Abstract
This manuscript reviews the requirements for acceptable compositional analysis of kidney stones using various biophysical methods. High-resolution X-ray powder diffraction crystallography and Fourier transform infrared spectroscopy (FTIR) are the only acceptable methods in our labs for kidney stone analysis. The use of well-constructed spectral reference libraries is the basis for accurate and complete stone analysis. The literature included in this manuscript identify errors in most commercial laboratories and in some academic centers. We provide personal comments on why such errors are occurring at such high rates, and although the work load is rather large, it is very worthwhile in providing accurate stone compositions. We also provide the results of our almost 90,000 stone analyses and a breakdown of the number of components we have observed in the various stones. We also offer advice on determining the method used by the various FTIR equipment manufacturers who also provide a stone analysis library so that the FTIR users can feel comfortable in the accuracy of their reported results. Such an analysis on the accuracy of the individual reference libraries could positively influence the reduction in their respective error rates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urinary tract stone compositional analysis has been cited as a historically necessary metric in defining the etiology and the required medical or surgical treatment of a patient’s stone disease. The physician treating urinary tract stone disease first tries to define the etiology of the disease by studying the reported urine and blood chemistries within a few days and then solidifies the differential analysis with the reported stone composition analysis that normally arrives in many weeks to months from the commercial labs. The attending physician usually accepts the urine and blood chemistries without question and attempts to treat the noted imbalances as the first line of understanding the disease etiology. The offending agents leading to crystallization in the urine may stand out from the normal values, but most of the blood and urine chemistries from the abundance of stone patients are not dramatically different from normal and the patient’s stone disease is then labeled as idiopathic stone disease (over 70% of all stone patients are labeled as idiopathic stone formers) [1, 2]. As reported by urologists and nephrologists at national meetings, it is often difficult to add compositional data into the understanding of the patient’s disease when the reporting of the stone compositional data can take as long as one to three months for the analysis data to be entered into the patient’s chart. An additional parameter in adding stone compositional data to the study of a patient’s disease is the potential for errors in the analysis data as described below and the uncertainty due to different interpretations from the blood and urine chemistries.
There have been a number of reports in the literature describing the poor quality and inaccurate stone compositional analysis from most commercial laboratories and some academic institutions in the US and in Europe [3–5]. Most stone-treating physicians utilize the stone compositional analysis to finally diagnose the patients’ disease, especially for idiopathic patients, and they also use the compositional analysis to define the proper medical and surgical intervention. If there is any error(s) in the compositional analysis, the attending physician can have a difficult time properly defining the etiology of the disease and especially the proper surgical and medical treatment for the stone patient.
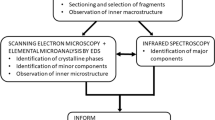
Historically, there have been a number of physical and chemical methods used in determining stone analysis (infrared spectroscopy, X-ray crystallography, micro CT, and electron microscopy). Chemical methods have had so many analysis errors associated with this method that have been universally removed from any modern stone analysis laboratory. Although X-ray crystallographic methods (XRD) are considered the gold standard in crystal identification, the method has some serious shortcomings limiting its routine use, including the amount of sample required, the amount of time it takes to collect adequate diffraction data, and the presence of highly trained technical staff. In addition, the method cannot easily define organic compounds (such as pharmaceutical drugs and over-the-counter, OTC, medications that are quickly metabolized). If the crystal structure has not been defined for the parent drug as well as for all of the metabolites, the XRD method fails. Fourier transform infrared spectroscopy (FTIR) is currently the accepted method of choice for stone analysis including drugs and other medications. However, FTIR is limited by the quality of the reference library available and the choice of computer algorithm chosen for matching sample spectral data with the reference library data. The choice of possible algorithms used in the mathematical matching methods allowed for study of a sample spectrum poorly matching with the reference libraries requires a more detailed analysis of the reasons for the poor match by a trained technician or the principal investigator of the analysis center. Some of the possible choices for the mathematical analysis methods to assist in the analysis of weak maxima, of a series of strong maxima overshadowing weak maxima, and first- and second-order derivatives of the spectral data for poorly defined maxima. Other methods such as chemical analysis have been shown to be far less accurate than FTIR and XRD, and in the case of electron microscopy, it takes a lot of time for sampling preparation but it can suffer from sampling errors.
