Abstract
Given the limited data on the predictive factors of residual kidney stone size after flexible ureteroscopy (fURS), this study aims to investigate the variables affecting residual stone size. The medical records of 642 patients without complications being treated for kidney stones with fURS between July 2014 and May 2022 were reviewed retrospectively, and the information of the 170 patients in whom residual stones were found was recorded. In addition to patient-specific factors and stone characteristics, length of postoperative hospital stay, postoperative fever, and preoperative antiaggregant use were evaluated. Of the 170 patients ultimately included in the study. The mean age was 51.56 (± 14.70). The mean stone size was 14.01 mm (± 5.75), the mean residual stone size was 7.04 mm (± 2.51), and the mean stone density was 829 Hounsfield units (± 395.06). The mean infundibulopelvic angle (IPA) was 49.37º (± 15.37), and 41.2% of the stones were non-opaque. The mean parenchymal thickness was 22.88 mm (± 5.55). 34 patients were on antiaggregant therapy. Preoperative stone size increases in stone density and decreases in IPA were found to be correlated with increase residual stone size (p < 0.001, p < 0.001, and p < 0.001, respectively). In addition, larger residual stones were observed after the fURS procedure in patients using anticoagulants and those without hydronephrosis (p = 0.02 and p = 0.016, respectively). Use of reliable predictive factors to forecast residual stone size after fURS may help to inform those treated and enable urologists to design rational surgical strategies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Many factors are considered in deciding whether a patient should undergo surgery for kidney stones. These factors include patient symptoms, the presence of urinary obstruction, decline in kidney function, and relevant risk [1]. Tremendous technological advances in imaging methods have increased the rate of diagnosis of urinary stone disease in the last three to four decades [2]. Currently, minimally invasive treatment options for kidney stones include extracorporeal shock wave lithotripsy (ESWL), flexible ureteroscopy (fURS), and percutaneous nephrolithotomy (PCNL).
The European Association of Urology’s urolithiasis recommendations state that ESWL and fURS are equally effective treatment techniques for the management of kidney stones smaller than 2 cm in diameter. For stones greater than 2 cm in diameter, PCNL is the treatment of choice, due to its superior rate of achievement of “stone-free” status. However, given the severe complications associated with PCNL procedures, including bleeding, perforation, and infection, the safety and effectiveness of the fURS method has gained increasing attention from endourologists [3]. Furthermore, as a result of advances in ureteroscope technology, the use of holmium:yttrium–aluminum–garnet lasers and efficient use of accessory instruments has led to the application of fURS in moderate and, to some extent, larger stones on a worldwide basis. The safety and comparable success rate of the technique relative to other available modalities has led many endourologists to apply this method on a more frequent basis [4, 5].
Present day endourological procedures have two primary goals: maximizing the removal of stone load and providing high stone-free rates in a single session. The need for secondary intervention to address residual stones could lead to greater morbidity among patients and pose additional costs to the healthcare system. Residual stone size is a major determinant of whether or not further treatment is called for after an initial procedure [6, 7].
Stone-free rates (SFRs) are an indicator of the success of retrograde intrarenal surgery (RIRS). Although many studies have investigated the factors affecting SFR, no study currently exists to explore the factors affecting residual stone size. This study seeks to fill this hole in the literature. Estimating residual stone size before operation will enable surgeons to develop a rational plan for treatment and follow-up and facilitate patient participation in the process.
Materials and methods
This study observed strict adherence to the guidelines on research ethics laid out in the Helsinki Declaration and was approved by the institute’s ethics review board (Approval No.: 80576354-050-99/210).
The medical records of 642 patients treated for kidney stones with RIRS between July 2014 and May 2022 in this clinic were reviewed retrospectively. Patients with preoperative ureteral stents, ureteral stenosis, intraoperative ureteral rupture or avulsion, or in whom a solitary kidney, ectopic kidney, or other anatomical anomaly was detected, were excluded from this study. Kidney–ureter–bladder radiography (KUB) and non-contrast computed tomography (NCCT) were performed in all cases to evaluate stone characteristics and the renal collecting system. Urinary system ultrasonography (USG) was performed when necessary. Preoperative stone size and postoperative residual stone size were measured using NCCT (for non-opaque stones) and KUB (for opaque stones). The infundibulopelvic angle (IPA) was calculated using the El-Bahnasy technique: i.e., measuring the internal angle between the ureteropelvic axis and the central axis of the lower pole infundibulum [8]. Parenchymal thickness was obtained by measuring the thickest region of transverse sections imaged in preoperative NCCTs.
Patients’ urine cultures were checked prior to operation. Patients with positive urine cultures were operated on after antibiotic treatment. All patients received a routine single dose of second-generation cephalosporins prophylactically during surgery. To evaluate the presence and size of residual stones 3 months after treatment, KUB and USG were used for opaque stones, and NCCT was used for non-opaque stones. Patients were considered stone-free if there were no residual stone fragments or if stone fragments were < 3 mm in diameter.
