media were renewed after 48 h of exposure, and the tetracycline concentrations were adjusted to the initial values. Because tetracycline is a photodegradable compound (Kummerer 2009), exposure tanks were covered with opaque plastic sheet to prevent the test substance from being degraded. Test organisms were divided in 4 groups with 10 individualized specimens each (1 fish/test vessel): one control treatment and three sublethal levels of exposure to tetracycline (5, 50, and 500 ng/L). The here adopted concentrations of exposure were in the same order of magnitude of the environmentally reported levels; i.e., tetracyclines have been found in surface waters in levels >110 ng/L; additionally, measured concentrations of the same compound were 16.5 ng/L (influent of an STP), 1.9 ng/L (effluent of the same STP), and 2.1 ng/L (river water) (Jia et al. 2009; Kummerer 2009). During the exposure period, parameters, such as temperature, pH, and dissolved oxygen concentrations, were monitored daily for test validation purposes.
Immediately after exposure, fish were sacrificed by immersion in an ice-cold (4 °C) water bath (Wilson et al. 2009) and immediately decapitated. Five individuals per treatment were used for biomarkers. From these individuals, liver, gills, and total head were isolated and homogenized (mechanical homogenizer Ystral, Germany) on ice-cold phosphate buffer 50 mM (pH 7.0) with 0.1 % Triton X-100 and centrifuged at 15,000g in a refrigerated centrifuge. Supernatants were recovered and stored at −80 °C until further analyses. The remaining individuals (5/treatment) were immersed in Bouin solution (24 h) for the histopathological determinations.
Biomarker Assays
Catalase (CAT) activity in liver tissue was measured spectrophotometrically after the decomposition of H2O2 to water and O2, which causes a decrease of absorbance at a wavelength of 240 nm monitored during 30 s. Enzymatic activities were expressed as µmol H2O2 consumed per minute per milligram of protein (Aebi 1984).
Glutathione-S-transferase (GST) activity in liver and gills was determined by spectrophotometry. GST catalyses the conjugation of the substrate 1-chloro-2,4-dinitrobenzene with reduced glutathione to form a thioeter, the formation of which was followed by an increment of absorbance at 340 nm. Results were expressed as nmol of thioeter produced per minute per milligram protein (Habig et al. 1974).
The extent of lipid peroxidation was measured in liver tissue by the quantification of thiobarbituric acid reactive substances (TBARS). TBARS were expressed as malondialdehyde (MDA) equivalents, calculated using an extinction coefficient of 1.56 × 105 M−1 cm−1. This methodology is based on the reaction of compounds such as malondialdehyde (formed by degradation of membrane lipids by free radical attack) with thiobarbituric acid (TBA) (Buege and Aust 1978).
The activity of acetylcholinesterase (AChE) in total head was determined by the increase of absorbance at a wavelength of 414 nm, which occurs when AChE hydrolyses the substrate acetylthiocholine forming choline; this compound reacts in turn with the Ellman’s reagent (5,5′-dithiobis-(2-nitrobenzoic acid) forming a complex that absorbs radiation in the above-mentioned wavelength (Ellman et al. 1961).
Protein concentration of samples was determined according to the methodology described by Bradford (1976) to express enzymatic activities taking into account the protein content of the analyzed tissues.
Histological Assessment
For histopathological analysis, entire fish were fixed in Bouin solution (24 h), decalcified (12 h), dehydrated through graded alcohols (70, 80, 90, and 100 %), embedded in paraffin wax (56–58 °C), and sectioned (5–7 μm) using a manual microtome (Reichert-Jung 2030, Boston LabCo, USA). Sections were stained with hematoxylin–eosin and examined at 100× and 200× by light microscopy (Olympus C×41, Olympus Corporation, Japan). Micrographs were taken using a digital camera (Olympus SC30, Olympus Corporation, Japan).
For description of histological changes and assessment of tissue alterations, a standardized method was used (Bernet et al. 1999). According to this method, pathological changes in gills and liver are grouped in five reaction patterns, namely circulatory, regressive, progressive, inflammatory, and neoplastic. The pathological importance of the observed alteration is defined as an “importance factor” (w), which was classified in 1, 2, or 3 classes corresponding to minimal, moderate, and severe pathological importance, respectively. The extension of the pathological change is rated with a “score value” (a) ranging from 0 to 6, meaning unchanged (0), mild occurrence (2), moderate occurrence, (4) and severe occurrence (6), respectively. The “organ index” (Iorg) results from the sum of the multiplied importance factors (Eq. 1), and score values of all changes found within the examined organs (IH or IG for liver and gills, respectively)
A high index indicates a severe degree of damage and allows a statistical comparison between the degrees of damage of the same organ in different individuals/treatments.
