Abstract
High-frequency recordings of valve opening behavior (VOB) in bivalves are often used to detect changes in environmental conditions. However, generally a single variable such as temperature or the presence of toxicants in the water is the focus. A description of routine VOB under non-stressful conditions is also important for interpreting responses to environmental changes. Here we present the first detailed quantitative investigation of the in-situ VOB of eastern oysters (Crassostrea virginica) to environmental variables typically not considered stressful. The VOB of eight individuals was monitored for seven weeks in a Louisiana estuary. We examined the relationships between VOB metrics (variance in mean % max opening among oysters, the probability of an oyster being closed, and the rate of valve closure), and temperature, salinity, chlorophyll-a (chl-a) concentration, the rate of change in those environmental variables, and the rate of change in water depth. Relationships were analyzed through statistical models including rates of change over 0, 0.25, 1-, 6-, 12-, and 24-hours. All the responses were best explained by the 12-hour time step model. The interaction effect between salinity and the rate of change of salinity had the greatest impact on variance in oysters’ behavior. Oysters closed faster at higher salinities and were more likely to be closed at lower chl-a concentrations. Significant interactions were found between many environmental variables, indicating a high level of complexity of oyster behavior in the natural environment. This study contributes to a better understanding of the impact of environmental conditions on oyster behavior and can help inform predictive tools for restoration initiatives and fisheries practices.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The eastern oyster, Crassostrea virginica, is an important species for habitat formation and ecosystem function in estuaries along the Atlantic and Gulf of Mexico coasts of North America. Like many other bivalves, they live most of their life attached to a substrate (rock, congener shell, wood piling, etc.) and are exposed to changes in environmental conditions that some mobile organisms may be able to avoid. Eastern oysters can tolerate large ranges and changes in temperature (Shumway 1996; Comeau et al. 2012; Marshall et al. 2021a), salinity (Shumway and Koehn 1982; Casas et al. 2018a; Marshall et al. 2021b), and dissolved oxygen (DO; Stickle et al. 1989; Coxe et al. 2023) and can survive starvation periods of several months (Comeau et al. 2012). With a wide geographical distribution, different populations can live in highly contrasting environmental conditions both in terms of average values and range of daily or seasonal variations (Beseres Pollack et al. 2011; Casas et al. 2018b). When changes occur rapidly and beyond the oysters’ capacity to osmoconform in time, oysters close their valves to seclude themselves from the surrounding water. For instance, oysters typically close when salinity drops too rapidly or when it falls below a certain threshold to avoid cellular damage caused by osmotic pressure changes (Hand and Stickle 1977; Shumway 1996; Casas et al. 2018a). The capacity of oysters to behaviorally respond to changes and the rate of change in environmental conditions through valve closure may be critical to their resilience in a changing coastal environment (Cloern et al. 2016). However, valve closure may also impact metabolism through a reduction in energy input from feeding and less energy efficient anaerobic pathways.
Bivalves open their valves to perform essential physiological functions such as feeding, respiration, reproduction, and excretion. When closed, the shell offers protection from predators and adverse environmental conditions. For a century, scientists have increasingly studied the valve opening behavior (VOB) of bivalves for two purposes: to better understand bivalve tolerance to ranges of environmental conditions or their rate of change (e.g., Shumway 1977a, b) and to use them as sentinels of environmental variability (Vereycken and Aldridge 2023). However, while past works have contributed to the description of the impact of single or a few environmental variables on VOB at the same time, analyses of the response of bivalves and oysters in particular to multiple environmental factors co-occurring in natural conditions are scarce. Knowledge is also lacking on the response of oysters to conditions within their tolerance range, i.e., ‘normal’ conditions. Many studies have focused on identifying thresholds of VOB response of oysters at the extreme of the tolerance range such as winter temperatures (Comeau et al. 2012; Clements et al. 2018), low pH (Clements et al. 2018), or hypoxic events (Coffin et al. 2021; Coxe et al. 2023); however, few reports exist of their behavior over long periods and under more average conditions. A better understanding of how oysters respond to a variety of changes in their natural environment will provide valuable information for the interpretation of VOB in both ecophysiological and environmental monitoring studies.
Valvometry techniques have evolved since the pioneering work of Nelson (1922), but research has continuously shown that the valve movements of oysters in response to environmental factors are complex. Oysters exhibit high inter-individual variability, which may be due to the generally low number of monitored individuals or failure to account for interactive effects. Valve closures of bivalves have been linked to tidal cycles (Nelson 1922; Sow et al. 2011), algal concentration (Higgins 1980), algal toxins (Nagai et al. 2006; Tran et al. 2010; Lavaud et al. 2021), chemical compounds (Kramer and Foekema 2001; Hartmann et al., 2016), acidification (Clements et al. 2018; Lassoued et al. 2021), dissolved oxygen concentration (DO; Porter and Breitburg 2016; Coffin et al. 2021), parasitic infections (Chambon et al. 2007), and sound (Charifi et al. 2017; Hubert et al. 2023). Most studies carried out in the field have related valve opening to a single factor. However, in the natural environment, oysters can experience multiple changes in conditions simultaneously, and the potential effect and interaction of multiple variables on the VOB of oysters and bivalves in general have rarely been studied (Hubert et al. 2023).
In this study, we aimed to describe the VOB of eastern oysters under typical environmental conditions in southeastern Louisiana, USA. A tray containing eight oysters was deployed under natural conditions and oyster valve movements were continuously recorded along with multiple environmental variables. We quantified the respective effects of water temperature, salinity, chlorophyll-a concentration, DO concentration, depth, their rate of change and their possible interactions on oyster VOB over a 7-week period. We hypothesized that interactions between environmental variables would be important drivers of oyster VOB. To test this assumption, we analyzed the data to determine how each environmental variable and their interactions may influence (i) inter-individual variability in VOB, (ii) the probability of oysters being closed, and (iii) the strength of the response through the rate of valve closure.
Materials and methods
Study site, oysters, and environmental variables
Oysters used in this study were the progeny of wild oysters (i.e., diploids) from Calcasieu Lake (29°47′6.00″N, 93°55′5.02″W) spawned during the summer of 2019 at the Louisiana Sea Grant Oyster Research Farm (LASGRF) in Grand Isle, LA (see Bodenstein et al. 2023 for details of larvae rearing). Once the spat height reached 6 mm, they were transferred to baskets on the longline system at the LASGRF. In January 2020 market-sized oysters (> 75 mm shell height) were transferred to Louisiana Universities Marine Consortium’s (LUMCON) De Felice Marine Center in Cocodrie, LA (29°15’14.10"N; 90°39’49.70"W).On 9 March 2021, shell height was measured for eight oysters, which were individually tagged, equipped with a valvometry system (see next section), and evenly placed in a tray box formed of two trays (50 × 50 × 10 cm each) securely tightened with cable ties to protect the oysters from predation. The trays were suspended horizontally from a pier located 42 m south of LUMCON’s environmental monitoring station so that the oysters were continuously submerged approximately 0.3 m off the bottom at 2 m depth (high tide). The study area is within a coastal saltmarsh dominated by a network of small bays connected by channels and is under the influence of the Gulf of Mexico to the South and runoff from various bayous (i.e., streams) in the North. Temperature (°C), salinity, chlorophyll-a concentration (µg L–1), DO concentration (mg L–1), and water depth (m) were recorded every fifteen minutes (0.25 h) by LUMCON’s environmental monitoring station located in a channel of western Terrebonne Bay (29°15.20’N, 90°39.80’W; data available at https://lumcon.edu/environmental-monitoring/).