Based on evaluations made 3 months after surgery, 170 patients with residual stones were ultimately included in this study. Pre- and postoperative laboratory and radiological parameters were compared.
All surgeries were performed under general anesthesia and in the lithotomy position. Under fluoroscopic guidance, a 0.038 Fr guide wire was inserted into the renal pelvis using a 9.5 Fr semi-rigid ureteroscope. The pelvicalyceal system was evaluated by retrograde pyelography. A 9.5/11.5 Fr ureteral access sheath (Cook Medical, Bloomington, IN, USA) was placed over the guidewire under fluoroscopy. The collecting system was entered by passing a 7.5 Fr fiber optic flexible ureteroscope (Storz FLEX-X2) through the UAS. Stones were fragmented using a holmium laser (273 µ fiber), and fragments > 3 mm in diameter were removed from the body using a nitinol basket (ZeroTip™, Cook Urological Inc.). One hundred seventy patients with residual stones > 3 mm in the third month after surgery, based on control results, had their laboratory and radiographic data examined.
SPSS v. 22.0 software was utilized for statistical analysis (SPSS Inc., Chicago, IL, USA). Means and standard deviations were displayed for continuous variables. The Spearman correlation test was applied between groups, and the Mann–Whitney U test was applied between categorical variables. The odds ratio and corresponding 95% confidence interval were used to express impact significance. In all statistical analyses, p < 0.05 was considered significant.
Results
Evaluation of the data obtained produced the following findings: of the 170 patients included in the study, 113 were male and 57 were female, and the mean age was 51.56 (± 14.70). The mean Charlson Comorbidity Index of the patients was 1.78 (0–8). Right-side RIRS was applied to 78 patients and left-side RIRS was applied to 92 patients. The mean stone size was 14.01 mm (± 5.75), the mean residual stone size was 7.04 mm (± 2.51), and the mean stone density was 829 Hounsfield units (± 395.06). The mean IPA was 49.37º (± 15.37), and 41.2% of the stones were non-opaque. The mean parenchymal thickness was 22.88 mm (± 5.55). Postoperative fever was observed in seven patients. Thirty-four patients were on antiaggregants, and 14 were on alpha-blockers (Table 1).
When the Spearman correlation test was performed between groups, residual stone size was observed to correlate positively with preoperative stone size and Hounsfield (stone) density (HU) (p < 0.01). A negative correlation was observed between IPA and residual stone size (p < 0.01). No correlation was observed between parenchymal thickness, body mass index, and residual stone size (Fig. 1).
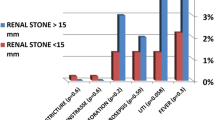
When the Mann–Whitney U test was applied for categorical variables, it was observed that residual stone size increased significantly in the absence of hydronephrosis (p = 0.015) and in antiaggregant users compared with nonusers (p = 0.02). It was determined that stone opacity, lower calyxel localization, number of stones, and previous stone surgery did not affect residual stone size (p = 0.059, p = 0.56, p = 0.86, and p = 0.13, respectively) (Fig. 2).
Discussion
Following its clinical introduction in the 1970s, the fURS method has enjoyed tremendous technical improvements and has grown to inhabit a pivotal role in the minimally invasive management of upper urinary tract calculi. The lower complication rates of fURS relative to both ESWL and PCNL have led to its offering as the first-line option in stone management guidelines, and its popularity has increased significantly among practising urologists [4, 9]. However, the increasing application of this modality, particularly in moderate and large stones, has highlighted the importance of improving SFRs. Studies on this marker of success in recent years have resulted in the development of scoring systems to predict SFRs after fURS [10, 11]. However, while these scoring systems have attempted to predict the occurrence of residual fragments following surgery, the factors affecting residual stone size have not been examined. The main finding of this study is that larger residual stone sizes correlate with larger preoperative stone size, higher HU, and lower IPA. The use of antiaggregants and the absence of hydronephrosis were also found to result in larger residual stone size.
