Abstract
Background
Autologous fat transfer is widely used in plastic surgery for both reconstructive and esthetic purposes. The aim of this study is to identify the results of autologous fat graft to the breast through the rate of fat necrosis, cyst formation, and calcification patterns to avoid unnecessary breast biopsies.
Methods
This prospective study included 30 patients, from September 2015 to February 2018. Baseline mammography was done for patients 40 years or older and ultrasound scans for those under 40 years old then repeated at 6 and 12 months postoperative.
Results
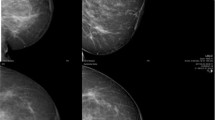
The age of the patients ranged 19–51 years old. The mean amount of the fat transferred to each breast was 252.17 cc with range of 100–410 cc. No major complications were recorded. Six months after breast lipofilling: 6 (25%) of 24 patients show multiple small anechoic and hypoechoic lesions (solid nodules) with disruption of surrounding normal breast tissue. Three patients showed cystic lesions of variable size. Microcalcifications detected in 4 mammograms (66.6%) of the 6 patients over 40 years old. One year after breast lipofilling: Cystic lesions increased to 5. Microcalcifications detected in 3 patients. The microcalcifications in the 4th patient had an increased amount of calcifications and progressed to macrocalcification.
Conclusion
Survival of fat cell grafts and the breast lipofilling complication depends on the techniques used to harvest and then injecting the fat into the receiver site. However, the preliminary results should be confirmed in larger series, and the radiographic follow-up of women undergoing breast lipofilling should be standardized to ensure reproducibility and improve patient safety.
Level of evidence
Level V, therapeutic study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Neuber first performed autologous fat transfer in 1893 to correct depressed scars on the face. The first report of autologous fat grafting for breast reconstruction was done on 1895, when Czerny transplanted a lipoma from the back to reconstruct a breast mound after a mastectomy defect. Ellenbogen challenged Peer’s initial report of a fat graft survival rate of 50% at 1 year in the 1980s with an improved technique and outcome. This together with the advent of liposuction in the 1980s broadened the applications and practice of fat grafting [1].
In 1987, Bircoll was the first surgeon who reported on autologous fat injection for breast augmentation using fat from liposuction [2]. Despite the popularity of breast lipofilling for cosmetic and the reconstructive indications to the female breast, the procedure has a long history surrounded by a great deal of controversy [3]. In 1987, the American Society of Plastic Surgeons prohibited the use of autologous fat grafting to the female breast because of concerns that it would interfere with subsequent cancer screening and that adipocytes may have tumor-promoting effects [4].
Fat grafting regained popularity after the confirmation by Coleman and Saboeiro that fat tissue can be transplanted safely if meticulous care is exercised in the preparation and transfer of fat cell [5]. Rigotti et al. in 2007 elucidated how lipoaspirate heals irradiated tissue through a process mediated by adipose-derived adult stem cells [6]. The transfer of fat tissue for breast lipofilling acts not only as a volume replacement, but also as a tissue regenerator [7].
The aim of this study is to identify the results of autologous fat graft to the breast through the rate of fat necrosis, cyst formation, and calcification patterns to avoid unnecessary breast biopsies.
Patients and methods
This prospective clinical study was conducted at the Department of Plastic Surgery, Menoufia University Hospitals, from September 2015 to February 2018. The ethical committee approved it on July 2016. It included 30 patients with age range from 19 to 51 years old.
All the patients signed for informed consent for photography before and after surgical procedures including publication for research issues.
Patients excluded from this study were those with history of breast cancer, with psychological instability with unrealistic expectations, and who are medically unfit. Baseline mammography was done for patients 40 years or older and breast ultrasound scans for those under 40 years old. All patients were prospectively followed up at 6 months and 1 year after the procedure (by mammography and ultrasound according to their age) to detect signs of fat necrosis, development of microcalcifications, areas of architectural distortion, development of abnormal cysts, and evaluation of the cyst contents. Final assessment was performed 1 year after the last session of fat transfer.
