Abstract
Purpose
This review highlights the importance of functional connectivity in pediatric neuroscience, focusing on its role in understanding neurodevelopment and potential applications in clinical practice. It discusses various techniques for analyzing brain connectivity and their implications for clinical interventions in neurodevelopmental disorders.
Methods
The principles and applications of independent component analysis and seed-based connectivity analysis in pediatric brain studies are outlined. Additionally, the use of graph analysis to enhance understanding of network organization and topology is reviewed, providing a comprehensive overview of connectivity methods across developmental stages, from fetuses to adolescents.
Results
Findings from the reviewed studies reveal that functional connectivity research has uncovered significant insights into the early formation of brain circuits in fetuses and neonates, particularly the prenatal origins of cognitive and sensory systems. Longitudinal research across childhood and adolescence demonstrates dynamic changes in brain connectivity, identifying critical periods of development and maturation that are essential for understanding neurodevelopmental trajectories and disorders.
Conclusion
Functional connectivity methods are crucial for advancing pediatric neuroscience. Techniques such as independent component analysis, seed-based connectivity analysis, and graph analysis offer valuable perspectives on brain development, creating new opportunities for early diagnosis and targeted interventions in neurodevelopmental disorders, thereby paving the way for personalized therapeutic strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Long before the advent of modern neuroscience, Aristotle (384–322 BC), the renowned Greek philosopher, recognized the significance of relationships in shaping the nature of things. He proposed that the world is not a collection of isolated entities but rather a tapestry of interconnected elements, each imbued with meaning by its interactions with others [1]. This relational perspective laid the foundation for understanding the brain architecture, where billions of neurons form a complex network of connections that enable the emergence of human consciousness and behavior.
In modern neuroscience, the concept of connectome, a comprehensive map of neural connections within the brain, has emerged as a powerful tool to explore the complex relationships between different brain regions [2]. Functional connectivity (FC), a key component of the connectome, assesses the temporal correlations in neural activity across brain areas, providing insights into how these regions cooperate to perform specific cognitive functions. FC studies have revealed that the brain is not a rigid collection of isolated modules but rather a dynamic and flexible network that can adapt and reorganize to meet the demands of changing tasks and environments [3].
The study of FC holds particular importance in pediatrics due to its unique relevance in understanding the development and functioning of the pediatric brain [4]. The brain undergoes a remarkable journey of development from its inception in fetal life to its full maturation in adolescence. During this intricate process, FC plays a pivotal role in shaping the neural circuitry and enabling the emergence of cognitive functions [5, 6]. FC studies in pediatrics contribute significantly to our understanding of neurodevelopment, aid in the early identification of developmental disorders, and have practical implications for personalized medicine, interventions, and educational practices [7]. This research is essential for promoting optimal brain health and well-being in children.
FC involves numerous technical details that can be challenging for pediatric radiologists to comprehend [8]. This method delves into the synchronized activity of different brain regions, requiring a nuanced understanding of complex neural interactions. Pediatric radiologists, who primarily specialize in medical imaging, may find it challenging to grasp the details of FC analyses, which often involve advanced neuroimaging techniques and statistical methods. Interpretation of connectivity patterns demands expertise in both radiology and neurosciences, and bridging this interdisciplinary gap can be particularly demanding. This study aims to elucidate the fundamental principles of FC, presenting them in a simplified manner to facilitate their comprehension. The goal is to empower pediatric radiologists with the knowledge needed to effectively interpret and incorporate FC analyses into their practice, fostering a more seamless integration of this valuable tool in pediatric neuroimaging.
FC imaging
Among the various techniques available for assessing FC, functional magnetic resonance imaging (fMRI) has emerged as the most prevalent method, owing to its unique advantages [9, 10]. Unlike electroencephalography (EEG) and magnetoencephalography (MEG), which directly measure electrical or magnetic brain signals, fMRI detects changes in blood oxygenation—a reliable indirect proxy for neuronal activity. The increased metabolic and oxygen demands during neuronal activation are met by an increase in local cerebral blood flow, a process known as neurovascular coupling. More specifically, the hemodynamic response is characterized by a small initial dip followed by a peak and a post-activation undershoot. During the peak, oxygenated hemoglobin in the activated area has a slightly lower magnetic susceptibility compared to deoxygenated hemoglobin in the vicinity. This subtle difference in magnetic susceptibility alters the local magnetic field, a change detected using multi-volume, dynamic, T2*-weighted pulse sequences. The ensuing signal, known as the BOLD (blood-oxygen-level-dependent) signal, serves as a reliable indicator of neuronal activation [10]. Age-related changes in the BOLD signal have been reported and are related to brain maturation. Compared to adults, the BOLD hemodynamic response to brain activation in infants is characterized by smaller amplitude and delayed time to peak, with preterm infants exhibiting an even more delayed response than full-term infants [11,12,13,14]. Furthermore, the BOLD signal characteristics change throughout childhood and adolescence as the brain continues to develop [15]. These age-dependent variations necessitate careful consideration when analyzing and interpreting FC across different pediatric age groups.
Resting-state functional magnetic resonance imaging (rs-fMRI) and task-based functional magnetic resonance imaging (task-fMRI) are two distinct approaches in functional MRI. Rs-fMRI, first described by Biswal et al. in 1995 [16], examines spontaneous, intrinsic brain activity during rest, capturing baseline FC between different brain regions. Large-scale resting-state networks (RSNs) have been identified in adult and pediatric populations that correspond to activation patterns commonly derived from task-based fMRI [17,18,19,20,21]. RSNs are dominated by low-frequency BOLD fluctuations, have age related distinct spatial patterns and time courses and are present even during sleep and anesthesia [18,19,20,21,22]. The brain remains active even when a person is not actively involved in specific mental or physical activities, and this intrinsic activity is characterized by complex patterns of communication between different regions. In contrast, task-fMRI involves performing specific cognitive or motor tasks during the fMRI scan, with the resulting BOLD signal indicating regions activated by the tasks. While rs-fMRI reveals patterns of correlated activity at rest, task-fMRI provides insights into the brain’s response to external stimuli or goal-directed activities. The BOLD signal in rs-fMRI reflects baseline connectivity, while in task-fMRI, it signifies changes in blood flow and oxygenation associated with the performance of specific cognitive or motor tasks. These complementary approaches contribute to a comprehensive understanding of both intrinsic and task-related aspects of FC.
Methods evaluating local function of specific brain areas are Amplitude of Low-Frequency Fluctuations (ALFF), Fractional Amplitude of Low-Frequency Fluctuations (fALFF) and regional homogeneity (ReHo). The ALFF is a metric utilized in rs-fMRI to quantify the intensity or amplitude of spontaneous low-frequency fluctuations in the BOLD signal [21]. It focuses on the magnitude of neural activity at rest within specific brain regions, typically in the low-frequency range of 0.01 to 0.1 Hz. In contrast, the fALFF is a variant that normalizes ALFF by dividing it by the total power across the entire frequency range, providing a more specific measure of the relative strength of low-frequency oscillations while mitigating the impact of non-neuronal physiological noise [19]. While neither directly mapping functional connections, both ALFF and fALFF serve as valuable tools in neuroimaging research, offering insights into regional neural activity and aiding in the identification of genuine neuronal fluctuations within the complex fMRI signal during resting-state [23]. ReHo uses the Kendall’s coefficient of concordance to evaluate the synchronization between the time series of a given voxel and its nearest neighbours [24].