Valve opening measurement
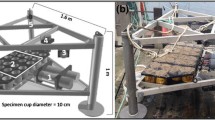
The oysters were equipped with a valvometry system (Nagai et al. 2006; Comeau et al. 2012) to monitor valve movements. UV light curing glue was used to attach a coated Hall element sensor (HW-300a, Asahi Kasei Corp, Chiyoda-ku, Tokyo, Japan; 0.5 g) to one valve at the maximum distance from the hinge (i.e., the ventral margin). A small magnet (4.8 mm diameter × 0.8 mm height; 0.1 g) was glued to the other valve, opposite the Hall sensor. Valve movements, measured by variation in the magnetic field between the sensor and the magnet (in µV), were recorded every 1 s using dynamic strain recording devices (DC 204R, Tokyo Sokki Kenkyujo Co., Shinagawa-ku, Tokyo, Japan). Every two weeks, the tray box was cleaned of fouling organisms and the oysters were checked to ensure that the sensors were still securely attached. The monitoring ended on 25 April 2021, at which point the oysters were notched, the adductor muscle was cut, and small calibration wedges (1–6 mm) were inserted between the valves at the ventral margin to derive a voltage versus gap calibration curve (R2 > 0.90) and convert voltage measurements into valve opening distances (VOD) for each individual oyster.
Valvometry data analysis
Data pre-processing
The valve opening distance (VOD) and shell height (SH) were combined to calculate the opening angle (OA) via the following equation: \(\text{OA}=\text{arcsin}\left(0.5\times\text{VOD}/\text{SH}\right)\times100\) (adapted from Wilson et al. 2005). There was detectable drift in voltage measurements over the course of the study, likely due to shell growth, which resulted in unrealistic measurements of opening angles (i.e., < 0 or > 100% of percent of maximum opening). To correct for this the maximum and minimum angle openings for each oyster were calculated over a centered rolling 48-hour window. This window was chosen after visual inspection of the data confirmed that all oysters completely opened and completely closed multiple times within any given 48-hour window. Drift-corrected values were then used to convert each OA to the percent of maximum (% max) opening over that 48-hour window so that when an oyster was completely open % max = 100 and when an oyster was completely closed % max = 0.
Oyster VOB data (% max) were matched to environmental data, which were collected every 15 min, using the date and time of the measurements. For example, environmental variables were measured at 00:00:00 and 00:15:00 on 9 March 2021, and the time between these two readings was designated as environmental interval 1. Oyster % max values recorded at or between 0:00:00 and 0:14:59 on 9 March 2021, were assigned to the environmental interval 1. To avoid pseudo-replication, % max values were summarized for each environmental interval into three variables: the mean of % max (hereafter mean % max) for that environmental interval, the percentage of time closed during that environmental interval – where closed was defined as % max ≤ 10%, and percent of time fully open during that environmental interval – where fully open was defined as % max ≥ 80%. Finally, we identified individual closure events for each oyster. These closure events were identified through a 5-step process. First, the change in % max values from the previous second was calculated. Second, the change was classified as positive (i.e., the valve opening became larger), negative (i.e., the valve opening narrowed), or no change. Third, consecutive periods of change in the same direction were combined. Fourth, the total % change in opening was calculated for all negative change groups by subtracting the % max value from the last second of the group from the % max value from the first second of the group. The rate of closure was then calculated by dividing this total % change by the number of seconds within the group. Finally, negative change groups were identified as a closure event if the total change in % max was ≥ 40%.
Initial visual inspection of the environmental data and oyster VOB indicated that oyster behavior may be related to the rate of change of temperature, salinity, chlorophyll-a concentration, and DO concentration. Therefore, for each of these environmental variables five additional variables were calculated: change from the previous 15-min time step (i.e., measurement recorded on the previous environmental interval), the rate of change over the past hour, as well as the rate of change over the past 6, 12, and 24 h. The rates of change were calculated using the equation:
where vt is the environmental variable value (temperature, salinity, or DO) for the current environmental interval and vt-x is the environmental variable value of the previous time interval with x = 0.25, 1, 6, 12, or 24 h.
In many systems, oyster VOB is tightly coupled to tidal cycles. However, in our study system, water depth varies both with tidal cycles and in response to on- and off-shore winds. As a result, behavioral rhythms associated with the ebb and flow of tides are regularly disrupted. Therefore, rather than accounting for tidally linked behaviors using a cyclical model, as several other studies have done (Tran et al. 2011), we included the rate of change in water depth as an explanatory variable in all models. This rate of change was calculated as described above, except only at the 6-hour interval.
Inter-individual variability in oyster behavior
Inter-individual correlations in oyster behavior (mean % max) was assessed in two ways. First, to assess if oyster behavior was correlated across the time series, we used variance ratio analysis (Schluter 1984) using the codyn package (Hallett et al., 2016). Significance was assessed by comparing the observed value to a null distribution generated via bootstrapping, where each oyster’s time series started at randomly assigned starting points (Hallett et al. 2014). Second, to determine whether environmental variables contributed to inter-individual variation, we calculated the variance between the mean % max values of all oysters in each environmental interval. We then fit 6 separate mixed effects models to test for a relationship between the variance in mean % max among oysters (% max2) and environmental variables in the current environmental interval and their rates of change at the 5 different time steps described above (0.25, 1, 6, 12, and 24 h). All models included the rate of change in water depth at the 6-hour interval as a proxy for tides and included all possible two-way interactions between all environmental variables and their rates of change as fixed effects (Table S2-S4). The oyster tray box was cleaned, and oysters were measured three times throughout the study. The period of time between each cleaning was considered a deployment. All the models also included deployment as a random variable.
All models were checked for multicollinearity using the check_collinearity function in the performance package in R (Lüdecke et al. 2021). Any model terms with moderate or high values of multicollinearity were removed from the model. In all cases, including both DO and chlorophyll-a concentrations (and their rates of change) introduced multicollinearity to the model. Since DO concentrations never reached critical values (> 4 mg L–1 throughout the monitoring; Table 1), DO and its associated rate of change variables were removed from all models. All predictor variables were scaled to adjust for variables having different ranges. The residuals of all models were examined to ensure compliance with model assumptions. Models were compared using the corrected Akaike information criterion (AICc) and all models within 2Δ AICc were considered well supported.
Environmental drivers of the rate of closure
To assess whether environmental conditions drove the rate of oyster closure, the dataset was filtered to retain only oysters and intervals where closure events occurred (see Sect. 2.3.1 for details). Multiple closure events within a single environmental interval did not occur. The relationship between the rate of oyster closure (absolute value of the slope) and environmental variables was assessed using mixed effects models with oyster ID as a random variable and a Beta distribution with logit link within the glmmTMB package (Brooks et al. 2017). The same models were assessed as with variability in oyster behavior and model residuals were examined using the DHARMa package (Hartig 2020). To meet model assumptions, the log of the rate of oyster closure was ultimately used as the response variable. Model fit was again compared using AICc.