Important landmarks of the breast included the midline, breast meridian, inframammary fold, and suprasternal notch (SSN)—nipple line, besides marking the donor sites for liposuction.
Surgical technique
Anesthesia
General anesthesia was used for 26 patients (86.7%) under general anaesthesia and 4 patients (13.3%) under local anaesthesia in the 1st session of the breast lipofilling. The 2nd session of the breast lipofilling was done under local anesthesia for all patients.
Patient positioning
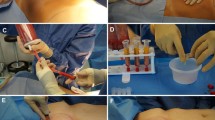
During breast lipofilling, patients were placed in a supine position with elevation of the upper half of the table 30° and arms abducted 90°. The same plastic surgery team using the Coleman technique did the surgery.
Operative requirements
3mm liposuction cannula, 50 ml luer-lock syringes, 20 ml luer lock syringes, 17 to 20 G blunt cannula, Luer -to- Luer connector or 3-way connector, centrifuge for the fat processing and sterile sleeve.
The ideal donor site is patient dependent, based on patient preference and availability of fat at the donor site. The fat graft was harvested from the abdomen, flanks, back, inner thigh, and arms. Donor sites were infiltrated (1 l normal saline, 1 ml epinephrine (1: 1000), 30 ml 1% xylocaine; “super wet” technique). We waited for approximately 20 min to allow for dispersion of the tumescent solution and to optimize the effect of the epinephrine to minimize the blood loss during liposuction. Fat was harvested using a 50-ml Luer lock syringe attached to a 3-mm harvest cannula. The aspirated fat was centrifuged at 3000 rpm for 3 min. Centrifugation allows separation into three phases: the supernatant (containing lysed cells), the precipitate (blood residuals), and the intermediate phase (purified adipocytes).
The purified fat was injected in multiple layers and multiple directions, from deep to superficial using a 20-G blunt cannula with 20-ml Luer lock syringe. The access for fat injection was at the middle point of the inframammary crease or the axillary tail of the breast and at the margin of the areola. The breast was softly massaged until the lump induced by the injection had disappeared.
Postoperative care
The patients were instructed to keep wearing the pressure garment for 4 weeks and received the postoperative medications, e.g., pain killers, and follow-up appointments.
All data were collected, tabulated, and statistically analyzed using SPSS 19.0 for windows (SPSS Inc., Chicago, IL, USA). Quantitative data were expressed as mean and standard deviation (X ± SD). Qualitative data were expressed as number and percentage (No & %). Parametric tests such as t test were used when data are followed or being transformed to normal distribution. Non-parametric tests such as chi-square were used when data did not follow normal distribution. P value was considered significant if it less than or equal to 0.05.
Results
The study included thirty patients, and the age of the patients ranges from 19 to 51 years old with mean age of 31.23 years. Twenty-four patients (80.0%) under 40 years old were radiologically assessed by breast ultrasound, and 6 patients (20.0%) over 40 years old radiologically assessed by mammogram. The mean body mass index (BMI) was 26.9 with range of 24–30. (Table 1).
Twenty-five patients (83.3%) underwent to one session of breast lipofilling, and 5 patients (16.6%) underwent to two sessions of further fat transfer to breast after 6 months of the 1st session.
Breast lipofilling was simultaneously done with other procedures in 21 patients (70%) including liposuction for other areas more than the needed amount of fat for breast lipofilling in 11 patients (36.7%), fat transfer to the buttock in 9 patients (30.0%), abdominoplasty in 8 patients (26.7%), scar revision for old breast reduction in 1 patient (3.3%), vertical thigh lift in 1 patient (3.3%), and arm lift in 1 patient (3.3%). Liposuction areas for the 1st session of breast lipofilling include flanks in 24 patients (80.0%), abdomen in 19 patients (63.3%), inner thigh in 7 patients (23.3%), and arms in 5 patients (16.7%).