Rs-fMRI is increasingly employed in pediatric studies because unlike task-based fMRI, allows for a comprehensive examination of the whole brain’s FC without imposing explicit cognitive demands, which is crucial, especially for younger children facing challenges in engaging with complex tasks [20, 25,26,27,28,29,30,31,32,33,34,35,36]. Resting-state activity can be studied in neonates using the “wrap and feed” method but beyond that period up to the age of 6 years and even later in children with developmental delay, involuntary movements or claustrophobia, drug induced sedation or even anesthesia are necessary. Pharmacologically induced modulations to the resting-state activity of the brain would be a challenge in rs-fMRI studies. Popular choices for sedation in the pediatric population include pentobarbital for deeper, more controlled sedation, particularly in very young or highly anxious children, and chloral hydrate for lighter sedation, ease of administration, and a potentially better safety profile in infants [37]. The effect of sedation with chloral hydrate appears to be age-related. Studies in neonates and young infants have demonstrated that sedation with chloral hydrate does not affect the identification or topology of RSNs, nor the amplitude of neural responses [12, 27, 38]. However, in school-age children, it induces a redistribution of the brain’s information processing and a decrease in both global and local efficiency in transferring information [39]. There are few comparative studies on the effect of sedative drugs on brain BOLD signal, but further research is necessary to determine the best option for rs-fMRI in children [37, 40,41,42].
FC modeling
The field of FC analysis encompasses a diverse range of approaches that offer complementary perspectives, enabling researchers to delve into the obscure aspects of neural communication and organization [20, 43, 44]. The choice of specific model often depends on the precise research question, the nature of the available data, and the underlying assumptions of the employed method. Some FC models posit a causal link between brain regions (a.k.a. effective connectivity), suggesting a directional flow of information from one region to another [45,46,47] while others focus on a simpler correlational relationship [16, 48, 49]. In most cases, there is no pre-existing model of the FC pattern, and the analysis is conducted in an unsupervised, data-driven manner [16, 48, 49]. However, some model-driven approaches involve testing one or more specific FC models to assess their ability to explain the acquired data [46, 47]. Further, FC analysis can be performed at the voxel level, examining the connectivity between individual small brain regions [49], or at the level of regions of interest (ROIs), focusing on the functional relationships between larger, defined brain regions [50]. Finally, some FC models concentrate on the regional connectivity within an ROI [24], while others aim to elucidate the global properties of the brain connectome, the comprehensive map of neural connections across the entire brain [50]. There are several popular FC analysis methodologies used in FC studies in pediatrics, each with its own advantages and limitations:
Independent component analysis (ICA)
ICA is a widely utilized methodology in pediatric rs-fMRI analysis. It stands out as a data-driven approach, meaning it doesn’t rely on predefined models or assumptions about the brain’s functional networks. Instead, ICA analyzes raw rs-fMRI data, aiming to identify patterns of activity that naturally emerge. The primary goal of ICA is to find spatially independent networks within the brain [49, 51] (Fig. 1). Spatial independence implies that the identified networks are not influenced by each other; they represent distinct patterns of activity. In simpler terms, it’s akin to isolating the sound of individual instruments in a symphony that play independently.
Resting-state networks were derived using ICA analysis applied to resting-state fMRI images from two distinct groups: a cohort of adults and a cohort of premature infants. Adult ICA networks, integrated within the CONN toolbox, were derived by applying ICA to a dataset comprising 497 subjects from the Human Connectome Project database. Conversely, the resting-state networks of premature infants were obtained by applying ICA to resting-state fMRI data collected from 8 premature infants around term equivalent age
ICA focuses on detecting regions of the brain that exhibit synchronized activity. This synchronization indicates that certain brain areas are activated together, suggesting a functional connection between them. What makes ICA particularly powerful is its ability to explore various levels of brain organization, from broad functional systems to more focused, task-specific networks. A prominent example is the default mode network (DMN), which is active during periods of rest or mind wandering [52]. This network is involved in self-referential processing, autobiographical memory, and internal thought processes. The salience network (SN) acts as a filter, detecting and responding to salient stimuli in the environment. It is involved in attention, arousal, and emotion regulation [53]. The executive control network is engaged in goal-directed cognitive tasks that require planning, decision-making, and working memory [54]. It is located in the prefrontal cortex, parietal cortex, and cingulate cortex. Other important networks include the auditory network, medial visual network, lateral visual network, sensorimotor cortex, dorsal visual stream (frontoparietal attention network), basal ganglia, limbic network, and precuneus network [20, 55].
In essence, ICA serves as a detective tool for neuroscientists. It sifts through the complex data generated by rs-fMRI scans, identifies unique patterns of synchronized brain activity, and reveals spatially independent networks. Its ability to work without preconceived notions is particularly valuable in understanding the pediatric brain development, shedding light on both large and small-scale functional networks.
Seed-based correlation analysis
Another popular methodology in pediatric rs-fMRI analysis is seed-based correlation analysis [8, 16, 20, 56]. This approach involves selecting a region of interest (ROI) within the brain and computing the correlation between the time series of this ROI and every other voxel in the brain (Fig. 2). Seed-based correlation analysis allows researchers to investigate FC between specific regions of interest and the rest of the brain. It has the advantage of being relatively straightforward to implement and interpret, making it a popular choice in both pediatric and adult rs-fMRI studies.
An illustration of seed-based correlation analysis employed to identify regions within the neonatal brain functionally connected with the left precentral gyrus. The neonatal brain was normalized and segmented based on the University of North Carolina (UNC) neonatal atlas (left). The correlation between the signal of each voxel in the brain and the average signal from the left precentral gyrus (seed area) is visualized as a color map in red, superimposed on the UNC neonatal brain template
Several metrics can be used to assess associations between time-series of brain areas, including cross-correlation coefficient, partial correlation and multiple regressions. Cross-correlation coefficient measures the linear relationship between two time-series and is simple to interpret but sensitive to noise. Partial correlation controls for the effects of other brain regions on the relationship between two time-series, making it more robust to noise but potentially less sensitive to true FC. Multiple regressions can control for the effects of multiple brain regions on the relationship between a target region and another region, enabling the capture of more complex relationships but at the cost of increased complexity.
Graph analysis
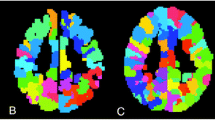
Graph theory-based analysis is a valuable method employed in pediatric rs-fMRI analysis, helping researchers unravel how the brain functions [57] (Fig. 3). Imagine the brain as a vast network of interconnected nodes (representing specific brain regions) and edges (symbolizing the connections between these regions). This approach allows scientists to explore the brain’s architecture in a unique way, focusing on understanding its organizational principles. Through graph theory, researchers measure various network properties such as degree (the number of connections a node has), clustering coefficient (indicating how tightly connected neighbors of a node are), path length (the number of connections needed to travel from one node to another), betweenness centrality, (the importance of a node in terms of its ability to connect other nodes), and modularity (a measure of the organization of a network into modules or communities) [58, 59]. These metrics provide valuable insights into the efficiency and structure of the brain’s networks.
