Abstract
Reef manta rays (Mobula alfredi) face threats from human exploitation and activity in several regions of the world and crucial information on the ecology of the species is needed. The species is observed at several sites in all parts of the archipelago of New Caledonia where anthropogenic influence is presumed to be minimal. This study is the first to investigate the population of New Caledonia and focuses on its characteristics and its habitat use. Photographs of reef manta rays were collected directly from the authors (14.4%) between 2017 and 2020 and gathered from recreational divers and snorkelers (85.6%) from 11 sites around New Caledonia. The authors used the unique ventral coloration patterns of the manta rays that were clearly identifiable from 1741 of these photographs to identify 391 individuals and record their physical characteristics (sex, injuries, and colour morph) and resighting rates. These results highlight the widespread distribution of the species in the archipelago (11 sites) with little connectivity between all aggregations sites, with only 5.4% of the individuals observed at more than one site. Strong and long-term site fidelity was recorded at all studied sites through re-sighting rates (52.2% overall) and residency analysis. The population also records the world highest known proportion of melanistic manta rays (43%) to date, and a balanced male: female ratio (1.0:1.15). The analysis of injuries reported that 44.8% of all reef manta rays identified (n = 391) had noticeable wounds or injuries with no significant difference in the proportions of males and females injured. From these injured individuals only 9.8% of injuries judged to be of anthropogenic origin and 29.7% from attempted predation. Our study complements and correlates previous findings on this population that revealed strong site fidelity and low connectivity. The different characteristics of this population of reef manta rays also emphasize that concerns about species in New Caledonia are limited and that such favourable context needs to be preserved as reference for conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The distribution patterns and habitat use of highly mobile marine species can be difficult to predict and understand. These animals are able to move and have the potential to disperse over large distances, although many use smaller home ranges than their dispersal capabilities (Mannocci et al. 2017). In the marine environment, the importance of ecological and environmental factors that limit a given species’ dispersal and result instead in long-term site fidelity patterns often remain unclear. For instance, habitat heterogeneity or topography (e.g. currents or deep-water channels) as well as habitat choice based on food resources or reproductive ecology can be the drivers of a fragmented population (Palumbi 1994; Bowen et al. 2016). The characterisation of a population range is crucial to identify effective management units for conservation purposes, especially for threatened species (Hueter et al. 2004).
Reef manta rays Mobula alfredi (Krefft 1868) are large, primarily coastal planktivores that are distributed in equatorial and tropical waters in the Indo-Pacific region (Couturier et al. 2012). Similar to many other elasmobranchs, reef manta rays have conservative life history traits: low fecundity, slow maturity and high longevity, which make them particularly sensitive to overexploitation (Couturier et al. 2012; Stevens 2016). Since 2011, the reef manta rays have been listed as vulnerable to extinction on the IUCN Red List (Marshall et al. 2022) due to over-harvesting pressure as a result of the high commercial value of their gill plates for use in non-traditional Chinese medicine (O’Malley et al. 2017). In response to important declines of manta ray populations around the world and the important lack of knowledge on the species (Ward-Paige et al. 2010), the number of studies on the ecology and behaviour of the species has increased considerably over the past decade (Stewart et al. 2018). A major part of previous research efforts focused on identifying population characteristics and structure, as well as movements and habitat use patterns (Stewart et al. 2018). These studies have shown that reef manta rays are highly mobile species capable of movements over a thousand of kilometres along a continuous coastline (up to 1150 km, Armstrong et al. 2019) and hundreds of kilometres between island chains (up to 450 km, Germanov & Marshall 2014). Large-scale movements seem to be mainly conditioned by variations in food resources (Sleeman et al. 2007; Dewar et al. 2008; Anderson et al. 2011a, b; Jaine et al. 2012; Couturier et al. 2018; Setyawan et al. 2018). On the contrary, when sufficient foraging opportunities persist or are seasonal within an area, reef manta rays tend to remain in or return to this area over extended periods of time, which is described as site fidelity or affinity. ‘Site fidelity’ is defined by Chapman et al. (2015) as the return of an individual to a location where it previously resided after an absence as long as or longer than the residency period. In the case of potential regular movements within a large home range including many sites, Couturier et al. 2011 applied the term ‘site affinity’. For example, strong evidence of site fidelity was observed in Hawaii (Deakos et al. 2011; Whitney et al. 2023), Mozambique (Marshall 2008), the Seychelles (Peel et al. 2020), Indonesia (Setyawan et al. 2020), French Polynesia (Carpentier et al. 2019) and eastern Australia (Couturier et al. 2011). In these studies, individuals were re-sighted over decadal periods, confirming long-term, and potentially life-long use of aggregation sites (e.g. Clark 2010; Setyawan et al. 2020; Carpentier et al. 2019). In New Caledonia, satellite telemetry also demonstrated constrained movements of the tagged individuals near the aggregation site of origin (Lassauce et al. 2023). This observation was based on relatively short deployment duration (approximatively 77 days), thus multiple years analyses of site fidelity and residency are necessary to fully understand these patterns.
