Abstract
Aims
Modifications of antimicrobials’ pharmacokinetic parameters have been reported in critically ill patients, resulting in a risk of treatment failure. We characterized amikacin pharmacokinetic variability in critically ill patients with ventilator-associated pneumonia (VAP) and evaluated several dosing regimens.
Methods
We conducted a prospective multicenter study in critically ill patients with presumptive diagnosis of Gram-negative bacilli (GNB) VAP. Patients empirically received imipenem and a single-dose of amikacin, which was administered as a 30-min infusion (20 mg/kg). Concentrations were measured 0.5, 1, 8, 16, and 24 h after beginning of infusion. Pharmacokinetic parameters were estimated using a population approach. Main pharmacodynamic target was a ratio ≥10 between the concentration achieved 1 h after beginning of infusion (C 1h) and the minimal inhibitory concentration of the liable bacteria (MIC). We simulated individual C 1h for several dosing regimens by Monte Carlo method and computed C 1h/MIC ratios for MICs from 0.5 to 64 mg/L.
Results
Sixty patients (47 males), median (range) age, and body weight, 61.5 years (28–84) and 78 kg (45–126), respectively, were included. Amikacin median C 1h was 45 mg/L (22–87). Mean value (between-patients variability) for CL, V1, Q, and V2 were 4.3 L/h (31 %), 15.9 L (22 %), 12.1 L/h (27 %), and 21.4 L (47 %), respectively. CL increased with CrCL (p < 0.001) and V1 with body weight (p < 0.001) and PaO2/FIO2 ratio (p < 0.001). With a 25 mg/kg regimen, the pharmacodynamic target was achieved in 20 and 96 % for a MICs of 8 and 4 mg/L, respectively.
Conclusion
Amikacin clearance was decreased and its volume of distribution was increased as previously reported. A ≥25 mg/kg single-dose is needed for empirical treatment of GNB-VAP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ventilator-associated pneumonia (VAP), defined as pneumonia occurring more than 48 h after the initiation of mechanical ventilation, is the most common hospital-acquired infection in the intensive care unit (ICU) [1]. Many previously published studies have shown that early and appropriate antibiotic therapy is associated with better outcome in critically ill patients with severe infections [2]. The latest guidelines of the American Thoracic Society and Infectious Diseases Society of America recommend that the empirical antibiotic therapy for late-onset pneumonia or in patients with risk factors for infection by multidrug-resistant pathogens should include a combination of an anti-pseudomonal β-lactam and an anti-pseudomonal fluoroquinolone or an aminoglycoside such as amikacin [3]. The superiority of once-daily dosing of aminoglycosides over multiple-daily dosing has been established [4]. However, no data supports a clinical benefit of a single-dose of aminoglycosides over multiple administrations. The rationale for an initial combination therapy is to broaden the antimicrobial spectrum of the empirical therapy; however, some data suggest adverse effects linked to repeated administrations, such as adaptive resistance or nephrotoxicity.
Amikacin, like other aminoglycosides, exhibits a concentration-dependent killing and produces a prolonged post-antibiotic effect. Previous clinical studies have shown that a ratio of 10 or more between the concentration achieved 1 h after the beginning of a 30-min infusion (C 1h) and the minimal inhibitory concentration (MIC) of the bacteria responsible of the infection was predictive of therapeutic success [5, 6]. Some authors recently suggested that a ratio between the area under the curve of concentration over time (AUC) and MIC (AUC/MIC) greater than 90 also has some predictive value for therapeutic success, with no difference in predictive capacities when compared to C 1h/MIC ratio [7]. Those pharmacodynamic targets may be difficult to reach in critically ill patients for several reasons. First, large interindividual variations of amikacin pharmacokinetic parameters have been reported [8–10], with variations of C 1h and AUC as a consequence. This variability is partially explained by total body weight and creatinine clearance, which are the most frequently reported covariates of amikacin pharmacokinetic parameters [8–11]. Second, the current breakpoint of MICs defined by the European Committee on Antimicrobial Susceptibility Testing (EUCAST) for Enterobacteriaceae species and Pseudomonas aeruginosa is 8 mg/L [12].
Few authors investigated the amikacin dosage needed in a single infusion setting to achieve the defined pharmacodynamic target in a homogenous group of patients hospitalized in ICU with suspected VAP.
