Abstract
Purpose
The primary aim was to examine and compare the increased risk of incident diabetes associated with second-generation antipsychotics (SGAs) and first-generation antipsychotics (FGAs), with and without adjusting for potential confounding factors. The secondary aim was to recalculate the relative risks of diabetes onset using a semi-symmetric bidirectional case-crossover (SSBC) design to adjust for time-trend bias.
Method
Prescription records (2005–2015) of antipsychotics were sourced from New Zealand Pharmaceutical Collections. The first-time diabetes diagnosis was extracted from the National Minimal Dataset. Relative risks (RRs) of diabetes onset were calculated using conditional logistic regression. Time-trend bias was corrected by recalculating the RR using a SSBC design.
Results
Among 645 individuals, the risk of diabetes onset is higher in SGA users (ARR = 8.72, 95% CI = [5.57, 13.67]) compared to FGA users (ARR = 5.68, 95% CI = [3.43, 9.39]). The increased risk of diabetes onset associated with quetiapine is higher (ARR = 7.47, 95% CI = [4.10, 13.62]), compared to haloperidol (ARR = 5.05, 95% CI = [2.91, 8.75]). However, the increased risk of diabetes onset associated with olanzapine (ARR = 2.27, 95% CI = [0.86, 5.98]) is insignificant after adjusting for concomitant use of effect modifiers and other antipsychotic drugs.
Conclusion
The results support that the magnitude of the risk of diabetes is higher with SGA use compared with FGA use, and the risk is higher when co-prescribed. Confounding by indication and time-varying confounders such as body mass index could bias the risk of onset of diabetes. Marginal structural models could provide more precise estimates of the risk of onset of diabetes following exposure to antipsychotics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antipsychotics are mainly indicated for the treatment of schizophrenia, bipolar disorder, psychotic depression and behavioural and psychological symptoms of dementia. There are two main classes of antipsychotics: the first-generation antipsychotics (FGAs) and the second-generation antipsychotics (SGAs) [1–3]. Clinical trials have demonstrated the link between antipsychotic exposure and incidence of diabetes, risk of cardiovascular events and increased mortality [4–9]. However, these trials were often based on populations of younger individuals aged between 18 and 65 years; people 65 and over with chronic medical conditions were often excluded.
In a real-world setting, individuals prescribed antipsychotics may be older, have higher comorbidity and are frailer and hence have a higher baseline risk of experiencing adverse drug effects than patients recruited in a clinical trial. An uncertainty exists about making causal inferences about the true effect of the risk of diabetes posed by antipsychotic drugs in older people due to uncontrolled confounding [10] and limited statistical power [11].
National-level studies examining the risk of diabetes mellitus with antipsychotic drug exposures are limited in New Zealand. The primary aim of this study was to perform a nationwide case-crossover analysis to examine the association of diabetes with antipsychotic exposure in a large population-based sample of older (age 65 and above) New Zealanders. Using a case-crossover design, we examined the risk of incidence diabetes in older individuals.
Methods
Approval to undertake this study was provided by the Human Ethics Research Committee of the University of Otago, New Zealand (approval number HD 16/022).
Data source
The New Zealand Ministry of Health maintains national collections of prescription use and hospital discharges. Individual records in these national collections include a unique seven-digit alphanumeric identifier, known as the National Health Index (NHI) identifier. The NHIs are encrypted in all datasets, but there is only one encrypted version of each NHI and is never changed. Therefore, we were able to link new data with datasets previously extracted. The pharmaceutical information database (Pharms) includes record of all prescription claims made by community pharmacists funded by PHARMAC, including the medicine name and prescription date. The National Minimum Dataset (NMDS) is a collection of hospital discharges for inpatients and day patients. Patients’ diagnoses are coded according to the International Classification of Diseases and Related Health Problems Tenth Revision, Australian Modification (ICD-10-AM).
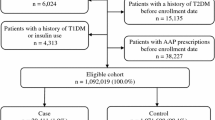
This study considered only drugs that are funded by PHARMAC. Only prescriptions of antipsychotics, effect modifiers of interests (i.e. diabetogenic drugs) and antidiabetic drugs are considered; the rest were excluded as they were irrelevant to our study. We excluded over-the-counter medicines and drugs not subsidised by PHARMAC. The process of case selection is shown in Fig. S1.