The movements and habitat use patterns of reef manta ray populations have been studied using different methods (or a combination of them), including photographic mark-recapture methods, acoustic and satellite telemetry, or genomic techniques (Stewart et al. 2018). Most studies on manta rays use photo-identification (photo-ID) to describe population characteristics (e.g. Deakos et al. 2011; Carpentier et al. 2019; Setyawan et al. 2020), assess connectivity (e.g. Beale et al. 2019; Germanov et al. 2019, Knochel et al. 2022) and estimate population size (e.g. Marshall et al. 2011; Harty et al. 2022; Setyawan et al. 2022). The cataloguing of individuals was made possible through the identification of each animal based on the variation in the ventral body pigmentation patterns (Marshall et al. 2011). Photo-identification offers an inexpensive, minimally invasive, and widely accessible method to build large databases of individuals and conduct long-term monitoring of these populations. Ocean exploring tourists or local enthusiasts (“citizen scientists”) have been informed on how to take research-grade photographs that can be used by experts to examine individual markings, compare to our national photo-identifications database, and either add new or identify already known individuals. For all identification, such photographs also enable the monitoring of characteristics such as injuries or predation marks, maturity stages and the colormorph. Such submission of photo-identifications by citizen scientists can enable the collection of large quantities of data over extended geographic scales, while also ensuring longevity of the monitoring program. This citizen science approach has proven highly effective for the study of manta rays because the species is attractive for recreational divers, benefiting researchers with important contributions to their study (e.g. Germanov et al. 2019).
New Caledonia is an archipelago located in the southern Pacific region where reef manta rays are sighted year-round at many sites (Lassauce, pers. comm.). In New Caledonia, fisheries effort is low (harvesting on average 0.26t/km2/year) in comparison to other Pacific islands, manta rays are not fished, and tourism development is still relatively insignificant (Guillemot et al. 2009). Anthropogenic influence on manta rays such as accidental capture or catches in ghost nets is presumed to be minimal. In fact, there is no records of neither bycatch nor observations of entangled or stranded manta rays. Nonetheless, manta rays are locally only protected in the exclusive economic zone since 2013 (GNC 2013) but under no current legal protection within coastal waters (10 nmi from the barrier reef). To date, the few published studies on the reef manta rays of New Caledonia reveal unprecedented deep diving behavior for reef manta rays using satellite telemetry at all studied sites in New Caledonia, potentially indicating the presence of important food resources in deeper (300–700 m depth) waters (Lassauce et al. 2020). Satellite telemetry also described horizontal movements where reef manta rays presented a strong site fidelity and an important affinity for coastal waters (Lassauce et al. 2023). Moreover, genetic analysis has revealed small scale differentiation between studied sites within New Caledonia waters, suggesting limited connectivity and high site fidelity causing detectable fine-scale genetic structure (Lassauce et al. 2022). Nonetheless, the population of reef manta rays of New Caledonia has never been described. Information on the population size, sex ratio, incidence of melanism, injury and predation rates remain unknown to date. These characteristics are necessary as baseline for monitoring and establish effective management strategies. For instance, investigating melanism add more perspective into the global documentation and may contribute to a better understanding of the underlying evolutionary origin of this trait. Similarly, revealing the proportion and the origin of injuries in a population help identifying key areas where conservation measures are required to ensure more effective protection. Finally, high site fidelity and limited connectivity were demonstrated through satellite telemetry and genetics in New Caledonia, yet long-term analysis of these trends are lacking, and the use of photo-identification will complete and help verify these findings over several years.
Our study aimed to provide a first insight into the population size, sex ratio, incidence of melanism, injury and predation rates, as well as patterns of connectivity, site fidelity and residency of reef manta rays of New Caledonia. We analysed decades of compiled photo-identifications data collected through both scientific surveys and citizen scientists photographs from known aggregation sites around the archipelago.
Methods
Study sites
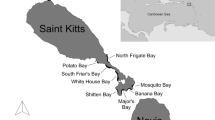
The archipelago of New Caledonia is divided into a so-called Main Island surrounded by the second largest barrier reef in the world (1660 km in length) and islands in the south (Isle of Pines), east (Loyalty Islands) and north (Belep Island). We chose to focus our data collection efforts at three main study areas (Fig. 1): Noumea, Touho and Ouvea. Noumea is in the south-eastern part of the Main Island and includes two aggregation sites along the barrier reef, approximatively 20 km off the coast: Dumbea Channel, a feeding station, and Boulari Channel, a cleaning station (24 km apart). Several dive clubs and other tourist operators from Noumea visit these sites daily. Touho is located in the northeast of the Main Island. Only one aggregation site in this area, a cleaning station, which is located on the barrier reef less than 5 km off the coast, at the northern tip of the Great Channel of Touho. This site is a 15- to 20-m-deep reef flat with several cleaning stations aggregating manta rays year-round. A dive club was present almost daily on the site between 2014 and 2017 before its operation ceased, after which nearly no visitors came to this site besides the authors. Ouvea is the most northerly of the Loyalty Islands, 90 km off the Main Island and separated by a 2000 m deep channel. There are two known aggregation sites: one located along the Southern reefs and the other at the Northern reefs of the island, called the Pleiades, that enclose the lagoon of Ouvea. These two sites are mainly cleaning stations with occasional feeding on the reef crest at a depth of 10 to 15 m. Tourist operators regularly frequent these cleaning stations on Ouvea. These three study areas were selected among others based upon two factors. First, these were the areas where reef manta rays are photographed the most due to the regular presence of tourist operators that ease the access to the sites for both recreational divers and scientists. Second, these areas, being distributed across New Caledonia, give a broad view of the connectivity within the archipelago. All other sites are defined as secondary in this study since data were scarcer and the authors were not able to complement the sampling effort. Data at these sites are only based on opportunistic collection of photo-identifications by citizen scientists, with no standardised surveys performed directly by the authors at these locations.