A standard pipeline for graph-based resting-state functional connectivity analysis is depicted. The processing steps encompass data acquisition, preprocessing, defining graph nodes typically through regions delineated in a brain atlas, extracting time-series data, defining edges via correlation analysis to produce a connectivity matrix, thresholding to generate an adjacency matrix, and finally, conducting graph analysis to compute various graph metrics
Graph analysis has showed that brain’s network structure demonstrates characteristics of a small-world network [60]. This configuration is characterized by high clustering, indicating the presence of densely interconnected local regions, and short average path lengths, reflecting efficient global communication between distant brain areas. In the context of neuroimaging data analysis, this organizational pattern suggests a balance between specialized processing within local neighborhoods and the ability for quick and efficient information transmission across the entire brain network. The small-world topology of the brain is considered advantageous for cognitive functions, allowing for both segregated processing and integrated communication between different regions.
By applying graph theory to pediatric rs-fMRI data, scientists gain a deeper understanding of the developing brain’s organization and connectivity. This analytical approach enables them to identify key features that characterize normal brain development and detect abnormalities or disruptions in these patterns. For example, changes in the clustering coefficient may indicate alterations in local processing, while variations in path length could suggest differences in the efficiency of information transfer between brain regions. Such insights contribute to our knowledge of pediatric brain function and provide a foundation for investigating neurological disorders that may manifest in childhood.
In essence, graph theory-based analysis serves as a powerful tool that translates complex brain activity into comprehensible patterns. It offers a unique lens through which researchers can explore the structural and functional aspects of the pediatric brain, shedding light on both the normal developmental processes and potential irregularities that may underlie neurological conditions.
It is worth noting that each of these methodologies has its own strengths and weaknesses. ICA can be sensitive to noise and artifacts, which can lead to inaccurate network identification. Other concerns include the potential fragmentation of a single network into subnetworks, contingent on the specified number of independent components, and the necessity of manual or computer-driven identification of specific networks when utilizing ICA. Seed-based correlation analysis relies on a priori selection of regions of interest, which may introduce bias into the analysis. Graph theory-based analysis requires careful consideration of network construction parameters, which can influence the results. Researchers must therefore carefully choose the appropriate methodology based on their research question, sample size, and data quality [8, 20, 44].
Special considerations in pediatric FC analysis
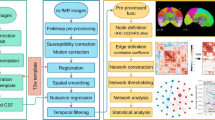
When analyzing rs-fMRI in pediatric populations, researchers face unique challenges that require specialized approaches. Preprocessing pipelines for pediatric data often require customization. For instance, smoothing kernel sizes should be adjusted to smaller values (e.g., 4–6 mm FWHM instead of the adult standard of 6–8 mm) to account for smaller brain structures. Denoising strategies may be more aggressive, potentially incorporating both ICA-based noise removal and nuisance regression approaches [61].
Motion artifacts, a primary concern due to children’s tendency to move more during scans, necessitate robust correction techniques [62]. Real-time motion correction capabilities that adjust for movement during the scan itself, if available, should be applied [63]. Advanced motion correction tools are essential for improving the quality of pediatric rs-fMRI data due to prevalent movement. Advanced software solutions include HALFpipe (Harmonized Analysis of Functional MRI pipeline) [64] and HCP pipelines for comprehensive preprocessing [65]. ICA-based approaches like ICA-AROMA [65] and ICA-FIX [66] help identify and remove motion artifacts. Combined techniques, such as motion scrubbing using stringent frame-wise displacement thresholds (sometimes as low as 0.2–0.3 mm) with temporal interpolation, are highly recommended. Physiological differences in children, such as higher respiratory and heart rates, can affect data quality. Advanced physiological noise correction techniques such as CompCor [66] or RETROICOR [67] should be customized for children to further enhance data integrity and ensure higher quality and reliability in FC studies.
Developmental factors play a crucial role in pediatric FC analysis. The brain’s ongoing development leads to greater variability in structure and function. The pediatric brain differs from the adult brain not only in size but also in tissue contrast and cortical folding patterns. These anatomical differences pose challenges for image processing and analysis, particularly in template selection and spatial normalization. The reduced availability of age-appropriate anatomical and functional templates for pediatric populations further complicates these issues. Large open-source neuroimaging datasets exist such as the NIH MRI study of normal brain development [68], the Baby Connectome Project [69] and the Developing Human Connectome Project [70]. Additionally, the National Institutes of Health (NIH) has announced the HEALthy Brain and Child Development Study (HBCD). However, there is still no universally accepted common brain template to define a common space for normalization similar to MNI in adults [71]. Popular choices are the UNC infant templates 0–1–2 [72], the JHU neonate atlases [73], and some age-specific atlases [74,75,76,77,78], including fetal atlases [79]. For custom template creation, Advanced Normalization Tools (ANTs) offers powerful options, allowing researchers to generate study-specific templates that accurately represent their sample’s characteristics [80].
FC applications
Resting-state functional magnetic resonance imaging studies have been conducted in pediatrics, including fetuses, infants, and children. These studies have explored various aspects of functional brain connectivity, such as tracking changes in FC patterns that occur during brain development, identifying subtle differences in FC patterns in children with neurological disorders, and assessing the effects of interventions on FC patterns in children.
The initial fetal resting-state study by Schopf et al. demonstrated a bilateral occipital network, robust connections between the medial and lateral prefrontal regions, and the presence of a lateralized (either right or left) network in the superior temporal cortical regions [81]. Subsequent investigations highlighted the developmental trajectory of the fetal functional connectome, a network of communicating brain regions during rest, which forms the basis for cognitive and behavioral abilities. Examination of the fetal functional connectome using rs-fMRI identified networks resembling those observed in adults, including the default mode network, the sensorimotor network, and the visual network [82]. Further studies characterized the maturation of the fetal functional connectome, revealing features such as small-worldness, modularity, and hubs [83, 84], along with gradient organization of connectivity within RSNs [31]. Associations were reported between fetal FC and factors such as gestational age [85] sex [86], prenatal exposure to lead [87], and preterm birth [88]. Furthermore alterations in FC of the developing brain have been reported in relation with maternal mental illness with hyperconnectivity in areas related with emotional regulation or connectivity disruptions related with vulnerability to develop schizophrenia [89]. These collective findings indicate the sensitivity of the fetal functional connectome to environmental influences and its potential to reflect individual differences in brain development and function.