The mean volume of the injected fat during the 1st session for right breast was 259.83 cc ranging from 125 to 410 cc and for left breast was 252.17 cc ranging from 100 to 410 cc. During the 2nd session, it was 120 cc for right breast ranging from 100 to 200 cc and for left breast 98 cc ranging from 90 to 100 cc.
The baseline radiological examination for all patients showed no radiographic abnormality. Twenty-four patients (80.0%) under 40 years old were radiologically assessed by breast ultrasound, and 6 patients (20.0%) over 40 years old radiologically assessed by mammogram.
Six months after breast lipofilling
Breast ultrasound showed no radiographic abnormality in 18 patients (60.0%). Multiple small anechoic and hypoechoic lesions (solid nodules) with disruption of surrounding normal breast tissue were found in 6 patients (25%); this percentage is from the total 24 patients under 40 years old. Total number of the solid nodules was 14: 5 were anechoic and 9 nodules were hypoechoic. The location of the nodules correlated with the area of the fat grafted. Three patients show cystic lesions of variable size, two are simple and one is complicated by calcified wall. Mammogram showed no radiographic abnormality in 2 patients (33.4%), and microcalcifications detected in 4 patients (66.6%); this percentage is from the total 6 patients over 40 years old. These small (2 mm) round deposits were either isolated or associated with small, pale fat nodules. This type of calcification appeared to have typical benign features: thin-walled calcifications in oil cysts or coarse irregular calcifications. One patient shows clear, well-focused images of cystic lesions most probably oil cyst.
One year after breast lipofilling
Breast ultrasound showed no newly formed lesions were found. Most of the detected nodules remained unchanged in shape and size after 1-year follow-up. Only in one patient, one nodule dissolved to cystic lesion (Fig. 1). Cystic lesions increase to 5; this means resorption of some nodules in two patients as they were 3 only after 6 months. Mammogram showed no radiographic abnormality in 2 patients (33.4%), and microcalcifications detected in 3 patients. Macrocalcification was found in one patient; this means the microcalcifications in the 4th patient had an increased amount of calcifications and progressed to macrocalcification. One cystic lesion was detected (Table 2, Figs. 2, 3, 4, and 5).
No immediate serious complications occurred in any patients during or after fat grafting procedures, such as hematoma, infection, cellulitis, or thromboembolism. The main complications were breast pain in 4 patients (13.3%), fat necrosis with yellowish discharge in 1 patient (3.3%), irregularities of the donor sites of liposuction in 3 patients (10.0%), fluid collection of the donor sites of liposuction in 2 patients (6.7%), asymmetry of both breast sizes in 1 patient (3.3%), and palpable mass in 1 patient (3.3%) by breast ultrasound diagnosed as lipo-necrotic cyst and drained through needle aspiration in the outpatient clinic with systemic oral antibiotic therapy, with no need of hospitalization.
The presence of a complication was associated with the volume of fat injected. The mean total volume of fat injected into the breasts was 322 ml for those patients who developed complications following the procedure, whereas the fat volume was 223 ml for those without complications (Table 3).
Every patient (at 6 months and 1 year postoperatively) was asked to answer a patient satisfaction questionnaire and to give a score from 1 (dissatisfied) to 4 (highly satisfied) regarding each of the following item: the new breast size, breast shape, breast symmetry, esthetic results, and overall satisfaction. This revealed highly satisfied in 9 patients (30.0%), satisfied in 11 patients (36.7%), neutral in 7 patients (23.3%), and dissatisfied in 3 patients (10.0%).
There was a close correlation in the percentage of the patient satisfaction as compared with the radiological finding and the mean amount of injected fat. 77.8% of the patients were highly satisfied observed in radiological negative patients, 63.6% were satisfied observed in radiological negative patients, 71.4% were neutral observed in radiological negative patients, and 33.3% were dissatisfied observed in radiological negative patients (Tables 4 and 5).