Sighting locations of reef manta rays (M. alfredi) in New Caledonia, South Pacific (inset) recorded by photo-identification between 1993 and 2020. The red line indicates the EEZ of New Caledonia. Stars indicate sites included in the dataset. Triangles indicate sites mentioned in the study but not in the dataset
Data collection
Reef manta rays’ photographs were collected either while SCUBA diving or free diving through the combination of opportunistic observations from local tourist operators, dive clubs, spearfishers and other users of the sea, and surveys performed by the authors. On the one hand, photographs were gathered from citizen scientists via communication campaign was launched in 2016 by the ‘Manta Initiative in New Caledonia’ program (www.facebook.com/initiativemantaNC), a collaboration between The Manta Trust (www.mantatrust.org), Conservation International (www.conservation.org) and the Aquarium des Lagons (www.aquarium.nc). This program aims to inform and promote citizen science in order to collect all already available images and encouraging everyone to participate by capturing new photographs. A training was offered to the dive club representatives, and they promoted the initiative by involving the customers. Communication materials were broadcasted to explain the photo-identification process and the conditions required to participate. Citizen science has been used in many studies to gather information on population characteristics of reef manta rays around the world (e.g. East Australia, Couturier et al. 2014, Indonesia, Germanov et al. 2019, French Polynesia, Carpentier et al. 2019). In order to account for the inconsistent sampling effort through time and enable comparisons between sites, we measured the sampling effort variable in ‘sighting days’ when at least one reef manta rays sighting was recorded at each site. On the other hand, the authors performed surveys at the three main sites between 2017 and 2020 at different periods of the year (Table S1). Sampling was opportunistic based on boat and staff availability A
Images were captured while SCUBA diving or free diving using underwater digital cameras. A team of 2–4 divers surveyed a site for a minimum of two dives when using SCUBA and between 4 and 8 h when free diving. The photographs that generated the data used for this study were collected from different periods depending on site. In Noumea, the earliest photograph was captured in 1993 and the latest was in December 2020. In Touho, the photographs were taken between January 2014 and August 2020. Photographs from Ouvea were dated from January 2005 to August 2020. Data collected at secondary sites ranged from 2017 to January 2020 in Mare, from January 2008 to November 2020 in Lifou, from 2003 to July 2020 in the Ile of Pines, from June 2019 and July 2020 in Boulouparis, from February 2015 and November 2017 in Bourail, from March 2010 and August 2019 in Pouembout, from March 2017 and 2019 in Ouano, and from September 2011 in Poindimie (Table S1).
Photo-identification data processing
A New Caledonian database was built in 2016 with identification entries only generated from photographs with a clear distinction of the spots and other patches located on the inter-brachial and sub-abdominal areas on the ventral surface of the animals. In addition, only photo-identifications with a precise date and location were entered in the database. All photographs that did not comply with these conditions were not retained at all in the database. Additional distinctive wound marks were also used to assist in the identification process. A sighting event constitutes a positive identification of an individual. Re-sightings of a known individual were recorded when the identification occurred at least 24 h after the previous identification (Marshall 2008). Two encounters of the same individuals but at two different sites within the same day would be considered as a re-sighting. The identification was processed visually by the author comparing the new photographs to the photo-identifications of each individual already recorded in the database. Once identified, distinctive characteristics such as the sex, the colour morph and injuries were recorded from all available images (following Marshall et al. 2011). The sex was determined by the presence or absence of claspers for male or female individuals, respectively (following Marshall et al. 2011). The maturity stage was classified threefold: newborn, juvenile and adults. Newborns were identified with unmarked body with no scratches (Marshall and Bennett 2010a, b) and the presence of creases along the junction between the pectoral fins and the body (Marshall 2008). Juvenile males were identified when claspers are not fully developed, and juvenile females were individuals bearing no mating scars on the pectoral fins. Finally, adults were males with fully developed claspers and females with mating scars. The presence of injuries was also recorded and marks of predators and of anthropogenic origin were distinguished independently, when possible, among all injuries. Predation attempts were identified by the appearance of clear shark bites (Marshall & Bennett 2010a) and/or the tail cut, while damaged or missing cephalic fins, clear cuts on the frontal edge of the body and/or clear propeller marks (Carpentier et al. 2019) were identified as being of anthropogenic origin. Maturity and disc width were also recorded but since the data showed high discrepancy among observers, we decided not to include them in our analyses.
Abundance estimations
Our approach to estimate the abundance of reef manta rays at each site is based on the method to estimate species richness through species accumulation curves. Following the Baker et al. (2006) approach, species discovery curves can be derived to the individual level to determine whether a population has been fully enumerated and estimate abundance of partially enumerated populations. In this study, we used the cumulative number of reef manta ray individuals identified at a given site plotted against cumulative field effort for this analysis, the dataset was confined to only periods when sampling effort was the highest and the most consistent: from 2017 to 2020 for Noumea and Ouvea, and 2014–2015 for Touho (Table S1). In addition, the abundance was only estimated at these three sites since only at these locations the effort was consistent enough to run this analysis. For this approach to be valid, assumptions require to be met: (1) observers continuously attempt to identify new individuals throughout the period; (2) all manta rays have equal chance to be identified; and (3) there are no significant additions to the population during the period that would cause true abundance to raise (Baker et al. 2006). In other words, the rate at which new individuals are discovered per unit effort should decline with time. The first assumption was met by confirming that at these sites during these particular periods, observers were trained and identified all reef manta rays present. For the second assumption, these sites are all cleaning stations and, being key habitat for reef manta rays, all individual of an area should be available to be identified given a sufficient sampling effort. The third assumption is met based on the fact that the number of manta rays that were born or died during the period prior to being identified is negligible.