Using data from a prospective clinical trial, we aimed (i) to characterize amikacin pharmacokinetic parameters and their variability in ICU patients with suspected VAP, (ii) to study covariates of amikacin pharmacokinetic parameters, and (iii) to evaluate several amikacin dosing regimens using Monte Carlo simulations.
Material and methods
Patients and sampling
This study is part of the IMPACT trial, a prospective multicenter clinical trial conducted between 2009 and 2011 in three ICUs, at Bichat university hospital, Paris, France, and Victor Dupouy hospital, Argenteuil, France (ClinicalTrials #NCT00950222).
Patients were included in the trial if they presented the following criteria: mechanical ventilation for more than 48 h, a clinical suspicion of Gram-negative bacilli VAP, risk factors for multidrug resistant bacteria, and if a microbiologic sample was obtained before initiation of antimicrobial therapy using blinded protected telescoping catheter or bronchoalveolar lavage. Clinical suspicion of VAP was defined by the onset of new lung infiltrates on chest radiography, fever greater than 38.3 °C, purulent tracheal aspirates, or a leukocytosis >10,000/mL. Risk factors for multidrug-resistant bacteria were antimicrobial therapy in the preceding 15 days or late-onset VAP (≥ 6 days) [3]. Patients were not eligible if they were younger than 18 year old and if they had renal failure requiring renal replacement therapy or received a treatment by imipenem or amikacin at the time of inclusion. Written consent was obtained from patients or their legal representative. The study was approved by the Ethics Committee of the Hôtel-Dieu university hospital (APHP, Paris).
All patients were treated with a combination of imipenem and amikacin. Imipenem was administered every 8 h over a 30-min infusion and should not be changed for the first 48 h. Doses ranged from 500 to 1000 mg, according to creatinine clearance (CrCL). Amikacin treatment consisted in one single 30-min infusion of a suggested dose of 20 mg/kg administered the first day of antimicrobial therapy. This dose was recommended by guidelines at the time of this study was designed [3]. De-escalation therapy was encouraged after isolation of the Gram-negative pathogen and obtaining the results of susceptibility tests.
Usual clinical and demographic characteristics were recorded at ICU admission, as well as ventilator parameters, two scores for assessment of severity (SOFA [13] and SAPS-II [14]) and routine biologic markers.
Blood samples were collected 0.5, 1, 8, 16, and 24 h after the onset of infusion. Exact times of beginning and end of infusion were recorded, as well as exact sampling times. Amikacin concentrations were determined by fluorescence polarization immunoassay using the amikacin Innofluor® kit [15]. The limit of quantification of the technique is 0.5 mg/L, and coefficients of variation for intra- and inter-assay were 4.1 % and 5.8 %, respectively.
Population pharmacokinetic analysis
Basic model
Population pharmacokinetic analysis was performed using the Stochastic Approximation Expectation Minimization (SAEM) algorithm in Monolix v4.2 (Lixoft, Orsay, France, available at http://www.lixoft.com). This algorithm handles concentrations below the limit of quantification to improve parameters estimation [16]. We used the total dose administered to each patient for model building.
Both one- and two-compartment(s) models with first order elimination were tested [8, 10, 11, 17].
We used an exponential random effects model for each pharmacokinetic parameter. We assumed the random effects to have a normal distribution with a mean of 0 and a variance of ω2. Correlation (ρ) between individual random effects was kept in the model if the estimated correlation coefficient was ≥0.2. The residual error model was supposed to be additive, proportional or combined, with a being the standard deviation of the additive component and b the standard deviation of the proportional component.
The best model was chosen using the Bayesian information criteria (BIC), derived for each model from the computation of likelihood by importance sampling [18].
Covariate model building
The influence of the following covariates at initiation of treatment on amikacin pharmacokinetic parameters was tested: three demographic variables (age, gender, and total body weight), two clinical variables (shock and edema score [19]), two severity scores (SOFA [13] and SAPS-II [14]), two ventilator-related parameters (PEEP and PaO2/FIO2 ratio), and three biochemical markers (serum albumin, total bilirubin and 4-h creatinine clearance). Four-hour creatinine clearance was calculated using serum creatinine level and a 4-h urine collection at the day of inclusion. This approach has been validated for monitoring renal function in critically ill patients [20]. Missing values for tested covariates were imputed to the median value observed in the analysis population. Steps for selection of covariate model are described in Online Resource 1.