Discussion
Graft survival percentage varies with the methods used to aspirate, prepare, and transplant the fat. It also varies with respect to the destination of the graft, with fat grafted into well-vascularized muscle surviving at a higher rate than fat grafted into a relatively oxygen-poor environment such as scarring tissue from previous breast surgery [3].
Although identification of the donor site is often based on the location of excess adipose tissue and the patient preference, identifying the optimal donor site will help guide surgical approaches. It is clear that there is no significant difference in the volume or viability of the grafted fat obtained from different donor sources [8].
Fat necrosis can be prevented by avoiding overfilling of tissues with fat grafts while respecting the “spaghetti” principle in a crisscross pattern from the deep to the superficial layers [9,10,11].
The technique of lipofilling must maximize survival of the fatty tissue, not only by minimizing trauma during harvesting and refinement but also by placing the living fatty tissue in small aliquots rather than large clumps. The less the grafted amount of fat with each passes of the cannula, the higher the percentage of the surface area of contact between the grafted fat and the recipient tissue. Fat necrosis and calcification can be avoided by increasing the fat survival rate, by the proximity of the transferred fat to blood supply areas at the recipient site. If fat is transferred in large volumes, some of the fat cells may be too far from a blood supply. This can lead to fat necrosis. Therefore, transplanting fat in large clumps should be avoided [12].
Recent findings on the mechanism of fat graft survival and regeneration suggested that fat particles with a more than 2 to 3 mm diameter could not be engrafted at 100%. Fat necrosis after grafting largely depends on the injection technique/volume and microenvironments of the recipient site [13].
Few studies have addressed the issue of the rate of autologous fat graft (AFG) absorption and the factors that may be associated with a higher or lower graft survival volume in the literature. Also, the mechanism of fat tissue survival and absorption is not fully understood. It is believed that fat tissue survives by nutrient diffusion from the serum within the first 48 h and then starts to regenerate [14].
It was thought that fat grafting to the breast could potentially interfere with breast cancer detection; however, no conclusive evidence of such interference has been found even there is no increase in rates of loco-regional recurrence, systemic recurrence, or second breast cancer [15, 16].
Cyst and calcification patterns in the subcutaneous and sub-glandular tissues where the fat grafts were injected can be differentiated from benign or malignant lesions. The features of fat necrosis, oil cysts, and calcification were identified by the presence of decreased signal intensity in the center of fat necrosis, which is the key to differentiation of fat necrosis from solid tumor. The cancerous tumor should not contain fat in the center of the lesion. Signal intensity of fat necrosis decreased on fat-suppressed images in contrast to the intensified signal of the mammary tissue or other breast lumps. Calcifications appear to be a low intensity circle of the signal around a lipo-necrotic cyst [17]. Complications are strictly related to the oily content of the transplanted tissue, where more TGs in the recipient site are clearly at the origin of a higher level of complications [7].
The number and size of nodules following fat transfer to the breast increases as the injection volume increases. As reported, the survival rate of the grafted fat is dependent on the total volume of fat used and the vascularity of the recipient site. We predicted that small amount of fat injected each time might improve the result [18].
There is an inverse relationship between the volume of fat that can be injected in a single session and the percentage fat graft survival two (i.e., the more fat grafted, the lower its survival rate). Most of the clinical studies have addressed the issue of the rate of autologous fat graft absorption and the factors that may be associated with a higher or lower graft survival volume through manipulating the harvesting techniques, fat manipulation, stem cells, and related approaches [19].
An extensive literature review indicated that the major complications observed after lipografting of the breast were related mainly to technical errors and to the wrong anatomic site of harvesting and implantation of the fat [20].
Kim [21] demonstrated in his study that the complications including fat necrosis and cyst formation occurred in 18 of the 102 patients (17.6%). The presence of a complication was associated with the volume of fat injected. The mean total volume of fat injected into the breasts was 67.5 cc for those patients who developed complications, whereas the fat volume was 45.2 cc for those without complications. Of the 18 patients with complications, 10 had fat necrosis and 8 had cystic lesions. Among them, only three patients complained of a palpable mass: pathologic examination confirmed as fat necrosis. All cases complained palpable mass which was conservatively managed with no other procedure like surgical excision or drainage, but regular follow-up was done.