Study population
We identified all individuals, aged 65 years and above, who had been diagnosed for the first time with diabetes, between 01 January 2005 and 31 December 2015. Individuals were considered to have new-onset diabetes if they had an ICD-10-AM code for diabetes mellitus (Table S1) and without a prescription for an antidiabetic medication (Table S2) within 2 years before the first-time diagnosis of diabetes. The day the individual was diagnosed with diabetes for the first time after 01 January 2005 is the index date.
Statistical analysis
Analyses was conducted using R version 3.2.4 Revised (2016-03-16 r7033). We used a case-crossover design [12–14], to study the population-level association between antipsychotic drug exposure (Table S2) and incident diabetes. Two 365-day observation periods were defined over a 730-day study period. The first observation period is the time interval 1–365 days before the index date, which is the case period. The control period is the time interval 366–730 days before the index date. For each individual, we summarised the history of antipsychotic exposure as SGA-only, FGA-only, using both and using neither during the observation periods. In a case-crossover design, time-invariant or slow-changing confounding variables are automatically balanced even if they are unknown, so we did not spell them out. Fast-changing confounding variables will need to be adjusted. Concomitant use of diabetogenic drugs (i.e. the effect modifiers) is an important time-varying confounding variable that can vary quickly over time, so we adjusted for the use of these drugs.
From the case-crossover design, for each individual, we analysed antipsychotic exposure and the combination of antipsychotic exposures (i.e. SGA-only, FGA-only or both drugs), over an observation period, and whether or not diabetes onset occurred at the end of the same observation period. Using conditional logistic regression (CLR), we were able to estimate the risk of incident diabetes associated with antipsychotic exposure/s and expressed the effect size as relative risk (RR). CLR accounts for the between-individual variation of health characteristics when estimating the changed risk with drug exposure and thus mitigates confounding. We used multivariable CLR to adjust for concomitant use of effect-modifying drugs that are known to increase the risk of diabetes (Table S2). The relative risk calculations were repeated to calculate the changed risk of incident diabetes associated with the use of each antipsychotic drug, with or without adjusting for concomitant use of other antipsychotic drugs and effect-modifying drugs.
As a secondary aim, we recalculated the RRs of diabetes onset using a semi-symmetric bidirectional case-crossover (SSBC) design [23] with 365-day observation periods based on the same cohort, to adjust for time-trend bias. The SSBC design is based on the work published by Navidi and Weinhandl [23]. Briefly, a case period, a pre-event control period and a post-event control period were defined for each individual. In this study, the case period is the time interval 1–365 days before the index date as for the conventional case-crossover design. The time interval 366–730 days before the index date is the pre-event control period, and the time interval 1–365 days after the index date is the post-event control period. For each individual, we summarised the use of antipsychotics during each observation period. When evaluating the relative risk, we randomly selected one of the two control periods as the reference period, with equal (50%) probability but subjected to the constraint that the reference period must be between 01 January 2005 and 31 December 2015.
Results
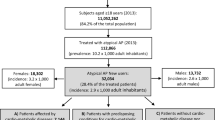
We found a total of 52,135 individuals with a first-time diagnosis of diabetes between 01 January 2005 and 31 December 2015. Among these individuals, 1842 of them had prescription records covering the time interval 2 years before the date of the first-time diagnosis of diabetes and, from these prescription records, have had at least one prescription of antipsychotics (SGA or FGA). Then, 1197 of them were excluded because they had received antidiabetic medicines at least once within 2 years before the first-time diabetes diagnosis. The 645 individuals remaining had a reliable date of first-time diabetes diagnosis and form the study cohort.