Initial observations of immature forms of RSNs have been reported in very preterm infants born at or even before 30 weeks gestation, who were scanned at their term equivalent postmenstrual age [29]. These findings have been supported by more recent studies that consistently reported adult-like RSNs in neonates [27, 90,91,92,93,94,95,96]. These early networks, although immature, show developmental changes over time, forming stronger interhemispheric connections between localized homotopic brain regions with increasing age. Rates of development vary across networks, with primary networks, such as sensorimotor, auditory, and visual networks, exhibiting an adult-like configuration before higher-order association networks like the language, the social interaction, the memory, and the executive control networks [89]. The emergence of RSNs in preterm infants represents a potential milestone in their brain development that not only mirrors their cognitive functions but also holds promise for their evaluation [26, 97,98,99,100,101]. Additionally, the application of resting-state functional connectivity (rs-FC) has yielded novel insights into the neurobiological basis of developmental abnormalities. Recent literature has implicated network-specific disruptions in RSNs architecture due to prematurity, correlating with neurological disturbances in preterm infants, such as autism spectrum disorders, attention deficit hyperactivity disorder, and Tourette syndrome [32, 97, 102,103,104]. In very preterm infants, the FC of the medial orbitofrontal cortex with the rest of the brain depends on body growth and the degree of prematurity [34]. Very premature and moderate-late preterm infants with low-grade intraventricular hemorrhage at around term-equivalent age may present functional impairment consistent with abnormal neurodevelopmental outcomes [23, 36]. Research on brain FC in preterm infants with hypoxic-ischemic encephalopathy (HIE) reveals significant abnormalities compared to healthy controls characterized by disrupted neural networks and atypical connectivity patterns, particularly in regions critical for cognitive and motor development [105]. Very preterm infants with diffuse white matter abnormalities exhibit reduced FC in the executive control and the frontoparietal networks [106]. Severe HIE cases exhibited decreased local efficiency and clustering coefficient in whole-brain networks compared to mild cases [107]. However, no significant differences were found in rs-FC between cooled HIE children without cerebral palsy and healthy controls at early school age, despite underlying structural differences [108]. Apart from morbidities related to prematurity, babies hospitalized in the intensive care unit undergo multiple invasive and painful procedures. Early pain exposure in preterm neonates has been associated with altered brain development and long-term neurodevelopmental consequences. Studies have shown that invasive procedures during neonatal intensive care can lead to decreased FC between key brain regions involved in pain processing, such as the thalamus, somatosensory cortex, and insular cortex [109]. These alterations are more pronounced in infants undergoing more invasive procedures and can affect neurodevelopmental outcomes at 24 months [109, 110]. Increased procedural pain exposure in neonatal intensive care unit have been associated with significant changes in brain structure and connectivity that include regions relevant for pain processing [111, 112]. Additionally, cumulative pain exposure has been linked to slower growth of brain structures, particularly in females [110]. Long-term effects include changes in somatosensory function, brain structure, and pain response later in life [113].
Research on functional brain connectivity in full-term neonates has revealed the developmental trajectories of RSNs. Although still immature at full-term birth, neonatal brain presents prominent rich club, small-world and modularity structure indicative of a high efficient and organized topology [25, 28, 114, 115]. Global and local efficiency keep increasing the first year of life due to the development of long-distance connections, while remaining stable during the second year [115]. In general, the early development of the human brain, from mid-gestation to the first two years of life, involves a shift in the growth processes of the functional network [116]. Before birth, the functional network is fragmented and separated, with short-range connections. After birth, it transitions to a more integrated state, emphasizing connections between local clusters and long-range edges. This post-birth phase enhances both global and local information transfer, responding to environmental stimuli and fostering refined responses and higher-order cognitive abilities. This aligns with the “local to distributed” developmental pattern, supporting the hypothesis of interactive specialization in the human brain [4, 5, 117]. Resting-state fMRI studies have been performed to evaluate perinatal morbidities such as hypoxic-ischemic encephalopathy (HIE), which, despite advances in neonatal care, remains one of the most common causes of neonatal mortality and morbidity. Depending on the severity, it can be associated with motor, cognitive, language, learning, and memory abnormalities. In relation with these clinical abnormalities, rs-fMRI studies have demonstrated that infants with severe HIE, compared with those with mild HIE, display abnormal local and global topological properties of the RSNs [105, 107]. Furthermore the degree of abnormality in RSNs was associated with acute exam findings and outcomes in neonatal acute brain injury, with total lack of network detection observed only in non-survivors [118].
Research indicates that the FC of a child’s brain becomes more similar to that of an adult as they age, with significant changes occurring during adolescence [5, 119]. This shift is characterized by a trend towards increased correlation strength between distant brain regions, reflecting a transition from a local to a more distributed architecture [5]. This findings suggests that there is a trend towards increased correlation strength between distant brain regions during this developmental phase [6]. This shift in FC reflects the maturation process, where higher-order cognitive functions are fine-tuned and specialized. The primitive architecture observed in certain brain networks in younger children gradually evolves into a more integrated and mature configuration during adolescence, contributing to the overall development of cognitive abilities [6].
Various brain diseases affecting children and adolescents, such as Autism Spectrum Disorder [30], Attention Deficit Hyperactivity Disorder [120], and Major Depressive Disorder [121], have been associated with abnormal rs-FC. However, this is not an exhaustive list, as epilepsy [122], bipolar disorder, anxiety disorders [33] and obsessive-compulsive disorder [123], among others, can also impact this population. Each of these conditions may exhibit distinct patterns of rs-FC alterations, emphasizing the need for further research to explore the potential of FC as a diagnostic and therapeutic tool for a diverse range of brain diseases in children and adolescents.
A representative example of the work that remains before FC becomes a clinical tool is in the field of pediatric epilepsy. Decreased connectivity in the default mode network (DMN) and the prefrontal-limbic system has been reported in children with epilepsy intractable to medical treatment [107, 124]. A systematic review on FC in children with focal epilepsy demonstrated abnormal interhemispheric connections and decreased connectivity within and between canonical functional networks, particularly the DMN [125]. However, findings across studies are inconsistent, with both increased and decreased FC observed in multiple brain regions [125]. Resting-state fMRI has also been used in the detection of epileptogenic areas. Increased ALFF and ReHo has been detected in the hippocampus and adjacent structures in mesial temporal lobe epilepsy, whereas increased ReHo in the thalamus has been reported in patients with generalized tonic-clonic seizures [126]. However, using FC mapping to predict surgical outcomes and identify epileptogenic tissue remains challenging [127]. Despite its promise, the wide variety of FC study methodologies currently limits clinical application, highlighting the need for longitudinal studies to understand network evolution during disease progression [125].
Conclusion
The exploration of brain FC in pediatrics through rs-fMRI has provided invaluable insights into the network dynamics of the developing brain. It has allowed researchers to unravel the complexity of neural interactions, shedding light on the maturation and organization of functional circuits in pediatric populations. FC methods have proven significant in understanding neurodevelopmental trajectories, identifying potential biomarkers for the early detection of neurological disorders, and informing targeted interventions. The integration of multi-modal imaging techniques and collaborative efforts across disciplines holds great potential for unlocking further secrets of pediatric brain function. Thus, despite the technical challenges, brain FC is a valuable tool that pediatric radiologists should embrace in their work. As the technology continues to develop and the challenges are addressed, this tool is likely to become increasingly important in the diagnosis, treatment, and understanding of neurological conditions in children.