Discovery curves of the cumulative number of individuals per sampling effort unit (days) were generated for each site using the Sample rarefaction (Mao’s tau) function (Colwell et al. 2004) implemented in the PAST 4.03 software (Hammer 2001). A total of five non-linear models (listed in Table S2) proposed within the Non-linear fit function in PAST were fit to our data by following Levenberg–Marquardt optimization and the best-fit model was chosen using the smallest AIC value (Table S2). Abundance estimates were indicated by the asymptote of the curve and 95% confidence intervals were based on 1999 bootstrap replicates (default value of the program).
Site fidelity and residency analysis
We investigate site fidelity using individual sighting histories such re-sighting rates and lengths of re-sightings periods. Based solely on sightings, our data do not allow us to know whether an individual stayed at or left a site between two sightings. Thus, we analysed residency, the uninterrupted occupation of an area by an individual during a given period, using models. Residency models, or models of lagged identification rates (LIR), defined as the probability of resighting an individual after a variable lag time, were fitted to our resighting data to compare patterns between sites. These statistical models are appropriate with the analysis of opportunistically sampled data, as it uses the recapture data to infer sampling effort (Whitehead 2001). Several studies used these statistical models on manta rays (Deakos et al. 2011; Germanov et al. 2019; Carpentier et al. 2019), whale sharks (e.g. Ramírez-Macías et al. 2012; Robinson et al. 2016; McCoy et al. 2018), and marine mammals (Whitehead 2001; Chabanne et al. 2017). We used the SOCPROG2.9 software (Whitehead 2009) to calculate LIR data and compare them to eight models with different closed and open population scenarios that includes variables such as emigration, mortality, and re-immigration (Table S3). In this case, the dataset was confined to only those periods when sampling effort was the highest and the most consistent from both, the authors and citizen scientists: from 2017 to 2020 for Noumea and Ouvea, and 2014–2015 for Touho (Table S1). For each site, the best-fit model was chosen by selecting the lowest value from the Akaike information criterion (AIC), when no overdispersion occurs in the data, or the quasi-Akaike information criterion (QAIC) when overdispersion occurs in the data. The differences in AIC or QIAC values between the best-fit model and any other ones (∆AIC or ∆QAIC) provide an indication of how the data support other models based on the following results (Whitehead 2007): ∆AIC or ∆QAIC < 2 = substantial support; ∆AIC or ∆QAIC 4 – 7 = considerably less support; and ∆AIC or ∆QAIC > 10 essentially no support. All chosen models were then bootstrapped 1999 times (default value of the program) to calculate standard errors (SE) and 95% confidence intervals (CI).
Statistical analysis
The comparisons of sex ratio, maturity stages, ratio of normal chevron colored vs melanistic individuals for each site and potential sex bias in movements were tested using Chi-squared (χ2) goodness of fit tests. Injury and attempted predation rate data were also compared using Chi-squared (χ2) tests for contingency tables to assess the relationship between these variables and the sex at each site. Differences in mean numbers of re-sighting events, mean re-sighting period and re-sighting events per individual were tested using analysis of variance (ANOVA). Student t-tests (equal variances) and Welch’s t-tests (unequal variances) were used for pairwise comparisons of means. Levene’s tests were used to test the homogeneity assumptions.
Results
Sampling effort
Sampling effort, as measured by number of sightings days with at least one sighting event, was not evenly distributed through locations and time at all sites in New Caledonia. Spatially, of a total of 654 sightings days, the largest sampling effort (91% of all sightings days) occurred at the three main study sites: Noumea (56.6%, 370 days), Touho (22.9%, 150 days) and Ouvea (11.5%, 75 days), while sampling effort at secondary sites ranged from 0.2% (1 day) at Poindimie and Bourail to 4.3% (28 days) at Mare. Temporally, these 654 sightings days were unevenly distributed from 1993 to 2020 (Table S1A). In Noumea (N = 370 days) and Ouvea (N = 75 days), most of the sampling effort was conducted between 2017 and 2020, with 55.7% and 85.3% of the total effort at these sites, respectively. In Touho (N = 150 days), sampling effort was the highest in 2014 and 2015, representing 85.3% of the total effort. This sampling heterogeneity was taken into account when analysing temporal and spatial patterns in our manta sightings as reported below.
Sightings distribution
A total of 1741 sighting records consisting of at least one photograph suitable for identification were collected from 1993 to 2020 for the reef manta ray. Photographs were taken by citizen scientists (85.6%) between 1993 and 2020 and the authors (14.4%) between 2017 and 2020 (Table S1B). These sightings originated from 11 sites around the archipelago of New Caledonia, although 94.4% were recorded at the three main study sites of Noumea, Ouvea and Touho. Noumea had the highest percentage of sightings (48.9% of total), with 851 sightings recorded in 370 days between 1993 and 2020. In Touho, 591 sightings were recorded in 150 days between 2014 and 2020, with 81% of those sightings recorded in 2014 and 2015 when a tourism boat was operational there. In Ouvea, we recorded 206 sightings from 75 days between 2014 and 2020, with 80.1% collected since 2017. At the eight secondary sites, a total of 93 sightings were recorded since 2003 in 59 days of successful survey effort. Mare and the Isle of Pines accounted for 37 and 27 sightings in 28 and 15 days, respectively, while the six other sites combined produced a total of 29 sighting events in 16 days. The average number of sightings per sampling effort unit was significantly different between sites (F(3,653) = 27.1, p < 0.001). The highest rate was recorded in Touho with 3.9 individuals per day and the lowest was in Noumea with 2.3, while in Ouvea the rate was 2.7 individuals per day (Table 1). When all data were pooled across New Caledonia, there was no significant difference in the number of males and females sighted (χ2(1, N = 1741) = 1.71, p > 0.05).
