Abstract
Purpose
To determine the diagnostic accuracy of 3D time of flight MR angiography (TOF-MRA), contrast enhanced MR angiography (CE-MRA), and T1-weighted high-resolution isotropic volume examination (THRIVE) at 3 T for the evaluation of intracranial aneurysm occlusion after endovascular treatment and to evaluate the usability of the THRIVE sequence in endovascular treatment follow-up.
Methods
In 3 T MR follow-up examinations of 66 aneurysms in 50 patients treated endovascularly, 3D TOF-MRA (index test), THRIVE (index test), and CE-MRA (reference standard) examinations were performed in a retrospective consecutive case series. Source images were classified as class 1, class 2, and class 3 according to the Raymond criteria using MIP (maximum intensity projection) techniques. The compatibility between sequences was evaluated with the Kappa test. The sensitivity and specificity were also calculated.
Results
In the evaluation of THRIVE and CE-MRA sequences, compatibility was determined in 61 cases in total, with an overall fit of 61/66 (92.42%). A statistically significant correlation was found between THRIVE and CE-MRA (p < 0.001, κ = 0.800). In the evaluation of TOF and CE-MRA sequences, compatibility was determined in 54 cases in total, and the overall fit was 54/66 (81.8%). A statistically significant agreement was found between TOF and CE-MRA (p < 0.001, κ = 0.502). Assuming that CE-MRA is a reference standard, the sensitivity and specificity of the TOF sequence were 44.4% and 97.9%, respectively, and the sensitivity and specificity of the THRIVE sequence were 77.8% and 97.9%, respectively.
Conclusion
The THRIVE sequence can be used as a noncontrast method for monitoring endovascularly treated intracranial aneurysms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Intracranial aneurysms have significant neurological mortality and morbidity. Treatment options include surgical intervention or endovascular therapy [1]. Endovascular treatment is the first-line treatment method in most aneurysms. The purpose of long-term follow-up for these aneurysms is to determine possible recurrence and to evaluate retreatment if necessary [2, 3]. Digital subtraction angiography (DSA) used for diagnostic procedures and posttreatment follow-up is a more sensitive method with a high accuracy rate when compared to all other imaging techniques and is considered the gold standard method. However, catheter angiography is an invasive diagnostic method with the disadvantages of thromboembolism, contrast agent reactions, nephrotoxicity, and insertion site complications [2, 3].
MRA (magnetic resonance angiography) used for the diagnosis and posttreatment follow-up of cerebral aneurysms has become an alternative diagnostic method to DSA [4, 5]. In addition to providing an accurate and rapid diagnosis of intracranial aneurysms, MRA is an alternative technique that is noninvasive, has no radiation risk, and does not carry the risks of conventional angiography [6].
Technically, PCA (phase contrast angiography), TOF (time of flight), and CE-MRA (contrast enhanced magnetic resonance angiography) are basic MRA methods [6]. The preferred method of imaging intracranial aneurysms is TOF-MRA. This provides high-quality images without contrast and has better resolution and an improved signal-to-noise ratio, although there is also the disadvantage of turbulent flow in carotid siphons and large aneurysms. The CE-MRA method is easier to interpret than the PC-MRA and TOF-MRA methods, and sensitivity artifacts are less common. With the CE-MRA method, 0.1–0.2 mmol/kg gadolinium-based intravenous (IV) contrast is obtained on T1-weighted images following a bolus injection. In a routine protocol, images are usually obtained using fast spoiled gradient recalled-based sequences [6]. Many studies comparing MRA and IA-DSA (intra-arterial digital subtraction angiography) have shown that MRA is the correct technique to demonstrate aneurysm residue or recanalization after endovascular coil embolization. In some studies, CE-MRA has been evaluated, while in others, 3D TOF or both techniques have been evaluated together [7].