Furthermore, a second arch gill filament section was measured using a free software program (MeasureIT, Olympus) in the parasagittal plane for secondary lamellar length (SLL), secondary lamellar width (SLW), interlamellar distance (ID), and basal epithelial thickness (BET). Three measurements (distal, central, and proximal regions, respectively) were made in each individual gill filament. The proportion of the secondary lamellae available for gas exchange [polyacrylamide gel electrophoresis (PAGE)] was averaged for each filament of an individual and calculated as:
These morphometric variables represent the major dimensions of gill tissue influencing the diffusion distance (gas exchange) in fish (Hughes and Perry 1976) and are correlated with the total gas-exchange area of fish gills (Stevens 1992).
Statistical Analysis
One-way analysis of variance (ANOVA) was run for each variable followed, if needed, by Dunnet multicomparison test to discriminate significantly different chemical treatments relative to the control. A significance level of 0.05 was applied to all analyses. The software used to run the statistical analysis was Sigmaplot 11 (Systat Software, USA). Values were presented as mean (±SE).
Results
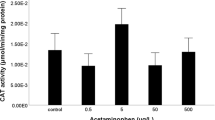
Exposure to increasing amounts of tetracycline was responsible for a dose-dependent significant increase in catalase (CAT) levels (one-way ANOVA F [3,16] = 7.62; p = 0.002) with significant differences being reported for all tested concentrations (Fig. 1a). TBARS levels were significantly decreased by tetracycline exposure in an apparent dose-dependent manner (one-way ANOVA F [3,16] = 6.53; p = 0.004) (Fig. 1b). Exposure to tetracycline lead to a non significant increase of GSTs in liver of exposed organisms (one-way ANOVA F [3,16] = 1.98; p = 0.158) (Fig. 1c). However, the results for the same parameter but measured in gill tissue showed a significant increase, which was only statistically significant for the highest tested concentration (one-way ANOVA F [3,16] = 10.52; p = 0.0004) (Fig. 1d). The activity of AChE was not significantly altered by tetracycline exposure (one-way ANOVA F [3,16] = 0.499; p = 0.688) (Fig. 1e).
Biomarker quantifications in G. holbrooki exposed to several concentrations of tetracycline. a CAT (in liver). b TBARS contents (in liver). c Glutathione S- transferase activity (in liver). d Glutathione S-transferase activity (in gills). e AChE activity (in total head). Values are the mean of five replicate organisms and correspondent SE bars. *Significant differences p < 0.05
The individuals from all exposed groups presented several histological alterations in gills and liver (Figs. 2, 3), but an overall increase in the prevalence, severity, and extension of the tissue lesions appeared to be dose related. Nonexposed individuals showed normal branchial and hepatic architecture. However, exposed individuals presented hypertrophy of the primary and secondary lamellae (Fig. 2a), fusion of the secondary lamellae (Fig. 2d), epithelial lifting (Fig. 2b), aneurisms (Fig. 2c), and necrotic alterations (Fig. 2f). Liver vascular congestion (Fig. 3c), inflammatory lesions (Fig. 3b), and generalized vacuolization (Fig. 3d) were frequently observed. Concerning the IG, significant differences were observed in the experimental groups (one-way ANOVA F [3,16] = 4.14; p = 0.042), namely, between the control group and the two higher concentrations tested (50 and 500 ng/L) (Dunnett test p < 0.05) (Fig. 4a). However, for IH no differences were recorded (one-way ANOVA F [3,16] = 2.60; p = 0.117) despite a non significant increase that was reported (Fig. 4b).
Observed pathological alterations (arrows) in gills of G. holbrooki after an acute exposure (96 h) to several concentrations of tetracycline. a Hypertrophy of the primary and secondary lamellae. b Epithelial lifting. c Aneurism. d Lamellar tip fusion. e Cellular proliferation of the primary epithelium. f Necrosis
No significant differences were recorded for the morphometric measurements of gills (i.e., BET one-way ANOVA F [3,16] = 2.88; p = 0.096; ID one-way ANOVA F [3,16] = 0.62; p = 0.620; SLL one-way ANOVA F [3,16] = 0.89; p = 0.483; and SLW one-way ANOVA F [3,9] = 2.79; p = 0.101) nor for the PAGE index (one-way ANOVA F [3,16] = 2.22; p = 0.155) (Fig. 5).