Individuals per site and estimated abundance
Using photo-identification, off the 1741 sightings, we recorded 391 reef manta ray individuals throughout the 11 known aggregation sites in New Caledonia. In Noumea, we counted a total of 153 reef manta rays at two aggregation sites: 82.3% at Boulari channel and 17.7% at Dumbea channel, with 14.4% of the Noumea individuals observed at both sites. In Touho, the total count was 72 reef manta rays, while in Ouvea, we recorded 116 individuals from two sites: Northern (16.5%) and Southern Pleiades (83.5%) with 13.3% observed at both sites. From the eight secondary sites we recorded a total of 66 individuals. Individual counts varied from one (in only one sighting) in Poindimie to 20 from both Mare and the Isle of Pines for 37 and 27 sighting events, respectively (Table S1).
The discovery curves describing the cumulative number of individuals recorded over time did not reach an asymptote for any of the sampling sites, indicating that these counts did not represent the whole population (Fig. 2). However, the curves for most sites, and especially for Touho and Noumea population, showed a slight decline in accumulation rate, suggesting that extrapolation to an asymptote might provide an abundance estimate (Baker et al. 2006). The best-fitting model (Table S2) for the Noumea and Touho was Hill’s equation and reached its asymptote at 252 [CI 210, 265] and 94 [74, 97] individuals, suggesting that, based on the current number of identified individuals, 60.7% [57.7, 72.9] and 76.6% [74.2, 97.3] of these groups were sampled, respectively. In Ouvea, the Michaelis–Menten model best-fitted the data and reached its asymptote at 238 [238, 239] individuals, suggesting that 47.5% [47.4, 47.6] of the population was sampled.
Discovery curves of the cumulative number of individuals of reef manta rays (M. alfredi) identified over the number of days of sighting events at main areas in New Caledonia: Noumea, Touho, Ouvea between January 1993 and December 2020 (black parts).Gray parts of the curves are model projections and dotted lines are 95%CI
Re-sighting rate and residency
A total of 222 reef manta rays (56.8%) of New Caledonia (N = 391) were re-sighted at least once (Table 1). In New Caledonia, the average resighting period, between the first and last sighting event for a given individual, was 1467 ± 1507 days (approximatively 4.0 ± 4.1 years). The longest re-sighting period was recorded for an adult male sampled in Noumea with 9997 days (approximatively 27.4 years) between the first observation in 1993, already adult at the time, and the last in 2020. Among the three main study sites, the site fidelity was the highest in Touho (N = 72) with 76.4% of individuals resighted at least once in 150 days of sampling and the lowest in Ouvea (N = 116) with 44.8% of individuals resighted in 75 days (Table 1). In Noumea (N = 153) 59.5% of individuals were resighted at least once in 370 days. On average each individual was resighted 9.1 ± 11.5 times, with a mean resighting period of 1947 ± 1776 days (approximatively 5.3 ± 4.9 years). The individual with the most re-sightings was an adult female recorded 64 times in 370 sightings days, with a resighting period of 4953 days (approximatively 13.6 years). In Touho (N = 72), 53 manta rays were observed more than once. On average, individuals from this site recorded 10.5 ± 9 re-sight events with a resighting period of 1196 ± 764 days (approximatively 3.3 ± 2.1 years). The most re-sighted individual was an adult male recorded 35 encounters in 50 sightings days within a period of 4596 days (approximatively 12.6 years). In Ouvea (N = 116), 45 manta rays were observed more than once. The number of re-sighting events was 3.3 ± 2.5 times on average and the mean re-sighting period of 1021 ± 1362 days (approximatively 2.8 ± 3.7 years). At this site, the maximum number of sightings was recorded for an adult male with 17 times in 75 sightings days over a period of 202 days. Finally, of the eight remaining sites, Mare (N = 20) and Isle of Pines (N = 14) counted eight and seven individuals that were sighted more than once, respectively. The average of re-sighting events per individual was 3.1 ± 1.4 and 3.8 ± 2 times with mean re-sighting period of 366 ± 63 days and 1345 ± 1429 days (approximatively 3.7 ± 3.9 years), respectively. The most re-sighted manta rays were both adult females with eight and six sighting events within 428 and 1153 days (approximatively 1.2 and 3.2 years), respectively.
The observed mean Lagged Identification Rate (LIR) shows the probability of re-sighting an individual after a variable lag time. Using the sightings data from each site, LIR revealed long-term residency as all trends indicated a levelling off over long-time lags (Fig. 3).
