Abstract
Background
Secondary rhinoplasty is a much more complex operation than primary rhinoplasty, because previously operated and fibrotic tissue is involved. The predictability of results, both aesthetic and functional, is thus inherently lower, demanding greater attention and safety. Even more intricate are those instances in which cartilaginous septum is absent, either in part or almost entirely. This comparative analysis targeted long-term outcomes of secondary rhinoseptoplasties using either auricular or rib cartilage as grafts.
Methods
All patients selected for study met the following criteria: (a) required secondary rhinoplasty for functional and/or cosmetic problems, (b) voids of septal cartilage, (c) follow-up of ~ 2 years, (d) availability of standard pre- and postoperative photos, (e) fair command of the Italian language, and (f) signed participatory consent. Each subject was randomly assigned to group 1 (costal cartilage graft for secondary rhinoplasty) or group 2 (auricular cartilage graft for secondary rhinoplasty). In follow-up, patients completed the Italian version of the FACE-Q rhinoplasty module. Anthropometric measurements were also acquired in AutoCAD for MAC. We determined angles of deviation pre- and postoperatively for comparison and analysed patient satisfaction levels by group using the chi-squared test for unpaired data. Two plastic surgeons reviewed all postoperative photographs of study subjects, rating them on a scale of 1–5.
Results
A total of 135 patients undergoing primary rhinoplasties between January 2008 and September 2019 in our Department of Plastic Surgery and meeting all inclusion criteria qualified for study. Mean patient age was 48.5 years, and mean follow-up time was 3 years. Pre- and postoperative FACE-Q values in group 1 differed significantly (p < 0.05). Anthropometric measurements revealed significant differences (p < 0.05) in pre- and postoperative angles of septal deviation determined for group 1 vs group 2. In long-term follow-up, group 1 maintained an angle close to 180° (p < 0.015), and results in group 1 (vs group 2) remained stable (p < 0.05). Members of group 2 underwent more secondary procedures by comparison (p < 0.05). Ultimately, the two reviewers determined that outcomes in group 1 (vs group 2) proved more satisfactory (p < 0.05).
Conclusions
This effort is the first to compare grafting of auricular and costal cartilage in secondary rhinoplasty procedures. Implants from the 5th rib are preferential to ensure satisfactory long-term outcomes and durable, natural aesthetics in patients lacking septal cartilage.
Level of evidence: Level V, Therapeutic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rhinoplasty is one of the most coveted interventions in cosmetic facial surgery and falls within the domain of several surgical specialties, particularly plastic surgery, maxillofacial surgery, and otolaryngology [1, 2]. It is also a procedure in which constant training is critical to ensure predictable and safe results [3,4,5]. Secondary rhinoplasty is a much more complex operation than primary rhinoplasty, because previously operated and fibrotic tissue is involved. The predictability of results, both aesthetic and functional, is thus inherently lower, demanding greater attention and safety. Even more intricate are those instances in which cartilaginous septum (the chief grafting site in rhinoplasty and a pivotal support structure for the nasal pyramid) is absent, either in part or almost entirely.
Primary causes for absence of septal cartilage are prior aggressive surgery, use of vasoconstrictors (such as cocaine), and malformations (such as cleft lip and palate). Such conditions call for in-depth planning of nasal pyramid restructuring and an obliging source of cartilage, one that enables functional/aesthetic results as durable and predictable as possible. Auricular concha has customarily provided the necessary cartilage, given its ease of access and the obscurity of resultant scars (posterior auricular, as in otoplasties). Unfortunately, it is patently clear that cartilage has memory that acts to preserve the original curvatures. Consequently, our group has increasingly gravitated to costal cartilage over the years [6].
This retrospective analysis of reconstructed nasal pyramids was intended to show that costal cartilage is the best choice for secondary rhinoplasty grafting, offering stable, safe, and predictable correction of functional and cosmetic problems.