Final model determination
We graphically studied the influence of covariates on their related pharmacokinetic parameters. Outliers were studied and excluded from analysis when there was a suspicion of incomplete data collection, in particular for creatinine clearance which was estimated using a 4-h urine collection. Patients for whom covariates had been imputed were also excluded. The covariate model was adjusted to the reduced population obtained after exclusion of these patients, and a backward selection method was used in order to obtain a final model in which all covariates had a p value < 0.05 using the likelihood-ratio test.
The coefficient estimated for creatinine clearance was compared to 1 using the likelihood ratio test.
Model evaluation
Evaluation of the final model was conducted using graphical methods. Basic goodness-of-fit plots were used, as well as the individual weighted residuals (IWRES) and the normalized prediction distribution errors (NPDE) over time and the visual predictive check (VPC). NPDE and VPC were generated using 500 Monte Carlo simulations.
Evaluation of doses by simulation
Using the estimated distribution of amikacin pharmacokinetic parameters in the final model with covariates, we simulated amikacin concentration obtained 1 h after the start of a 30-min infusion and AUC for 1000 patients by Monte Carlo simulation for several dosing regimen. This timing for amikacin sampling is commonly used for studying amikacin efficacy [17]. For pharmacokinetic simulations, we randomly re-sampled 1000 vectors of covariates among those observed in the patients included in the analysis and simulated individual pharmacokinetic parameters from their estimated distribution in the final model with covariates. Simulated dose regimens were the following: 20, 25, 30, 35, and 40 mg/kg. We computed the probability to achieve a C 1h/MIC ratio ≥ 10 and an AUC/MIC ratio ≥ 90 for MICs ranging from 0.25 to 64 mg/L for each or these dosing regimens. These values are usually observed in clinical practice (http://www.eucast.org/mic_distributions/), with an 8 mg/l susceptibility breakpoint for Enterobacteriaceae species and P. aeruginosa.
Results
Patients’ characteristics
Of the 61 patients included in the IMPACT trial, one had a kinetic profile which was not compatible with a unique injection and was withdrawn from analysis. Thus, data from 60 patients were available for modeling. Patients’ characteristics are presented in Table 1. Median (min-max) age and total body weight were respectively 61.5 years (28–84) and 78 kg (45–126), and most patients were males (n = 47, 78 %). At inclusion, 26 patients had septic shock (43 %). Median SAPS-II and SOFA scores were 42 (19–90) and 7 (2–17), respectively, and median 4-h creatinine clearance was 82 mL/min (4–412).
Pharmacokinetic data and modeling
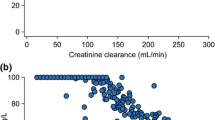
A total of 291 values of amikacin concentrations were available, with a median of 5 per patient (3–5). Median amikacin dose administered was 20 mg/kg (11–28). Median serum concentration observed 1 h after the beginning of infusion was 45 mg/L (22–87). Individual observed concentrations are presented in Fig. 1. Fourteen values (4.8 %) of amikacin concentration were below the limit of quantification.
Serum concentrations were best described by a two-compartment model with a combined residual error model (Online Resource 2). Correlations between estimates in the basic model were ≥ 0.2 between all individual random effects, and thus kept in the analysis. In this model, amikacin clearance (CL) was estimated to 4.0 L/h, central volume of distribution (V1) to 15.3 L, peripheral volume of distribution (V2) to 22.1 L, and inter-compartmental clearance (Q) to 12.2 L/h (Table 2). Their relative standard errors were satisfactory, all being <10 %. The inter-individual variability of pharmacokinetic parameters ranged from 30 % for Q to 60 % for CL. Estimates of inter-individual variability of pharmacokinetic parameters were also satisfactory (Table 2). The goodness of fit plots of this model without covariate did not show any model deficiency (data not shown).