References
Stedman JM (2013) Aristotle and Modern Cognitive psychology and neuroscience: an analysis of similarities and differences. J Mind Behav 34:121–132
Behrens TEJ, Sporns O (2012) Human connectomics. Curr Opin Neurobiol 22:144–153. https://doi.org/10.1016/j.conb.2011.08.005
Friston KJ (2011) Functional and effective connectivity: a review. Brain Connect 1:13–36. https://doi.org/10.1089/brain.2011.0008
Vértes PE, Bullmore ET (2015) Annual research review: growth connectomics - the organization and reorganization of brain networks during normal and abnormal development. J Child Psychol Psychiatry 56:299–320. https://doi.org/10.1111/jcpp.12365
Fair DA, Cohen AL, Power JD et al (2009) Functional brain networks develop from a local to distributed organization. PLoS Comput Biol 5:e1000381. https://doi.org/10.1371/journal.pcbi.1000381
Hoff GEA-J, Van den Heuvel MP, Benders MJNL et al (2013) On development of functional brain connectivity in the young brain. Front Hum Neurosci 7:650. https://doi.org/10.3389/fnhum.2013.00650
Freilich ER, Gaillard WD (2010) Utility of functional MRI in pediatric neurology. Curr Neurol Neurosci Rep 10:40–46. https://doi.org/10.1007/s11910-009-0077-7
Li K, Guo L, Nie J et al (2009) Review of methods for functional brain connectivity detection using fMRI. Comput Med Imaging Graph 33:131–139. https://doi.org/10.1016/j.compmedimag.2008.10.011
Baert AL (1999) Functional MRI, 1st edition. Springer, Berlin Heidelberg
Forster BB, MacKay AL, Whittall KP et al (1998) Functional magnetic resonance imaging: the basics of blood-oxygen-level dependent (BOLD) imaging. Can Assoc Radiol J 49:320–329
Kozberg M, Hillman E (2016) Neurovascular coupling and energy metabolism in the developing brain. Prog Brain Res 225:213–242. https://doi.org/10.1016/bs.pbr.2016.02.002
Arichi T, Fagiolo G, Varela M et al (2012) Development of BOLD signal hemodynamic responses in the human brain. NeuroImage 63:663–673. https://doi.org/10.1016/j.neuroimage.2012.06.054
Anderson AW, Marois R, Colson ER et al (2001) Neonatal auditory activation detected by functional magnetic resonance imaging. Magn Reson Imaging 19:1–5. https://doi.org/10.1016/s0730-725x(00)00231-9
Brauer J, Neumann J, Friederici AD (2008) Temporal dynamics of perisylvian activation during language processing in children and adults. NeuroImage 41:1484–1492. https://doi.org/10.1016/j.neuroimage.2008.03.027
Harris JJ, Reynell C, Attwell D (2011) The physiology of developmental changes in BOLD functional imaging signals. Dev Cogn Neurosci 1:199–216. https://doi.org/10.1016/j.dcn.2011.04.001
Biswal B, Yetkin FZ, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34:537–541. https://doi.org/10.1002/mrm.1910340409
Smith SM, Fox PT, Miller KL et al (2009) Correspondence of the brain’s functional architecture during activation and rest. Proc Natl Acad Sci U S A 106:13040–13045. https://doi.org/10.1073/pnas.0905267106
Fukunaga M, Horovitz SG, van Gelderen P et al (2006) Large-amplitude, spatially correlated fluctuations in BOLD fMRI signals during extended rest and early sleep stages. Magn Reson Imaging 24:979–992. https://doi.org/10.1016/j.mri.2006.04.018
Zou Q-H, Zhu C-Z, Yang Y et al (2008) An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF. J Neurosci Methods 172:137–141. https://doi.org/10.1016/j.jneumeth.2008.04.012
Smitha K, Akhil Raja K, Arun K et al (2017) Resting state fMRI: a review on methods in resting state connectivity analysis and resting state networks. Neuroradiol J 30:305–317. https://doi.org/10.1177/1971400917697342
Yang H, Long X-Y, Yang Y et al (2007) Amplitude of low frequency fluctuation within visual areas revealed by resting-state functional MRI. NeuroImage 36:144–152. https://doi.org/10.1016/j.neuroimage.2007.01.054
Vincent JL, Patel GH, Fox MD et al (2007) Intrinsic functional architecture in the anaesthetized monkey brain. Nature 447:83–86. https://doi.org/10.1038/nature05758
Argyropoulou MI, Astrakas LG, Xydis VG et al (2020) Is low-grade intraventricular hemorrhage in very preterm infants an innocent condition? Structural and functional evaluation of the brain reveals regional neurodevelopmental abnormalities. Am J Neuroradiol 41:542–547. https://doi.org/10.3174/ajnr.A6438
Zang Y, Jiang T, Lu Y et al (2004) Regional homogeneity approach to fMRI data analysis. NeuroImage 22:394–400. https://doi.org/10.1016/j.neuroimage.2003.12.030
De Asis-Cruz J, Bouyssi-Kobar M, Evangelou I et al (2015) Functional properties of resting state networks in healthy full-term newborns. Sci Rep 5. https://doi.org/10.1038/srep17755
Della Rosa PA, Canini M, Marchetta E et al (2021) The effects of the functional interplay between the default Mode and Executive Control Resting State Networks on cognitive outcome in preterm born infants at 6 months of age. Brain Cogn 147:105669. https://doi.org/10.1016/j.bandc.2020.105669
Doria V, Beckmann CF, Arichi T et al (2010) Emergence of resting state networks in the preterm human brain. Proc Natl Acad Sci U S A 107:20015–20020. https://doi.org/10.1073/pnas.1007921107
Fransson P, Åden U, Blennow M, Lagercrantz H (2011) The functional architecture of the infant brain as revealed by resting-state fMRI. Cereb Cortex 21:145–154. https://doi.org/10.1093/cercor/bhq071
Fransson P, Skiöld B, Horsch S et al (2007) Resting-state networks in the infant brain. Proc Natl Acad Sci U S A 104:15531–15536. https://doi.org/10.1073/pnas.0704380104
Khandan Khadem-Reza Z, Shahram MA, Zare H (2023) Altered resting-state functional connectivity of the brain in children with autism spectrum disorder. Radiol Phys Technol 16:284–291. https://doi.org/10.1007/s12194-023-00717-2
Moore JW, Wilson S, Oldehinkel M et al (2023) Gradient organisation of functional connectivity within resting state networks is present from 25 weeks gestation in the human fetal brain. https://doi.org/10.7554/eLife.90536.1. eLife 12:
Posner J, Park C, Wang Z (2014) Connecting the dots: a review of resting connectivity MRI studies in attention-deficit/hyperactivity disorder. Neuropsychol Rev 24:3–15. https://doi.org/10.1007/s11065-014-9251-z
Sylvester CM, Barch DM, Corbetta M et al (2013) Resting state functional connectivity of the ventral attention network in children with a history of depression or anxiety. J Am Acad Child Adolesc Psychiatry 52:1326–1336e5. https://doi.org/10.1016/j.jaac.2013.10.001