Environmental drivers of oyster closure
To assess whether environmental conditions drove whether or not oysters closed their valves, the larger dataset was subset to include only periods where an oyster was fully opened or closed. The probability of an oyster being closed was assessed using logistic regression with oyster ID as a random variable via the glmmTMB package (Brooks et al. 2017). The same models were assessed as with the rate of valve closure. Model residuals were examined using the DHARMa package (Hartig 2020). Model fit was again compared using the Akaike Information Criterion corrected for small sample size (AICc).
Results
Environmental conditions and summary of oyster behavior
Throughout the 48 days of the study, environmental conditions varied, but would not be expected to induce a significant stress response in oysters (Table 1; Fig. 1). DO concentration was negatively correlated with water temperature and positively correlated with chlorophyll-a concentration (Table S1). As a result, DO concentration was removed from further analyses. Oysters spent most of their time either opening or closing their valves (Table 2) rather than in a fully open or fully closed position. Seven of the eight oysters we studied spent more time fully opened than fully closed, and all 8 oysters spent more time either fully or partially open than fully closed (Table 2). When oysters were closing their valves, they did so with an average closure rate of 9.7% per second (± 12.1%, sd).
Dynamics of the variance between oyster % max values of the 8 oysters monitored (a, g), % max of a single individual (b, h; per-second measures of % max averaged over the 15 min environmental intervals), % change in water depth (c, i), salinity (d, j), chlorophyll a concentration (Chl a; e, k), and temperature (f, l) over the entire study period (left) and over one week (right) between 9 March and 26 April 2021. PSU = practical salinity unit
Inter-individual variation in oyster behavior
Oyster behavior was significantly correlated among individuals throughout the course of the study (Observed Variance Ratio: 3.3, null mean and 95% confidence intervals: 1, 0.92–1.1). Despite this overall correlation between oyster behavior, variance in mean % max among oysters varied over the course of the study (Fig. 1a). The 12-hour time step model was the best model to explain changes in variance in oyster behavior (Table 3). Overall, this model explained a large proportion of the variance between oysters (R2 = 0.48). However, the fixed effects alone explain a much lower proportion (R2 = 0.07).
Rate of valve closure
The 12-hour time step model was the best model to explain how quickly oysters closed their valves (Table 4). Overall, this model explains the variability in closing rate well (Conditional R2 = 0.60).
Probability of an oyster being closed
The 12-hour time step model was the best model to explain the probability of an oyster being closed (Table 5). Overall, this model does a good job of explaining the probability of an oyster being closed (Tjur’s R2 = 0.56) and the fixed effects alone account for more than half of that probability (Tjur’s R2 = 0.50).
Environmental influence on oyster behavior
All the studied environmental variables influenced at least one aspect of valve opening behavior (Fig. 2a-c). The interaction between salinity and its rate of change had the largest effect on the variance among oysters’ behavior (Fig. 2a). Salinity also had the greatest influence on the rate of valve closure (Fig. 2b). Chlorophyll-a concentration was the variable that had the largest effect size on the probability of an oyster being closed (Fig. 2c). The effects of all the environmental variables on the three aspects of oyster behavior we examined were modified, to some extent, by interactions. Below we describe only the interactions that alter the general pattern between the environmental variable and oyster behavior. The significant interactions that modify the intensity of the relationship between the environmental variables and oyster behavior but do not change the general pattern of response (Fig. 2a-c, Tables S2-4) are not discussed in the following sections.
Effect size (β terms from the best-fit models) of single- and two-way interactions of environmental predictors (salinity (S), temperature (T), chlorophyll-a (Chl-a), their rates of change (Δ), and the rate of change of water depth (ΔDepth)) on the variance in oyster behavior ((% max/100)2; a), rate of valve closure (b), and probability of an oyster being closed (c). A positive effect size means that a higher value of the variable is associated with higher variability, more time spent closed, or a faster rate of closure. Error bars represent 95% confidence intervals. The color of each model term is scaled from smallest to largest effect size within each model
Chlorophyll-a concentration
When considered on its own, higher chlorophyll-a (chl-a) concentrations (~ 25 µg L–1) decreased variance in oysters’ behavior (β = −0.0032, 95% confidence interval (CI) = −0.0039 to −0.0026, t31000 = −9.20, p < 0.001). Only the interaction with the rate of change of water depth altered the direction of the relationship between the variance and chl-a concentration so that when water depth was decreasing (i.e., tide was ebbing) there was no relationship between chl-a concentration and variance in oysters’ behavior (β = −0.04, 95% CI = 0.02 to 0.06, t31000 = −15.00, p < 0.001; Fig. 3a). Oysters closed their valves more rapidly at higher chl-a concentrations than at lower concentrations (β = 0.11, 95% CI = 0.01 to 0.22, z = 2.05, p = 0.04). This relationship is modified by salinity so that at lower salinities (~ 8) there is no relationship between closure rate and chl-a concentrations, but at higher salinities (~ 20) there is a strong positive relationship with oysters closing faster in higher chl-a concentrations (β = 0.19, 95% CI = 0.08 to 0.3, z = 3.34, p < 0.001). Chl-a concentration had the largest effect of all variables on the probability of an oyster being closed with oysters being more likely to be closed when chl-a concentration was low (~ 4 µg L–1; β = −1.10, 95% CI = −1.20 to −0.90, z = −12.00, p < 0.001). This relationship was weaker at higher salinities (> 15; β = 0.96, 95% CI = 0.77 to 1.15, z = 9.80, p < 0.001; Fig. 3b) or when salinity was rapidly decreasing (β = −0.83, 95% CI = −1.02 to −0.64, z = −8.60, p < 0.001; Fig. 3c), as oysters were less likely to be closed at low chl-a concentrations under those conditions.
Rate of change of chlorophyll-a concentration
Rising chl-a concentration increased variance among oysters (β = 0.0023, 95% CI = 0.0017 to 0.0029, t31000 = 7.60, p < 0.001; Fig. 2a). Conversely, the rate of change in chl-a concentration did not significantly influence the rate of valve closure (β = 0.036, 95% CI = −0.069 to −0.142, z = 0.67, p = 0.5; Fig. 2b). While rising chl-a concentration increased the probability that an oyster would be closed (β = 0.39, 95% CI = 0.23 to 0.55, z = 4.80, p < 0.001; Fig. 2c), this relationship was dramatically reduced at high salinities (β = −0.42, 95% CI = −0.56 to −0.28, z = 6.00, p < 0.001) and reversed when the water temperature was cooler (β = 0.72, 95% CI = 0.57 to 0.88, z = 9.00, p < 0.001).