Among the 12 studied covariates, eight were significantly associated with the individual Bayes estimates of pharmacokinetic parameters (Online Resource 1). The best model included four covariates (Online resource 1): creatinine clearance for CL, total body weight and PaO2/FIO2 ratio for V1, and creatinine clearance for V2. Two patients presented a low creatinine clearance but a high amikacin clearance. Their individual fits were satisfactory, and we hypothesized that their urine collection was incomplete. They were excluded from the analysis. In another patient, amikacin clearance was estimated to 11 mL/min, but creatinine clearance was missing and had been imputed to the median value. The five patients in whom creatinine clearance was not available were excluded. The four-covariate model was adjusted to the 53 remaining patients.
Creatinine clearance was no longer a significant covariate for V2 (p = 0.4), and the final model included three covariates: creatinine clearance (p < 0.001) for CL, total body weight (p < 0.001), and PaO2/FIO2 ratio (p < 0.001) for V1 (Table 2). The coefficient for creatinine clearance on CL was significantly different from 1 (p < 0.001). The variations of pharmacokinetic parameters according to covariates are presented in Fig. 2. All three of them were positively correlated with the pharmacokinetic parameters. Amikacin clearance was estimated in the final model at 4.3 L/h (72 mL/min), V1 at 15.9 L, and V2 at 21.4 L (Table 2). Variance–covariance matrix between the individual random effects is presented in Online Resource 3.
Changes in amikacin pharmacokinetic parameters with respect to covariates in the final model. Circles correspond to observed values; red curves represent the changes of pharmacokinetic parameters according to estimated covariate model; dashed lines correspond to the median observed values. P/F, PaO2/FIO2
Goodness-of-fit plots did not show any model misspecification. The IWRES and NPDE were centered to zero and did not show any trend over time (Online Resource 4). The VPC did not show any model deficiency (Fig. 3).
Pharmacokinetic simulations
Probabilities to achieve a C 1h/MIC ratio ≥ 10 according to the single-dose of amikacin simulated and MICs are presented in Fig. 4. A 20 mg/kg dose was sufficient to achieve the pharmacokinetic target in 100 % of patients for a MIC of 2 mg/L or less. With this dose, only 80 % and 4 % of patients achieved the target for a MIC of 4 and 8 mg/L, respectively. With a 25 mg/kg simulated infusion, the probability to reach the target was 96 % for a MIC of 4 mg/L and 20 % for a MIC of 8 mg/L. With the 40 mg/kg regimen, the probability to achieve a C 1h/MIC ratio ≥10 was 80 % for a MIC of 8 mg/L. Online Resource 5 presents the boxplots of the 1000 simulated C 1h/MIC ratio for a MIC of 8 mg/L, for doses from 20 to 40 mg/kg.
Results were similar when considering the target of AUC/MIC ≥ 90 (Online Resource 6). With a dose of 20 mg/kg, 90 % of patients achieved the target for a MIC of 2 mg/L or less, but this proportion was below 20 % for a MIC value of 8 mg/L. Simulations for a dose of 25 mg/kg showed that 69 % of patients achieved the pharmacodynamic target for a MIC of 4 mg/L, but only 22 % did for a MIC of 8 mg/L. An increase to 40 mg/kg led to an increase of the probability to reach the pharmacodynamic target to 90 % and 52 % for a MIC of 4 or 8 mg/L, respectively.
Discussion
The present study confirms that the 20 mg/kg single-dose regimen traditionally used is not adequate based on the analyzed pharmacokinetic and pharmacodynamic indices and that amikacin dosing regimen should be increased to at least 25 mg/kg for critically ill patients when initiating empirical amikacin therapy for VAP caused by a Gram-negative rod. This increase has been recently suggested by Taccone et al. [17]. To our knowledge, few data supported this recommendation [21, 22]. Those studies did not use a population approach and were performed in patients with severe sepsis or septic shock, thereby gathering patients with different varieties of infection. Although the influence of the nature of infection has not been thoroughly examined, some authors suggested that the clinical diagnosis might influence the pharmacokinetic parameters of aminoglycosides [10]. We focused our analysis on a homogenous group of patients. Our simulation-based analysis, using data from a prospective multicenter trial and a solid methodology, confirms the results of Taccone et al. [17], and suggest that a higher initial dose is necessary, at least in patients requiring mechanical ventilation.