The T1-weighted high-resolution isotropic volume examination (THRIVE) sequence SENSE is an optimized, fast, T1-weighted 3D imaging technique that combines large volumes and homogeneous fat suppression. 3D T1-weighted imaging is a method that uses parallel signal acquisition techniques that provide fast high-resolution scans and allow contrast-enhanced imaging of the liver, intraabdominal organs, small intestine, breast, prostate, and pancreas. The THRIVE sequence provides high-resolution isotropic images and only requires the patient to hold his or her breath for a short breath holding time, providing suitable images in dynamic liver MR. The THRIVE sequence is an important technique that enables detailed imaging of neuroanatomical structures with or without dynamic contrast. Tartari et al. [8] used contrast and noncontrast THRIVE sequences to visualize atherosclerotic plaques in the extracranial segments of the carotid artery.
The aim of this study was to compare TOF-MRA, CE-MRA, and THRIVE techniques at 3 T for the evaluation of intracranial aneurysm occlusion after endovascular treatment and to evaluate the usability of THRIVE sequences in endovascular treatment follow-up.
Materials and methods
Study population
A retrospective evaluation was made of patients who underwent endovascular intracranial aneurysm treatment between January 2012 and November 2018 in Bursa Uludag University Faculty of Medicine, Department of Radiology. The study included patients with scans obtained via 3 T MR with the appropriate technique and protocol and patients who underwent 3D TOF-MRA, THRIVE, and CE-MRA sequences in the same session. Patients with insufficient MRA sequence analyses, patients who previously had artifacts due to intracranial surgical material, and patients aged < 18 years were excluded from the study. Patients in the entire universe were evaluated in this study. A total of 66 aneurysms in 50 patients in a consecutive case series were included in the study. The demographic data of the patients were obtained from the Avicenna Hospital Information Management Systems program used in our hospital.
Technique
With the diagnostic imaging performed using the DSA device (AXIOM Artis, Siemens, Erlangen, Germany) in the Interventional Radiology Department, the location and dimensions of the aneurysm were determined.
MRA was performed using a 3 T MR unit (Achieva TX, Philips, Eindhoven, Holland). In all patients, vascular access was opened from the antecubital vein using a 20-gauge needle before the examination. After positioning on the table, the patient was connected to an MR compatible injection (Spectris, Medrad, Pittsburgh, PA) and prepared for CE-MRA, which was set as reference standard. A standard head coil was used in the MRA technique. The patients underwent 3D TOF-MRA, THRIVE, and, most recently, CE-MRA. For CE-MRA, the contrast material with 0.2 ml/kg gadolinium was administered via the antecubital vein with a contrast rate of 2.5 mL/sec. After the administration of the contrast agent, an injection of 20 ml of 0.9% saline injection was made at a rate of 2 ml/sec. The technical parameters of MRA are shown in Table 1. The 3D TOF-MRA, THRIVE, and CE-MRA sequences were evaluated by creating 3D MIP (maximum intensity projection) and 3D VR (volume rendered), each in Centricity RIS-I Plus and Centricity PACS (GE, Fairfield, Connecticut, USA) programs registered at workstations in the radiology department. All images (TOF-MRA, THRIVE, and CE-MRA) were evaluated by two experienced neuroradiologists (EM, BH) until an agreement was reached. The MRA images of the patients treated with endovascular aneurysms were classified as class 1, class 2, and class 3 on the Raymond scale according to the degree of occlusion. The Raymond criteria are described in Fig. 1. Sample cases are shown in Figs. 2, 3, 4, and 5.
Raymond criteria used to evaluate the success of the treatment of patients after endovascular treatment [26]. a Class 1, Complete occlusion b Class 2, Residual neck c Class 3, Residual aneurysm
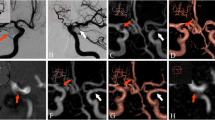
After the endovascular treatment of a 56-year-old male patient basilar truncus fusiform aneurysm, TOF-MRA (a) complete occlusion (class 1), CE-MRA (b) residual neck (class 2, white arrow), THRIVE (c) residual neck were evaluated (white arrow). Anterior Posterior (AP) DSA (D) image shows residual neck (class 2)
Statistical analysis
Data entry and statistical analyses were performed using SPSS 20.0 (IBM SPSS Inc., Chicago, IL, USA) statistical software. Frequency distributions and percentages were calculated for descriptive analysis, and averages are shown with the standard deviation. The aggregation between sequences in distinguishing Raymond criteria was evaluated with the Kappa test and the following parameters: κ = 0–0.2 inadequate agreement, κ = 0.21–0.4 agreement, κ = 0.41–0.6 moderate agreement, κ = 0.61–0.8 substantial agreement, κ = 0.81–1.00 almost perfect agreement, and κ = 1.00 perfect agreement. The sensitivity and specificity of THRIVE and TOF-MRA to CE-MRA were calculated. A value of p < 0.05 was considered statistically significant.