Discussion
The present work showed significant increases in the activities of the two antioxidant enzymes, CAT and GST, in liver and gills, respectively. The most prominent was CAT, the enhancement of which was observed for all three tested concentrations. GST isoenzymes were not equally responsive, and their overall activity suffered only a significant increase at high levels of tetracycline exposure. TBARS levels were decreased after tetracycline exposure. It is possible to hypothesize that given the significant increase in CAT activity, overproduction of hydrogen peroxide may have occurred; however, the absence of effects in terms of the conjugation pathway (namely, involving GST) or in oxidative damage (namely, lipoperoxidation with increase in TBARS content) suggest that despite the occurrence of oxidative effects, a full scenario of oxidative stress is not likely after exposure to tetracycline. In fact, the absence of lipid peroxidation reinforces this assumption. TBARS levels of animals exposed to tetracycline were significantly decreased compared with control (nonexposed) organisms. This is a somewhat interesting result since the occurrence of oxidative stress associated with tetracycline exposure, previously reported in the literature (Dong et al. 2012), may be signaled by an increase of lipoperoxidation byproducts. The presence of such compounds, including MDA, can be reflected in increased TBARS in exposed organisms if subjected to oxidative stress. In contrast, in our case, TBARS contents seem to have been decreased after tetracycline exposure, which is the opposite effect if one was expecting the occurrence of oxidative damage. The metabolism of tetracycline can indeed result in the formation of reactive oxygen species (ROS; Dong et al. 2012), and, consequently, tetracycline may be responsible for the establishment of pro-oxidative conditions by decreasing the activity of antioxidant enzymes, and by decreasing the levels of intracellular reduced GSH, with the co-occurrence of lipid peroxidation (Asha et al. 2007). Effects of tetracycline on CAT activity of living organisms have already been shown by previous studies. Tetracycline (and another antibiotic of the tetracycline class, e.g., chlortetracycline) have been involved in the establishment of oxidative stress conditions, which elicited increased levels of CAT activity in earthworms (Dong et al. 2012). The involvement of the glutathione pathway in the metabolism of tetracycline is important. Oxytetracycline, a compound sharing important similarities with tetracycline in terms of chemical structure and antibiotic activity, has been shown to exert pro-oxidative effects in fish (namely, Oncorhynchus mykiss) with significant increases of GST activities in several tissues of exposed fish (Yonar 2012).
More than evidencing an oxidative stress scenario, the results obtained may indicate a significant increase in the global efficacy of the antioxidant defense system and of its protective role to serve as a response to the pro-oxidant effect elicited by tetracycline. The increased expression of the here assessed enzymatic forms constitutes an adaptation response of organisms exposed to increasing amounts of tetracycline. The protective nature of this response was evidenced by the significant decrease of oxidative damage as shown by the decrease of membrane lipid byproducts (TBARS) resulting from oxidative aggression. This typology of response has been already described in the literature and can indeed serve as basis for the monitoring of the ecotoxicological activity of a large number of compounds in aquatic organisms (e.g., Sturve et al. 2008; Ramos et al. 2014). The observed response minimized or prevented oxidative damage (including lipid peroxidation, a result reflected by the observed decrease of TBARS levels as reported in other studies (Ozmen et al. 2008; Gravato et al. 2010; Ezemonye and Ikpesu 2011). The decrease in TBARS levels after exposure to tetracycline may indicate that the endogenous production of ROS was efficiently scavenged. Furthermore, the obtained results can also imply the enhancement of phase II metabolic reactions combined with the previously discussed antioxidant effect. This can be concluded from the increase of GST activity. This finding may be related with the tendency, as shown by gill tissue, to preferentially eliminate tetracycline, as well as ROS, to protect cells from lipid peroxidation (Ozmen et al. 2008; Gravato et al. 2010; Ezemonye and Ikpesu 2011). However, one cannot discard the potential antioxidant paradoxical effect, especially as shown by the decrease of the TBARS content, of tetracycline when interpreting the obtained data concerning antioxidant defenses. Tetracycline shows evident similarities, in terms of chemical structure, with vitamin E, a well-known antioxidant compound. Vitamin E and its analogues, including minocycline, are used as antioxidant compounds in diseases in which the involvement and production of ROS has already been described (Kraus et al. 2005; Soory 2008).
The effect of tetracycline in the cholinesterasic activity of exposed fish was, at the least, negligible. This may suggest that tetracycline does not interact with this enzyme under the proposed experimental conditions. As reported for other species (i.e., the rodent species guinea pig), tetracycline can disturb the parasympathetic nervous function (Said et al. 1995). Some policyclic compounds (namely, polycyclic aromatic hydrocarbons), derived from combustion of fossil fuels and other materials (e.g., wood), are reputed cholinesterasic inhibitors (Kang and Fang 1997). Other policyclic compounds, including pharmaceutical drugs (e.g., tacrine) are therapeutically used as cholinesterasic inhibitors to improve cognitive functions (Spilovska et al. 2013). However, the lack of effects observed in our study might suggest that this impairment of the cholinergic pathway may have not occurred in G. holbrooki. Furthermore, the duration of exposure adopted herein may not be optimal to ascertain the effects in terms of cholinesterasic activity. Inhibition of cholinesterases has been reported for a large set of specific compounds such as anticholinesterasic pesticides and heavy metals (Nunes 2011). However, several pharmaceutical drugs, including the antibiotic furazolidone, have been implicated in similar responses (Tu et al. 2009); in contrast, this same study showed that somewhat similar antibiotic compounds, such as enrofloxacine, were not capable of significantly altering AChE activity, thus suggesting a drug-specific mode of action that requires further study.