A comparative study of different initial amikacin dosing regimen would confirm our conclusions. It would also allow to study the impact of higher doses on renal function. Nephrotoxicity is a well-known adverse effect of aminoglycoside therapy. However, this effect has been associated with duration of therapy [23]. Available studies suggest that short-course treatments would allow to minimize toxicity while keeping maximal efficacy [24]. In the majority of severe infections, aminoglycosides are administered as a single infusion in combined antimicrobial therapy. Higher single doses would therefore have potentially limited or no effect on renal function, as suggested by recent studies [21, 22]. It should also be noticed that neuromuscular blockade might have an increased frequency when using higher doses of aminoglycosides [25]. Its main manifestation is respiratory weakness, and this side effect is reversible. It would have a limited impact on patients with mechanical ventilation.
Another result is that amikacin clearance and volume of distribution were estimated to approximately 70 mL/min and 37 L, respectively. These values were in accordance with published data on ICU patients. This confirms the issues observed in antimicrobial pharmacokinetic parameters in critically ill patients [26, 27], i.e., a decrease of drug clearance and an increase of volume of distribution [28–32]. Significant covariates were 4-h creatinine clearance for amikacin clearance, and total body weight and PaO2/FIO2 ratio for central volume.
The association between total body weight and volume of distribution has already been reported [9, 10, 33], and administered amikacin doses are commonly adjusted to total body weight. It is still not clear which measure of body size best describes pharmacokinetic parameters. A recent meta-analysis explored the relationship between drug clearance and body size metrics [34]. The authors found that lean body weight to the exponent 2/3 was more suitable for describing the relationship between drug clearance and body size. However, they did not question the link between the volume of distribution and body size, and this conclusion may not apply for all pharmacokinetic parameters. As body size was not collected in the IMPACT trial, we could not investigate the effect of this weight metrics or of body mass index on amikacin pharmacokinetic parameters. For hydrophilic drugs such as aminoglycosides, data on the influence of lean body weight and drug volume of distribution are lacking and would be of interest.
The BIC was not improved by adding total body weight as a covariate for V2. This is quite unexpected, and V2 might represent a weight-independent compartment in which amikacin accumulates. It has previously been shown that total urine recovery of amikacin is not complete 24 h after its administration and that aminoglycosides including amikacin accumulates in the kidney [35, 36].
We found a high interindividual variability in amikacin pharmacokinetic parameters as previously reported in studies performed among critically ill patients [8, 37]. Significant covariates reduced this variability, with a 50 % maximal decrease for amikacin CL in our study. To our knowledge, no published covariate model could fully explain the observed interindividual variability. It is therefore highly probable that inside diagnosis-homogenous groups of patients, subpopulations exist that are not individualized by usual covariates. Such variability makes the choice of the optimal dosing strategy extremely challenging from an individual perspective. Considering the absence of severe adverse effect expected in a single-dose setting, mechanically ventilated patients in intensive care units should receive a high amikacin dose in order to maximize the probability of pharmacodynamic target attainment. Further studies should furthermore focus on identification of covariates allowing for reducing the high unexplained interindividual variability.
The main limitation of this study is that it was restricted to a limited number of patients who had a suspicion on VAP. Therefore, the results may not apply to patients who do not require mechanical ventilation. Nevertheless, the French surveillance network of nosocomial infections recently reported that about 60 % of patients hospitalized in ICU would require mechanical ventilation during their stay [38]. Another limitation is the use of the MIC breakpoint for Monte Carlo simulations. To our knowledge, no published data report the distribution of MICs for Gram-negative bacilli responsible for infection in intensive care units. As bacteria involved in infection of critically ill patients frequently have higher MICs than those isolated from patients hospitalized in other wards, we did not use published MICs distributions that gather strains obtained from all wards. A recent study of MIC breakpoints based on simulations suggested that pharmacokinetic–pharmacodynamic breakpoints are similar to those defined by the EUCAST or the CLSI [39]. In the empirical setting, the worst-case assumption should be preferred when initiating an antimicrobial therapy. This approach is currently used for pharmacokinetic-pharmacodynamic simulations [17, 40, 41]. However, it is likely that the majority of Gram-negative rods involved in critical infections have a MIC below 8 mg/L. For a MIC of 4 mg/L, 96 % of patients achieved a C 1h/MIC ratio ≥10 after a dose of 25 mg/kg in our simulations.