Modeled interactions between chlorophyll-a (chl-a) concentration and the rate of change of water depth on the variance in oysters’ behavior (a), and between chl-a concentration and salinity (b) and chl-a concentration and the rate of change of salinity (c) on the probability of an oyster being closed. Plotted values are conditional effects generated using the ggpredict function from the ggeffects package in R (Lüdecke 2018). PSU = practical salinity units. Shading represents the 95% confidence interval
Temperature
Similar to chl-a concentrations, warmer temperatures (~ 26 °C) decreased variance among oysters (β = −0.0013, 95% CI = −0.0025 to −0.0013, t31000 = −6.20, p < 0.001). However, several other environmental variables influenced this relationship (Table S2). When salinity was higher, temperature had no effect on variance (β = 0.0021, 95% CI = 0.0014 to 0.0028, t31000 = 6.00, p < 0.001; Fig. 4a) and when salinity was rapidly increasing, variance increased at warmer temperatures (β = 0.0055, 95% CI = 0.0048 to 0.0061, t31000 = 16.00, p < 0.001; Fig. 4b). Temperature alone had no effect on the rate of valve closure (β = −0.023, 95% CI = −0.105 to 0.059, z = −0.54, p = 0.54) but it did influence how salinity (Fig. 4c) and the rate of change of temperature (Fig. 4d) affect the rate of valve closure. At low temperatures (~ 14 °C), oysters closed more quickly in high salinities (> 20) than in low salinities (< 8), but at high temperatures (~ 26 °C), oysters closed at a similar rate, regardless of salinity (β = −0.17, 95% CI = −0.20 to −0.15, z = −13.00, p < 0.001; Fig. 4c). The interactive effect of the rate of change of temperature was small, but significant (β = 0.16, 95% CI = 0.27 to −0.06, z = − 3.6, p = 0.002). Oyster closure rate decreased with increasing temperature when water temperatures were steady or rising, but when water temperatures were falling valve closure rates were higher at higher temperatures (> 23 °C) (Fig. 4d). Warmer temperatures also decreased the probability that an oyster would be closed (β = −0.55, 95% CI = −0.70 to −0.41, z = −7.5, p < 0.001), but several other variables interacted with temperature to influence this pattern (Table S4). When salinity was low (< 5; Fig. 4e), or chl-a concentration was rapidly increasing (Fig. 4f), this pattern reversed, and the oysters were more likely to be closed at warmer temperatures. There was no relationship between water temperature and the probably that an oyster would be closed when temperatures were decreasing, or when salinity or water depth were increasing (Table S4).
Modeled interactions between temperature and salinity (a) and temperature and the rate of change of salinity (b) on the variance in oysters’ behavior, between temperature and salinity (c) and temperature and the rate of change of temperature (d) on the rate of valve closure, and between temperature and salinity (e) and temperature and the rate of change of chlorophyll-a concentration (f) on the probability of an oyster being closed. Plotted values are conditional effects generated using the ggpredict function from the ggeffects package in R (Lüdecke 2018). PSU = practical salinity units. Shading represents the 95% confidence interval
Rate of change of temperature
Rising temperatures increased variance among oysters (β = 0.0054, 95% CI 0.0049 to 0.0060, t31000 = −19.00, p < 0.001, Fig. 2a) and decreased the rate of valve closure (β = −0.15, 95% CI = −0.25 to −0.04, z = −2.77, p = 0.01; Fig. 2b). The effect of rising temperatures on the rate of valve closure was modified by temperature, as discussed in the previous section. The rate of change in water temperature also had no direct effect on the probability of an oyster being closed (Table S4), but the interaction between the rate of change in water temperature and temperature did (β = −0.28, 95% CI = −0.43 to −0.12, z = −3.50, p < 0.001). At lower temperatures, oysters were more likely to be closed when temperatures were rising rapidly; the reverse was true when temperatures were warmer.
Salinity
At higher salinities (~ 23 and above) variance in behavior was higher (β = 0.0037, 95% CI = 0.0028 to 0.0047, t31000 = 7.60, p < 0.001; Fig. 2a). The relationship between variance and salinity was significantly modified by the rate of change of salinity (β = 0.0065, 95% CI = 0.0057 to 0.0073, t31000 = 17.00, p < 0.001; Fig. 5a) so that when salinity was decreasing oyster VOB was less variable at higher salinities than at lower salinities (~ 4). This constituted the largest effect on inter-individual variability (Fig. 2a). The rates of change of water depth (β = −0.0044, 95% CI = −0.0050 to −0.0039, t31000 = −16.00, p < 0.001; Fig. 5b) and temperature (β = −0.0034, 95% CI = −0.0040 to −0.00285, t31000 = −12.00, p < 0.001; Fig. 5c) also interacted significantly with salinity. There was no relationship between variance and salinity when water depth or temperature were rising. Oysters closed faster at higher salinities (β = 0.33, 95% CI = 0.24 to 0.42, z = 2.74, p < 0.001; Fig. 2b) and this amounted to the largest effect on rate of valve closure. Finally, the probability of an oyster being closed was higher at higher salinities (Fig. 2c). This relationship was altered by chl-a concentration (β = 0.96, 95% CI = 0.77 to 1.15, z = 9.81, p < 0.001; Fig. 5d) as well as the rates of change of chl-a concentration (β = −0.42, 95% CI = −0.56 to −0.28, z = −6.02, p < 0.001; Fig. 5f) and salinity (β = 0.89, 95% CI = 0.71 to 1.08, z = 9.30, p < 0.001, Fig. 5e) in opposite ways. At higher salinities, high (> 12 µg L–1) or decreasing chl-a concentrations or rising salinities increased the probability that an oyster would be closed; at lower salinities, low (< 6 µg L–1) or rising chl-a concentrations or decreasing salinities increased the probability that an oyster would be closed.
Rate of change of salinity
The rate of change of salinity had no direct effect on variance among oysters (β = −0.00015, 95% CI = −0.00071 to 0.00041, t31000 = −0.52, p = 0.6; Fig. 2a) but decreased the rate of valve closure (β = −0.23, 95% CI = −0.35 to −0.11, z = −3.75, p < 0.001; Fig. 2b) and the probability that an oyster would be closed (β = −0.31, 95% CI = −0.46 to −0.16, z = −4.04, p < 0.001; Fig. 2c). The rate of change of salinity interacted significantly with other environmental variables to influence variance (Table S2), the rate of valve closure (Table S3), and the probability that an oyster would be closed (Table S4); all these interactions are described in the above sections.
Rate of change of water depth
Rising water depth increased variance among oysters (β = 0.0058, 95% CI = 0.0053 to 0.0063, t31000 = 22.00, p < 0.001; Fig. 2a) and the probability that an oyster would be closed (β = −0.70, 95% CI = −0.01 to −0.04, z = −4.80, p < 0.001; Fig. 2c), but had no effect on the rate of valve closure (Fig. 2b). The rate of change of water depth interacted significantly with other environmental variables to influence variance among oysters (Table S2), the rate of valve closure valve closure (Table S3), and the probability that an oyster would be closed (Table S4); all these interactions are described above.