Results
Sixty-six aneurysms of 50 patients, comprising 15 males and 35 females with a mean age of 50.28 ± 13.33 years were evaluated. The age group classification showed 13 patients (26%) aged ≤ 39 years, 30 patients (60%) aged 40–64 years, and 7 patients (14%) aged ≥ 65 years. Anterior system localization was observed in 55 (83.3%) aneurysms, and 11 (16.7%) were in the posterior system. With respect to size, 52 (78.8%) were defined as small aneurysms (≤ 10 mm), 12 (18.2%) as large aneurysms (≤ 25 mm, > 10 mm), and 2 (3%) as giant aneurysms (> 25 mm). While 54 (81.8%) of the cases were saccular aneurysms, 10 (15.8%) were fusiform, and 2 (3%) were fenestrated aneurysms. The size of the aneurysms varied between 3 and 40 mm, with an average size of 10.21 ± 6.92 mm.
Of the aneurysms treated with endovascular treatment, 36 (54.8%) were located in the internal carotid artery, 2 (3%) in the anterior cerebral artery, 10 (15.2%) in the middle cerebral artery, 1 (1.5%) in the posterior cerebral artery, 2 (3%) in the vertebral artery, 9 (13.6%) in the basilar artery (13.6%), 4 (6.1%) in the anterior communicating artery, and 2 (3%) in the posterior communicating artery. A coil was used in 8 cases (12.1%), a stent was used in 37 cases (56.1%), and both coils and stents were used in 21 cases (31.8%).
Assuming that CE-MRA is a reference method, the sensitivity and specificity of the TOF sequence were 44.4% and 97.9%, respectively, and the sensitivity and specificity of the THRIVE sequence were 77.8% and 97.9%, respectively (95% confidence intervals).
When the THRIVE and CE-MRA sequences of the 66 cases were analyzed, of the 48 cases evaluated as complete occlusion with CE-MRA, 47 demonstrated complete occlusion via THRIVE, and 1 case was evaluated as residual neck. Of the 17 cases classified as residual neck with CE-MRA, the THRIVE sequence data indicated that 14 of these cases had residual neck and 3 had full occlusion. In 1 case diagnosed with residual aneurysm by CE-MRA, the evaluation via THRIVE revealed complete occlusion. Compatibility was determined in 61 cases in total, and the overall fit was 61/66 (92.42%). A statistically significant correlation was found between THRIVE and CE-MRA (p < 0.001, κ = 0.800) (Table 2).
In the comparison of 66 cases studied with TOF and CE-MRA sequences, 47 of the 48 cases evaluated as complete occlusion with CE-MRA were classified as complete occlusion with TOF, and 1 case was evaluated as residual neck. Of the 17 cases evaluated as residual neck with CE-MRA, 10 were classified as complete occlusion, and 7 were classified as residual neck when analyzed by TOF. While 1 case was evaluated as residual aneurysm with CE-MRA, no residual aneurysm was detected in the TOF sequence, and 1 case was classified as residual neck. Compatibility was determined in 54 cases in total, and the overall fit was 54/66 (81.8%). The flow of cases is highlighted in Figure 6. A statistically significant agreement was found between TOF and CE-MRA (p < 0.001, κ = 0.502) (Table 3).
In the comparison of the 66 cases examined via TOF and THRIVE sequences, 50 of 51 cases evaluated as complete occlusion with THRIVE were classified as complete occlusion with TOF, and 1 case was evaluated as residual neck. Of the 15 cases evaluated as residual neck with THRIVE, 8 were classified as residual neck and 7 were classified as full occlusion when analyzed in TOF. Overall compatibility was present in 58/66 cases (87.88%). A statistically significant agreement was found between TOF and THRIVE (p < 0.001, κ = 0.594) (Table 4). There was not any adverse event from performing the examinations.