Groen et al. [22] in a review of 33 studies reported 461 complications in a total of 5502 patients. The reported total complication rate was 8.4% including nodules/masses (11.5%), cyst formation (6.9%), hematoma (6.3%), calcifications (5.2%), fat/lipo-necrosis (4%), granulomas (3.6%), infections/cellulitis (0.8%), seroma (0.8%), donor site infections (0.7%0, abscess (0.6%), pneumothorax (0.2%), and delayed wound healing (0.1%).
In our study, the patient satisfaction and presence of a complication were associated with the volume of fat injected. The mean total volume of fat injected into the breasts was 322 cc for those patients who developed complications following the procedure, whereas the fat volume was 223 cc or those without complications (Table 4).
Conclusion
The clinical outcome of breast lipofilling depends on the technique used during its all stages; according to the published literature, there is no clear guidance to the optimal technique at each stages of fat grafting. The using of low suction pressure during liposuction, using of large bore-sized harvesting cannulas, applying of low centrifugation forces, decreasing of the shear stress during injection, injecting of small amount of fat, and optimizing of the amount of fat injected to the capacity of the recipient breast were noted to improve the grafted fat survival rate.
The higher the survival rate of the fat grafts, the lesser the complication rate following breast lipofilling. Fat grafting to the breast can be associated with complications such as infection, lipo-necrotic cysts, fat necrosis, and calcifications. Such calcifications produce distinct radiologic features that may be differentiated from breast cancer, and no reports suggest an increased risk of malignancy associated with fat grafting. The highest incidence of infectious and radiologic complications is associated with bolus fat injection. Finally, it is still very difficult to predict the percentage of the fat graft survival. The need to standardize the autologous fat grafting technique is critical [23]. We acknowledge some weaknesses in the present study. First, because of the difficulty of obtaining preoperative images, the study sample was relatively small. Second, one cannot rule out systemic base in the interpretation of ultrasound and mammograms because they were taken by different mammography systems with different image quality. The number of cases in our study was small, and the follow-up period was also inadequate. Therefore, further studies are required to examine the problems, which may potentially occur after autologous fat grafting.
References
Rosing JH, Wong G, Wong MS, Stevenson TR, Pu LL (2011) Autologous fat grafting for primary breast augmentation: a systematic review. Aesthet Plast Surg 35:882–890
Bircoll M, Novack BH (1987) Autologous fat transplantation employing liposuction techniques. Ann Plast Surg 18:327–329
Wilson H, Spear SL (2013) Fat grafting to the breast. Plastic surgery 3rd edition. Peter C. Neligan. Elsevier Inc 5:581–604
ASPRS Ad-Hoc Committee on New Procedures (1987) Report on autologous fat transplantation. Plast Surg Nurs 7:140–141
Coleman SR, Saboeiro AP (2007) Fat grafting to the breast revisited: safety and efficacy. Plast Reconstr Surg 119:775–785
Rigotti G, Marchi A, Galie M et al (2007) Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: a healing process mediated by adipose-derived adult stem cells. Plast Reconstr Surg 119:1409–1422
Zocchi ML (2017) Large volume breast fat transfer: technical evolution and safety aspects based on over 800 cases and 26 years of follow-up. Eur J Plast Surg 40:367–382
Strong AL, Cederna PS, Rubin GP et al (2015) The current state of fat grafting: a review of harvesting, processing, and injection techniques. Plast Reconstr Surg 136:897–912