A high percentage of commercial pathology laboratories or pathology sections in major hospital-based pathology labs using FTIR for stone analysis continue to report inaccurate stone compositional results [3–5]. Such errors in compositional analysis are directly related to the poor quality and lack of completeness of the online computer reference library(s). When FTIR instruments are sold, the manufacturer often supplies (at a cost) a stone compositional reference library developed or purchased by the instrument manufactures. The source of the reference library is usually not reported, but many of the reference standards in the libraries have been derived from human stone samples. It is unknown if the reference standards have been analyzed by another biophysical technique to verify their accuracy or if the developer often uses visual or chemical data for labeling the composition. Human stones are always comprised by more than one component and it is very rare that the human stones used as standards are 100% pure. The use of ANY human stones in the development of a reference library (for either XRD or FTIR methods) is a primary contributor to the errors attributed to the FTIR method. In creating a reference library using human stone materials, the pathology laboratories have created an inaccurate reference library that is used for the compositional analysis of human stones. To circumvent this problem, it is advised to use pure chemical components to create a spectroscopic reference data set [6].
Experimental methods summarized in Ref. [6]
Materials
Briefly, our reference library was developed by either purchasing or synthesizing the needed pure standard components (Table 1). The components were each verified for composition and structure using XRD before being used in the development of the FTIR spectral library. Before developing a computer method for stone component admixtures, two-component admixtures of the pure components were weighed at 10% increments and FTIR spectra taken. These spectral data were compared with the spectral data from a locally developed software program that digitally produced the spectral data for all of the pure components admixed at 10% mixtures from 10% component A–90% component B and from 90% A to 10% B for two-component admixtures and similarly for three-component admixtures. A series of studies were conducted at 5, 10, and 15% admixtures to determine at what % admixtures that we felt comfortable in accepting accurate FTIR analysis. In some admixtures, we could have accepted 5% mixing, but in all cases, we could accept a 10% admixture in differential stone compositional analysis; so, 10% admixing is our accepted policy. In our reports, we label our 10% observations as a possible component present in the stone as the value of 10% composition is still rather low but, yet, we want to inform the physician of such observations.
Stone analysis
Stone analysis has been routinely performed using FTIR and occasionally verified using XRD. We have found a dramatically reduced need to use XRD over the 35+ years of stone analysis conducted in our labs as the compounds used in the generation of our FTIR reference library have continued to increase in the number of components added and in the number of possible admixtures. Our current FTIR spectral reference library approaches 2000 spectra. We also recommend that commercial libraries from the major chemical manufacturers be purchased and referred to when normal stone components do not appear to fulfill an accurate compositional analysis. When the spectral analysis provided by the matching routines is not within our defined goodness-of-fit acceptance levels, then one of our staff contacts the attending physician for the specific stone patient and requests additional information on prescribed medications or known over-the counter (OTC) medications routinely used by the patient. The information on the OTC medications used by the patient often requires repeated questioning of the patient before such information is obtained. If the prescribed or the OTC compounds are not already in the FTIR reference library, then a sample is obtained from the hospital pharmacy or from the vendor used by the patient in the case of OTC compounds. The FTIR spectra of the drugs are added to our FTIR reference library. Even if the stones contain a metabolite of the drug, the FTIR method is based on an analysis of the chemical bonding in the compound and the majority of the bonding chemistry is maintained in most metabolites and the stone can be positively characterized.
Development of a stone reference library
The details of the development of the stone compositional reference library for FTIR are described above and in additional detail in Ref. [6]. The composition of our FTIR reference library is described in Table 1 for the pure core components, Table 2 lists the various drug components [7–11], and Table 3 lists the unusual stone or false stone components we have seen [12].
