Abstract
Purpose
Although contrast-enhanced three-dimensional T2 fluid-attenuated inversion recovery (3D T2-FLAIR) images are useful for assessing various neuronal diseases, physiological enhancement of the circumventricular organs on the images have not been investigated. We aimed to assess the physiological appearance of the circumventricular organs on contrast-enhanced 3D T2-FLAIR images.
Methods
We studied 3-T MR images of the brain of 30 individuals with no apparent brain abnormalities. In ten areas of the brain, the degree of contrast enhancement on 3D T2-FLAIR and magnetization-prepared rapid gradient-echo (MPRAGE) images was evaluated using a 4-point grading system. The pre- and post-contrast mean contrast ratios (CRs) of the anterior pituitary gland, median eminence, and pineal gland were compared.
Results
On post-contrast 3D T2-FLAIR images, marked enhancement was most frequently scored in the median eminence, followed by the choroid plexus, posterior pituitary gland, and pineal gland. In 10 of the 30 cases, the vascular organ of the lamina terminalis and the area postrema were enhanced but the subcommissural organ was not. The difference in the mean pre- and post-contrast CRs of the median eminence and pineal gland was statistically significant, while that of the anterior pituitary gland was not.
Conclusion
On contrast-enhanced 3D T2-FLAIR images, the circumventricular organs show variable enhancement. Our findings help to recognize physiological and abnormal enhancement of brain structures on contrast-enhanced 3D T2-FLAIR images.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The circumventricular organs (CVOs) are small structures lining the cavity of the third ventricle (the neurohypophysis, vascular organ of the lamina terminalis, subfornical organ, pineal gland, and subcommissural organ) and of the fourth ventricle (area postrema) [1,2,3]. The choroid plexus has been included in the CVOs [2, 4]. In their capillary endothelial morphology and permeability, they differ from other parts of the brain. This and the presence of neural elements in contact with fenestrated capillaries allow the secretion of brain-derived products into the peripheral circulation (neurosecretion) and render CVOs the recipients of blood-born information that can then be transmitted to the brain [1, 3]. These structures are thought to be an important link in the regulation of metabolic and endocrine functions [1,2,3,4].
Clinically, the neurohypophysis and pineal gland of the CVOs are involved in pathologic conditions such as pituitary diabetes insipidus, and germ cell and pineal parenchymal tumors. Chordoid gliomas and papillary tumors of the pineal region are thought to originate in the vascular organ of the lamina terminalis and the subcommissural organ, respectively [5, 6]. One of the diagnostic criteria for disorders of the neuromyelitis optica spectrum has focused on the area postrema [7].
Contrast-enhanced two-dimensional (2D) or three-dimensional (3D) T2 fluid-attenuated inversion recovery (T2-FLAIR) images are useful for evaluating abnormal enhancement due to pathologic conditions, especially the breakdown of the blood-brain barrier (BBB), and the blood-meningeal and the blood-nerve barriers in patients with ischemic stroke, multiple sclerosis, leptomeningeal diseases, and facial neuritis [8,9,10,11,12]. Post-contrast pericortical enhancement has been observed on 3D T2-FLAIR images of older individuals with normal cognition, mild cognitive impairment, and dementia [13]. While contrast-enhanced 3D gradient-echo MR techniques [e.g., magnetization-prepared rapid gradient-echo (MPRAGE)] are widely used to evaluate brain lesions, 3D T2-FLAIR imaging is more sensitive to low gadolinium (Gd) concentrations, and it more effectively suppresses signals in the intracranial vessels [10] and cerebrospinal fluid (CSF) flow artifacts [14, 15].
Several studies have demonstrated normal enhancement of CVOs on contrast-enhanced 2D T1-weighted images [16, 17]. Normally, existing CVOs could represent a diagnostic pitfall and be mistaken with inflammatory or neoplastic enhancing lesions. Enhancement patterns of CVOs on 3D (volumetric) MR images such as 3D T2-FLAIR or 3D T1-MPRAGE have, to the best of our knowledge, not yet been studied. After having observed CVOs in routine contrast-enhanced 3D T2-FLAIR MRI, we sought to evaluated whether CVOs could effectively be demonstrated using this MRI sequence and sought to compare it to 3D T1-MPRAGE sequences.