Discussion
Treated aneurysms should be followed up for recurrence and residues, for which there are a few basic modalities. DSA has risks such as invasive interference complications, reactions due to the use of an iodinated contrast agent, and exposure to ionizing radiation. MRA with or without contrast is frequently preferred in the follow-up of endovascularly treated aneurysms due to the absence of ionizing radiation; it is the least invasive method, and there are none of the complications associated with DSA [9, 10]. Performing MRA examination with a contrast agent provides more accurate imaging of the current in the aneurysmal sac. Another advantage of CE-MRA is the short TE time and low number of magnetic susceptibility artifacts [11]. Some studies have shown that CE-MRA is better than TOF-MRA. In a study by Leclerc et al., while CE-MRA sensitivity was 100% in residual neck detection in the follow-up of coiled anterior communicating artery aneurysms, it was found to be 60% for TOF-MRA [12]. In this study, TOF, THRIVE and CE-MRA sequences were evaluated in the comparison of occlusions of endovascularly treated aneurysms. The results we found in comparing the THRIVE sequence with CE-MRA are important for predicting the usability of the technique in neurovascular radiology. The use of the THRIVE sequence in neurovascular practice is limited and relatively new. To the best of our knowledge, there are no data that indicate that the THRIVE sequence has been used in intracranial aneurysms. Due to its noncontrast feature, it can be used as an alternative to TOF or in combination with TOF. In particular, the high sensitivity value compared to TOF shows the superiority of the technique in detecting residues and recurrences.
Despite the advantages of MRA, it has some disadvantages. These include poor visualization of small vessels, overestimation of stenosis, view-to-view variations, false positives, false negatives, and vessel overlap. Extravascular subacute thrombus can be used as an example for false positivity. Intraluminal thrombus or intimal flap may be obscured if surrounded by hyperintense flowing blood. This is an example of false negativity [13].
The calculation of the sensitivity and specificity of MRA after endovascular aneurysm treatment is based on the acceptance of DSA as the gold standard [12, 14, 15]. However, there are also studies where only CE-MRA and the TOF method have been used, and DSA is not accepted as the gold standard for follow-ups. In a study by Shankar et al. [16], 134 aneurysms, which were coiled, were followed up with CE-MRA in the 3rd month, 15th month, 3rd year, and 5th year, but no comparison was made with DSA for grading. This and similar studies are important for predicting long-term MRA follow-up [16,17,18,19]. It was also emphasized in the Shankar study that CE-MRA may be higher in terms of classification compared to DSA. For example, it was stated that an endovascularly treated aneurysm could be reported as a residual neck (class 2) with CE-MRA and defined as complete occlusion (class 1) in DSA control. This was thought to be due to the technique and physical principles of CE-MRA compared to those of DSA. Another highlighted issue in that study was that the aneurysm neck recurrence residues were evaluated more accurately with CE-MRA than with DSA [16]. In the current study, DSA was not accepted as the gold standard, and the different MRA sequences were compared for compatibility with respect to one other.
Complete occlusion rates have been reported to be approximately 50% in follow-up after endovascular treatment [12, 14, 15, 17, 16]. However, these studies included coiled aneurysms and did not include patients treated with stents. In the current study of 66 cases, 58 (73%) were classified as complete occlusion by CE-MRA, 57 (86%) by TOF-MRA, and 51 (77%) by a THRIVE sequence. Coil or stent application was performed in all of these cases.
In the study conducted by Kauffman et al. [18], TOF-MRA and CE-MRA examinations performed under favorable conditions were shown to be better than DSA in aneurysm occlusion assessment. This is important in that it allows safe and noninvasive follow-up of endovascularly treated aneurysms with MRA without DSA examination. As supporting evidence in this regard, the work of some authors has shown that TOF-MRA is more sensitive than DSA in coiled aneurysms [18,19,20,21,22]. Kauffman et al. [18] reported that sensitivity was sufficient for the evaluation of aneurysm residue or recurrence considered as class 2 or class 3. TOF-MRA shows the coil load well. Therefore, for CE-MRA, which can be examined with less contrast, the examination is more valuable in terms of treatment evaluation. In the study conducted by Leclerc et al. [12], very high compatibility was found between CE-MRA and DSA in the posttreatment follow-up of anterior communicating artery aneurysms, with CE-MRA sensitivity of 100% in residual neck detection and 60% in TOF MRA. In a large-scale meta-analysis study by Kewe et al. [23], the specificity rates for TOF-MRA and CE-MRA were found to be 90% and 91.9%, respectively.