The gills are multitasking organs and are responsible for several physiological functions such as respiration, osmotic, and ionic balance, acid-base regulation, excretion of nitrogenous wastes, and neurotransmission modulation (Evans et al. 2005). Moreover, because gills are in direct contact with water, they are among the most sensitive organs to changes of water quality caused by xenobiotics in the aquatic compartment (Poleksic and Mitrovic-Tutundzic 1994; Fernandes and Mazon 2003; Brunelli et al. 2011). Furthermore, other important features of gills include a large surface and a short diffusion distance allowing effective gas exchange (Sayed et al. 2012). Their delicate structure usually responds to environmental changes by nonspecific structural alterations depending on the dose and duration of exposure, especially in cases of sublethal levels of aquatic pollutants (Nero et al. 2006).
The histopathological changes of gills reported in the present study were epithelial lifting, fusion of the secondary lamellae, gills hypertrophy, aneurisms, and a few necrosis signs. It is well known that the lifting of the secondary epithelium could produce a significant decrease of the gas surface exchange area, which, together with the hypertrophy of gills epithelium, could result in an increased distance between water and blood and thus consequently compromising the respiratory function (e.g., Sayed et al. 2012). Although fishes can increase their respiration rate to compensate for this oxygen uptake impairment (Fernandes and Mazon 2003), these gill structural changes usually result in an ionic imbalance in blood parameters and red cells of fish (Wood and Soivio 1991; Poleksic and Mitrovic-Tutundzic 1994). These tissue lesions are, however, considered reversible and are interpreted as an adaptive defense response of fish because the decrease of the gas-exchange area of gills also works as an enhanced barrier to the entry of pollutants (Poleksic and Mitrovic-Tutundzic 1994; Fernandes and Mazon 2003). Aneurysms are, however, pathological alterations with more severity and are considered as a specific reaction of the gills to toxic substances (Temmink et al. 1983). Aneurysms occur due to an increase of blood in the lamellae resulting in pillar cell damage and loss of vascular integrity (Stentiford et al. 2003; Martinez et al. 2004). Necrotic lesions are degenerative tissue alterations, usually irreversible, that could contribute to the partial or total loss function of the organ (Nero et al. 2006). The histopathological alterations observed herein are considered nonspecific and, with the exception of necrosis, reversible lesions, at least if the toxicant is withdrawn (Poleksic and Mitrovic-Tutundzic 1994). Furthermore, these types of alterations can occur for a wide variety of aquatic xenobiotics. However, fish gill alterations usually result in hypoxia, respiratory failure, and ionic and acid-base imbalances (Alazemi et al. 1996; Yasser and Naser 2011) because the individuals are more susceptible to secondary infections and even death (Hawkins et al. 1984). Modern immunological and molecular techniques also showed that neuroendocrine cells present on fish gill epithelium, and usually involved in the paracrine regulation of gill blood flow and cardiorespiratory control through their role in oxygen sensing (Evans et al. 2005), can be seriously damaged by exposure to environmental xenobiotics, namely, to metals (Brunelli et al. 2011; De Domenico et al. 2011). However, fish seem to be able to trigger some physiological adaptations interfering with specific neuroendocrine control mechanisms located in gills that enable their long-term survival (De Domenico et al. 2013). This seems likely under conditions similar to those adopted herein because a significant higher gill pathological index was observed for animals exposed to the greater concentrations (50 and 500 ng/L), although the morphometric measurements suggest that gas-exchange efficacy was not yet compromised.
Liver of fish is especially susceptive to environmental variations and is extremely sensitive to waterborne pollutants (Fernandes et al. 2008). Liver is an important storage organ, the primary function of which in fish is detoxification (Olsson et al. 1996). The main hepatic alterations observed in the present study were dilation of sinusoids, hemorrhagic signs, inflammatory processes, and generalized vacuolization. Dilation in the hepatic capillaries and inflammatory foci were already reported as nonspecific hepatic lesions in organisms exposed to subtoxic concentrations of pesticides and heavy metals (e.g., Cengiz and Unlu 2006). Vacuolization can be a degenerative process resulting from metabolic damage related to contaminant exposure (Camargo and Martinez 2007). Several studies have reported a similar finding, with the onset of vacuolization, in fish exposed to heavy metals (Van Heerden et al. 2004; Van Dyk et al. 2007), herbicides (Olurin et al. 2006), and insecticides (Cengiz and Unlu 2006). Tetracycline was responsible for hepatic steatosis in dogs (Amacher and Martin 1997), a condition that was also reported in rodents exposed to tetracycline (Machado et al. 2003; Shabana et al. 2012). Liver damage has been also related with exposure to a similar compound, such as oxytetracycline; this substance was shown to cause deleterious effects in liver of fish (Bruno 1989). Oxytetracycline was also responsible for the induction of toxicity in the liver of rats, such as increased lipid peroxidation markers (TBARS and lipid hydroperoxides) and significantly decreased activities of superoxide dismutase, CAT, and glutathione peroxidase (Pari and Gnanasoundari 2006). As previously mentioned, liver is the primary detoxification organ in fish, and the hepatic changes suggest mobilization of some type of defensive mechanism in an endeavor to detoxify tetracycline. However, because no statistical alterations were observed in the hepatic index, this suggests that our observations correspond to an early stage of this detoxification process.