Materials and methods
Subjects
Our institutional review board approved this retrospective study; informed patient consent was waived. Our study population included 30 patients (20 women, 10 men, age range 25–86 years, mean age 63.4 years). They were selected from among consecutive early-stage cancer patients referred for magnetic resonance (MR) screening for brain metastasis. Inclusion criteria were normal MR brain findings, availability of digital MR data including contrast-enhanced 3D T2-FLAIR and MPRAGE images, and absence of motion artifacts on the MR images, a history of neurologic disease, head trauma, chemotherapy, neurosurgery, and apparent neurologic symptoms during 12-month follow-up.
MR imaging protocol
All studies were performed using a 3-T MR imaging scanner (Magentom Trio a Tim; Siemens, Erlangen, Germany) and a 12-channel head coil. Before contrast injection, T1- (TR, 600 ms; TE, 8.5 ms; excitations, 1) and T2-weighted (TR, 3600 ms; TEeff, 96 ms; excitation, 2; echo train length, 7) imaging and 3D T2-FLAIR sequences with sampling perfection with application-optimized contrasts by using different flip angle evolutions (SPACE) were performed. T1- and T2-weighted images were acquired with a slice thickness of 5 mm. Immediately after the intravenous injection of gadopentetate dimeglumine (ProHance, 0.1 mmol/kg body weight; Bracco Diagnostics, Milan, Italy), T1-weighted, MPRAGE, and 3D T2-FLAIR images were obtained in this order.
The imaging parameters for 3D T2-FLAIR were TR 6000 ms, TEeff 420 ms, inversion time 2000 ms, echo train length 142, imaging time 4 min 26 s, field of view (FOV) 230 × 230 mm, matrix 256 × 256, and section thickness 0.9 mm. For MPRAGE, they were TR 1900 ms, TEeff 4.7 ms, inversion time 900 ms, imaging time 4 min 18 s, FOV 230 × 230 mm, matrix 256 × 256, and section thickness 0.9 mm. To reduce the imaging time, we used sagittal 3D planes covering the entire brain with each pulse sequence.
Image evaluation
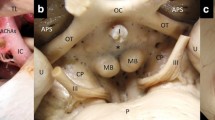
Referring to the anatomic location of the CVOs [1,2,3,4], two neuroradiologists (H.U., T.H.) independently evaluated contrast enhancement on 3D T2-FLAIR and MPRAGE images in ten intracranial structures, i.e., the anterior and posterior pituitary gland, the pituitary stalk, the median eminence, the vascular organ of the lamina terminalis (organum vasculosum laminae terminalis (OVLT)), the subfornical organ, the pineal gland, the subcommissural organ, the area postrema, and the choroid plexus (Fig. 1). Although not a CVO, the anterior pituitary gland was included in their evaluation because it is a well-enhanced structure very close to the CVOs on conventional MR images. All images were assessed on a picture archiving and communication system (PACS) workstation. Pre-contrast T1-weighted and 3D T2-FLAIR images were simultaneously reviewed as the reference. For image evaluation of the 3D MR images, sagittal source and axial and coronal multiplanar reconstruction images were available. The 3D T2-FLAIR images were always analyzed in conjunction with the corresponding MPRAGE images. The readers independently graded contrast enhancement; final judgments were based on their consensus.
Schema of the ten evaluated midline brain structures. The two radiologists reviewed the anterior (1) and posterior (2) pituitary gland, the pituitary stalk (3), the median eminence (4), the pineal gland (5), the vascular organ of the lamina terminalis (organum vasculosum laminae terminalis (OVLT)) (6), the subfornical organ (7), the subcommissural organ (8), the area postrema (9), and the choroid plexus (10). All but the anterior pituitary gland are circumventricular organs
The structures of interest were assessed with reference to their anatomical location [1,2,3,4]. To evaluate the subfornical organ, the inferior surface of the cerebral fornix at the origin of the right and left columns of the fornix, located at the meeting point of the lateral ventricles and the third ventricle, was inspected. For the subcommissural organ, the anterior and inferior surface of the posterior commissure, above the opening of the cerebral aqueduct, was assessed. For the area postrema, the dorsal medulla at the junction of the fourth ventricle and the central canal of the spinal cord was inspected. The choroid plexi in the third and fourth ventricles were identified and assessed. The readers compared enhancement of each brain structure on pre- and post-contrast 3D FLAIR images using a 4-point scale where grade 3 means marked, grade 2 moderate, grade 1 slight, and grade 0 no enhancement (Figs. 2 and 3). To evaluate the enhancement of each structure on post-contrast MPRAGE images, they used the same 4-point scale. As no pre-contrast MPRAGE images were available, pre-contrast T1-weighted images were the reference for post-contrast MPRAGE images.