The main purpose of this study was to compare the THRIVE sequence results with those of TOF and CE-MRA. When the THRIVE and CE-MRA sequences were compared, there was compatibility in a total of 61/66 cases (92.42%). A statistically significant correlation was found between CE-MRA and THRIVE (p < 0.001, κ = 0.800). In a comparison of the THRIVE sequence with the TOF-MRA sequence, compatibility was determined in 58/66 cases (87.88%). A statistically significant correlation was found between TOF-MRA and THRIVE (p < 0.001, κ = 0.595). The substantial agreement of the noncontrast THRIVE sequence with CE-MRA is a valuable finding in detecting aneurysm recurrence on noncontrast sequences.
The THRIVE sequence SENSE is an optimized, fast, T1-weighted 3D imaging technique that combines large volume and homogeneous fat suppression. THRIVE is a sequence name unique to the Philips 3 T MR (Achieva TX, Philips, Eindhoven, Holland). The name of this sequence is known at SIEMENS as VIBE and at General Electric (GE) as LAVA. 3D T1-weighted imaging is a method that uses parallel signal acquisition techniques, providing fast high-resolution, contrast-enhanced imaging of the liver, intra-abdominal organs, small intestine, breast, prostate, and pancreas. The THRIVE sequence provides high-resolution isotropic images and only requires the patient to hold his or her breath for a short time, providing suitable images in dynamic liver MR examination. After contrast injection, the THRIVE sequence is an important technique that allows dynamic examination of the breast or detailed imaging of neuroanatomic structures without contrast. Fat suppression reduces magnetic susceptibility artifacts and may minimize artifacts caused by a coil or stent. Tartari et al. [8] used the contrast and noncontrast THRIVE sequence to visualize atherosclerotic plaques in the extracranial segments of the carotid artery. In addition, Panyaping et al. [24] compared the contrast-enhanced THRIVE sequence with MR venography in cases of dural venous sinus thrombosis. In that study, MR venography examination of dural venous sinuses in 98 patients was compared with contrast-enhanced 3D THRIVE results. Compared to the 11 dural sinus thromboses detected in the MR venography examination, the 3D THRIVE examination revealed a sensitivity of 81.8% and a specificity of 92%.
To the best of our knowledge, there are no data that indicate that the THRIVE sequence has been used in intracranial aneurysms. The perfect match determined between THRIVE and CE-MRA in the current study is important in that a THRIVE examination, which can be performed without contrast, gives almost similar results to that of CE-MRA. The THRIVE sequence can be safely chosen for patients with impaired renal function. Allergic reactions caused by the use of contrast media are eliminated, and there is no risk of nephrogenic systemic fibrosis. In venous structures, a hyperintense appearance can be observed in the CE-MRA examination, especially when the contrast timing is not suitable, and can be seen with the entry slice phenomenon or flow-related enhancement. This may be a disadvantage because it causes venous overlap in occlusion grading [25]. This is particularly evident at the level of the skull base and in middle cerebral artery aneurysms. Venous overlap with similar features can be seen in the THRIVE sequence. The current study can be considered valuable in that it demonstrates the applicability of the THRIVE sequence in intracranial aneurysms. However, more comprehensive studies are needed in this regard.
There were some limitations to this study. First, the sample size was small. Second, data were collected retrospectively. Third, MRA was not compared to DSA. Fourth, there was no evaluation of posttreatment follow-up processes and changes in aneurysm occlusion rates. Finally, no classification was performed according to stent types.
Conclusion
MRA can be considered an important method in the follow-up of intracranial aneurysms treated with endovascular aneurysms compared to the invasive method of DSA. In this study, the THRIVE sequence was used in addition to the standard MRA examination, and the findings demonstrate that the THRIVE sequence can be safely chosen for patients with impaired renal function. Further studies are needed with larger patient groups to highlight the importance of the THRIVE sequence.