Lagged identification rates (LIR ± SE) of reef manta rays (M. alfredi) from each of the main study areas: Noumea (N = 104; 2017–2020), Touho (N = 61; 2014–2015) and Ouvea (N = 96; 2017–2020. Best-fitting models for each area are denoted in Table S4
Lagged Identification Rate (LIR), calculated within each study site, showed a net decrease at time lags from 1 to approximatively 200 days at Touho and to approximatively 100 days at Noumea and Ouvea. Following this decrease, the LIR showed slight increase after approximatively 370 days in Noumea, 340 days in Touho, and twice, after 100 days and 390 days in Ouvea. This suggests that the manta rays stayed seasonally in the site. The LIR then remained stable at increasing time lags indicating the return of these individuals to the site in the following months. Models fitted to the LIR substantially supported the data based on the ∆QAIC (< 2) for Noumea and Ouvea (due to over-dispersion in the data) and the ∆AIC (< 2) for Touho (no over-dispersion in the data) (Table 2). The model H consisting of the following parameters: emigration, re-immigration and mortality (includes permanent emigration) was the best fit for Noumea and Touho. On average, a daily abundance of 20.83 (± SE = 3.72; 95% CI 14.94–29.94) and 22.40 (± SE = 4.08; 95% CI 12.16–28.01) manta rays may be found within the site of Noumea (N = 104) and Touho (N = 61), respectively. These individuals were estimated to occupy these sites for periods of 31.47 (± SE = 15.52; 95% CI 9.03–72.62) and 139.57 (± SE = 53.14; 95% CI 7.04–171.04) days and be away for periods of 23.68 (± SE = 8.34; CI 10.24–43.70) and 175.57 (± SE = 686.69; CI 5.74–959.69) days, respectively. The mortality rates were negligible for both sites since values were close to zero (< 0.001). Models E and F best fitted the data from Ouvea (N = 96). The model F revealed a daily abundance of 39.79 (± SE = 13.94; 95% CI 18.89–73.84) manta rays. Residence times for individuals in Ouvea could not be estimated with precision with the model F as the sampling size was too small, and mortality estimated by the model E was also negligible (< 0.001). Other models also explained the data to a lesser degree (∆QAIC or ∆AIC between 4 and 7) and indicated similar results (Table S3).
Connectivity
A total of 21 reef manta rays (5.4%) have been observed at two or three different sites. Among these 21 individuals, six recorded two movements between sites and one recorded four (Fig. 4; Table S4). Overall, there were no distinguishable routine movement patterns. These movements were recorded by 10 males and 11 females, with no significant bias in the sex ratio (1.0: 1.1, χ2(1, N = 21) = 0.04, p > 0.05) and four (19%) were juveniles. The two sites with the highest number of rays connecting at least once were between two areas on the same coastline: Noumea and Touho (approximatively 320 km shortest distance through the water) with five individuals and time interval between re-sighting ranging from 141 to 2328 days. Two of them made the return trip from Noumea to Touho within a minimum time interval of 354 and 567 days. Three manta rays were sighted at both Touho and Ouvea, with the two areas being located approximatively 115 km apart and with a 2000-m-deep channel in between. Two of them connected from Ouvea to Touho and one from Touho to Ouvea. Six manta rays were sighted at the Isle of Pines and either Touho (N = 3, approximatively 300 km shortest distance through water) or Noumea (N = 3, approximatively 100km). Connections between the Isle of Pines were observed in both directions with time intervals ranging from 89 to 446 days between sightings at the two sites. Only two individuals were recorded moving from Ouvea to Noumea (approximatively 260 km). Only two individuals were sighted at 3 sites. One connected from Lifou to Ouvea to Noumea and the other moved from Poindimie to Touho to Noumea. Only the individuals sighted in Mare (N = 20) were not sighted at another site during the study period.
Injuries and attempted predations (or bite marks)
Of the total of 391 identified reef manta rays in New Caledonia, 175 (44.8%) had noticeable wounds or injuries (Fig. 5). Among these individuals (N = 175), the majority (67.4%) showed marks of natural predation attempts, while 9.8% had injuries assumed to be of anthropogenic origin. Off the total of reef manta rays (N = 391), this translates to 116 (29.2%) individual that bore bite-related injuries and 17 (4.3%) had injuries assumed to be of anthropogenic origin. The origin of the injuries of the remaining 42 (22.8%) individuals could not be identified. There was no significant difference in the proportions of males and females injured (χ2(1, N = 170) = 0.14, p > 0.05), and neither was there a difference between sex in signs of attempted predation (χ2(1, N = 118) = 0.51, p > 0.05). Between sites, significant differences were observed when comparing the proportions of injured (χ2(3, N = 175) = 22.7, p < 0.001) and predated (χ2(3, N = 170) = 48.6, p < 0.001) manta (Fig. 5). Ouvea (N = 116) and Touho (N = 72) each had a significantly higher proportion of injured individuals with 54.3% and 52.8%, respectively, than Noumea (N = 153) with 38.6%. The proportion of manta rays recorded with bite marks was significantly higher in Ouvea (43.1%) than in Noumea (23.5%) (χ2(1, N = 86) = 11.6, p < 0.001). The differences in proportions of predated individuals between Touho (33.3%) and the two other sites were not significantly different. Pooling the data from the other remaining sites (N = 68) into an “Other Sites” category, both the proportion of injured manta rays (21.4%) and the percentage of predated individuals (10.7%), were significantly the lowest compared to the three main sites.