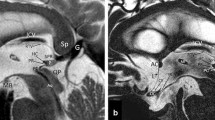
MR images of a 42-year-old woman. Compared to the pre-contrast 3D T2-FLAIR sagittal image (a), on the post-contrast sagittal image (b), the median eminence (large arrow) and the upper part of the posterior pituitary gland (large open arrowhead) are markedly enhanced (grade 3). The anterior pituitary gland is not enhanced (grade 0). Respectively, slight (grade 1) and moderate (grade 2) enhancement is observed in the pituitary stalk and pineal gland (medium arrow). The structure at the inferior surface of the cerebral fornix may be the subfornical organ (small open arrowhead); however, this is not clear because the structure is contiguous with the choroid plexus (small arrow). On the post-contrast 3D T2-FLAIR coronal image (c), the median eminence is also markedly enhanced (large arrow). The post-contrast MPRAGE image (d) shows marked (grade 3) enhancement of the median eminence, pituitary stalk, and anterior and posterior pituitary gland and moderate enhancement of the pineal gland. An area of the lamina terminalis (rightwards double arrow) is weakly enhanced on the post-contrast (b) compared with the pre-contrast 3D T2-FLAIR sagittal image (a); both observers scored enhancement as slight (grade 1). The subcommissural organ is not enhanced on either the post-contrast 3D T2-FLAIR or the MPRAGE image (b, d, gray arrow); both observers scored enhancement as absent (grade 0)
MR images of a 54-year-old man. Compared to the pre-contrast 3D T2-FLAIR image (a), on the post-contrast 3D T2-FLAIR image (b), the median eminence (large arrow) is markedly enhanced (grade 3) although the anterior pituitary gland is not enhanced (grade 0). The putative subfornical organ (open arrowhead) contiguous with the choroid plexus (small arrow) is visualized. On the post-contrast 3D T2-FLAIR image (b), an enhanced area is seen in the lamina terminalis (rightwards double arrow); both observers judged that the structure is the organum vasculosum laminae terminalis with moderate (grade 2) enhancement. These structures are more clearly seen on the post-contrast 3D T2-FLAIR than the post-contrast MPRAGE image (b, c, double arrow). Compared to the pre-contrast 3D T2-FLAIR image (d), on post-contrast 3D T2-FLAIR sagittal (e) and axial (f) images (large arrow), the area postrema is enhanced in the dorsal medulla near the obex. It is not clearly visualized on the post-contrast MPRAGE image (g, large arrow). Although the vessel structures are markedly enhanced on the post-contrast MPRAGE image (c, g), they are not enhanced on the post-contrast 3D T2-FLAIR image (b, e, f)
One neuroradiologist (T.H.) recorded the contrast ratio (CR) of the anterior pituitary gland (CRAPG), median eminence (CRME), and pineal gland (CRPG) on 3D T2-FLAIR images before and after contrast administration using the formulae:
where SIAPG, SIME, SIPG, and SICC are the signal intensity (SI) inside the region of interest (ROI) of the anterior pituitary gland, the median eminence, the pineal gland, and the splenium of the corpus callosum, respectively. A circular ROI was drawn in each structure. The ROI diameter was 2 mm for the anterior pituitary gland, 1 mm for the median eminence, 2 mm for the pineal gland, and 5 mm for the splenium of the corpus callosum. We did not address the other structures because their solid components were too small for assessment.
Statistical analysis
The level of interobserver agreement with respect to the degree of contrast enhancement was determined by calculating the κ coefficient (κ < 0.20 = poor, κ 0.21–0.40 = fair, κ 0.41–0.60 = moderate, κ 0.61–0.80 = good, κ 0.81–0.90 = very good, κ > 0.90 = excellent agreement). CRAPG, CRME, and CRPG on pre- and post-contrast 3D T2-FLAIR images were compared with the paired t test. A two-tailed p value of 0.05 or less was considered significant.
Results
Neither reader was able to identify the subfornical organ as an isolated structure on post-contrast MPRAGE- and 3D T2-FLAIR images. In all the 30 subjects, an enhanced structure at the inferior surface of the cerebral fornix was contiguous with the choroid plexus (Figs. 2 and 3).