Overview of more than 35 years of our stone analysis experience
Over the nearly 35-plus years that our lab has been conducting kidney stone analyses, we have analyzed 88,768 kidney stones (we have omitted bladder stones, various gallbladder, and dental salivary stones). As shown in Fig. 1, we have found that 41% of stones are comprised by a single component, 42% of all stones contain two components, and 17% of all stones have three identifiable components. Figure 2 showsthe dominant stone components for stones analyzed in our lab. The most frequent stone type was calcium oxalates (COM + COD = 61%), followed by basic calcium phosphates (15%), uric acid (12%), struvite (6%), brushite (3%), cystine (2%), and matrix (1%). We have found an increasing number of stones associated with prescription and the OTC medications as a component of stones; however, the incidence of these is roughly only 0.1% of our total number of stones we have characterized. XRD is not useful in drug stone analysis because the stone composition is often a drug metabolite and/or the drug is not purely crystalline in nature.
Number of detected stone components per kidney stone. A summary of 88,768 stones over 35 years resulted in 36,721 stones having one component, 37,065 stones having two components, and 14,982 stones with three components. A stone component inclusion must be greater than 10% to be included in this comparison
Primary stone component incidence. The groups represented here are a summary of the primary components detected in single-component and two- or three-component stones based on the analysis of 88,768 stones over 35 years. COM calcium oxalate monohydrate, COD calcium oxalate dihydrate, BCP basic calcium phosphate, UA uric acid: includes uric acid (10.1%) and uric acid dihydrate (1.5%), STR struvite; includes newberyite, BR brushite, CYS cystine, and MTX protein matrix; also includes hemoglobin. Drug stones (RX) and unknown (UN) account for 0.1 and 0.2%, respectively. Cholesterol stones were excluded from this graphic due to the non-kidney origin, but account for roughly 1% of the stones analyzed (946 stones for a total 89,714 to date)
One of the major difficulties in designing a reference library for kidney stone analysis is that stones contain a matrix component for around 2–3% of total stone weight (including proteins, lipids, etc.) [13, 14]. The spectral analysis of organic polymers such as proteins, glycoproteins, and lipoproteins is also very difficult in FTIR and it cannot easily discriminate between proteins such as hemoglobin and albumin as the identification of proteins is based on a review of the broad absorbance maxima in the FTIR region for the amide 1 and amide 2 structures in all proteins. FTIR is definitely not the ideal method to determine the percent composition of selected proteins due to the strong absorbance characteristics of all proteins in the amide 1 and 2 infrared wavelength regions.
Importance of accurate stone analysis in determining the pathogenesis of a patient’s stone disease
Clinical correlation road map with stone composition
As noted in Fig. 3 (A gift to Dr. Neil Mandel from the late Dr. Jacob Lemann, then Chief of the Nephrology Division, Department of Medicine, Medical College of Wisconsin), the stone composition determination is pivotal to determining the pathogenesis of the various types of stone disease. Stone analysis is clearly highlighted very early in the four vertical divisions in the flowchart as a correct and accurate stone analysis is so critical to the possible choices for the understanding of the pathogenesis of the patient’s disease. Errors described in this paper and in all of the review papers cited here can easily translate to misdiagnosis of the etiology of a patient’s disease. This process becomes more complicated if the stone appears to be composed of an unknown mineral or unknown organic material [11, 12]. As stated above, a discussion with the attending physician on prescribed medications and OTC medications and any comorbid diseases usually resolves FTIR uncertainties when the FTIR spectra are added to the reference libraries.
Clinical correlation road map with stone composition mandates. A flowchart entitled “Evaluation of the Patient with Suspected Nephrolithiasis” was provided to Dr. N. Mandel by the late Dr. J. Lemann. The flowchart provides a road map for the evaluation of patients with suspected stone disease and all evaluations include stone analysis as an integral part of the diagnosis. *Hyperuricemia annotation: elevated urine uric acid has also been associated with guaifenesin ingestion. To ensure accurate stone analysis, both the physician and the patient are queried for all medications, including OTC, vitamins, and diet supplements
Conclusions and discussion
In summary, we have highlighted that an accurate and complete reference library is critical for a correct compositional analysis of kidney stones. The review articles cited within this manuscript have correctly described the error rates in stone analysis within commercial analysis laboratories. I stipulate that the high error rates are due to improper and inaccurate reference libraries for both FTIR and XRD methods. The use of FTIR as the chosen method is a good trend in the stone analysis field, but all of the positive attributes of using FTIR fall short in its choice when the reference library is not as accurate as it needs to be. This is probably the primary reason for the errors reported to physicians in charge of the surgical and medical care of the stone patient. As the reference library data are matched to the experimental spectral data of the unknown sample, the similarity of some chemical bonding chemistry among the various stone components, the uncertainty can only be reviewed and decisions made by well-trained technical staff including the director of the laboratory. Discussions with the stone patient’s physician are often very helpful in removing uncertainty and, of course, in providing additional facts on comorbid diseases and prescribed and OTC medications taken by the patient. Such discussions are not only helpful in the interpretation of the stone analysis FTIR spectra, but also help in modifying the dynamic nature of any good reference library.