Mouka V, Drougia A, Xydis VG et al (2019) Functional and structural connectivity of the brain in very preterm babies: relationship with gestational age and body and brain growth. Pediatr Radiol 49:1078–1084. https://doi.org/10.1007/s00247-019-04412-6
Argyropoulou MI, Astrakas LG, Xydis VG et al (2020) Is low-Grade Intraventricular Hemorrhage in very Preterm infants an innocent Condition? Structural and functional evaluation of the Brain reveals Regional Neurodevelopmental abnormalities. AJNR Am J Neuroradiol 41:542–547. https://doi.org/10.3174/ajnr.A6438
Argyropoulou MI, Xydis VG, Drougia A et al (2022) Structural and functional brain connectivity in moderate-late preterm infants with low-grade intraventricular hemorrhage. Neuroradiology 64:197–204. https://doi.org/10.1007/s00234-021-02770-3
Bernal B, Grossman S, Gonzalez R, Altman N (2012) FMRI under sedation: what is the best choice in children? J Clin Med Res 4:363–370. https://doi.org/10.4021/jocmr1047w
Ball G, Pazderova L, Chew A et al (2015) Thalamocortical connectivity predicts Cognition in Children Born Preterm. Cereb Cortex 25:4310–4318. https://doi.org/10.1093/cercor/bhu331
Wei Z, Alcauter S, Jin K et al (2013) Graph theoretical analysis of sedation’s effect on whole brain functional system in school-aged children. Brain Connect 3:177–189. https://doi.org/10.1089/brain.2012.0125
Gemma M, de Vitis A, Baldoli C et al (2009) Functional magnetic resonance imaging (fMRI) in children sedated with propofol or midazolam. J Neurosurg Anesthesiol 21:253–258. https://doi.org/10.1097/ANA.0b013e3181a7181d
Martin E, Joeri P, Loenneker T et al (1999) Visual processing in infants and children studied using functional MRI. Pediatr Res 46:135–140. https://doi.org/10.1203/00006450-199908000-00001
Coull JT, Jones MEP, Egan TD et al (2004) Attentional effects of noradrenaline vary with arousal level: selective activation of thalamic pulvinar in humans. NeuroImage 22:315–322. https://doi.org/10.1016/j.neuroimage.2003.12.022
Soares JM, Magalhães R, Moreira PS et al (2016) A hitchhiker’s guide to functional magnetic resonance imaging. Front Neurosci 10:515. https://doi.org/10.3389/fnins.2016.00515
Yang J, Gohel S, Vachha B (2020) Current methods and new directions in resting state fMRI. Clin Imaging 65:47–53. https://doi.org/10.1016/j.clinimag.2020.04.004
Kline RB (2011) Principles and practice of structural equation modeling, 3rd edn. Guilford Press, New York, NY, US
Ding M, Chen Y, Bressler SL (2006) Granger Causality: Basic Theory and Application to Neuroscience. Handbook of Time Series Analysis. Wiley, Ltd, pp 437–460
Friston KJ, Harrison L, Penny W (2003) Dynamic causal modelling. NeuroImage 19:1273–1302. https://doi.org/10.1016/s1053-8119(03)00202-7
Lee MH, Hacker CD, Snyder AZ et al (2012) Clustering of resting state networks. PLoS ONE 7:e40370. https://doi.org/10.1371/journal.pone.0040370
Calhoun VD, Adali T, Pearlson GD, Pekar JJ (2001) A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp 14:140–151. https://doi.org/10.1002/hbm.1048
Bullmore E, Sporns O (2009) Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 10:186–198. https://doi.org/10.1038/nrn2575
Calhoun VD, de Lacy N (2017) Neuroimaging Clin N Am 27:561–579. https://doi.org/10.1016/j.nic.2017.06.012. Ten Key Observations on the Analysis of Resting-state Functional MR Imaging Data Using Independent Component Analysis
Menon V (2023) 20 years of the default mode network: a review and synthesis. Neuron 111:2469–2487. https://doi.org/10.1016/j.neuron.2023.04.023
Uddin LQ (2015) Salience processing and insular cortical function and dysfunction. Nat Rev Neurosci 16:55–61. https://doi.org/10.1038/nrn3857
Vincent JL, Kahn I, Snyder AZ et al (2008) Evidence for a Frontoparietal Control System revealed by intrinsic functional connectivity. J Neurophysiol 100:3328–3342. https://doi.org/10.1152/jn.90355.2008
Beckmann CF, DeLuca M, Devlin JT, Smith SM (2005) Investigations into resting-state connectivity using independent component analysis. Philos Trans R Soc Lond B Biol Sci 360:1001–1013. https://doi.org/10.1098/rstb.2005.1634
van den Heuvel MP, Hulshoff Pol HE (2010) Exploring the brain network: a review on resting-state fMRI functional connectivity. Eur Neuropsychopharmacol 20:519–534. https://doi.org/10.1016/j.euroneuro.2010.03.008
Sporns O (2018) Graph theory methods: applications in brain networks. Dialogues Clin Neurosci 20:111–121
Sporns O (2010) Networks of the Brain, 1st edition. Mit Pr, Cambridge, Mass
Rubinov M, Sporns O (2010) Complex network measures of brain connectivity: uses and interpretations. NeuroImage 52:1059–1069. https://doi.org/10.1016/j.neuroimage.2009.10.003
Bassett DS, Bullmore ET (2017) Small-world brain networks revisited. Neuroscientist 23:499–516. https://doi.org/10.1177/1073858416667720
Golestani AM, Chen JJ (2024) Comparing data-driven physiological denoising approaches for resting-state fMRI: implications for the study of aging. Front Neurosci 18:1223230. https://doi.org/10.3389/fnins.2024.1223230
Zaitsev M, Maclaren J, Herbst M (2015) Motion artifacts in MRI: a complex problem with many partial solutions. J Magn Reson Imaging 42:887–901. https://doi.org/10.1002/jmri.24850
Maclaren J, Herbst M, Speck O, Zaitsev M (2013) Prospective motion correction in brain imaging: a review. Magn Reson Med 69:621–636. https://doi.org/10.1002/mrm.24314
Waller L, Erk S, Pozzi E et al (2022) ENIGMA HALFpipe: interactive, reproducible, and efficient analysis for resting-state and task-based fMRI data. Hum Brain Mapp 43:2727–2742. https://doi.org/10.1002/hbm.25829
Glasser MF, Sotiropoulos SN, Wilson JA et al (2013) The minimal preprocessing pipelines for the human Connectome Project. NeuroImage 80:105–124. https://doi.org/10.1016/j.neuroimage.2013.04.127
Behzadi Y, Restom K, Liau J, Liu TT (2007) A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. NeuroImage 37:90–101. https://doi.org/10.1016/j.neuroimage.2007.04.042
Glover GH, Li TQ, Ress D (2000) Image-based method for retrospective correction of physiological motion effects in fMRI: RETROICOR. Magn Reson Med 44:162–167. https://doi.org/10.1002/1522-2594(200007)44:1%3C162::aid-mrm23%3E3.0.co;2-e