Materials and methods
All study candidates met the following criteria: (a) secondary rhinoplasty performed for functional and/or cosmetic concerns, (b) partial or subtotal septal voids, (c) postoperative follow-up ~ 2 years in duration, (d) standard pre- and postoperative photographic documentation, (e) good command of the Italian language, and (f) signed participatory consent. Informed consent by each patient included permission for photographic publication. All subjects underwent maxillofacial computed tomography (CT) in advance of surgical procedures. Cottle’s classification [7] served to evaluate levels of obstruction. In each of our patients, obstruction was confined to area 2 (valve area) or area 4 (anterior turbinate area). The following were grounds for patient exclusion: (a) primary rhinoplasty, (b) intact septal cartilage, (c) follow-up < 2 years, (d) complications from secondary rhinoplasty, (e) invalid pre- or postoperative photographic images, and (f) inability to complete the FACE-Q questionnaire [8]. Data gathering for analysis focused on patient age, sex, duration of follow-up, specific surgical procedure, nature of cartilage used, graft type, means of incision, and level of patient satisfaction. All procedures were performed by the same surgical team, adhering to ethical standards of institutional and/or national research committees and the amended 1964 Declaration of Helsinki or comparable ethical standards.
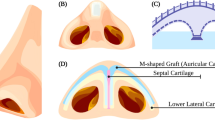
This study was comparative by design, stratifying patients according to graft utilised for secondary rhinoplasty, either 5th costal cartilage (Fig. 1) (group 1) or cartilage from auricular concha (group 2). Cartilage was obtained from donor sites in the course of secondary rhinoplasty, the goal being sound cosmetic and functional outcomes. The same two-surgeon team performed all procedures, positioning respective grafts within septal pockets after efforts made to straighten them and restructure nasal pyramids. Every patient completed the Italian version of the FACE-Q rhinoplasty module, administered via email ~ 2 years postoperatively. We used Cakir’s theory (Fig. 2) [9] of polygons to schematise the aesthetic subunits of the nose in the preoperative checklist as in Fig. 1. Anthropometric measurements were generated in AutoCAD (https://www.autodesk.com/products/autocad/overview) for MAC (Autodesk Inc, San Rafael, CA, USA) from frontal photos taken prior to surgery and at final follow-up visit. One line was drawn from pretarsal fold (A) to maximum projection of dorsal deviation (B) and another from point B to nasal tip (C), depicting the angle of deviation (Fig. 3). Nasal tip projection was measured in profile view using Goode’s method, again relying on AutoCAD for angle calculations. All collected data were stored in an EXCEL file.
In theory, dorsal angles should be 180°. This is precisely what we achieved in each patient at the close of secondary rhinoplasty. By evaluating angles of deviation, we were able to gauge graft performance (auricular vs costal) in reaching a stable aesthetic and functional reconstruction of nasal pyramid. We compared pre- and postoperative levels of patient satisfaction with nasal appearance by group, using the chi-squared test for unpaired data. Two plastic surgeons reviewed all postoperative photos of study subjects, rating images on a scale of 1–5. Data analysis was driven by SPSS software (v25.00 for Windows; SPSS Inc, Armonk, NY, USA), with significance set at p < 0.05. Values were expressed as numbers and percentages (categorical data) or means and standard deviations (quantitative data). Fisher’s exact test was applied to compare categorical data of the two groups.
FACE-Q rhinoplasty module
The 2016 pre- and postoperative FACE-Q rhinoplasty module [10] is a rigorously developed, nasal surgery–specific reporting instrument for patient outcomes proven by Rasch measurement theory analysis. The latter has prompted refinement of a 10-item Satisfaction with Nose Scale and a 5-item Satisfaction with Nostrils Scale. Person separation indices and Cronbach α values were 0.91 and 0.96, respectively, for Satisfaction with Nose and 0.89 and 0.96, respectively, for Satisfaction with Nostrils. All items showed ordered thresholds and good item fits. This module may be used to incorporate patient perspectives in outcome assessments conducted for clinical practice, research, and quality improvement.