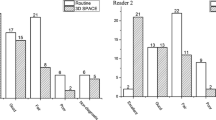
A summary of the qualitative analysis results of the nine other brain structures on post-contrast MPRAGE and 3D T2-FLAIR images is presented in Fig. 4a, b, respectively. Interobserver agreement for contrast enhancement on MPRAGE and 3D T2-FLAIR images was good to excellent (κ = 0.78–1.0) (Fig. 4). On MPRAGE images, enhancement of the anterior and posterior pituitary gland, the pituitary stalk, the median eminence, and the choroid plexus was marked (grade 3) in all the cases; the OVLT and the subcommissural organ were not enhanced on any of the images (grade 0). In 6 (20%) of the 30 cases, enhancement of the area postrema was moderate (grade 2) or slight (grade 1).
Distribution of the enhancement grade in nine brain structures on MPRAGE (a) and 3D T2-FLAIR (b) images. Numbers in parentheses indicate the κ coefficient for interobserver agreement with respect to the degree of contrast enhancement. OVLT organum vasculosum laminae terminalis, SCO subcommissural organ
Among the brain structures evaluated on 3D T2-FLAIR images, marked enhancement (grade 3) was observed most often in the median eminence, followed by the choroid plexus and the posterior pituitary and the pineal glands. In 19 cases (63%), there was slight to marked focal enhancement (grades 1–3) in the upper part of the posterior pituitary gland. Enhancement of the pituitary stalk was graded as absent (grade 0) in 12 (40%), as slight (grade 1) in 17 (57%), and as moderate (grade 2) in 1 (3%) case. The degree of enhancement of the pineal gland on 3D T2-FLAIR and MPRAGE images varied. On 3D T2-FLAIR images, the OVLT and the area postrema were enhanced in 10 of the 30 cases (33%); enhancement of the anterior pituitary gland was scored as absent (grade 0) in 25 of 30 (83%) and as slight (grade 1) in the other 5 cases (Figs. 2, 3, and 4). The subcommissural organ was not enhanced on post-contrast 3D T2-FLAIR images.
Quantitative assessment of pre- and post-contrast values on 3D T2-FLAIR images showed that they were 1.38 ± 0.17 and 1.31 ± 0.19 for CRAPG, 1.15 ± 0.08 and 1.91 ± 0.33 for CRME, and 1.20 ± 0.21 and 1.69 ± 0.27 for CRPG. The difference was statistically significant (p < 0.0001) only for CRME and CRPG.
Discussion
Our study showed various enhancement of the CVOs on post-contrast 3D T2-FLAIR images. On the 3D T2-FLAIR images, enhancement was most frequently observed in the median eminence, followed by the choroid plexus and the posterior pituitary and the pineal glands. The subcommissural organ never showed any contrast enhancement. These results were similar to those of the previous study using sagittal 2D T1-weighted, 2.5-mm-thick images [17]. Human CVOs are generally formed by a rich capillary network made up of fenestrated endothelial cells and are devoid of a BBB [1,2,3,4]. Each CVO has characteristic anatomy and vessel permeability. Neurohormones from tuberohypophyseal fibers reaching the median eminence from the hypothalamus are transported to the adenohypophysis through the hypophyseal portal system [2]. Electron-microscopic studies of the median eminence revealed many capillary fenestrations, cytoplasmic vesicles or pits, and wide pericapillary labyrinths between the parenchymal and capillary basal laminae, resulting in high permeability [18,19,20,21,22]. In humans, the capillary network of the pineal gland can be divided into several areas [2]. In its central and dorsal parts, pinealocytes are assembled in lobules surrounded by a network of large capillaries; capillary loops identical to those seen in other CVOs originate there and penetrate to the center of the lobule. The inferior part of the gland is vascularized by small-caliber capillaries and pinealocytes are arranged diffusely. Consequently, in the pineal gland, some vessels may lack the BBB while others may be less permeable [2]. On the other hand, unlike the other CVOs, the vessels of the subcommissural organ have no fenestrated walls in humans, and the BBB is functional [2]. Thus, the permeability difference of the CVOs must have affected their appearance on post-contrast 3D T2-FLAIR images.
On 3D T2-FLAIR images, the posterior pituitary gland often manifested focal enhancement limited to its upper part. The capillary network of the posterior lobe receives neurohypothalamic hormones, vasopressin and ocytocin, via a nerve tract originating in the paraventricular and supraoptic nuclei; it courses through the median eminence to reach the blood vessels of the lobe [2]. Thus, the localized enhancement of the posterior lobe on 3D T2-FLAIR images may reflect the permeability of the secretory nerve endings.