Modeled interactions between salinity and its rate of change (a), salinity and the rate of change of water depth (b), and salinity and the rate of change of temperature (c) on the variance in oysters’ behavior, and between salinity and the chlorophyll-a (chl-a) concentration (d), salinity and its rate of change (e) and salinity and the rate of change of chl-a concentration (f) on the probability of an oyster being closed. Plotted values are conditional effects generated using the ggpredict function from the ggeffects package in R (Lüdecke 2018). PSU = practical salinity units. Shading represents the 95% confidence interval
Discussion
In this study, we present the first quantitative evaluation of the effects of multiple environmental variables on the VOB of eastern oysters under typical estuarine conditions in Louisiana, United States. The simultaneous and continuous monitoring of VOB and temperature, salinity, chlorophyll-a (chl-a) concentration, DO concentration, and the rate of change in these variables and in water depth allowed the characterization of the complexity of the response of oysters to fluctuations in surrounding environmental conditions. As VOB directly influences basic physiological functions such as respiration and feeding in bivalves, which in turn determine growth and reproduction success (Payton et al. 2017; Casas et al. 2018a; Tonk et al. 2023), our findings provide valuable insight into the interpretation of physiological data, the improvement of bioenergetic model simulations, and the management of fisheries and restoration initiatives.
Each of the four environmental variables examined and their rate of change influenced oyster VOB separately, but several significant two-way interactions between variables were found to influence the direction or the magnitude of these relationships. This highlights the complexity of the response of oysters to environmental factors, particularly as scientists in past decades have focused on relating VOB to single environmental conditions (Clements et al. 2018; Lassoued et al. 2021; Tran et al. 2010; Lavaud et al. 2021; Kramer and Foekema 2001; Hartmann et al., 2016; Chambon et al. 2007; Charifi et al. 2017; Hubert et al., 2023). Our study encompassed close to two months of continuous recording, corresponding to 4,608 data points for environmental factors and more than 4 million gape angle values for each oyster (averaged each 15 min to match the resolution of the environmental data). No extreme or known thresholds that represent conditions outside of the eastern oyster’s tolerance range where mortality is expected were observed: temperature remained well below 30 °C, salinity never fell below 3.0, and chl-a concentration never decreased below 3.7 µg L–1 (Table 1). As such, these results are good indicators of conditions in which oysters are known to thrive in Louisiana. Moreover, despite being somewhat restricted in time, our monitoring captured the overall range and variability in average environmental conditions encountered throughout a year (Lowe et al., 2017).
Among the three VOB metrics analyzed (variance in oysters’ behavior, rate of valve closure, and probability of an oyster being closed), variance in oysters’ behavior was the least affected by environmental variation as shown by the lowest range of effect sizes on this metric (Fig. 2). This observation is noteworthy considering the relatively small sample size (n = 8). Synchronism in valve opening in eastern oysters has previously been reported in relation to changes in temperature and light intensity (Comeau et al. 2012). Variance in oysters’ behavior did increase when environmental variables tended toward physiologically stressful conditions. The interaction between salinity and its rate of change (Fig. 5a) had the largest effect on variance in oysters’ behavior. Variation between individuals was more important at high salinity when salinity was increasing and at low salinity when salinity was decreasing. Conversely, low variance was observed at low salinity when salinity was increasing and at high salinity when salinity was decreasing. This pattern can be explained by the fact that the differences between the physiological capacity of individual oysters are likely to be revealed under conditions at the extremes of the species tolerance range. This is also evidenced by previous studies describing population differences in tolerance to various salinities in the northern Gulf of Mexico (Marshall et al. 2021b; Swam et al. 2022).
Salinity was also, by far, the main factor influencing the rate of valve closure in our study (Fig. 2b). This result underscores the role of salinity as a major driver of oyster ecophysiology even within a range of typical spring salinity conditions in southeastern Louisiana estuaries in which oysters generally thrive (3.2–23.9; Table 1; Fig. 1; Lowe et al., 2017). Salinity is a well-documented driver of the ecological and physiological responses of eastern oysters (Loosanoff 1953; Shumway 1996; Casas et al. 2018a; Marshall et al. 2021b; Swam et al. 2022), and field studies have indicated that low salinity events (< 5) may cause mortality events in Louisiana or adjacent waters (La Peyre et al., 2013; Gledhill et al. 2020). In our study, oysters were less likely to be closed at lower salinities (< 5) than at higher salinities (> 17). This observation contradicts the expectation that oysters would close when exposed to lower salinity. Moreover, as salinity remained above 3.2 and did not change abruptly over the course of this monitoring (which may not be outside the oyster’s tolerance range), oysters may have acclimated to such gradual changes. This aligns with observations by Marshall et al. (2021b), who recorded no mortality from oysters gradually exposed to a salinity of 2. Slower rates of valve closure were also measured at low salinities, which could reflect negative effects of low salinity on cellular metabolism (through disruption of intracellular ion and acid base regulation), as was reported for gill ciliary activity (van Winkle 1972) and clearance rates (Casas et al. 2018a) at the same salinity (5). Additionally, the rate of closure may have been affected indirectly by salinity through the presence of predators around the cages. Oyster predators include black drums, mud and blue crabs, and shell drilling snails, which are usually more abundant at higher salinity (White and Wilson 1996; Brown and Richardson 1988; Brown et al. 2008).
Food availability is usually considered not limiting for oysters in southeast Louisiana given elevated phytoplankton concentrations (D’Sa 2014; Turner et al. 2019). Oysters closed their valves more rapidly at higher chl-a concentrations, possibly to unclog their gills. Despite high concentrations (mean of 10 µg L–1 ± 3.7 sd), lower chl-a concentrations increased the probability of an oyster being closed (~ 4 µg L–1; Fig. 2c). Most bivalves typically close their valves or drastically reduce their gaping amplitude to decrease clearance rates at low chl-a concentrations (Strohmeier et al. 2009; Comeau et al. 2012; Tonk et al. 2023) to conserve energy. Furthermore, the interaction between chl-a concentration and salinity (Fig. 5d) also had a large effect on the probability of an oyster being closed (Fig. 2c). Oysters remained open at low salinity when chl-a concentration was high but not at low chl-a concentrations. This effect on VOB could be mechanistically linked to the energetic physiology of oysters through a trade-off between being open and feeding (accumulating energy) versus being closed to avoid osmotic stress and fast (depleting energy). As osmoconformers, salinity variations trigger a physiological response involving the transport or synthesis of amino acids and ions, which may incur high energy expenditure (although no clear quantitative data exist to our knowledge). Oysters generally close their valves for these physiological processes to take place gradually (Hand and Stickle 1977; McFarland et al. 2013). During these closing phases, no feeding occurs, which was identified as the main effect of low salinity on oysters’ energy budgets (Lavaud et al. 2017). So, as salinity changes, valve closure could be controlled by the energetic status of oysters, which could determine whether they remain open to fuel the energetic demand from osmoconforming or close and rely on existing reserves. A similar mechanism was hypothesized in a bioenergetic model to account for the impact of salinity on the energy budget of oysters in the region (Lavaud et al. 2017). Our results confirm this assumption. The probability of an oyster being closed even seemed to increase at median chl-a concentration (10%; Fig. 5d), suggesting a threshold in the balance between energy uptake and consumption despite the high food availability in these estuarine conditions. Adding support to this hypothesis is the fact that clearance rates are higher at high salinity (above 6–9; Casas et al. 2018a); as oysters trap more food (and non-food) particles through their gills, they may need to close to process and ingest large amounts of food at higher chl-a concentration. Recently, Ledoux et al. (2023) measured glycogen content along with the gaping response of mussels exposed to acoustic stress, but neither acute nor longer-term correlations were found. Further investigations of bivalve’s responses to salinity could provide valuable insights into the suggested links between VOB and the energetic physiology of oysters.