Percentages of males and females reef manta rays (M. alfredi) bearing one or more injuries (left) and attempted predation marks (right) at all sites in New Caledonia (N = 391): Noumea (N = 153), Touho (N = 72), Ouvea (N = 116) and Other sites (combining Mare, Isle of Pines, Lifou, Pouembout, Boulouparis, Ouano, Bourail and Poindimie, N = 68). Letters indicate pairwise significative differences (p < 0.05) in proportion of injured or predated individuals between sites
Sex ratio
Of the 391 reef manta rays recorded from New Caledonia, 50.4% were identified as females, 44% as males and 5.6% were undetermined (Table 1). This male: female ratio (1.0:1.15) showed no significant bias (χ2(1, N = 391) = 2.84, p > 0.05). Noumea (N = 153) was the only site with a significantly biased sex ratio towards females (1.0:1.47, χ2(1, N = 153) = 5.42, p < 0.05), though we noted that 10 individuals from this site were unsexed. In Touho, Ouvea and all the remaining sites, we did not find any significant differences in the numbers of males to females. Touho had 34 males, 35 females and 3 unsexed individuals (1.0:1.03, χ2(1, N = 72) = 0.14, p > 0.05); Ouvea had 60 males, 50 females and 6 unsexed rays (1.2:1.0, χ2(1, N = 116) = 1.17, p > 0.05) and all the eight remaining sites combined had 22 males, 31 females and 3 unsexed individuals (1.0:1.41, χ2(1, N = 68) = 1.61, p > 0.05).
Melanism
Overall (N = 391 individuals), there were significantly more chevron-coloured reef manta rays (57.0%) than melanistic ones (43.0%) (1.33:1.0, χ2(1, N = 391) = 7.74, p < 0.01) (Table 1). However, this trend was significant only among individual from Noumea, with 58.2% chevron and 41.8% melanistic manta rays (1.39:1.0, χ2(1, N = 153) = 4.08, p < 0.05), but not in Touho (1.25:1.0, χ2(1, N = 72) = 0.35, p > 0.05) and Ouvea (1.0:1.0, χ2(1, N = 116) = 0, p = 1), where there was no significant bias in the ratio of colour morphs.
Maturity stage
Over the total of reef manta rays identified (N = 391), by the end of this study (2020), 80.8% were adults while 16.6% were juveniles. The maturity stage of the remaining 2.6% could not be identified. Off the 316 adults, only five individuals were first sighted as juvenile and re-sighted later as adults. Among the juveniles, none were identified as new-born. The average period between the first sighting as a juvenile and the first sighting as an adult is 1170.4 ± 598 days (approximatively 3.2 ± 1.6 years). Among the adults, there was no significant differences in the numbers of males to females (1.0:1.0, χ2 (1, N = 302) = 0.01, p > 0.05) Off the 65 juveniles, the number of females was significantly higher than the number of males (1.0:2.2, χ2 (1, N = 65) = 8.88, p < 0.01), with twice as much females than males. Among sites, a significative difference in the proportion of juveniles was recorded (χ2 (1, N = 65) = 13.7, p < 0.01) with the highest identified in Noumea (N = 153) with 24.5%, and the lowest in Ouvea with 6%. In Touho (N = 72), 10 juveniles were counted. Finally, out of the eight remaining sites, juveniles were sighted at five of them for a total count of 11 individuals, including that 6 were recorded in Mare.
Discussion
This study provides the first insight into the population characteristics, movements, and occurrence patterns of reef manta rays in New Caledonia through the collection of photo-identifications provided in major part via citizen science. We describe strong and long-term site fidelity among studied sites and limited connectivity between them. We also report the presence of reef manta rays in most parts of the archipelago with the highest proportion of melanistic individuals documented worldwide to date.
Sampling effort and sighting distribution
Reef manta rays were sampled at various locations (N = 11) scattered around New Caledonia territory. While this provides an indication that manta rays are largely distributed in New Caledonian coastal waters, this only describes the data generated through filed surveys and mainly citizen science, and thus, does not account for all individuals located at sites that remained unsampled. The Exclusive Economic Zone of New Caledonia expand over 1,422,543 km2, home to numerous islands and reefs that are potential habitats for reef manta rays. A campaign of aerial surveys (Laran et al. 2016) of marine megafauna (including manta rays) over New Caledonia waters, undertaken in 2014, confirmed the presence of manta rays at isolated reefs, such as Petrie Reef and Astrolabe Reefs, at other islands, such as Belep Islands, and other locations along the Main Island (Laran et al. 2016). Increasing and geographically extending our sampling effort by reaching more observers at these locations will help precise the distribution of the population.
Individual count and estimated abundance
The overall individual count from data collected from 1993 to 2020 (in 654 days) at 11 sites around New Caledonia consisted of 391 reef manta rays. Locally, the current count in Noumea represents almost two thirds of the model estimation although sampling effort at this site was two times higher than it was at Touho. In comparison, the total number of individuals identified in Touho constitutes three quarter of the estimated abundance. In Ouvea, since the discovery curve did not show a decline which suggest that that extrapolation to an asymptote might provide biased estimation (Baker et al. 2006). These results combined with evidence of strong site fidelity (see paragraph 4.3), suggest that the population could be structured into multiple partially independent groups. This finding correlates with both results recorded via satellite telemetry (Lassauce et al. 2023) and genetic analyses (Lassauce et al. 2022) which revealed strong site fidelity pattern for reef manta rays in New Caledonia. This prevents us from investigating manta ray population size of New Caledonia as a whole, sampling all aggregation sites in New Caledonia would require a sampling effort beyond our logistic capacities. Global effort to estimate individual counts vary greatly among location (Table 3) from the largest documented population in the Maldives with 4,411 individuals counted in 54,605 sightings over 12 years (Harris et al. 2020) to a minimum of 305 reef manta rays in 11,111 sightings in Southern Japan (Kashiwagi 2014). In comparison to these previous findings, our results seem to indicate that New Caledonia is home for a rather small population, but these estimates could be in great part reviewed by increasing the sample size in this archipelago. In addition, the small proportion of juvenile identified in this study indicates that a segregation based on maturity stages is occurring in the use area. On this regard, Setyawan et al. (2022) present example of how to separately estimate the populations of reef manta rays in two different areas, taking into account varying survey efforts (which may influence sighting probability) and environmental conditions. Further efforts to estimate abundance and assess demographic dynamics should rather be concentrated at each known aggregation site using more robust modelling approaches based on regular and more intensive sampling effort (Deakos et al. 2011; Marshall et al. 2011; Couturier et al. 2014) or genetic analysis (Venables et al. 2021).