Evans AC, Brain Development Cooperative Group (2006) The NIH MRI study of normal brain development. NeuroImage 30:184–202. https://doi.org/10.1016/j.neuroimage.2005.09.068
Howell BR, Styner MA, Gao W et al (2019) The UNC/UMN Baby Connectome Project (BCP): an overview of the study design and protocol development. NeuroImage 185:891–905. https://doi.org/10.1016/j.neuroimage.2018.03.049
Eyre M, Fitzgibbon SP, Ciarrusta J et al (2021) The developing human Connectome Project: typical and disrupted perinatal functional connectivity. Brain 144:2199–2213. https://doi.org/10.1093/brain/awab118
Dufford AJ, Hahn CA, Peterson H et al (2022) (Un)common space in infant neuroimaging studies: a systematic review of infant templates. Hum Brain Mapp 43:3007–3016. https://doi.org/10.1002/hbm.25816
Shi F, Yap P-T, Wu G et al (2011) Infant brain atlases from neonates to 1- and 2-year-olds. PLoS ONE 6:e18746. https://doi.org/10.1371/journal.pone.0018746
Oishi K, Mori S, Donohue PK et al (2011) Multi-contrast human neonatal brain atlas: application to normal neonate development analysis. NeuroImage 56:8–20. https://doi.org/10.1016/j.neuroimage.2011.01.051
Fonov V, Evans AC, Botteron K et al (2011) Unbiased average age-appropriate atlases for pediatric studies. NeuroImage 54:313–327. https://doi.org/10.1016/j.neuroimage.2010.07.033
Evans AC, Janke AL, Collins DL, Baillet S (2012) Brain templates and atlases. NeuroImage 62:911–922. https://doi.org/10.1016/j.neuroimage.2012.01.024
Sanchez CE, Richards JE, Almli CR (2012) Age-specific MRI templates for pediatric neuroimaging. Dev Neuropsychol 37:379–399. https://doi.org/10.1080/87565641.2012.688900
Molfese PJ, Glen D, Mesite L et al (2021) The Haskins pediatric atlas: a magnetic-resonance-imaging-based pediatric template and atlas. Pediatr Radiol 51:628–639. https://doi.org/10.1007/s00247-020-04875-y
Wilke M, Holland SK, Altaye M, Gaser C (2008) Template-O-Matic: a toolbox for creating customized pediatric templates. NeuroImage 41:903–913. https://doi.org/10.1016/j.neuroimage.2008.02.056
Ciceri T, Casartelli L, Montano F et al (2024) Fetal brain MRI atlases and datasets: a review. NeuroImage 292:120603. https://doi.org/10.1016/j.neuroimage.2024.120603
Avants BB, Tustison NJ, Song G et al (2011) A reproducible evaluation of ANTs similarity metric performance in brain image registration. NeuroImage 54:2033–2044. https://doi.org/10.1016/j.neuroimage.2010.09.025
Schöpf V, Kasprian G, Brugger PC, Prayer D (2012) Watching the fetal brain at rest. Int J Dev Neurosci 30:11–17. https://doi.org/10.1016/j.ijdevneu.2011.10.006
Turk E, van den Heuvel MI, Benders MJ et al (2019) Functional connectome of the fetal brain. J Neurosci 39:9716–9724. https://doi.org/10.1523/JNEUROSCI.2891-18.2019
van den Heuvel MI, Turk E, Manning JH et al (2018) Hubs in the human fetal brain network. Dev Cogn Neurosci 30:108–115. https://doi.org/10.1016/j.dcn.2018.02.001
De Asis-Cruz J, Andersen N, Kapse K et al (2021) Global Network Organization of the fetal functional connectome. Cereb Cortex 31:3034–3046. https://doi.org/10.1093/cercor/bhaa410
Thomason ME, Dassanayake MT, Shen S et al (2013) Cross-hemispheric functional connectivity in the human fetal brain. Sci Transl Med 5:173ra24. https://doi.org/10.1126/scitranslmed.3004978
Wheelock MD, Hect JL, Hernandez-Andrade E et al (2019) Sex differences in functional connectivity during fetal brain development. Dev Cogn Neurosci 36:100632. https://doi.org/10.1016/j.dcn.2019.100632
Thomason ME, Hect JL, Rauh VA et al (2019) Prenatal lead exposure impacts cross-hemispheric and long-range connectivity in the human fetal brain. NeuroImage 191:186–192. https://doi.org/10.1016/j.neuroimage.2019.02.017
Thomason ME, Scheinost D, Manning JH et al (2017) Weak functional connectivity in the human fetal brain prior to preterm birth. Sci Rep 7:39286. https://doi.org/10.1038/srep39286
Gao W, Lin W, Grewen K, Gilmore JH (2017) Functional connectivity of the infant human brain: plastic and modifiable. Neuroscientist 23:169–184. https://doi.org/10.1177/1073858416635986
Smyser CD, Snyder AZ, Neil JJ (2011) Functional connectivity MRI in infants: exploration of the functional organization of the developing brain. NeuroImage 56:1437–1452. https://doi.org/10.1016/j.neuroimage.2011.02.073
Vogel AC, Power JD, Petersen SE, Schlaggar BL (2010) Development of the brain’s functional network architecture. Neuropsychol Rev 20:362–375. https://doi.org/10.1007/s11065-010-9145-7
Lee W, Morgan BR, Shroff MM et al (2013) The development of regional functional connectivity in preterm infants into early childhood. Neuroradiol 55 Suppl 2:105–111. https://doi.org/10.1007/s00234-013-1232-z
Smyser CD, Inder TE, Shimony JS et al (2010) Longitudinal analysis of neural network development in preterm infants. Cereb Cortex 20:2852–2862. https://doi.org/10.1093/cercor/bhq035
Fransson P, Skiöld B, Engström M et al (2009) Spontaneous brain activity in the newborn brain during natural sleep-an fMRI study in infants born at full term. Pediatr Res 66:301–305. https://doi.org/10.1203/PDR.0b013e3181b1bd84
Cao M, Huang H, He Y (2017) Developmental Connectomics from Infancy through Early Childhood. Trends Neurosci 40:494–506. https://doi.org/10.1016/j.tins.2017.06.003
Gilmore JH, Knickmeyer RC, Gao W (2018) Imaging structural and functional brain development in early childhood. Nat Rev Neurosci 19:123–137. https://doi.org/10.1038/nrn.2018.1
Bouyssi-Kobar M, De Asis-Cruz J, Murnick J et al (2019) Altered functional Brain Network Integration, Segregation, and modularity in infants born very Preterm at Term-Equivalent Age. J Pediatr 213:13–21e1. https://doi.org/10.1016/j.jpeds.2019.06.030
Wheelock MD, Austin NC, Bora S et al (2018) Altered functional network connectivity relates to motor development in children born very preterm. NeuroImage 183:574–583. https://doi.org/10.1016/j.neuroimage.2018.08.051
Alcauter S, Lin W, Keith Smith J et al (2014) Development of thalamocortical connectivity during infancy and its cognitive correlations. J Neurosci 34:9067–9075. https://doi.org/10.1523/JNEUROSCI.0796-14.2014
Sylvester CM, Smyser CD, Smyser T et al (2018) Cortical functional connectivity Evident after birth and behavioral inhibition at Age 2. Am J Psychiatry 175:180–187. https://doi.org/10.1176/appi.ajp.2017.17010018