In the analysis, we also explored the potential relationships between the measured environmental variables and the probability of oysters opening, but most monitored variables had little effect. This can be expected as the closing of valves secludes the animal from the surrounding water, making the organism unable to assess any change in environmental conditions. Some authors mentioned that once closed, bivalves may “test the water” before re-opening or re-open slowly (Kramer and Foekema 2001; Tran et al. 2010). The individual VOB dynamics in the present study clearly showed that oysters re-opened quickly (between consecutive 15-min intervals) and at wide angles when doing so (Figure S1), indicating that the animals did not slightly open their valves to evaluate environmental conditions before opening again. The main difference between our study and the previous work mentioned above is that we conducted our investigations under conditions thought to be within the physiological tolerances of the animals. Exposure to harsher conditions, such as toxic algae blooms, hurricanes or freshwater discharge from river diversions, may produce different results. Nevertheless, a closed oyster may well perceive thermal variations, as shell valves do not act as thermal barriers. This may also explain why the interactive effect of temperature and water depth had the strongest effect on the probability of opening. In another study on eastern oysters in Canada, Comeau et al. (2012) reported a correlation between temperature and valve re-opening after a long period of ‘quiescence’ over the winter. After being either closed or slightly open during the quiescent phase, the oysters abruptly awakened and opened to maximum angles when temperature rose. Further investigations, possibly including measurements of anaerobic metabolic products (e.g., alanine and succinate concentrations), could help to better understand the factors leading to the re-opening of oysters following closure.
Because DO concentration and water depth were correlated with other variables analyzed, we did not investigate their role on oyster VOB in our analysis (but the role of the rate of change in water depth on oyster VOB was investigated, see Sect. 2.3.1). Like chl-a concentration, DO concentration did not reach any known threshold impacting oyster physiology, typically assumed around 2 mg L–1 (Porter and Breitburg 2016; Coxe et al., 2023). However, this threshold is known to vary with temperature, activity level, and exposure time (Deutsch et al., 2020; Seibel et al., 2021). Combining VOB and metabolic rate measurements could provide valuable insight into the effect of DO concentrations on VOB, particularly as increases in temperatures, hypoxic events, and freshwater runoff from river management are expected in the future (Rabalais and Turner 2019). The water depth in Louisiana estuaries is strongly impacted by winds and weather systems, which is illustrated by the broken cyclical pattern in this variable (Fig. 1) and could explain why we did not observe a clear tidal pattern in VOB. The monitoring of spawning activity could also provide valuable insight to interpret VOB (Payton et al. 2017), although such analysis is usually destructive. More studies of the VOB of oysters, and generally sessile bivalves, in relation with variations in environmental conditions could help to better identify thresholds (possibly population or geographically specific) in the physiological tolerance range of organisms triggering a behavioral response. Extreme or prolonged events affecting oyster off-bottom aquaculture could be managed through real-time warning systems as is being done with other bivalve species that trigger pollution alerts (Vereycken and Aldridge 2023). This knowledge can also be used as an input for bioenergetics models as VOB directly influences energy acquisition (Lavaud et al. 2017, 2021). As we aim to understand and explain the complex effects of environmental variables and their interactions on oyster VOB and ultimately on their life traits, such a mechanistic approach could be a valuable tool to integrate this complexity.
Data availability
Data collected during this study may be shared upon reasonable request.
References
Beseres Pollack JB, Kim HC, Morgan EK, Montagna PA (2011) Role of flood disturbance in natural oyster (Crassostrea virginica) population maintenance in an estuary in South Texas, USA. Estuaries Coasts 34:187–197. https://doi.org/10.1007/s12237-010-9338-6
Bodenstein S, Callam BR, Walton WC, Rikard FS, Tiersch TR, La Peyre JF (2023) Survival and growth of triploid eastern oysters, Crassostrea virginica, produced from wild diploids collected from low-salinity areas. Aquaculture 564:739032. https://doi.org/10.1016/j.aquaculture.2022.739032
Brooks ME, Kristensen K, Van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9(2):378–400. https://doi.org/10.3929/ethz-b-000240890
Brown KM, George GJ, Peterson GW, Thompson BA, Cowan JH (2008) Oyster predation by black drum varies spatially and seasonally. Estuaries Coasts 31:597–604. https://doi.org/10.1007/s12237-008-9045-8
Brown KM, Richardson TD (1988) Foraging ecology of the southern oyster drill Thais haemastoma (Gray): constraints on prey choice. J Exp Mar Biol Ecol 114(2–3):123–141. https://doi.org/10.1016/0022-0981(88)90133-5
Casas SM, Filgueira R, Lavaud R, Comeau LA, La Peyre MK, La Peyre JF (2018b) Combined effects of temperature and salinity on the physiology of two geographically-distant eastern oyster populations. J Exp Mar Biol Ecol 506:82–90. https://doi.org/10.1016/j.jembe.2018.06.001
Casas SM, Lavaud R, La Peyre MK, Comeau LA, Filgueira R, La Peyre JF (2018a) Quantifying salinity and season effects on eastern oyster clearance and oxygen consumption rates. Mar Biol 165:1–13. https://doi.org/10.1007/s00227-018-3351-x
Chambon C, Legeay A, Durrieu G, Gonzalez P, Ciret P, Massabuau JC (2007) Influence of the parasite worm polydora sp. on the behaviour of the oyster Crassostrea gigas: a study of the respiratory impact and associated oxidative stress. Mar Biol 152(2):329–338. https://doi.org/10.1007/s00227-007-0693-1
Charifi M, Sow M, Ciret P, Benomar S, Massabuau JC (2017) The sense of hearing in the Pacific oyster, Magallana Gigas. PLoS ONE 12(10):e0185353. https://doi.org/10.1371/journal.pone.0185353
Clements JC, Comeau LA, Carver CE, Mayrand É, Plante S, Mallet AL (2018) Short-term exposure to elevated pCO2 does not affect the valve gaping response of adult eastern oysters, Crassostrea virginica, to acute heat shock under an ad libitum feeding regime. J Exp Mar Biol Ecol 506:9–17. https://doi.org/10.1016/j.jembe.2018.05.005