The data obtained herein shows a comprehensive set of effects that occurred after exposure to realistic levels of tetracycline. Despite being obtained after a short-term exposure, obtained data show significant alterations in fish physiology with the involvement of multiple organs and biochemical pathways. From this set of data, it is possible to infer the occurrence of oxidative-based effects that were preceded and/or accompanied by alterations at the tissue level. Despite not being apparently connected, at least from a mechanistic standpoint, it is important to stress the co-occurrence of adaptive alterations and damages at the subcellular biochemical level and those at the cellular and tissue levels. Consequently, it is important to assume the complimentary role of both type of biomarkers that were validate herein to be used in future research involving the assessment of the effects of pharmaceutical drugs in the aquatic environment. Furthermore, the issue of ecological relevance must be raised and discussed. The major driving criterion used to select the range of concentrations to which test organisms were exposed was ecological relevance, i.e., all concentrations should be close to levels of tetracycline already reported in monitoring studies in aquatic ecosystems. Considering that all concentrations were of the same order of magnitude as those documented in the wild, it is possible to anticipate the risk to which wild organisms may be subjected in moderately polluted waters. However, the underlying mechanisms of tetracycline toxicity in fish still require further studies. The type of acute exposure addressed here does not necessarily reflect the chronic exposure to which organisms may be exposed in the wild. Not only may the extent of effects may be conditioned by longer exposure periods, it is possible that other types of toxic alterations may occur, as a consequence of bioaccumulation processes, that are more likely to happen for longer periods of time.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 6:105–121
Alazemi BM, Lewis JW, Andrews EB (1996) Gill damage in the fresh water fish Gnathonemus petersii (family: Mormyridae) exposed to selected pollutants: an ultrastructural study. Environ Technol 17(3):225–238
Amacher DE, Martin BA (1997) Tetracycline-induced steatosis in primary canine hepatocyte cultures. Fundam App Toxicol 40(2):256–263
Asha KK, Sankar TV, Nair PGV (2007) Effect of tetracycline on pancreas and liver function of adult male albino rats. J Pharmacy Pharmacol 59:1241–1248
Barceló D (2003) Emerging pollutants in water analysis. Trends Anal Chem 22(10):15–16
Bernet D, Schmidt H, Meier W, Burkhardt-Holm P, Wahli T (1999) Histopathology in fish: proposal for a protocol to assess aquatic pollution. J Fish Dis 22(1):25–34
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Brunelli E, Mauceri A, Maisano M, Bernabo I, Giannetto A, De Domenico E et al (2011) Ultrastructural and immunohistochemical investigation on the gills of the teleost, Thalassoma pavo L., exposed to cadmium. Acta Histochem 113:201–213
Bruno DW (1989) An investigation into oxytetracycline residues in Atlantic salmon, Salmo salar L. J Fish Dis 12(2):77–86
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Cabello FC (2006) Heavy use of prophylactic antibiotics in aquaculture: a growing problem for human and animal health and for the environment. Environ Microbiol 8(7):1137–1144
Cabral A, Marques C (1999) Life history, population dynamics and production of eastern mosquito fish, Gambusia holbrooki (Pisces, Poeciliidae) in rice fields of the lower Mondego River Valley, Western Portugal. Acta Oecol 20(6):607–620
Camargo MMP, Martinez CBR (2007) Histopathology of gills, kidney and liver of a neotropical fish caged in an urban stream. Neotrop Ichthyol 5(3):327–336
Castiglioni S, Bagnati R, Calamari D, Fanelli R, Zuccato E (2005) A multiresidue analytical method using solid-phase extraction and high-pressure chromatography tandem mass spectrometry to measure pharmaceuticals of different therapeutic classes in urban waste waters. J Chromatogr A 1092(2):206–215