Results
A total of 135 patients (men, 101; women, 34) undergoing primary rhinoplasties between January 2008 and April 2019 in our Department of Plastic Surgery and having met all inclusion criteria were enrolled for study (Table 1), divided into 2 groups: group one 71 patients, group two 64 patients. Demographic data and surgical details are shown in Table 2. Mean patient age was 48.5 years, and mean follow-up time was 3 years (range, 2–8 years). All patients underwent secondary rhinoplasties to correct nasal deviation, collapse, or deformity, with C-shaped, S-shape, saddle distortion, or hypoprojection. All showed preoperative nasal obstruction of varying severity. Anthropometric measurements revealed significant differences (p < 0.05) between pre- and postoperative angles of anterior dorsal deviation in group 1 (Figs. 4 and 5) vs group 2 (Table 3). In long-term follow-up, group 1 maintained an angle close to 180°. Differences in pre- and postoperative FACE-Q values for group 1 were significant (p < 0.05) (Table 4), and results for group 1 (vs group 2) remained stable during long-term follow-up (p < 0.05). Members of group 2 also underwent more secondary procedures by comparison (p < 0.05) (Table 2). The two reviewers enlisted determined that outcomes in group 1 proved more satisfactory than those of group 2 (p < 0.05) (Table 5).
Forty-four-year-old man with dorsal and hypoprojection of the dorsum, valves collapse, functional and cosmetic problems, and absence of the septal cartilage because of use of cocaine. Preoperative photos: A–D. Postoperative photos after nasal reconstruction with 5th costal cartilage with 5 years follow-up: E–H
Twenty-nine-year-old woman with previous correction of cleft lip and palate and previous rhinoseptoplasty, with saddle and deviated dorsum, with functional and cosmetic problems. Preoperative photos: A–D. Postoperative photos after nasal reconstruction with 5th costal cartilage with 5 years follow-up: E–H
Discussion
The purpose of this study was to assess secondary rhinoplasty procedures, steering all plastic surgeons, maxillofacial surgeons, and otolaryngologists who perform such revisions towards the most suitable approach. It is obvious that auricular cartilage has several advantages, such as ease of removal and the potential for scar concealment (in posterior auricular region). For years, this site has actually served as the main donor source in patients with deficient septal cartilage. Unfortunately, auricular cartilage is not only quite thin, but it is also curved and therefore less inclined to reconstructive stability over time. The present study is the first clinical analysis to underscore this weakness and encourage other reconstructive options for nasal septum, leaving its utility for alar revisions (similar thickness and shape) unchallenged.
In the literature, there are several histologic studies comparing costal and auricular cartilage. Buckwalter et al. [11] have confirmed that chondrocytes provide the macromolecular framework for a tissue matrix composed of collagens, proteoglycans, and non-collagenous proteins. Collagen types II, IX, and XI create a fibrillary meshwork instilling tissue form, tensile stiffness, and strength. Other shapes are thereby not readily imposed, and the original curvature tends to prevail. Buyuklu and colleagues [12] have also compared auricular and costal cartilage after crushing, demonstrating that auricular chondrocytes largely retain viability and afford better structural preservation. To our knowledge, there are no published studies exploring histologic aspects of auricular and costal cartilage or any differences in structure, memory, or adaptation once manipulated. Perhaps this will follow at some point in time and deliver guidance on their use.
Some sources have argued that auricular cartilage is the best solution for secondary rhinoplasties, regardless of surgical indications. After review of 197 secondary or tertiary rhinoplasties, Pascali et al. [13] have concluded that grafted auricular cartilage is the preferred means of correcting aesthetic and functional residuals from ineffective primary or secondary procedures. Our research group is not swayed by this study, given the less tenable results presented herein. Auricular cartilage does not ensure stable reconstruction of some aesthetic nasal subunits (more central) and will not remain straight over time, owing to its initial shape and thickness. This is especially true if major structural revisions must be performed in the total or subtotal absence of septum.
The same research group [14] (Gentile and Cervelli) cited above has reported a case of secondary reconstruction using the 11th rib. We are not in agreement with this strategy either, because the 11th rib is more sharply curved than the 5th rib (typically accessed to reconstruct nasal pyramid, particularly septum). In fact, this is the only case involving the 11th rib to appear in the literature. Lee et al. [15] have similarly endorsed (based on 22 cases) auricular cartilage as a versatile source of grafting material in primary and secondary rhinoplasties. Aside from the small number of cases entailed, the authors did not investigate an alternate donor site for comparison. Ultimately, many studies conducted in recent years have established the importance of costal cartilage [16,17,18,19,20,21,22], showing its versatility and long-term stability in even extreme scenarios of nasal pyramid reconstruction. Finally, tissue constraints must always be borne in mind. Unlike auricular sites, there is simply ample availability of costal cartilage. Probably the future of surgical interventions in which rib cartilage will be used will be to exploit frozen cartilage taken from a cadaver, despite a cost [23, 24].