The characteristics of the study sample are shown in Fig. 1. Our study sample has 113 more females than males, and a vast majority of them are New Zealand-European, and only a small proportion of them are Māori. Descriptive statistics (Figs. 1 and S2 and Table 1) support that antipsychotics are more likely to be used within the case period (i.e. 1–365 days the first-time diagnosis of diabetes), compared to the control period. Rate ratio analysis (Fig. 2) with or without adjusting for concomitant use of effect-modifying drugs indicates that higher risk of incident diabetes is associated with antipsychotic exposure. The risk of diabetes onset is higher with SGA use (ARR 8.72, 95% CI [5.57, 13.67]) compared to FGA use (ARR 5.68, 95% CI [3.43, 9.39]), and the risk increases further if both SGA and FGA are co-prescribed. Adjusting for concomitant uses of effect-modifying drugs lowered the rate ratios but had no effect on the outcome. The increased risk of diabetes onset associated with quetiapine is higher (Table 1, ARR 7.47, 95% CI [4.10, 13.62]), compared to haloperidol (ARR 5.05, 95% CI [2.91, 8.75]). However, the increased risk of diabetes onset associated with olanzapine (ARR 2.27, 95% CI [0.86, 5.98]) is insignificant after adjusting for concomitant use of effect modifiers and other antipsychotic drugs.
As a secondary aim, we adjusted for time-trend bias by recalculating the relative risks using SSBC design based on the protocol published by Navidi and Weinhandl [23]. The results support the general trend observed in the primary aim that higher risk of diabetes onset is associated with antipsychotic exposure (Fig. 2, Table 2), and risk of diabetes onset is higher in SGA users compared to FGA users with and without co-adjusting for concomitant uses of effect modifiers and other antipsychotic drugs. The increased risk of diabetes onset associated with olanzapine is still insignificant after adjusting for concomitant use of effect modifiers and other antipsychotic drugs, as well as time-trend bias (Table 2).
However, after adjusting for time-trend bias by using SSBC design, the relative risk of diabetes onset associated with concomitant use of SGA and FGA is lower compared to using only SGA and is only marginally higher compared to using only FGA (Fig. 2). This counterintuitive finding is related to the fact that SGA users commonly started to use both SGA and FGA within the year after the diagnosis if not adhering to it or discontinuing (Fig. S2). In SSBC design, reference period antipsychotic use include use of antipsychotics 1 year after the diagnosis; any post-event antipsychotic use will therefore reduce the relative risk.
Discussion
The present study examined the risk of diabetes in a large population-based cohort of older people exposed to antipsychotics. This study supports the risk of incident diabetes posed by exposure to antipsychotic drugs. The findings show an increased risk of incident diabetes ranging from 3.4 to 9.4 for FGAs and from 5.6 to 13.7 for SGAs after adjusting for concomitant use of effect modifiers (i.e. drugs that increase the risk of incident diabetes). After adjusting for time-trend bias, the increased risk of incident diabetes ranges from 7.6 to 22.6 for FGAs and from 4.3 to 12.7 for SGAs after adjusting for effect modifiers. Previous studies have highlighted and compared the risk of diabetes posed by FGAs and SGAs [15–17]. The results confirm that the magnitude of the risk of diabetes is higher with SGAs compared with FGAs and higher when co-prescribed.
Our study also found that if antipsychotics are not discontinued after the first-time diagnosis of diabetes, the most common drug switching is to change from using SGA only to using both SGA and FGA. This may explain why the relative risk of diabetes onset does not increase when SGA and FGA are co-prescribed, when a SSBC design is used to adjust for time-trend bias and post-event prescriptions are accounted for. A possible explanation is that individuals may switch from SGA to FGA in the year after the diagnosis because FGAs are perceived as being less diabetogenic than SGAs.
Pharmacological mechanisms [18] underpinning the association of antipsychotics with diabetes have highlighted that obesity and metabolic syndrome are the major drivers. Olanzapine and clozapine with the greatest effect on weight gain are known to be highly diabetogenic compared to risperidone that has relatively a less impact on inducing weight gain [19, 20]. Interestingly, our study favours the trend that risperidone is more diabetogenic compared to olanzapine (Tables 1 and 2).
Pharmacoepidemiological studies have highlighted that the use of both FGAs and SGAs is associated with onset of diabetes [21, 22]. Challenges in these studies include the use of cross-sectional designs, variation in the definition of incident diabetes and lack of adherence data. A case-crossover design mitigates confounding from unknown time-invariant confounding variables. The case-crossover design is similar to a matched case-control design. A matched case-control design is typically used to control confounding and is accomplished by matching an individual in the case group with a similar individual in the control group based on the values of potential confounder variables. Case-control matching requires accurate identification of potential confounders, but in reality, potential confounders are not always known or measured. The advantage of using case-crossover design is that comparisons are made using the same individual, the comparisons are matched without dealing with time-invariant confounders, and hence, the issue of unidentified or unmeasured biological and psychosocial confounders is avoided.