Cengiz E, Unlu E (2006) Sublethal effects of commercial deltamethrin on the structure of the gill, liver and gut tissues of mosquitofish, Gambusia affinis: a microscopic study. Environ Toxicol Pharmacol 21:246–253
Cerqueira MA, Vieira FN, Ferreira RV, Silva JF (2005) The water quality of the Cértima River Basin (Central Portugal). Environ Monit Assess 111:297–306
Chambers HF (2001) Cloramphenicol, tetracyclines, macrolides, clindamycin and streptogramins. In: Katzung BG (ed) Basic and clinical pharmacology, 8th edn. McGraw-Hill, New York, pp 774–783
Chen WR, Huang CH (2009) Transformation of tetracyclines mediated by Mn(II) and Cu(II) ions in the presence of oxygen. Environ Sci Technol 43(2):401–407
Cleuvers M (2003) Aquatic ecotoxicity of pharmaceuticals including the assessment of combination effects. Toxicol Lett 142(3):185–194
Daughton CG, Ternes TA (1999) Pharmaceuticals and personal care products in the environment: agents of subtle changes? Environ Health Perspect 107(6):907–938
De Domenico E, Mauceri A, Giordano G, Maisano M, Gioffrè G, Natalotto A et al (2011) Effects of “in vivo” exposure to toxic sediments on juveniles of sea bass (Dicentrarchus labrax). Aquat Toxicol 105:688–697
De Domenico E, Mauceri A, Giordano D, Maisano M, Giannetto A, Parrino V et al (2013) Biological responses of juvenile European sea bass (Dicentrarchus labrax) exposed to contaminated sediments. Ecotoxicol Environ Saf 97:114–123
Deblonde T, Cossu-Leguilla C, Hartemann P (2011) Emerging pollutants in wastewater: a review of the literature. Int J Hyg Environ Health 214:442–448
Díaz-Cruz MS, Alda MJL, Barceló D (2003) Environmental behavior and analysis of veterinary and human drugs in soils, sediments and sludge. Trends Anal Chem 22(6):340–351
Dong L, Gao J, Xie X, Zhou Q (2012) DNA damage and biochemical toxicity of antibiotics in soil on the earthworm Eisenia fetida. Chemosphere 89(1):44–51
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Evans DH, Piermarini PM, Choe KP (2005) The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol Rev 85:97–177
Ezemonye LI, Ikpesu TO (2011) Evaluation of sub-lethal effects of endosulfan on cortisol secretion, glutathione S-transferase and acetylcholinesterase activities in Clarias gariepinus. Food Chem Toxicol 49(9):1898–1903
Fatta-Kassinos D, Meric S, Nikolaou A (2011) Pharmaceutical residues in environmental waters and wastewater: current state of knowledge and future research. Anal Bioanal Chem 399:251–275
Fernandes MN, Mazon AF (2003) Environmental pollution and fish gill morphology. In: Val AL, Kapoor BG (eds) Fish adaptations. Science, Enfield, pp 203–231
Fernandes C, Fontaínhas-Fernandes A, Rocha E, Salgado MA (2008) Monitoring pollution in Esmoriz-Paramos lagoon, Portugal: liver histological and biochemical effects in Liza saliens. Environ Monit Assess 145(1–3):315–322
Gravato C, Guimarães L, Santos J, Faria M, Alves A, Guilhermino L (2010) Comparative study about the effects of pollution on glass and yellow ells (Anguilla anguilla) from the estuaries of Minho, Lima and Douro Rivers (NM Portugal). Ecotoxicol Environ Saf 73(4):524–533
Gu C, Karthikeyan KG (2005) Interaction of tetracycline with aluminum and iron hydrous oxides. Environ Sci Technol 39(8):2660–2667
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione-S-transferases—the first enzymatic step in mercapturic acid formation. J Biol Chem 249(22):7130–7139
Halling-Sorensen B, Nielson BN, Lanzky PF, Ingerslev F, Lutzhoft HCH, Jorgensen SE (1998) Occurrence, fate and effects of pharmaceutical substances in the environment—a review. Chemosphere 36(2):357–393
Halling-Sorensen B, Sengelov G, Tjornelund J (2002) Toxicity of teracyclines and tetracycline degradation products to environmentally relevant bacteria, including selected tetracycline-resistant bacteria. Arch Environ Contam Toxicol 44:7–16
Hawkins WE, Overstreet RM, Provancha MJ (1984) Effects of space shuttle exhaust plumes on gills of some estuarine fishes: a light and electron microscopic study. Gulf Res Rep 7(4):297–309
Hirsch R, Ternes R, Haberer K, Kratz KL (1999) Occurrence of antibiotics in the aquatic environment. Sci Total Environ 225(1–2):109–118
Hughes GM, Perry SF (1976) Morphometric study of trout gills: a light-microscope method suitable for the evaluation of pollutant action. J Exp Biol 64:447–460