The anthropometric study we carried out, evaluating angles of deviation, allowed us to verify the overall straightness achieved over time. This was primarily due to use of the 5th rib to reconstruct or support septum via spreader and extension grafts. On the other hand, septal cartilage drifted from 180° in procedures involving auricular grafts, failing to maintain inner straightness and stability after at least 2 years of follow-up. We considered the 2-year mark a minimum duration for assessing durability. In our study subjects, spreader grafts to straighten septum anteriorly and provide better aesthetics or septal extension grafts to achieve caudal elongation of septum were largely applied, most notably in cases of previously removed posterior septum, columellar strut grafting, required stabilisation of nasal pyramid, or augmented projection of nasal tip. A distinct disadvantage of donor rib cartilage is the chest scar (3–4 cm), more visible in hairless men. In women, the scar is immediately concealed within the inframammary sulcus. For this study, we selected only patients needing septal reconstruction, excluding those with intact nasal septa or revisions of alar cartilage alone (for example).
Limitations of our investigation include its single-centre, comparative, and non-prospective design and a lack of postoperative instrumental studies for the entire patient sampling. At present, however, it remains the only effort comparing two grafting sites in a sizeable number of patients. Corroborative research on the utility 5th rib cartilage in aesthetic and functional nasal revisions will hopefully materialise going forward.
Conclusions
Our data demonstrate that grafting of costal cartilage reduces the risk of further revision procedures, renders outcomes more predictable and safe, and confers aesthetic/functional stability, despite the inherent perils of secondary rhinoplasty.
In our experience, the 5th rib cartilage is preferred for reconstructing the nasal pyramid. Our department no longer harvests auricular cartilage for this purpose. Ours is ostensibly the first comparative study to demonstrate superiority of costal cartilage grafts in patients facing secondary rhinoplasties. We consider it the optimal approach in this setting, ensuring satisfactory long-term outcomes and durable, natural aesthetics.
References
Persichetti P, Cogliandro A, Barone M (2013) Nasal aesthetics: a cross-cultural analysis. Plast Reconstr Surg 132(4):664e–665e
Broer PN, Buonocore S, Morillas A, Liu J, Tanna N, Walker M, Ng R, Persing JA (2012) Nasal aesthetics: a cross-cultural analysis. Plast Reconstr Surg 130(6):843e–850e
Barone M, Cogliandro A, Salzillo R, Colapietra A, Alessandri Bonetti M, Morelli Coppola M, List E, Ciarrocchi S, Tenna S, Persichetti P (2019) Role of spreader flaps in rhinoplasty: analysis of patients undergoing correction for severe septal deviation with long-term follow-up. Aesthetic Plast Surg 43(4):1006–1013
Gode S, Benzer M, Uslu M, Kaya I, Midilli R, Karci B (2018) Outcome of in situ septoplasty and extracorporeal subtotal septal reconstruction in crooked noses: a randomized self-controlled study. Aesthetic Plast Surg 42(1):234–243
Barone M, Cogliandro A, Salzillo R, List E, Panasiti V, Tenna S, Persichetti P (2019) Definition of “Gender Angle” in Caucasian population. Aesthetic Plast Surg 43(4):1014–1020
Fedok FG (2016) Costal cartilage grafts in rhinoplasty. Clin Plast Surg 43(1):201–212. https://doi.org/10.1016/j.cps.2015.08.002
Cottle MH, Loring RM, Fischer GG, Gaynon IE (1958) The maxilla-premaxilla approach to extensive nasal septum surgery. AMA Arch Otolaryngol 68:301–313
Barone M, Cogliandro A, Di Stefano N, Aronica R, Tambone V, Persichetti P (2017) Linguistic validation of the “FACE-Q Rhinoplasty Module” in Italian. Eur Arch Otorhinolaryngol 274(3):1771–1772