Data from well-conducted studies indicate that larger stone size and higher HU values decrease SFRs and increase the need for auxiliary procedures after fURS. [12,13,14]. This investigation found that residual stone size increased alongside preoperative stone size and HU. Koç and Yıldırım found that SFRs decreased as kidney parenchymal thicknesses rose [15, 16]. However, no significant correlation was observed between parenchymal thickness and residual stone size in the current study. Reşorlu, Jesseb, Danilovic, and Dresner found that a decrease in IPA correlated with an increase in the SFRs of patients with lower pole stones who underwent RIRS. In their literature review, Karim et al. likewise showed that a decrease in IPA corresponded to an increase in SFR [17,18,19,20,21]. The current study confirms this observation once more, and an increase in the mean size of residual stones was noted in those with lower IPA values. Using the potential effect of hydronephrosis on final SFR values after fURS as another parameter, the S.T.O.N.E. scoring system [size, topography (location), obstruction, number, and evaluation (of HU)] defined by Molina and Ito et al.’s nomogram indicate a negative correlation between SFR and the degree of hydronephrosis [22, 23]. In the current study, it was observed that a reduction in residual stone size was significant in patients with hydronephrosis. Yıldırım and Tanik showed that SFRs were not affected by stone opacity. [16, 24]. The current study corroborates these findings. A meta-analysis by Özman et al. conjectured that the number of stones, and of lower calyx stones in particular, had an effect on the SFR; however, an evaluation of the findings of the current study did not indicate any correlation between the mean size of residual stones and the number of stones. Finally, while Altay and Turna could not show any positive correlation between the use of anticoagulants and SFRs after fURS, Yıldırım et al. demonstrated that the use of anticoagulants reduced SFRs. [25, 26]. The current study did not find that mean residual stone size was significantly larger in those using anticoagulants.
This study is not free from limitations. Major limitations may include the retrospective nature of the study design and the relatively low number of cases included and evaluated. However, considering the limited number of publications focusing on the factors that could affect the presence and size of residual stones after fURS, it is the authors’ belief that these findings will contribute to the existing literature and represent reliable clinical implications for practising urologists.
As the first study to evaluate the predictive factors of residual fragment size after fURS, these findings hold a certain value in that they enable surgeons to make reliable and rational management plans by choosing appropriate alternatives. This approach will not only increase SFRs but also decrease the need for additional interventions, improving the health of those treated and minimizing the economic impact such procedures would entail.
Conclusion
The findings of this study show that increases in preoperative stone size and HU and decreases in IPA are all associated with larger residual stone sizes. Residual stone fragments were likewise larger in patients on anticoagulant therapy and those without hydronephrosis following this procedure. These findings may help endourologists using fURS to foresee larger residual fragments, the presence of which could affect patients’ quality of life and necessitate additional procedures. These factors could be considered good predictors of stone-free status and assist individuals in making rational treatment decisions.
Data availability
The data that support the findings of this study areavailable on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Goldsmith ZG, Lipkin ME (2012) When (and how) to surgically treat asymptomatic renal stones. Nat Rev Urol 9:315–320. https://doi.org/10.1038/NRUROL.2012.43
Boyce CJ, Pickhardt PJ, Lawrence EM et al (2010) Prevalence of urolithiasis in asymptomatic adults: objective determination using low dose noncontrast computerized tomography. J Urol 183:1017–1021. https://doi.org/10.1016/J.JURO.2009.11.047
Chung DY, Kang DH, Cho KS et al (2019) Comparison of stone-free rates following shock wave lithotripsy, percutaneous nephrolithotomy, and retrograde intrarenal surgery for treatment of renal stones: a systematic review and network meta-analysis. PLoS One 14:e0211316. https://doi.org/10.1371/JOURNAL.PONE.0211316
Ordon M, Urbach D, Mamdani M et al (2014) The surgical management of kidney stone disease: a population based time series analysis. J Urol 192:1450–1456. https://doi.org/10.1016/J.JURO.2014.05.095
Lee MC, Bariol SV (2011) Evolution of stone management in Australia. BJU Int 108(Suppl 2):29–33. https://doi.org/10.1111/J.1464-410X.2011.10695.X
Emmott AS, Brotherhood HL, Paterson RF et al (2018) Complications, re-ıntervention rates, and natural history of residual stone fragments after percutaneous nephrolithotomy. J Endourol 32:28–32. https://doi.org/10.1089/END.2017.0618