I have written in some detail how we built our FTIR reference library and how we validated our method of admixing stone components at 10% increments. Although the actual methods vary from one stone analysis center to another, I firmly believe that the use of human stone samples in the creation of components of a reference library should be avoided at all cost. The use of unknowns as reference standards for the determination of composition of other stone samples definitely seems to be a violation of the scientific methods. The methods we used to prepare our FTIR reference standard library are, indeed, very time-consuming and can be expensive in terms of personnel time allocations and the needed personnel scientific expertise, but the efforts are worthwhile and the quality of the reference library is most satisfying.
We have provided data on our 35+ years of experience in kidney stone analysis as another educational review of our database. My stone analysis laboratory initially began with the exclusive use of XRD and only put the FTIR method in place when we could accurately define an FTIR reference library that allowed for the highest accuracy and a validated mathematical method that allowed for an accurate % admixture formulation.
It would be advisable that any purchase of a new FTIR instrument also be accompanied by the purchase of the manufacturer's online kidney stone reference library only after the authors of the library are identified and the methods used to generate the library are fully acceptable to the laboratory directors. For those labs already doing stone analysis, such questions are still worth asking and such answers might help the lab analysis directors determine the actual projected error rates in their reported clinical data on the compositional reports. The impact of incorrect kidney stone analysis can only be guessed at with respect to the effect on the individual patient. In the case of prescribed drugs, OTC medications, or false stones, the surgical or medical plan for treatment can be put into a state of uncomfortable uncertainty for the attending physician and the patient.
References
Zisman AL, Evan AP, Coe FL, Worcester EM (2015) Do kidney stone formers have a kidney disease? Kidney Int 88:1240–1249. doi:10.1038/ki.2015.254
Gambaro G, Croppi E, Coe F, Lingeman J, Moe O, Worcester E, Buchholz N, Bushinsky D, Curhan GC, Ferraro PM, Fuster D, Goldfarb DS, Heilberg IP, Hess B, Lieske J, Marangella M, Milliner D, Preminger GM, Reis Santos JM, Sakhaee K, Sarica K, Siener R, Strazzullo P, Williams JC, Consensus Conference Group (2016) Metabolic diagnosis and medical prevention of calcium nephrolithiasis and its systemic manifestations: a consensus statement. J Nephrol 29:715–734
Krambeck AE, Lingeman JE, McAteer JA, Williams Jr JC (2010) Analysis of mixed stones is prone to error: a study with US laboratories using micro CT for verification of sample content. Urol Res 38:469–475. doi:10.1007/s00240-010-0317-y
Hesse A, Kruse R, Geilenkeuser W, Schmidt M (2005) Quality control in urinary stone analysis: results of 44 ring trials (1980–2001). Clin Chem Lab Med 43:298–303
Siener R, Buchholz N, Daudon M, Hess B, Knoll T, Osther PJ, Reis-Santos J, Sarica K, Traxer O, Trinchieri A, EAU Section of Urolithiasis (EULIS) (2016) Quality assessment of urinary stone analysis: results of a multicenter study of laboratories in Europe. PLoS One 11:e0156606. doi:10.1371/journal.pone.0156606
Mandel IC, Mandel NS (2007) Structural and compositional analysis of kidney stones Chapter 5: (eISBN 978-1-59259-972-1). In: Stoller ML, Meng MV (eds) Urinary stone disease: the practical guide to medical and surgical management. Humana Press, New York, pp 69–81
Wirth GJ, Teuscher J, Graf JD, Iselin CE (2006) Efavirenz-induced urolithiasis. Urol Res 34:288–289