Cloern JE, Abreu PC, Carstensen J, Chauvaud L, Elmgren R, Grall J, Greening H, Johansson JOR, Kahru M, Sherwood ET, Xu J (2016) Human activities and climate variability drive fast-paced change across the world’s estuarine–coastal ecosystems. Glob Change Biol 22(2):513–529. https://doi.org/10.1111/gcb.13059
Coffin MR, Clements JC, Comeau LA, Guyondet T, Maillet M, Steeves L, Winterburn K, Babarro JM, Mallet MA, Haché R, Poirier LA (2021) The killer within: endogenous bacteria accelerate oyster mortality during sustained anoxia. Limnol Oceanogr 66(7):2885–2900. https://doi.org/10.1002/lno.11798
Comeau LA, Mayrand E, Mallet A (2012) Winter quiescence and spring awakening of the Eastern oyster Crassostrea virginica at its northernmost distribution limit. Mar Biol 159(10):2269–2279. https://doi.org/10.1007/s00227-012-2012-8
Coxe N, Casas SM, Marshall DA, La Peyre MK, Kelly MW, La Peyre JF (2023) Differential hypoxia tolerance of eastern oysters from the northern Gulf of Mexico at elevated temperature. J Exp Mar Biol Ecol 559:151840. https://doi.org/10.1016/j.jembe.2022.151840
Deutsch C, Penn JL, Seibel B (2020) Metabolic trait diversity shapes marine biogeography. Nature 585(7826):557–562. https://doi.org/10.1038/s41586-020-2721-y
D’Sa EJ (2014) Assessment of chlorophyll variability along the Louisiana coast using multi-satellite data. GIScience Remote Sens 51(2):139–157. https://doi.org/10.1080/15481603.2014.895578
Gledhill JH, Barnett AF, Slattery M, Willett KL, Easson GL, Otts SS, Gochfeld DJ (2020) Mass mortality of the Eastern Oyster Crassostrea virginica in the western Mississippi sound following unprecedented Mississippi River flooding in 2019. J Shellfish Res 39(2):235–244. https://doi.org/10.2983/035.039.0205
Hallett LM, Hsu JS, Cleland EE, Collins SL, Dickson TL, Farrer EC, Gherardi LA, Gross KL, Hobbs RJ, Turnbull L, Suding KN (2014) Biotic mechanisms of community stability shift along a precipitation gradient. Ecology 95(6):1693–1700. https://doi.org/10.1890/13-0895.1
Hallett LM, Jones SK, MacDonald AAM, Jones MB, Flynn DF, Ripplinger J, Slaughter P, Gries C, Collins SL (2016) Codyn: an R package of community dynamics metrics. Methods Ecol Evol 7(10):1146–1151. https://doi.org/10.1111/2041-210X.12569
Hand SC, Stickle WB (1977) Effects of tidal fluctuations of salinity on pericardial fluid composition of the American oyster Crassostrea virginica. Mar Biol 42:259–271. https://doi.org/10.1007/BF00397750
Hartig F (2020) DHARMa: residual diagnostics for hierarchical (multi-level/mixed) regression models. R Package Version 0.3, 3(5)
Hartmann JT, Beggel S, Auerswald K, Stoeckle BC, Geist J (2016) Establishing mussel behavior as a biomarker in ecotoxicology. Aquat Toxicol 170:279–288. https://doi.org/10.1016/j.aquatox.2015.06.014
Higgins PJ (1980) Effects of food availability on the valve movements and feeding behavior of juvenile Crassostrea virginica (Gmelin). I. Valve movements and periodic activity. J Exp Mar Biol Ecol 45(2):229–244. https://doi.org/10.1016/0022-0981(80)90060-X
Hubert J, van der Burg AD, Witbaard R, Slabbekoorn H (2023) Separate and combined effects of boat noise and a live crab predator on mussel valve gape behavior. Behav Ecol 34(3):495–505. https://doi.org/10.1093/beheco/arad012
Kramer KJ, Foekema EM (2001) The Musselmonitor® as Biological Early Warning System. In: Butterworth FM, Gonsebatt ME, Gunatilaka A (eds) Biomonitors and biomarkers as indicators of environmental change: a handbook, vol 2. Springer, Boston, MA, pp 59–87. https://doi.org/10.1007/978-1-4615-1305-6_4
La Peyre MK, Eberline BS, Soniat TM, La Peyre JF (2013) Differences in extreme low salinity timing and duration differentially affect eastern oyster (Crassostrea virginica) size class growth and mortality in Breton Sound, LA. Estuar Coast Shelf Sci 135:146–157. https://doi.org/10.1016/j.ecss.2013.10.001
Lassoued J, Padín XA, Comeau LA, Bejaoui N, Pérez FF, Babarro JM (2021) The Mediterranean mussel Mytilus galloprovincialis: responses to climate change scenarios as a function of the original habitat. Conserv Physiol 9(1):coaa114. https://doi.org/10.1093/conphys/coaa114
Lavaud R, Durier G, Nadalini JB, Filgueira R, Comeau LA, Babarro JM, Michaud S, Scarratt M, Tremblay R (2021) Effects of the toxic dinoflagellate Alexandrium catenella on the behaviour and physiology of the blue mussel Mytilus edulis. Harmful Algae 108:102097. https://doi.org/10.1016/j.hal.2021.102097
Lavaud R, La Peyre MK, Casas SM, Bacher C, La Peyre JF (2017) Integrating the effects of salinity on the physiology of the eastern oyster, Crassostrea virginica, in the northern Gulf of Mexico through a dynamic Energy Budget model. Ecol Model 363:221–233. https://doi.org/10.1016/j.ecolmodel.2017.09.003
Lüdecke D (2018) Ggeffects: tidy data frames of marginal effects from regression models. J Open Source Softw 3(26):772. https://doi.org/10.21105/joss.00772
Lüdecke D, Ben-Shachar MS, Patil I, Waggoner P, Makowski D (2021) Performance: an R package for assessment, comparison and testing of statistical models. J Open Source Softw 6(60):3139. https://doi.org/10.21105/joss.03139
Ledoux T, Clements JC, Comeau LA, Cervello G, Tremblay R, Olivier F, Chauvaud L, Bernier RY, Lamarre SG (2023) Effects of anthropogenic sounds on the behavior and physiology of the Eastern oyster (Crassostrea virginica). Front Mar Sci 10:1104526. https://doi.org/10.3389/fmars.2023.1104526
Loosanoff VL (1953) Behavior of oysters in water of low salinities. Proc Nat Shellfisheries Association 43:135–151
Lowe MR, Sehlinger T, Soniat TM, La Peyre MK (2017) Interactive effects of water temperature and salinity on growth and mortality of eastern oysters, Crassostrea virginica: a meta-analysis using 40 years of monitoring data. J Shellfish Res 36(3):683–697. https://doi.org/10.2983/035.036.0318
Marshall DA, Casas SM, Walton WC, Rikard FS, Palmer TA, Breaux N, La Peyre MK, Beseres Pollack J, Kelly MW, La Peyre JF (2021b) Divergence in salinity tolerance of northern Gulf of Mexico eastern oysters under field and laboratory exposure. Conserv Physiol 9(1):coab065. https://doi.org/10.1093/conphys/coab065
Marshall DA, Coxe NC, La Peyre MK, Walton WC, Rikard FS, Beseres Pollack J, Kelly MW, La Peyre JF (2021a) Tolerance of northern Gulf of Mexico eastern oysters to chronic warming at extreme salinities. J Therm Biol 100:103072. https://doi.org/10.1016/j.jtherbio.2021.103072