In conclusion, amikacin pharmacokinetic parameter values were similar to those previously reported. This study confirms recent results for the determination of the initial amikacin dose required in critically ill patients. An empirical dose of 25 mg/kg or more is needed to achieve the amikacin pharmacodynamic predictors of clinical efficacy in Gram-negative bacilli infections. Other studies are needed to prospectively evaluate these conclusions in order to improve the management of patients with severe infections.
References
Bouadma L, Wolff M, Lucet JC (2012) Ventilator-associated pneumonia and its prevention. Curr Opin Infect Dis 25(4):395–404
Zahar JR, Timsit JF, Garrouste-Orgeas M, Francais A, Vesin A, Descorps-Declere A, Dubois Y, Souweine B, Haouache H, Goldgran-Toledano D, Allaouchiche B, Azoulay E, Adrie C (2011) Outcomes in severe sepsis and patients with septic shock: pathogen species and infection sites are not associated with mortality. Crit Care Med 39(8):1886–1895
Zahar JR, Timsit JF, Garrouste-Orgeas M, Francais A, Vesin A, Descorps-Declere A, Dubois Y, Souweine B, Haouache H, Goldgran-Toledano D, Allaouchiche B, Azoulay E, Adrie C (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171(4):388–416
Barza M, Ioannidis JP, Cappelleri JC, Lau J (1996) Single or multiple daily doses of aminoglycosides: a meta-analysis. BMJ 312(7027):338–345
Moore RD, Lietman PS, Smith CR (1987) Clinical response to aminoglycoside therapy: importance of the ratio of peak concentration to minimal inhibitory concentration. J Infect Dis 155(1):93–99
Kashuba AD, Nafziger AN, Drusano GL, Bertino JS Jr (1999) Optimizing aminoglycoside therapy for nosocomial pneumonia caused by gram-negative bacteria. Antimicrob Agents Chemother 43(3):623–629
Zelenitsky SA, Harding GK, Sun S, Ubhi K, Ariano RE (2003) Treatment and outcome of Pseudomonas aeruginosa bacteraemia: an antibiotic pharmacodynamic analysis. J Antimicrob Chemother 52(4):668–674
Delattre IK, Musuamba FT, Nyberg J, Taccone FS, Laterre PF, Verbeeck RK, Jacobs F, Wallemacq PE (2010) Population pharmacokinetic modeling and optimal sampling strategy for Bayesian estimation of amikacin exposure in critically ill septic patients. Ther Drug Monit 32(6):749–756
Bacopoulou F, Markantonis SL, Pavlou E, Adamidou M (2003) A study of once-daily amikacin with low peak target concentrations in intensive care unit patients: pharmacokinetics and associated outcomes. J Crit Care 18(2):107–113
Romano S, Fdez DM, de Gatta M, Calvo V, Mendez E, Dominguez-Gil A, Lanao JM (1998) Influence of clinical diagnosis in the population pharmacokinetics of amikacin in intensive care unit patients. Clin Drug Investig 15(5):435–444
Lugo G, Castaneda-Hernandez G (1997) Relationship between hemodynamic and vital support measures and pharmacokinetic variability of amikacin in critically ill patients with sepsis. Crit Care Med 25(5):806–811
EUCAST Breakpoint tables for interpretation of MICs and zone diameters. Consulted on 03/02/2013, available at : http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/Breakpoint_table_v_3.1.pdf
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (sepsis-related organ failure assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European society of intensive care medicine. Intensive Care Med 22(7):707–710
Le Gall JR, Lemeshow S, Saulnier F (1993) A new simplified acute physiology score (SAPS II) based on a European/North American multicenter study. JAMA 270(24):2957–2963
White LO, Holt HA, Reeves DS, MacGowan AP (1997) Evaluation of innofluor fluorescence polarization immunoassay kits for the determination of serum concentrations of gentamicin, tobramycin, amikacin and vancomycin. J Antimicrob Chemother 39(3):355–361
Samson A, Lavielle M, Mentré F (2006) Extension of the SAEM algorithm to left-censored data in non-linear mixed-effects model: application to HIV dynamics model. Comput Stat Data Anal 51(3):1562–1574