Our study found preferential prescribing of quetiapine and risperidone over olanzapine. This preferential prescribing may have been influenced by clinical guidelines and previous studies that have implicated higher risk of diabetes associated with olanzapine compared to risperidone. We adjusted for concomitant medications in the CLR model, i.e. effect modifiers likely to induce diabetes. The effect sizes were smaller, but this did not impact the overall findings of increased risk of diabetes posed by both FGAs and SGAs.
However, it is unclear from observational and experimental studies about the exact temporal relationship (i.e. the time lag) between exposure to antipsychotic and the risk of developing diabetes. A prospective cohort study would perhaps shed light on time-dependent risk associated with antipsychotic drug exposure. However, our findings are in agreement with previous pharmacoepidemiological approaches showing an increased risk of diabetes associated with antipsychotic exposures, and the magnitude of effect size reported higher with SGAs is also consistent as reported in these studies.
Case-crossover designs are typically used to evaluate transient exposures with immediate and transient effects to ensure that the effects of any time-varying confounding variables are negligible [22], and therefore, we were concerned that the effects of changes to individual frailty scores and body mass index (BMI) scores may have biased the risk of diabetes onset. Hence, a potential limitation of our study is that there may be confounding that is residual as we did not control for these important time-varying confounders. Also, lack of data on lifestyle factors is another major limitation of this study.
We plan to address the issue of frailty and BMI changes by applying published statistical methodologies such as marginal structural modelling or covariate adjustment, to a complete set of health care data that allows time-varying confounders to be quantified.
Conclusion
We found that antipsychotic drug exposure increases the risk of diabetes onset in older New Zealanders. The risk of new-onset diabetes was shown to be lowest in individuals using FGAs. The risk of diabetes was found to be higher for quetiapine compared to haloperidol. We plan to use marginal structural models to provide unbiased estimates of the risk of antipsychotic exposures on onset of diabetes.
References
Stahl SM (1999) Selecting an atypical antipsychotic by combining clinical experience with guidelines from clinical trials. The Journal of clinical psychiatry 60(Suppl 10):31–41
Jakovljevic M (2009) New generation vs. first generation antipsychotics debate: pragmatic clinical trials and practice-based evidence. Psychiatr Danub 21(4):446–452
Lambert M, Schimmelmann BG, Schacht A, Suarez D, Haro JM, Novick D, Wagner T, Wehmeier PM, Huber CG, Hundemer HP, Dittmann RW, Naber D (2011) Differential 3-year effects of first- versus second-generation antipsychotics on subjective well-being in schizophrenia using marginal structural models. J Clin Psychopharmacol 31(2):226–230. doi:10.1097/JCP.0b013e3182114d21
Buse JB, Cavazzoni P, Hornbuckle K, Hutchins D, Breier A, Jovanovic L (2003) A retrospective cohort study of diabetes mellitus and antipsychotic treatment in the United States. J Clin Epidemiol 56(2):164–170
Caro JJ, Ward A, Levinton C, Robinson K (2002) The risk of diabetes during olanzapine use compared with risperidone use: a retrospective database analysis. The Journal of clinical psychiatry 63(12):1135–1139
Feldman PD, Hay LK, Deberdt W, Kennedy JS, Hutchins DS, Hay DP, Hardy TA, Hoffmann VP, Hornbuckle K, Breier A (2004) Retrospective cohort study of diabetes mellitus and antipsychotic treatment in a geriatric population in the United States. J Am Med Dir Assoc 5(1):38–46
Fuller MA, Shermock KM, Secic M, Grogg AL (2003) Comparative study of the development of diabetes mellitus in patients taking risperidone and olanzapine. Pharmacotherapy 23(8):1037–1043
Ulcickas Yood M, Delorenze GN, Quesenberry CP Jr, Oliveria SA, Tsai AL, Kim E, Cziraky MJ, McQuade RD, Newcomer JW, L'Italien GJ (2011) Association between second-generation antipsychotics and newly diagnosed treated diabetes mellitus: does the effect differ by dose? BMC psychiatry 11:197. doi:10.1186/1471-244x-11-197