In one third of our cases, the vascular organ of the lamina terminalis and the area postrema showed relatively low enhancement on 3D T2-FLAIR images. Some neurons in the former organ are osmoreceptors, being sensitive to the osmotic pressure of the blood, and project to the supraoptic nucleus and paraventricular nucleus to regulate the activity of vasopressin-secreting neurons [2]. The latter organ detects toxins in the blood and acts as a vomit-inducing center [2]. The vascular organ of the lamina terminalis is rudimentary and the area postrema atrophies with age [23]. A previous study using sagittal 2D T1-weighted, 2.5-mm-thick images showed enhancement of the vascular organ of the lamina terminalis in 34% of cases [17]. This result is equivalent with that of our 3D FLAIR, 0.9-mm-thick images, although we did not observe enhancement of this structure on MPRAGE, 0.9-mm-thick images. Others [16, 17] reported enhancement of the area postrema on sagittal 2D T1-weighted, 2.5- or 3-mm-thick images; its frequency was reported to be 2% of cases [17]. In our series, such enhancement was observed in 33% of cases on 3D T2-FLAIR and in 20% on MPRAGE images.
Although the vascular structure of the subfornical organ is similar to that of other CVOs [2], it was not clearly identified as an isolated structure on our contrast-enhanced 3D T2-FLAIR- and MPRAGE images. In humans, it is identified as a 1-mm gray nodule on the ventral surface of the fornix at the foramina of Monro [24]. As capillaries of the subfornical organ are contiguous with those of the choroid plexus [25], enhancement from the choroid plexus to the subfornical organ renders identification of this small structure difficult.
The enhanced appearance of the CVOs between the 3D T2-FLAIR and the MPRAGE images was quite different. We hypothesize that this is due to the difference of contrast enhancement mechanisms in the tissue between the two MR sequences: both vascular and interstitial (extravascular) space enhancement on post-contrast MPRAGE images; only interstitial (extravascular) space enhancement on post-contrast 3D T2-FLAIR images. Compared with MPRAGE sequence, 3D T2-FLAIR sequence more effectively suppresses signals in the vessels and is more sensitive to low Gd concentrations [10]. The characteristics of 3D T2-FLAIR imaging contribute to its superiority in the depiction of low Gd concentrations leaked from the BBB into the interstitial (extravascular) space.
Our qualitative and quantitative evaluations showed that the anterior pituitary gland was not or only slightly enhanced on 3D T2-FLAIR images. We hypothesize that the lack of intense enhancement of the anterior lobe of the pituitary on 3D T2-FLAIR images is attributable to T2 shortening due to a high Gd concentration. The estimated vascular concentration following the administration of the conventional dose of Gd is approximately 0.4–0.6 mmol/l [26]. Fukuoka et al. [10] suggested that T2 shortening occur on 3D T2-FLAIR images at Gd concentration of more than 0.5 mmol/l; this may explain the signal decrease we observed in the anterior pituitary gland. Alternatively, this phenomenon may be explicable by the flow in the anterior pituitary gland. The anterior pituitary gland features a hypophyseal portal system comprised of sinusoidal capillaries with 30–40-μm-diameter openings in the endothelium to allow the passage of 7.5–25-μm-diameter red and white blood cells and of various serum proteins. These large openings may induce flow in the leaked sinusoid spaces. Since 3D T2-FLAIR sequences are highly flow-sensitive [10], flow in the sinusoid can be expected to reduce the signal in these spaces. Further studies are needed to clarify the unexpected phenomenon for the anterior pituitary gland on post-contrast 3D T2-FLAIR images.
As the CVOs are small structures lining the cavity of the third and fourth ventricles, they may be misinterpreted as lesions when evaluating intracranial tumor dissemination or granulomatous diseases. One of the diagnostic criteria for disorders of the neuromyelitis optica spectrum has focused on the area postrema [7]. When assessing them on post-contrast 3D T2-FLAIR images, the physiological enhancement of the area postrema might be mistaken as abnormal enhancement associated with the disorders. The recognition of physiologic enhancement of the CVOs on post-contrast 3D T2-FLAIR images will lead to accurate diagnosis.