Delay E, Guerid S (2015) The role of fat grafting in breast reconstruction. Clin Plast Surg 42(3):315–323
Delay E, Garson S, Toussoun G et al (2009) Fat injection to the breast: technique, results, and indications based on 880 procedures over 10 years. Aesthet Surg J 29:360–376
Delaporte T, Delay E, Toussoun G, Delbaere M, Sinna R (2009) Reconstruction mammaire par transfert graisseux exclusif: a propos de 15 cas consecutifs. Ann Chir Plast Esthet 54(4):303–316
Li XQ, Guo X (2015) Progressive fat necrosis after breast augmentation with autologous lipotransfer: a cause of long-lasting high fever and axillary lymph node enlargement. Aesthet Plast Surg 39:386–390
Khouri RK, Eisenmann-Klein M, Cardoso E, Cooley BC, Kacher D, Gombos E, Baker TJ (2012) Brava and autologous fat transfer is a safe and effective breast augmentation alternative: results of a 6-year, 81-patient, prospective multicenter study. Plast Reconstr Surg 129:1173–1187
Atik B, Oztürk G, Erdogan E, Tan O (2006) Comparison of techniques for long-term storage of fat grafts: an experimental study. Plast Reconstr Surg 118:1533–1537
Kronowitz SJ, Mandujano CC, Liu J, Kuerer HM, Smith B et al (2016) Lipofilling of the breast does not increase the risk of recurrence of breast cancer: a matched controlled study. Plast Reconstr Surg 137:385–393
Simonacci F, Betrozzi N, Greieco M, Grignaffini E et al (2016) Autologous fat transplantation for breast reconstruction: a literature review. Ann Med Surg 12:94–100
Dan NZ, Qing FL, Hua L, Xie Y, Zheng S (2008) Autologous fat grafting to the breast for cosmetic enhancement: experience in 66 patients with long-term follow up. Plast Reconst Aesth Surg 61:792–798
Hongyan W, Yuxin J, Hua M, Zhu Q, Dai Q, Qi K (2010) Sonographic identification of complications of cosmetic augmentation with autologous fat obtained by liposuction. Ann Plast Surg 64:385–389
Zocchi ML, Zuliani F (2008) Bicompartmental breast lipostruc-turing. Aesthet Plast Surg 32(2):313–328
Kim HY, Bok KJ, Dae HL, Dong WL (2014) Autologous fat graft in the reconstructed breast: fat absorption rate and safety based on sonographic identification. Arch Plast Surg 41:740–747
Groen JW, Negenborn VL, Twisk DJ, Rizopoulos D, Ket JCF, Smit JM et al (2016) Autologous fat grafting in oncoplastic breast reconstruction: a systematic review on oncological and radiological safety, complications, volume retention and patient/surgeon satisfaction. J Plast Reconstr Aesthet Surg 69:742–764
Yoshimura K, Coleman SR (2015) Complications of fat grafting. How they occur and how to find, avoid, and treat them. Clin Plast Surg 42:383–388
Sterodimas A, Huanquipaco JC, de Souza Filho S, Bornia FA, Pitanguy I (2009) Autologous fat transplantation for the treatment of Parry-Romberg syndrome. J Plast Reconstr Aesthet Surg 62(11):e424–e426
Author information
Authors and Affiliations
Contributions
Ahmed Abdelsalam Atia: Concept and design, definition of intellectual content, literature search, data acquisition, and manuscript review
Fouad M. Ghareeb: Clinical studies, experimental studies, data analysis, and manuscript preparation
Mohammed G. Ellabban: Design, clinical studies, experimental studies, and manuscript editing
Dalia M. Elsakka: Clinical studies, experimental studies, data analysis, and manuscript preparation
Souzan Fouad Omar: Design, data analysis, statistical analysis, and manuscript review
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. For this kind of retrospective study, formal consent is not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Atia, A.A., Ghareeb, F.M., Ellabban, M.G. et al. Clinical and radiological assessment of autologous fat transfer to the breast. Eur J Plast Surg 43, 139–146 (2020). https://doi.org/10.1007/s00238-019-01588-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00238-019-01588-w