Availability of data and material
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Molyneux A, Kerr R, Stratton I, Sandercock P, Clarke M, Shrimpton J, Holman R, International Subarachnoid Aneurysm Trial Collaborative G (2002) International Subarachnoid Aneurysm Trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised trial. Lancet 360(9342):1267–1274. https://doi.org/10.1016/s0140-6736(02)11314-6
Cognard C, Weill A, Castaings L, Rey A, Moret J (1998) Intracranial berry aneurysms: angiographic and clinical results after endovascular treatment. Radiology 206(2):499–510. https://doi.org/10.1148/radiology.206.2.9457205
Cognard C, Weill A, Spelle L, Piotin M, Castaings L, Rey A, Moret J (1999) Long-term angiographic follow-up of 169 intracranial berry aneurysms occluded with detachable coils. Radiology 212(2):348–356. https://doi.org/10.1148/radiology.212.2.r99jl47348
Wilms G, Bosmans H, Demaerel P, Marchal G (2001) Magnetic resonance angiography of the intracranial vessels. Eur J Radiol 38:10–18. https://doi.org/10.1016/S0720-048X(01)00285-6
Berteloot D, Leclerc X, Leys D, Krivosic R, Pruvo JP (1999) Cerebral angiography: a study of complications in 450 consecutive procedures. J Radiol 80(8):843–848
Kapsalaki EZ, Rountas CD, Fountas KN (2012) The role of 3 Tesla MRA in the detection of intracranial aneurysms. Int J Vasc Med 2012:792834–792839. https://doi.org/10.1155/2012/792834
Agid R, Schaaf M, Farb R (2012) CE-MRA for follow-up of aneurysms post stent-assisted coiling. Interv Neuroradiol 18(3):275–283. https://doi.org/10.1177/159101991201800305
Tartari S, Rizzati R, Righi R, Deledda A, Capello K, Soverini R, Benea G (2011) High-resolution MRI of carotid plaque with a neurovascular coil and contrast-enhanced MR angiography: one-stop shopping for the comprehensive assessment of carotid atherosclerosis. AJR Am J Roentgenol 196(5):1164–1171. https://doi.org/10.2214/AJR.10.4751
Trossbach M, Hartmann M, Braun C, Sartor K, Hahnel S (2004) Small vessel stents for intracranial angioplasty: in vitro evaluation of in-stent stenoses using CT angiography. Neuroradiology 46(6):459–463. https://doi.org/10.1007/s00234-004-1205-3
Gonner F, Heid O, Remonda L, Nicoli G, Baumgartner RW, Godoy N, Schroth G (1998) MR angiography with ultrashort echo time in cerebral aneurysms treated with Guglielmi detachable coils. AJNR Am J Neuroradiol 19(7):1324–1328
Lovblad KO, Yilmaz H, Chouiter A, San Millan Ruiz D, Abdo G, Bijlenga P, de Tribolet N, Ruefenacht DA (2006) Intracranial aneurysm stenting: follow-up with MR angiography. J Magn Reson Imaging 24(2):418–422. https://doi.org/10.1002/jmri.20642
Leclerc X, Navez JF, Gauvrit JY, Lejeune JP, Pruvo JP (2002) Aneurysms of the anterior communicating artery treated with Guglielmi detachable coils: follow-up with contrast-enhanced MR angiography. AJNR Am J Neuroradiol 23(7):1121–1127
Tsuruda J, Saloner D, Norman D (1992) Artifacts associated with MR neuroangiography. AJNR Am J Neuroradiol 13(5):1411–1422
Gauvrit JY, Leclerc X, Pernodet M, Lubicz B, Lejeune JP, Leys D, Pruvo JP (2005) Intracranial aneurysms treated with Guglielmi detachable coils: usefulness of 6-month imaging follow-up with contrast-enhanced MR angiography. AJNR Am J Neuroradiol 26(3):515–521
Agid R, Willinsky RA, Lee SK, Terbrugge KG, Farb RI (2008) Characterization of aneurysm remnants after endovascular treatment: contrast-enhanced MR angiography versus catheter digital subtraction angiography. AJNR Am J Neuroradiol 29(8):1570–1574. https://doi.org/10.3174/ajnr.A1124