Our study has some limitations. First, we assessed the MR images of patients with confirmed or suspected early-stage cancer. However, as none had neurological symptoms, abnormal brain MR imaging findings, and apparent neurologic symptoms in the course of 1-year follow-up, we think that our data reflect the physiological appearance of the organs on contrast-enhanced 3D T2-FLAIR images. Second, we did not randomize the order of the two 3D MR sequences. After contrast-material injection, we obtained T1-weighted, MPRAGE, and 3D T2-FLAIR images in this order, consequently, we cannot rule out post-injection timing bias. Third, we did not include pre-contrast MPRAGE images because the acquisition time for 3D MR images is relatively long. Therefore, to assess contrast enhancement, we used pre-contrast T1-weighted images as the reference for post-contrast MPRAGE images. Fourth, we did not evaluate the contrast-enhanced findings of 3D FLAIR sequences in other venders. The widely used 3D FLAIR sequences are the variable flip angle refocusing pulse, fast spin-echo sequences with brand names such as SPACE (sampling perfection with application-optimized contrasts by using different flip angle evolutions; Siemens, Erlangen, Germany), VISTA (volume isotropic turbo spin-echo acquisition; Philips Healthcare, Best, the Netherlands), and Cube (GE Healthcare, Milwaukee, WI). Since these 3D FLAIR sequences have similar imaging characteristics [27], we think that our results can be applied to the sequences in other venders.
Conclusion
Post-contrast 3D T2-FLAIR images showed various enhancements in the CVOs. Among the CVOs, marked enhancement on post-contrast 3D T2-FLAIR images was most frequently seen in the median eminence, followed by the choroid plexus, posterior pituitary gland, and pineal gland. In one-third cases, the vascular organ of the lamina terminalis and the area postrema was enhanced. Post-contrast 3D T2-FLAIR and 3D T1-MPRAGE displayed a different sensitivity in detecting enhancing CVOs. Normally, enhancing structures such as the CVOs may be routinely observed on MRI studies and should not be mistaken for enhancing inflammatory or neoplastic lesions.
References
Johnson AK, Gross PM (1993) Sensory circumventricular organs and brain homeostatic pathways. FASEB J 7:678–686
Duvernoy HM, Risold PY (2007) The circumventricular organs: an atlas of comparative anatomy and vascularization. Brain Res Rev 56:119–147
Sisó S, Jeffrey M, González L (2010) Sensory circumventricular organs in health and disease. Acta Neuropathol 120:689–705
Ganong WF (2000) Circumventricular organs: definition and role in the regulation of endocrine and autonomic function. Clin Exp Pharmacol Physiol 27:422–427
Bielle F, Villa C, Giry M, Bergemer-Fouquet AM, Polivka M, Vasiljevic A, Aubriot-Lorton MH, Bernier M, Lechapt-Zalcman E, Viennet G, Sazdovitch V, Duyckaerts C, Sanson M, Figarella-Branger D, Mokhtari K, RENOP (2015) Chordoid gliomas of the third ventricle share TTF-1 expression with organum vasculosum of the lamina terminalis. Am J Surg Pathol 39:948–956
Hasselblatt M, Blümcke I, Jeibmann A et al (2006) Immunohistochemical profile and chromosomal imbalances in papillary tumours of the pineal region. Neuropathol Appl Neurobiol 32:278–283
Wingerchuk DM, Banwell B, Bennett JL, Cabre P, Carroll W, Chitnis T, de Seze J, Fujihara K, Greenberg B, Jacob A, Jarius S, Lana-Peixoto M, Levy M, Simon JH, Tenembaum S, Traboulsee AL, Waters P, Wellik KE, Weinshenker BG, International Panel for NMO Diagnosis (2015) International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 85:177–189
Latour LL, Kang DW, Ezzeddine MA, Chalela JA, Warach S (2004) Early blood-brain barrier disruption in human focal brain ischemia. Ann Neurol 56:468–477
Absinta M, Vuolo L, Rao A, Nair G, Sati P, Cortese ICM, Ohayon J, Fenton K, Reyes-Mantilla MI, Maric D, Calabresi PA, Butman JA, Pardo CA, Reich DS (2015) Gadolinium-based MRI characterization of leptomeningeal inflammation in multiple sclerosis. Neurology 85:18–28