Ghousheh AI, Groth TW, Fryjoff KM, Wille DF, Mandel NS, Roddy JT, Durkee CT (2013) Urolithiasis in patients on high dose felbamate. J Urol 189:1865–1869. doi:10.1016/j.juro.2012.12.032
Ettinger B, Weil E, Mandel NS, Darling S (1979) Triamterene-induced nephrolithiasis. Ann Intern Med 91:745–746
Assimos DG, Langenstroer P, Leinbach RF, Mandel NS, Stern JM, Holmes RP (1999) Guaifenesin- and ephedrine-induced stones. J Endourol 13:665–667
Kolbach-Mandel AM, Mandel NS, Cohen SR, Kleinman JG, Ahmed F, Mandel IC, Wesson JA (2016) Guaifenesin stone matrix proteomics: a protocol for identifying proteins critical to stone formation. Urolithiasis. doi:10.1007/s00240-016-0907-4
Avallone M, Kolbach-Mandel A, Mandel I, Mandel N, Dietrich P, Wesson J, Davis C (2015) Polyisobutylene urolithiasis due to ileal conduit urostomy appliance: An index case. J Endourol Case Rep 1(1):41–43
Boyce WH, Garvey FK (1956) The amount and nature of the organic matrix in urinary calculi: a review. J Urol 76:213–227
Khan SR, Atmani F, Glenton P, Hou Z, Talham DR, Khurshid M (1996) Lipids and membranes in the organic matrix of urinary calcific crystals and stones. Calcif Tissue Int 59:357–365
Acknowledgements
We gratefully acknowledge the financial support provided by the Department of Veterans Affairs for NM through their PhD Research Career Scientist Program as a Senior Research Career Scientist. The VA Merit Review Programs for NM 5453-02P, 5455-01P, and 5455-12P, and in part with resources and the use of facilities at the Clement J. Zablocki Department of Veterans Affairs Medical Center, Milwaukee, WI. We also thank the long-term support of our National VA Crystal Identification first by VA Central Office Department of Pathology, Washington DC and later by our local VA Medical Center with payback dollars per sample analysis from the referring VAMC. Funding was also obtained through parts of several grants (NM) from the National Institutes of Health/National Institute for Diabetes, Digestive, and Kidney, RO1 DK30579, DK064616, and R21 DK062739. The Mandel International Stone and Molecular Analysis Center, MIS.MAC is administered by the Medical College of Wisconsin, Milwaukee, WI with Dr. Mandel as the Director. MIS.MAC continues to operate even though the VA Crystal ID Center closed down due to local financial shortfalls for the Medical Center. I would especially like to acknowledge the valuable contributions by my son, Ian Mandel, in the design and development of the FTIR spectral reference library. Ian did all of the needed synthesis of components that were not commercially available. He also constructed the computer algorithm for admixing the spectra for the various admixtures. The manuscript for the polyisobutylene stone [12] was only possible due to Ian’s persistence in unraveling polymer spectra from the Aldrich reference library and his background in polymer research conducted at the University of Wisconsin, Madison, WI, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflicts of interest to report.
Human studies
The summary data for stone analyses reported in this manuscript was approved by the Clement J. Zablocki IRB as protocol 5455-09 approved through 2016. No informed consents were necessary for the epidemiologic computer review surveys as all patient identifiers were expunged from the reviews.
Additional information
I. C. Mandel: Deceased June 5, 2016.
Submitted to Special Issue of Urolithiasis on the "Physico-chemical, Biochemical and Biological Mechanisms of Stone-Formation" November 24, 2016.
Rights and permissions
About this article
Cite this article
Mandel, N.S., Mandel, I.C. & Kolbach-Mandel, A.M. Accurate stone analysis: the impact on disease diagnosis and treatment. Urolithiasis 45, 3–9 (2017). https://doi.org/10.1007/s00240-016-0943-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-016-0943-0