Taccone FS, Laterre PF, Spapen H, Dugernier T, Delattre I, Layeux B, De Backer D, Wittebole X, Wallemacq P, Vincent JL, Jacobs F (2010) Revisiting the loading dose of amikacin for patients with severe sepsis and septic shock. Crit Care 14(2):R53
Lavielle M, Mentré F (2007) Estimation of population pharmacokinetic parameters of saquinavir in HIV patients with the MONOLIX software. J Pharmacokinet Pharmacodyn 34(2):229–249
Gomez CM, Cordingly JJ, Palazzo MG (1999) Altered pharmacokinetics of ceftazidime in critically ill patients. Antimicrob Agents Chemother 43(7):1798–1802
Pickering JW, Frampton CM, Walker RJ, Shaw GM, Endre ZH (2012) Four h creatinine clearance is better than plasma creatinine for monitoring renal function in critically ill patients. Crit Care 16(3):R107
Galvez R, Luengo C, Cornejo R, Kosche J, Romero C, Tobar E, Illanes V, Llanos O, Castro J (2011) Higher than recommended amikacin loading doses achieve pharmacokinetic targets without associated toxicity. Int J Antimicrob Agents 38(2):146–151
Mahmoudi L, Mohammadpour AH, Ahmadi A, Niknam R, Mojtahedzadeh M (2013) Influence of sepsis on higher daily dose of amikacin pharmacokinetics in critically ill patients. Eur Rev Med Pharmacol Sci 17(3):285–291
Bertino JS Jr, Booker LA, Franck PA, Jenkins PL, Franck KR, Nafziger AN (1993) Incidence of and significant risk factors for aminoglycoside-associated nephrotoxicity in patients dosed by using individualized pharmacokinetic monitoring. J Infect Dis 167(1):173–179
Drusano GL, Ambrose PG, Bhavnani SM, Bertino JS, Nafziger AN, Louie A (2007) Back to the future: using aminoglycosides again and how to dose them optimally. Clin Infect Dis 45(6):753–760
Snavely S, Hodges G (1984) The neurotoxicity of antibacterial agents. Ann Intern Med 101(1):92–104
Roberts JA, Lipman J (2009) Pharmacokinetic issues for antibiotics in the critically ill patient. Crit Care Med 37(3):840–851, quiz 859
Pea F, Viale P, Furlanut M (2005) Antimicrobial therapy in critically ill patients: a review of pathophysiological conditions responsible for altered disposition and pharmacokinetic variability. Clin Pharmacokinet 44(10):1009–1034
Lode H, Grunert K, Koeppe P, Langmaack H (1976) Pharmacokinetic and clinical studies with amikacin, a new aminoglycoside antibiotic. J Infect Dis 134(SUPPL):S316–S322
Tormo C, Abad FJ, Ronchera-Oms CL, Parra V, Jimenez NV (1995) Critically-ill patients receiving total parenteral nutrition show altered amikacin pharmacokinetics. Clin Nutr 14(4):254–259
Marik PE (1993) Aminoglycoside volume of distribution and illness severity in critically ill septic patients. Anaesth Intensive Care 21(2):172–173
Bressolle F, Gouby A, Martinez JM, Joubert P, Saissi G, Guillaud R, Gomeni R (1996) Population pharmacokinetics of amikacin in critically ill patients. Antimicrob Agents Chemother 40(7):1682–1689
Marik PE, Havlik I, Monteagudo FS, Lipman J (1991) The pharmacokinetic of amikacin in critically ill adult and paediatric patients: comparison of once- versus twice-daily dosing regimens. J Antimicrob Chemother 27(Suppl C):81–89
Fernandez M, de Gatta MD, Victoria Calvo M, Ardanuy R, Dominguez-Gil A, Lanao JM, Moreno SR (2009) Evaluation of population pharmacokinetic models for amikacin dosage individualization in critically ill patients. J Pharm Pharmacol 61(6):759–766
McLeay SC, Morrish GA, Kirkpatrick CM, Green B (2012) The relationship between drug clearance and body size: systematic review and meta-analysis of the literature published from 2000 to 2007. Clin Pharmacokinet 51(5):319–330
Edwards CQ, Smith CR, Baughman KL, Rogers JF, Lietman PS (1976) Concentrations of gentamicin and amikacin in human kidneys. Antimicrob Agents Chemother 9(6):925–927
French MA, Cerra FB, Plaut ME, Schentag JJ (1981) Amikacin and gentamicin accumulation pharmacokinetics and nephrotoxicity in critically ill patients. Antimicrob Agents Chemother 19(1):147–152