Fukuoka H, Hirai T, Okuda T et al (2009) Comparison of the added value of contrast-enhanced 3D fluid-attenuated inversion recovery and magnetization-prepared rapid acquisition of gradient echo sequences in relation to conventional postcontrast T1-weighted images for the evaluation of leptomeningeal diseases at 3T. AJNR Am J Neuroradiol 31:868–873
Hirai T, Ando Y, Yamura M, Kitajima M, Hayashida Y, Korogi Y, Yamashita T, Yamashita Y (2005) Transthyretin-related familial amyloid polyneuropathy: evaluation of CSF enhancement on serial T1-weighted and fluid-attenuated inversion recovery images following intravenous contrast administration. AJNR Am J Neuroradiol 26:2043–2048
Lim HK, Lee JH, Hyun D, Park JW, Kim JL, Lee H, Park S, Ahn JH, Baek JH, Choi CG (2012) MR diagnosis of facial neuritis: diagnostic performance of contrast-enhanced 3D-FLAIR technique compared with contrast-enhanced 3D-T1-fast-field echo with fat suppression. AJNR Am J Neuroradiol 33:779–783
Freeze WM, Schnerr RS, Palm WM, Jansen JF, Jacobs HI, Hoff EI, Verhey FR, Backes WH (2017) Pericortical enhancement on delayed postgadolinium fluid-attenuated inversion recovery images in normal aging, mild cognitive impairment, and Alzheimer disease. AJNR Am J Neuroradiol 38(9):1742–1747
Naganawa S, Koshikawa T, Nakamura T, Kawai H, Fukatsu H, Ishigaki T, Komada T, Maruyama K, Takizawa O (2004) Comparison of flow artifacts between 2D-FLAIR and 3D-FLAIR sequences at 3 T. Eur Radiol 14:1901–1908
Chagla GH, Busse RF, Sydnor R, Rowley HA, Turski PA (2008) Three-dimensional fluid attenuated inversion recovery imaging with isotropic resolution and nonselective adiabatic inversion provides improved three-dimensional visualization and cerebrospinal fluid suppression compared to two-dimensional FLAIR at 3 Tesla. Investig Radiol 43:547–551
Williams KD, Dean B, Drayer BP (1990) Demonstration of the area postrema with contrast-enhanced MR. AJNR Am J Neuroradiol 11:733–734
Horsburgh A, Massoud TF (2013) The circumventricular organs of the brain: conspicuity on clinical 3T MRI and a review of functional anatomy. Surg Radiol Anat 35:343–349
Cazalis M, Dayanithi G (1987) Requirements for hormone release from permeabilized nerve endings isolated from the rat neurohypophysis. J Physiol 390:71–91
Gross PM, Sposito NM, Pettersen SE, Fenstermacher JD (1986) Differences in function and structure of the capillary endothelium in the supraoptic nucleus and pituitary neural lobe of rats. Neuroendocrinology 44:401–407
Gross PM, Blasberg RG, Fenstermacher JD, Patlak CS (1987) The microcirculation of rat circumventricular organs and pituitary gland. Brain Res Bull 18:73–85
Gross PM, Wall KM, Pang JJ, Shaver SW, Wainman DS (1990) Microvascular specializations promoting rapid interstitial solute dispersion in nucleus tractus solitarius. Am J Phys 259:l131–l1138
Shaver SW, Pang JJ, Wainman DS, Wall KM, Gross PM (1992) Morphology and function of capillary networks in subregions of the rat tuber cinereum. Cell Tissue Res 267:437–448
Schünke M, Schulte E, Schumacher U, Voll M, Wesker K (2006) PROMETHEUS LernAtlas der Anatomie. Georg Thieme Verlag, Stuttgart, Germany
Mark MH, Farmer PM (1984) The human subfornical organ: an anatomic and ultrastructural study. Ann Clin Lab Sci 14:427–442
Dellmann HD, Simpson JB (1976) Regional differences in the morphology of the rat subfornical organ. Brain Res 116:389–400
Strouse PJ, Prince MR, Chenevert TL (1996) Effect of the rate of gadopentetate dimeglumine administration on abdominal vascular and soft-tissue MR imaging enhancement patterns. Radiology 201:809–816
Naganawa S (2015) The technical and clinical features of 3D-FLAIR in neuroimaging. Magn Reson Med Sci 14:93–106
Acknowledgments
We are grateful to Ms. Ursula Petralia for assistance with English editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Azuma, M., Hirai, T., Kadota, Y. et al. Circumventricular organs of human brain visualized on post-contrast 3D fluid-attenuated inversion recovery imaging. Neuroradiology 60, 583–590 (2018). https://doi.org/10.1007/s00234-018-2023-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-018-2023-3