Yood MU, DeLorenze G, Quesenberry CP Jr, Oliveria SA, Tsai AL, Willey VJ, McQuade R, Newcomer J, L’Italien G (2009) The incidence of diabetes in atypical antipsychotic users differs according to agent—results from a multisite epidemiologic study. Pharmacoepidemiol Drug Saf 18(9):791–799. doi:10.1002/pds.1781
Montgomery JH, Byerly M, Carmody T, Li B, Miller DR, Varghese F, Holland R (2004) An analysis of the effect of funding source in randomized clinical trials of second generation antipsychotics for the treatment of schizophrenia. Control Clin Trials 25(6):598–612. doi:10.1016/j.cct.2004.09.002
Peluso MJ, Lewis SW, Barnes TR, Jones PB (2012) Extrapyramidal motor side-effects of first- and second-generation antipsychotic drugs. The British journal of psychiatry : the journal of mental science 200(5):387–392. doi:10.1192/bjp.bp.111.101485
Delaney JA, Suissa S (2009) The case-crossover study design in pharmacoepidemiology. Stat Methods Med Res 18(1):53–65. doi:10.1177/0962280208092346
Maclure M (1991) The case-crossover design: a method for studying transient effects on the risk of acute events. Am J Epidemiol 133(2):144–153
Chang CH, Chen HC, Lin JW, Kuo CW, Shau WY, Lai MS (2011) Risk of hospitalization for upper gastrointestinal adverse events associated with nonsteroidal anti-inflammatory drugs: a nationwide case-crossover study in Taiwan. Pharmacoepidemiol Drug Saf 20(7):763–771. doi:10.1002/pds.2140
Schulte PF, Bocxe JT, Doodeman HJ, van Haelst IM, Cohen D (2016) Risk of new-onset diabetes after long-term treatment with clozapine in comparison to other antipsychotics in patients with schizophrenia. J Clin Psychopharmacol 36(2):115–119. doi:10.1097/jcp.0000000000000465
Rubin DM, Kreider AR, Matone M, Huang YS, Feudtner C, Ross ME, Localio AR (2015) Risk for incident diabetes mellitus following initiation of second-generation antipsychotics among Medicaid-enrolled youths. JAMA Pediatr 169(4):e150285. doi:10.1001/jamapediatrics.2015.0285
Tandon R (2002) Safety and tolerability: how do newer generation “atypical” antipsychotics compare? The Psychiatric quarterly 73(4):297–311
Miron IC, Baroana VC, Popescu F, Ionica F (2014) Pharmacological mechanisms underlying the association of antipsychotics with metabolic disorders. Current health sciences journal 40(1):12–17. doi:10.12865/chsj.40.01.02
Nielsen J, Skadhede S, Correll CU (2010) Antipsychotics associated with the development of type 2 diabetes in antipsychotic-naive schizophrenia patients. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology 35(9):1997–2004. doi:10.1038/npp.2010.78
2004) Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care 27(2):596–601
Baker RA, Pikalov A, Tran QV, Kremenets T, Arani RB, Doraiswamy PM (2009) Atypical antipsychotic drugs and diabetes mellitus in the US Food and Drug Administration adverse event database: a systematic Bayesian signal detection analysis. Psychopharmacol Bull 42(1):11–31
DuMouchel W, Fram D, Yang X, Mahmoud RA, Grogg AL, Engelhart L, Ramaswamy K (2008) Antipsychotics, glycemic disorders, and life-threatening diabetic events: a Bayesian data-mining analysis of the FDA adverse event reporting system (1968-2004). Annals of clinical psychiatry : official journal of the American Academy of Clinical Psychiatrists 20(1):21–31. doi:10.1080/10401230701844612
Navidi W, Weinhandl E (2002) Risk set sampling for case-crossover designs. Epidemiology 13(1):100–105
Acknowledgments
The authors would like to thank the Analytical Services, Ministry of Health of New Zealand for providing the datasets and the Research in Pharmacoepidemiology (RiPE) group, School of Pharmacy, University of Otago for providing clinical data management and support. All patient data used in this study are de-identified.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Nishtala, P.S., Chyou, Ty. Real-world risk of diabetes with antipsychotic use in older New Zealanders: a case-crossover study. Eur J Clin Pharmacol 73, 233–239 (2017). https://doi.org/10.1007/s00228-016-2158-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-016-2158-2