Çakır B, Öreroğlu AR, Daniel RK (2016) Surface aesthetics and analysis. Clin Plast Surg 43(1):1–15
Klassen AF, Cano SJ, East CA, Baker SB, Badia L, Schwitzer JA, Pusic AL (2016) Development and psychometric evaluation of the FACE-Q scales for patients undergoing rhinoplasty. JAMA Facial Plast Surg. 18(1):27–35
Buckwalter JA, Mankin HJ (1998) Articular cartilage: tissue design and chondrocyte-matrix interactions. Instr Course Lect 47:477–486 (PMID: 9571449)
Buyuklu F, Hizal E, Yilmaz Z, Sahin FI, Cakmak O (2011) Viability of crushed human auricular and costal cartilage chondrocytes in cell culture. J Craniomaxillofac Surg 39(3):221–225. https://doi.org/10.1016/j.jcms.2010.03.013
Pascali M, Gentile P, Di Pasquali C, Bocchini I, Cervelli V (2016) The auricular cartilage in 197 secondary and tertiary rhinoplasties. J Craniofac Surg 27(2):339–344. https://doi.org/10.1097/SCS.0000000000002380
Gentile P, Cervelli V (2009) Nasal dorsum reconstruction with 11th rib cartilage and auricular cartilage grafts. Ann Plast Surg 62(1):63–66. https://doi.org/10.1097/SAP.0b013e31817433dc
Lee M, Callahan S, Cochran CS (2011) Auricular cartilage: harvest technique and versatility in rhinoplasty. Am J Otolaryngol. 32(6):547–52. https://doi.org/10.1016/j.amjoto.2010.11.008
Cochran CS, Gunter JP (2010) Secondary rhinoplasty and the use of autogenous rib cartilage grafts. Clin Plast Surg 37(2):371–382. https://doi.org/10.1016/j.cps.2009.11.001
Marin VP, Landecker A, Gunter JP (2008) Harvesting rib cartilage grafts for secondary rhinoplasty. Plast Reconstr Surg 121(4):1442–1448. https://doi.org/10.1097/01.prs.0000302467.24489.42
Jang YJ, Kim DY (2016) Treatment strategy for revision rhinoplasty in Asians. Facial Plast Surg 32(6):615–619. https://doi.org/10.1055/s-0036-1594254
Calvert JW, Patel AC, Daniel RK (2014) Reconstructive rhinoplasty: operative revision of patients with previous autologous costal cartilage grafts. Plast Reconstr Surg 133(5):1087–1096. https://doi.org/10.1097/PRS.0000000000000119
Miranda N, Larocca CG, Aponte C (2013) Rhinoplasty using autologous costal cartilage. Facial Plast Surg 29(3):184–192. https://doi.org/10.1055/s-0033-1346999
Gunter JP, Cochran CS, Marin VP (2008) Dorsal augmentation with autogenous rib cartilage. Semin Plast Surg 22(2):74–89. https://doi.org/10.1055/s-2008-1063567
Liang X, Wang K, Malay S, Chung KC, Ma J (2018) A systematic review and meta-analysis of comparison between autologous costal cartilage and alloplastic materials in rhinoplasty. J Plast Reconstr Aesthet Surg 71(8):1164–1173. https://doi.org/10.1016/j.bjps.2018.03.017
Mohan R, Shanmuga Krishnan RR, Rohrich RJ (2019) Role of fresh frozen cartilage in revision rhinoplasty. Plast Reconstr Surg 144(3):614–622. https://doi.org/10.1097/PRS.0000000000005996
Rohrich RJ, Abraham J, Alleyne B, Bellamy J, Mohan R (2022) Fresh frozen rib cartilage grafts in revision rhinoplasty: a 9-year experience. Plast Reconstr Surg. https://doi.org/10.1097/PRS.0000000000009203
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design.
Corresponding author
Ethics declarations
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Due to the retrospective nature of this study, approval from the ethics committee was not required.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
The authors affirm that human research participants provided informed consent for sharing their data and publication of their images.
Competing interests
Mauro Barone, Annalisa Cogliandro, and Paolo Persichetti declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Barone, M., Cogliandro, A. & Persichetti, P. Use of auricular versus costal cartilaginous grafts for secondary rhinoplasty procedures: comparison of long-term outcomes. Eur J Plast Surg 46, 343–350 (2023). https://doi.org/10.1007/s00238-022-02028-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00238-022-02028-y