Rogers CE, Sylvester CM, Mintz C et al (2017) Neonatal Amygdala Functional Connectivity at Rest in Healthy and Preterm Infants and early internalizing symptoms. J Am Acad Child Adolesc Psychiatry 56:157–166. https://doi.org/10.1016/j.jaac.2016.11.005
Church JA, Fair DA, Dosenbach NUF et al (2009) Control networks in paediatric Tourette syndrome show immature and anomalous patterns of functional connectivity. Brain 132:225–238. https://doi.org/10.1093/brain/awn223
Redcay E, Moran JM, Mavros PL et al (2013) Intrinsic functional network organization in high-functioning adolescents with autism spectrum disorder. Front Hum Neurosci 7:573. https://doi.org/10.3389/fnhum.2013.00573
Greene DJ, Koller JM, Robichaux-Viehoever A et al (2015) Reward enhances tic suppression in children within months of tic disorder onset. Dev Cogn Neurosci 11:65–74. https://doi.org/10.1016/j.dcn.2014.08.005
Wang Y, Wang Y, Hua G et al (2023) Changes of functional Brain Network in neonates with different degrees of hypoxic-ischemic Encephalopathy. Brain Connect 13:427–435. https://doi.org/10.1089/brain.2022.0073
He L, Parikh NA (2015) Aberrant executive and frontoparietal functional connectivity in very preterm infants with diffuse White Matter abnormalities. Pediatr Neurol 53:330–337. https://doi.org/10.1016/j.pediatrneurol.2015.05.001
Li H-X, Yu M, Zheng A-B et al (2019) Resting-state network complexity and magnitude changes in neonates with severe hypoxic ischemic encephalopathy. Neural Regen Res 14:642–648. https://doi.org/10.4103/1673-5374.247468
Spencer APC, Goodfellow M, Chakkarapani E, Brooks JCW (2024) Resting-state functional connectivity in children cooled for neonatal encephalopathy. Brain Commun 6:fcae154. https://doi.org/10.1093/braincomms/fcae154
Tortora D, Severino M, Di Biase C et al (2019) Early Pain exposure influences functional brain connectivity in very Preterm neonates. Front Neurosci 13:899. https://doi.org/10.3389/fnins.2019.00899
Schneider J, Duerden EG, Guo T et al (2018) Procedural pain and oral glucose in preterm neonates: brain development and sex-specific effects. Pain 159:515–525. https://doi.org/10.1097/j.pain.0000000000001123
Ranger M, Grunau RE (2014) Early repetitive pain in preterm infants in relation to the developing brain. Pain Manag 4:57–67. https://doi.org/10.2217/pmt.13.61
Duerden EG, Grunau RE, Guo T et al (2018) Early Procedural Pain is Associated with regionally-specific alterations in Thalamic Development in Preterm neonates. J Neurosci 38:878–886. https://doi.org/10.1523/JNEUROSCI.0867-17.2017
Walker SM (2019) Long-term effects of neonatal pain. Seminars in fetal and neonatal Medicine 24. https://doi.org/10.1016/j.siny.2019.04.005
Cao M, He Y, Dai Z et al (2017) Early development of functional network segregation revealed by connectomic analysis of the preterm human brain. Cereb Cortex 27:1949–1963. https://doi.org/10.1093/cercor/bhw038
Gao W, Gilmore JH, Giovanello KS et al (2011) Temporal and spatial evolution of brain network topology during the first two years of life. PLoS ONE 6. https://doi.org/10.1371/journal.pone.0025278
Zhao T, Xu Y, He Y (2019) Graph theoretical modeling of baby brain networks. NeuroImage 185:711–727. https://doi.org/10.1016/j.neuroimage.2018.06.038
Johnson MH (2000) Functional brain development in infants: elements of an interactive specialization framework. Child Dev 71:75–81. https://doi.org/10.1111/1467-8624.00120
Boerwinkle VL, Sussman BL, Manjón I et al (2022) Association of network connectivity via resting state functional MRI with consciousness, mortality, and outcomes in neonatal acute brain injury. Neuroimage Clin 34:102962. https://doi.org/10.1016/j.nicl.2022.102962
Jolles DD, van Buchem MA, Crone EA, Rombouts SARB (2011) A comprehensive study of whole-brain functional connectivity in children and young adults. Cereb Cortex 21:385–391. https://doi.org/10.1093/cercor/bhq104
Zhao Q, Li H, Yu X et al (2017) Abnormal resting-state functional connectivity of insular subregions and disrupted correlation with working memory in adults with attention Deficit/Hyperactivity disorder. Front Psychiatry 8:200. https://doi.org/10.3389/fpsyt.2017.00200
Singh MK, Gotlib IH (2014) The neuroscience of Depression: implications for Assessment and intervention. Behav Res Ther 62:60–73. https://doi.org/10.1016/j.brat.2014.08.008
Boerwinkle VL, Mirea L, Gaillard WD et al (2020) Resting-state functional MRI connectivity impact on epilepsy surgery plan and surgical candidacy: prospective clinical work. J Neurosurg Pediatr 1–8. https://doi.org/10.3171/2020.1.PEDS19695
Armstrong CC, Moody TD, Feusner JD et al (2016) Graph-theoretical analysis of resting-state fMRI in pediatric obsessive-compulsive disorder. J Affect Disord 193:175–184. https://doi.org/10.1016/j.jad.2015.12.071
Widjaja E, Zamyadi M, Raybaud C et al (2013) Impaired default mode network on resting-state FMRI in children with medically refractory epilepsy. AJNR Am J Neuroradiol 34:552–557. https://doi.org/10.3174/ajnr.A3265
Feng X, Piper RJ, Prentice F et al (2024) Functional brain connectivity in children with focal epilepsy: a systematic review of functional MRI studies. Seizure 117:164–173. https://doi.org/10.1016/j.seizure.2024.02.021
Chen Z, An Y, Zhao B et al (2017) The value of resting-state functional magnetic resonance imaging for detecting epileptogenic zones in patients with focal epilepsy. PLoS ONE 12:e0172094. https://doi.org/10.1371/journal.pone.0172094
Constable RT, Scheinost D, Finn ES et al (2013) Potential use and challenges of functional connectivity mapping in intractable epilepsy. Front Neurol 4:39. https://doi.org/10.3389/fneur.2013.00039
Funding
This study received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Informed consent
The requirement for informed consent was waived by the Institutional Review Board due to the retrospective nature of the study.
Conflict of interest
The authors declare that they have no conflicts of interest related to this research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Argyropoulou, M.I., Xydis, V.G. & Astrakas, L.G. Functional connectivity of the pediatric brain. Neuroradiology (2024). https://doi.org/10.1007/s00234-024-03453-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00234-024-03453-5