Joubert P, Bressolle F, Gouby A, Doucot PY, Saissi G, Gomeni R (1999) A population approach to the forecasting of amikacin plasma and urinary levels using a prescribed dosage regimen. Eur J Drug Metab Pharmacokinet 24(1):39–46
Savey A, Machut A (2012) Surveillance des infections nosocomiales en réanimation adulte. Réseau REA-Raisin, France, Résultats 2011. Available at http://www.invs.sante.fr/%20fr/Publications-et-outils/Rapports-et-syntheses/Maladies-infectieuses/2013/Surveillance-des-infections-nosocomiales-en-reanimation-adulte. In: ed
Frei CR, Wiederhold NP, Burgess DS (2008) Antimicrobial breakpoints for gram-negative aerobic bacteria based on pharmacokinetic-pharmacodynamic models with Monte Carlo simulation. J Antimicrob Chemother 61(3):621–628
Carlier M, Noe M, De Waele JJ, Stove V, Verstraete AG, Lipman J, Roberts JA (2013) Population pharmacokinetics and dosing simulations of amoxicillin/clavulanic acid in critically ill patients. J Antimicrob Chemother 68(11):2600–2608
de Montmollin E, Bouadma L, Gault N, Mourvillier B, Mariotte E, Chemam S et al. (2014) Predictors of insufficient amikacin peak concentration in critically ill patients receiving a 25 mg/kg total body weight regimen. Intensive Care Med
Acknowledgments
This study was funded by the Contrat d’Initiation à la Recherche Clinique 2006 (Assistance Publique-Hôpitaux de Paris, Département de la Recherche Clinique et du Développement, CRC 06049)
Authors’ contributions
FM, LAL, MW, and OP conceived the study. AF, MW, and OP provided medical care to trial participants. LM performed the pharmacological assays. CB, CC, CL, and FM performed the statistical analysis. CB, FM, MW, and OP wrote the first draft of the manuscript. All authors read and agreed with the manuscript in its published version.
Conflict of interest
All authors declare no support from any organization for the submitted work; no financial relationship with any organization that might have an interest in the submitted work in the previous 3 years; no other relationships or activity that could appear to have influenced the submitted work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
Covariate model building. (DOCX 54.9 kb)
Online Resource 2
Selection of structural and residual error models. (DOCX 29.7 kb)
Online Resource 3
Variance – covariance matrix between individual random effects estimated in the final model. (DOCX 29.5 kb)
Online Resource 4
Evaluation graphics for the final model. A, observed vs. population (left) and individual (right) predicted concentrations; B, individual weighted residuals (IWRES, left) and normalized predictions distribution errors (NPDE, right) versus time. BQL, below limit of quantification. (DOCX 66.3 kb)
Online Resource 5
Distribution of the C1h/MIC ratio according to amikacin dose for a MIC of 8 mg/L. Concentrations achieved 1 hour after the beginning of a 30-minute infusion (C1h) were simulated for 1000 patients using Monte Carlo method from the final pharmacokinetic model. The box and whisker plots display the median values and 25th and 75th percentiles of the distribution; the whiskers extend to the10th and 90th percentiles. The horizontal black line represent the usual target of a C1h/MIC ratio = 10. MIC, minimal inhibitory concentration. (DOCX 1.24 mb)
Online Resource 6
Probability to achieve the pharmacokinetic target (AUC/MIC ≥ 90) according to the dosing regimen and the theoretical minimum inhibitory concentration of the strain in the Monte Carlo simulation. AUC, Area under the curve ; MIC, minimal inhibitory concentration. (DOCX 1.24 mb)
Rights and permissions
About this article
Cite this article
Burdet, C., Pajot, O., Couffignal, C. et al. Population pharmacokinetics of single-dose amikacin in critically ill patients with suspected ventilator-associated pneumonia. Eur J Clin Pharmacol 71, 75–83 (2015). https://doi.org/10.1007/s00228-014-1766-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-014-1766-y