Koras O, Bozkurt IH, Karakoyunlu AN et al (2021) Retrospective analysis of the factors affecting intraoperative and immediate postoperative complications of retrograde intrarenal surgery classified by the clavien and satava grading systems. J Endourol 35:1764–1772. https://doi.org/10.1089/end.2021.0238
Elbahnasy AM, Clayman RV, Shalhav AL et al (1998) Lower-pole caliceal stone clearance after shockwave lithotripsy, percutaneous nephrolithotomy, and flexible ureteroscopy: impact of radiographic spatial anatomy. J Endourol 12:113–119. https://doi.org/10.1089/END.1998.12.113
Türk C, Petřík A, Sarica K et al (2016) EAU guidelines on interventional treatment for urolithiasis. Eur Urol 69:475–482. https://doi.org/10.1016/J.EURURO.2015.07.041
Özman O, Akgül HM, Başataç C et al (2022) Recent scoring systems predicting stone-free status after retrograde intrarenal surgery; a systematic review and meta-analysis. Cent European J Urol 75:72. https://doi.org/10.5173/CEJU.2022.0277
Polat S, Danaciolgu YO, Yarimoglu S et al (2023) External validation of the current scoring systems and derivation of a novel scoring system to predict stone free rates after retrograde intrarenal surgery in patients with cumulative stone diameter of 2–4 cm. Actas Urol Esp 47:211–220. https://doi.org/10.1016/j.acuroe.2022.08.015
Abd El Hamed AM, Elmoghazy H, Aldahshoury M et al (2017) Single session vs two sessions of flexible ureterosopy (FURS) for dusting of renal pelvic stones 2–3 cm in diameter: does stone size or hardness play a role in number of sessions to be applied?". Turk J Urol 43:158–161. https://doi.org/10.5152/TUD.2017.61257
Elbakary MR (2022) Factors affecting use of flexible ureteroscope in large renal stones; stone size or stone composition. Urol Ann 14:336–339. https://doi.org/10.4103/UA.UA_13_22
Ghani KR, Wolf JS (2015) What is the stone-free rate following flexible ureteroscopy for kidney stones? Nature reviews urology. Nat Rev Urol. https://doi.org/10.1038/nrurol.2015.74
Koc E, Kamaci D, Gok B et al (2021) Does the renal parenchymal thickness affect the efficacy of the retrograde intrarenal surgery? A prospective cohort study. Urolithiasis 49:57–64. https://doi.org/10.1007/S00240-020-01185-8/TABLES/5
Yildirim Ü, Ezer M, Uslu M et al (2023) Can additional variables be used to predict stone-free rates following retrograde intrarenal surgery? Anticoagulants and parenchyma thickness: a detailed examination. Urolithiasis 51:1–7. https://doi.org/10.1007/S00240-022-01404-4/FIGURES/2
Resorlu B, Oguz U, Resorlu EB et al (2012) The impact of pelvicaliceal anatomy on the success of retrograde intrarenal surgery in patients with lower pole renal stones. Urology 79:61–66. https://doi.org/10.1016/J.UROLOGY.2011.06.031
Karim SS, Hanna L, Geraghty R, Somani BK (2020) Role of pelvicalyceal anatomy in the outcomes of retrograde intrarenal surgery (RIRS) for lower pole stones: outcomes with a systematic review of literature. Urolithiasis 48:263. https://doi.org/10.1007/S00240-019-01150-0
Jessen JP, Honeck P, Knoll T, Wendt-Nordahl G (2014) Flexible ureterorenoscopy for lower pole stones: influence of the collecting system’s anatomy. J Endourol 28:146–151. https://doi.org/10.1089/END.2013.0401
Dresner SL, Iremashvili V, Best SL et al (2020) Influence of lower pole infundibulopelvic angle on success of retrograde flexible ureteroscopy and laser lithotripsy for the treatment of renal stones. J Endourol 34:655–660. https://doi.org/10.1089/END.2019.0720
Danilovic A, Rocha BA, Torricelli FCM et al (2019) Size is not everything that matters: preoperative CT predictors of stone free after RIRS. Urology 132:63–68. https://doi.org/10.1016/J.UROLOGY.2019.07.006
Molina WR, Kim FJ, Spendlove J et al (2014) The S.T.O.N.E. Score: a new assessment tool to predict stone free rates in ureteroscopy from pre-operative radiological features. Int Braz J Urol 40:23–29. https://doi.org/10.1590/S1677-5538.IBJU.2014.01.04
Ito H, Sakamaki K, Kawahara T et al (2015) Development and internal validation of a nomogram for predicting stone-free status after flexible ureteroscopy for renal stones. BJU Int 115:446–451. https://doi.org/10.1111/BJU.12775
Tanik S, Zengin K, Albayrak S et al (2015) The effectiveness of flexible ureterorenoscopy for opaque and non-opaque renal stones. Urol J 12:2005–2009
Turna B, Stein RJ, Smaldone MC et al (2008) Safety and efficacy of flexible ureterorenoscopy and holmium:YAG lithotripsy for intrarenal stones in anticoagulated cases. J Urol 179:1415–1419. https://doi.org/10.1016/J.JURO.2007.11.076
Altay B, Erkurt B, Albayrak S (2017) A review study to evaluate holmium:YAG laser lithotripsy with flexible ureteroscopy in patients on ongoing oral anticoagulant therapy. Lasers Med Sci 32:1615–1619. https://doi.org/10.1007/S10103-017-2289-4/TABLES/3
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Mehmet Uslu, Ümit Yıldırım, Mehmet Ezer, İsmet Bilger Erihan, Kemal Sarıca. The first draft of the manuscript was written by Mehmet Uslu and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uslu, M., Yıldırım, Ü., Ezer, M. et al. Residual fragment size following retrograde intrarenal surgery: a critical evaluation of related variables. Urolithiasis 51, 100 (2023). https://doi.org/10.1007/s00240-023-01478-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00240-023-01478-8