Jia A, Xiao Y, Hu J, Asami M, Kunikane S (2009) Simultaneous determination of tetracyclines and their degradation products in environmental waters by liquid chromatography–electrospray tandem mass spectrometry. J Chromatogr A 1216:4655–4662
Jones OAH, Voulvoulis N, Lester JN (2002) Aquatic environmental assessment of the top 25 English prescription pharmaceuticals. Water Res 36(20):5013–5022
Kang JJ, Fang H (1997) Polycyclic aromatic hydrocarbons inhibit the activity of acetylcholinesterase purified from electric eel. Biochem Biophys Res Commun 238(2):367–369
Kraus RL, Pasieczny R, Lariosa-Willingham K, Turner MS, Jiang A, Trauger JW (2005) Antioxidant properties of minocycline: neuroprotection in an oxidative stress assay and direct radical scavenging activity. J Neurochem 94(3):818–827
Kummerer K (2009) Antibiotics in the aquatic environment—a review—part I. Chemosphere 75:417–434
Machado ALS, Brandão AAH, da Silva CMOM, da Rocha RF (2003) Influence of tetracycline in the hepatic and renal development of rat’s offspring. Braz Ach Biol Technol 46(1):47–51
Martinez CBR, Nagae MY, Zaia CTBV, Zaia DAM (2004) Morphological and physiological acute effects of lead in the neotropical fish Prochilodus lineatus. Braz J Biol 64(4):797–807
Michael I, Rizzo L, McArdell CS, Manaia CM, Merlin C, Schwartz T et al (2013) Urban wastewater treatment plants as hotspots for the release of antibiotics in the environment: a review. Water Res 47(3):957–995
Nero V, Farwell A, Lee LEJ, Van Meer T, MacKinnon MD, Dixon DG (2006) The effects of salinity on naphthenic acid toxicity to yellow perch: gill and liver histopathology. Ecotoxicol Environ Saf 65(2):252–264
Nico L, Fuller P (2013) Gambusia holbrooki. USGS Nonindigenous Aquatic Species Database, Gainesville, FL. http://nas.er.usgs.gov/queries/Factsheet.aspx?speciesID=849. Accessed: May 4, 2013
Nunes B (2011) The use of cholinesterases in ecotoxicology. Rev Environ Contam Toxicol 212:29–59
Nunes B, Gaio AR, Carvalho F, Guilhermino L (2008) Behaviour and biomarkers of oxidative stress in Gambusia holbrooki after acute exposure to widely used pharmaceuticals and a detergent. Ecotoxicol Environ Saf 72(2):341–354
Olsson PE, Larsson A, Haux C (1996) Influence of seasonal changes in water temperature on cadmium inducibility of hepatic and renal metallothionein in rainbow trout. Mar Environ Res 42:41–44
Olurin KB, Olojo EAA, Mbaka GO, Akindele AT (2006) Histopathological responses of the gill and liver tissues of Clarias gariepinus fingerlings to the herbicide, glyphosate. AJB 5:2480–2487
Organisation for Economic Co-operation and Development (1992) OECD guidelines for the testing of chemicals, Section 2-fish, early-life stage toxicity test (guideline no. 210). OECD Publishing. doi:10.1787/9789264070103-en
Ozmen M, Ayas Z, Gungurdu A, Ekmekci GF, Yerli S (2008) Ecotoxicological assessment of water pollution in Sariyar Dam Lake, Turkey. Ecotoxicol Environ Safe 70(1):167–173
Pari L, Gnanasoundari M (2006) Influence of naringenin on oxytetracyclie mediated oxidative damage in rat liver. Basic Clin Pharmacol Toxicol 98(5):456–461
Péry AR, Gust M, Vollat B, Mons R, Ramil M, Fink G et al (2008) Fluoxetine effects assessment on the life cycle of aquatic invertebrates. Chemosphere 73(3):300–304
Poleksic V, Mitrovic-Tutundzic V (1994) Fish gills as a monitor of sublethal and chronic effects of pollution. In: Müller R, Lloyd R (eds) Sublethal and chronic effects of pollutants on freshwater fish. Fishing New Books, Oxford
Ramos AS, Antunes SC, Gonçalves F, Nunes B (2014) The gooseneck barnacle (Pollicipes pollicipes) as a candidate sentinel species for coastal contamination. Arch Environ Toxicol 66(317–326):1
Ribeiro SHM (2012) Estimativa dos benefícios da melhoria na qualidade da água no Cértima Master thesis [in Portugese]. University of Aveiro, Portugal
Said AA, Matsuki N, Kasuya Y (1995) Effects of aminoglycoside antibiotics on cholinergic autonomic nervous transmission. Pharmacol Toxicol 76(2):128–132
Sarmah AK, Meyer MT, Boxall AB (2006) A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 65(5):725–759