McFarland K, Donaghy L, Volety AK (2013) Effect of acute salinity changes on hemolymph osmolality and clearance rate of the non-native mussel, Perna Viridis, and the native oyster, Crassostrea virginica. Southwest Fla Aquat Invasions 8(3):299–310. https://doi.org/10.3391/ai.2013.8.3.06
Sow M, Durrieu G, Briollais L, Ciret P, Massabuau JC (2011) Environ Monit Assess 182(1):155–170. https://doi.org/10.1007/s10661-010-1866-9. Water quality assessment by means of HFNI valvometry and high-frequency data modeling
Nagai K, Honjo T, Go J, Yamashita H, Oh SJ (2006) Detecting the shellfish killer Heterocapsa Circularisquama (Dinophyceae) by measuring bivalve valve activity with a Hall element sensor. Aquaculture 255(1–4):395–401. https://doi.org/10.1016/j.aquaculture.2005.12.018
Nelson TC (1922) Report of the Department of Biology. Annual Report of the New Jersey State Agricultural Experiment Station for the year ending June 30, 1920, 42:289
Payton L, Sow M, Massabuau JC, Ciret P, Tran D (2017) How annual course of photoperiod shapes seasonal behavior of diploid and triploid oysters, Crassostrea gigas. PLoS ONE 12(10):e0185918. https://doi.org/10.1371/journal.pone.0185918
Porter ET, Breitburg DL (2016) Eastern oyster, Crassostrea virginica, valve gape behavior under diel-cycling hypoxia. Mar Biol 163(10):1–12. https://doi.org/10.1007/s00227-016-2980-1
Rabalais NN, Turner RE (2019) Gulf of Mexico hypoxia: past, present, and future. Limnol Oceanogr Bull 28(4):117–124. https://doi.org/10.1002/lob.10351
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Schluter D (1984) A variance test for detecting species associations, with some example applications. Ecology 65(3):998–1005. https://doi.org/10.2307/1938071
Seibel BA, Andres A, Birk MA, Burns AL, Shaw CT, Timpe AW, Welsh CJ (2021) Oxygen supply capacity breathes new life into critical oxygen partial pressure (P crit). J Exp Biol 224(8):jeb242210. https://doi.org/10.1242/jeb.242210
Shumway SE (1977a) Effect of salinity fluctuation on the osmotic pressure and Na+, Ca2+ and Mg2+ ion concentrations in the hemolymph of bivalve molluscs. Mar Biol 41(2):153–177. https://doi.org/10.1007/BF00394023
Shumway SE (1977b) The effect of fluctuating salinity on the tissue water content of eight species of bivalve molluscs. J Comp Physiol 116(3):269–285. https://doi.org/10.1007/BF00689036
Shumway SE (1996) Natural environmental factors. In: Kennedy VS, Newell RI, Eble AE (eds) The eastern oyster Crassostrea virginica, vol 467. Maryland Sea Grant, College Park, p 513
Shumway SE, Koehn RK (1982) Oxygen consumption in the American oyster Crassostrea virginica. Mar Ecol Prog Ser 9(1):59–68. https://doi.org/10.3354/meps009059
Stickle WB, Kapper MA, Liu LL, Gnaiger E, Wang SY (1989) Metabolic adaptations of several species of crustaceans and molluscs to hypoxia: tolerance and microcalorimetric studies. Biol Bull 177(2):303–312. https://doi.org/10.2307/1541945
Strohmeier T, Strand Ø, Cranford P (2009) Clearance rates of the great scallop (Pecten maximus) and blue mussel (Mytilus edulis) at low natural seston concentrations. Mar Biol 156:1781–1795. https://doi.org/10.1007/s00227-009-1212-3
Swam LM, Couvillion B, Callam B, La Peyre JF, La Peyre MK (2022) Defining oyster resource zones across coastal Louisiana for restoration and aquaculture. Ocean Coastal Manage 225:106178. https://doi.org/10.1016/j.ocecoaman.2022.106178
Tonk L, Witbaard R, van Dalen P, Cheng CH, Kamermans P (2023) Applicability of the gape monitor to study flat oyster (Ostrea edulis) feeding behaviour. Aquat Living Resour 36:6. https://doi.org/10.1051/alr/2022021
Tran D, Haberkorn H, Soudant P, Ciret P, Massabuau JC (2010) Behavioral responses of Crassostrea gigas exposed to the harmful algae Alexandrium minutum. Aquaculture 298(3–4):338–345. https://doi.org/10.1016/j.aquaculture.2009.10.030
Tran D, Nadau A, Durrieu G, Ciret P, Parisot JP, Massabuau JC (2011) Field chronobiology of a molluscan bivalve: how the moon and sun cycles interact to drive oyster activity rhythms. Chronobiol Int 28(4):307–317. https://doi.org/10.3109/07420528.2011.565897
Turner RE, Swenson EM, Milan CS, Lee JM (2019) Spatial variations in chlorophyll a, C, N, and P in a Louisiana estuary from 1994 to 2016. Hydrobiologia 834:131–144. https://doi.org/10.1007/s10750-019-3918-7
Van Winkle W Jr (1972) Ciliary activity and oxygen consumption of excised bivalve gill tissue. Comp Biochem Physiol Part A: Physiol 42(2):473–485. https://doi.org/10.1016/0300-9629(72)90126-0
Vereycken JE, Aldridge DC (2023) Bivalve molluscs as biosensors of water quality: state of the art and future directions. Hydrobiologia 850:231–256. https://doi.org/10.1007/s10750-022-05057-7
White ME, Wilson EA (1996) Predators, pests, and competitors. In: Kennedy VS, Newell RI, Eble AE (eds) The eastern oyster Crassostrea virginica. Maryland Sea Grant, College Park, pp 559–580
Wilson R, Reuter P, Wahl M (2005) Muscling in on mussels: new insights into bivalve behaviour using vertebrate remote-sensing technology. Mar Biol 147(5):1165–1172. https://doi.org/10.1007/s00227-005-0021-6
Acknowledgements
We would like to thank Kim de Mutsert, two anonymous reviewers and an external reviewer for their comments. The authors thank Amanda Fontenot for providing verified environmental monitoring data. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Funding
Funding for this study included LA Sea Grant, the U.S. Geological Survey South Central Climate Adaptation Science Center, the Louisiana Department of Wildlife and Fisheries, the U.S. Geological Survey, LUMCON and the Louisiana Fish and Wildlife Cooperative Research Unit.
Author information
Authors and Affiliations
Contributions
Funding for the study was acquired by Stephanie Archer, Megan La Peyre, and Jerome La Peyre. Study conceptualization and methodology were performed by Romain Lavaud, Stephanie Archer, Megan La Peyre, Sandra Casas, and Jerome La Peyre. Formal analysis, data curation, visualization, and original draft writing were conducted by Romain Lavaud and Stephanie Archer. Software and resources were provided by Stephanie Archer. All authors contributed to the investigation and manuscript review and editing.
Corresponding author
Ethics declarations
Competing interests
We declare no conflict of interest.
Animal welfare
No ethics approval was needed for this study.
Additional information
Communicated by P. Ramey-Balci.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lavaud, R., Archer, S.K., La Peyre, M.K. et al. In-situ valve opening response of eastern oysters to estuarine conditions. Mar Biol 171, 174 (2024). https://doi.org/10.1007/s00227-024-04488-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04488-1