Shankar JJ, Lum C, Parikh N, dos Santos M (2010) Long-term prospective follow-up of intracranial aneurysms treated with endovascular coiling using contrast-enhanced MR angiography. AJNR Am J Neuroradiol 31(7):1211–1215. https://doi.org/10.3174/ajnr.A2064
Buhk JH, Kallenberg K, Mohr A, Dechent P, Knauth M (2008) No advantage of time-of-flight magnetic resonance angiography at 3 Tesla compared to 1.5 Tesla in the follow-up after endovascular treatment of cerebral aneurysms. Neuroradiology 50(10):855–861. https://doi.org/10.1007/s00234-008-0413-7
Kaufmann TJ, Huston J 3rd, Cloft HJ, Mandrekar J, Gray L, Bernstein MA, Atkinson JL, Kallmes DF (2010) A prospective trial of 3 T and 1.5 T time-of-flight and contrast-enhanced MR angiography in the follow-up of coiled intracranial aneurysms. AJNR Am J Neuroradiol 31(5):912–918. https://doi.org/10.3174/ajnr.A1932
Urbach H, Dorenbeck U, von Falkenhausen M, Wilhelm K, Willinek W, Schaller C, Flacke S (2008) Three-dimensional time-of-flight MR angiography at 3 T compared to digital subtraction angiography in the follow-up of ruptured and coiled intracranial aneurysms: a prospective study. Neuroradiology 50(5):383–389. https://doi.org/10.1007/s00234-007-0355-5
Majoie CB, Sprengers ME, van Rooij WJ, Lavini C, Sluzewski M, van Rijn JC, den Heeten GJ (2005) MR angiography at 3 T versus digital subtraction angiography in the follow-up of intracranial aneurysms treated with detachable coils. AJNR Am J Neuroradiol 26(6):1349–1356
Brunereau L, Cottier JP, Sonier CB, Medioni B, Bertrand P, Rouleau P, Sirinelli D, Herbreteau D (1999) Prospective evaluation of time-of-flight MR angiography in the follow-up of intracranial saccular aneurysms treated with Guglielmi detachable coils. J Comput Assist Tomogr 23(2):216–223. https://doi.org/10.1097/00004728-199903000-00009
Boulin A, Pierot L (2001) Follow-up of intracranial aneurysms treated with detachable coils: comparison of gadolinium-enhanced 3D time-of-flight MR angiography and digital subtraction angiography. Radiology 219(1):108–113. https://doi.org/10.1148/radiology.219.1.r01mr06108
Kwee TC, Kwee RM (2007) MR angiography in the follow-up of intracranial aneurysms treated with Guglielmi detachable coils: systematic review and meta-analysis. Neuroradiology 49(9):703–713. https://doi.org/10.1007/s00234-007-0266-5
Panyaping T, Wongpanich G, Sangfai O, Tritanon O (2018) Comparison of contrast-enhanced 3D spoiled gradient-echo high-resolution T1-weighted sequences versus contrast-enhanced MR venography for detection of dural venous sinus thrombosis. Rama Med J 41:44–53
Metens T, Rio F, Baleriaux D, Roger T, David P, Rodesch G (2000) Intracranial aneurysms: detection with gadolinium-enhanced dynamic three-dimensional MR angiography-initial results. Radiology 216(1):39–46. https://doi.org/10.1148/radiology.216.1.r00jl1139
Roy D, Milot G, Raymond J (2001) Endovascular treatment of unruptured aneurysms. Stroke 32(9):1998–2004. https://doi.org/10.1161/hs0901.095600
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of Bursa Uludag University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Requirement for informed consent was waived by IRB of Bursa Uludag University.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mırçık, E., Hakyemez, B. New technique: the use of the THRIVE sequence in the follow-up of patients who received endovascular intracranial aneurysm treatment. Neuroradiology 63, 399–407 (2021). https://doi.org/10.1007/s00234-020-02527-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-020-02527-4