Sayed AEDH, Mekkawy IA, Mahmoud UM (2012) Histopathological alterations in some body organs of adult Clarias gariepinus (Burchell, 1822) exposed to 4-nonylphenol, zoology. In: Garcia MD (ed) zoology. InTech, West Palm Beach, pp 163–184
Shabana MB, Ibrahim HM, Khadre SEM, Elemam MG (2012) Influence of rifampicin and tetracycline administration on some biochemical and histological parameters in albino rats. J Basic App Zool 65(5):299–308
Snavely SR, Hodges GR (1984) The neurotoxicity of antibacterial agents. Ann Intern Med 101(1):92–104
Soory M (2008) A role for non-antimicrobial actions of tetracyclines in combating oxidative stress in periodontal and metabolic diseases: a literature review. Open Dent J 2:5–12
Spilovska K, Korabecny J, Kral J, Horova A, Musilek K, Soukup O et al (2013) 7-Methoxytacrine-adamantylamine heterodimers as cholinesterase inhibitors in Alzheimer’s disease treatment—synthesis, biological evaluation and molecular modeling studies. Molecules 18:2397–2418
Stentiford GD, Longshaw M, Lyons BP, Jones G, Green M, Feist SW (2003) Histopathological biomarkers in estuarine fish species for the assessment of biological effects of contaminants. Mar Environ Res 55(2):137–159
Stevens DE (1992) Gill morphometry of the red drum, Sciaenops ocellatus. Fish Physiol Biochem 10(2):169–176
Sturve J, Almroth BC, Forlin L (2008) Oxidative stress in rainbow trout (Oncorhynchus mykiss) exposed to sewage treatment plant effluent. Ecotoxicol Environ Safe 70(3):446–452
Temmink JHM, Bowmieister PJ, Jong P, van der Berg JHJ (1983) An ultra-structural study of chromate-induced hyperplasia in the gill of rainbow trout, Salmo gairdneri. Aquat Toxicol 4(2):165–179
Thomas RJ (1994) Neurotoxicity of antibacterial therapy. South Med J 87(9):869–874
Tu HT, Silvestre F, Scippo M-L, Thome J-P, Phuong NT, Kestemont P (2009) Acetylcholinesterase activity as a biomarker of exposure to antibiotics and pesticides in the black tiger shrimp (Penaeus monodon). Ecotoxicol Environ Saf 72(5):1463–1470
van den Booggard AE, Stobberingh EE (1999) Antibiotic usage in animals: impact on bacterial resistance and public health. Drugs 58(4):589–607
Van Dyk JC, Pieterse GM, Van Vuren JHJ (2007) Histological changes in the liver of Oreochromis mossambicus (Cichlidae) after exposure to cadmium and zinc. Ecotoxicol Environ Safe 66:432–440
Van Heerden D, Vosloo A, Nikinmaa M (2004) Effects of short-term, copper exposure on gill structure metallothionein and hypoxia-inducible factor-1a (HIF-1a) levels in rainbow trout (Oncorhynchus mykiss). Aquat Toxicol 69:271–280
Verlicchi P, Galleti A, Petrovic M, Barceló D (2010) Hospital effluents as a source of emerging pollutants: an overview of micropollutants and sustainable treatment options. J Hydrol 389(3–4):416–428
Wang QQ, Yates SR (2008) Laboratory study of oxytetracycline degradation kinetics in animal manure and soil. J Agric Food Chem 56(5):1683–1688
Wilson JM, Bunte RM, Anthony J, Carty AJ (2009) Evaluation of rapid cooling and tricaine methanesulfonate (MS222) as methods of euthanasia in zebrafish (Danio rerio). J Am Assoc Lab Anim Sci 48(6):785–789
Wood CM, Soivio A (1991) Environmental effects on gill function: an introduction. Physiol Zool 64:1–3
Yasser AG, Naser MD (2011) Impact of pollutants on fish collected from different parts of Shatt Al-Arab River: a histopathological study. Environ Monit Assess 181(1–4):175–182
Yonar ME (2012) The effect of lycopene on oxytetracycline-induced oxidative stress and immunosuppression in rainbow trout (Oncorhynchus mykiss, W.). Fish Shellfish Immunol 32(6):994–1001
Acknowledgments
This research was supported by the European Regional Development Fund through the COMPETE—Operational Competitiveness Program and by national funds through the Foundation for Science and Technology under the projects PEst-C/MAR/LA0015/2013 and PTDC/AMB/70431/2006.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nunes, B., Antunes, S.C., Gomes, R. et al. Acute Effects of Tetracycline Exposure in the Freshwater Fish Gambusia holbrooki: Antioxidant Effects, Neurotoxicity and Histological Alterations. Arch Environ Contam Toxicol 68, 371–381 (2015). https://doi.org/10.1007/s00244-014-0101-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-014-0101-z