Site fidelity and residency
Site fidelity as defined by Chapman et al. (2015) or site affinity as defined by Couturier et al. (2014) is characterised by the return of an individual to a previously occupied site after an absence as long as or longer than the residency period. This behaviour is common and has been documented for elasmobranchs (Bansemer & Bennett 2009, 2011; Hearn et al. 2010; Bessudo et al. 2011; Chapman et al. 2015). The habitat use and home range vary depending on the mobility capacity of a species (Chapman et al. 2015).
The present study shows high levels of site fidelity with rates ranging from 43 to 75% of the manta rays sighted more than once within an average re-sighting period of four years. The longest re-sighting period in this study reached over 27 years. The time interval between the first and last sighting event for a given individual (i.e. re-sighting period as per Marshall 2008) is strongly influenced by sampling effort, which in our study is inconsistent through time and among areas. However, it remains an indicator of a high tendency for reef manta rays to remain or return to a specific site. This observation was also supported by the lagged identification rate analysis which indicated that some individuals occupied a site following emigration and re-immigration patterns with the tendency to remain at or return to the same site (Whitehead 2009). Results of genetic analysis on this population also converge with these conclusions (Lassauce et al. 2022). In fact, genetic differentiation was detected between the studied sites suggesting limited migration and assortative mating (Lassauce et al. 2022). Similarly, in Hawaii, genomic analysis between small island populations revealed high residency. Previous research indicated that reef manta rays were able of large-scale movements across different types of habitats: continuous (along coastlines; e.g. Armstrong et al. 2019) or fragmented (between island chains; e.g. Germanov & Marshall 2014). However, these movements do not reflect a homogeneous use of the whole extend of the potential home range. Instead, the reef manta ray tends to concentrate their movements within preferential sites as described through the use of satellite telemetry in New Caledonia (Lassauce et al. 2023) and in various studies around the world (e.g. Hawaii Deakos et al. 2011; Mozambique Marshall 2008; Red Sea, Braun et al. 2015; Indonesia, Germanov et al. 2019, Setyawan et al. 2020; French Polynesia, Carpentier et al. 2019). In comparison to our findings, the highest re-sighting rate was reported in Maupiti, French Polynesia, where 90% of the individuals were sighted more than once (N = 51, Carpentier et al. 2019). In Nusa Penida, Indonesia, Germanov et al. (2019) recorded a re-sighting rate of 82% (N = 624). In Maui, Hawaii, Deakos et al. (2011) found 73% (N = 290). In Bird’s Head Seascape, Indonesia, Setyawan et al. (2020) counted 46.7% (N = 1,375) and Couturier et al. (2014) documented 63% at Lady Elliot, East Australia (N = 716). In addition, investigations on the genetic structure of populations of reef manta rays in archipelago showed fine scale differentiation in New Caledonia (Lassauce et al. 2022) and Hawaii (Whitney et al. 2023). These results indicates that the population of New Caledonia can be described as a metapopulation. As defined by Akçakaya et al. (2007), the term “metapopulation” indicates a set of geographically discrete sub-populations occupying the same region among which exchanges of individuals are limited. This hypothesis was also suggested by Setyawan et al. (2020) to describe the population of reef manta rays in Rajat Ampat, Indonesia.
Habitat selection is based on highest quality and suitability, thus, factors as the cost of changing territories and the heterogeneity in territory quality would result in a greater site fidelity (Switzer et al. 1993). Reproductive success is often the main driver to qualify high suitability of the habitat. As aforementioned (see previous paragraph), the life history of reef manta rays highly depends on their foraging success resulting in the species spending most of their time foraging or in search for foraging grounds (Dewar et al. 2008; Anderson et al. 2011a, b; Jaine et al. 2012; Couturier et al. 2011, 2012, 2018, Armstrong 2016). Although our main studied sites are cleaning stations, high rates of site fidelity could be explained by sufficient year-long foraging opportunities at nearby sites (Deakos et al. 2011). In fact, Barr and Abelson (2019) demonstrated that the reef manta ray chose foraging over cleaning when environmental conditions yield sufficient plankton concentrations, also implying a proximity between these cleaning and feeding sites. During our field operations, feeding events were observed near the cleaning station but were neither regular nor predictable, suggesting the potential existence of more prolific grounds. Given the geomorphology of the region, this species might also use deep offshore waters where the presence of plankton is possible. This is also favourited by the existence of upwelling events occurring sporadically during the summer in the south-west off the archipelago that bring nutrients and contribute to biological activity (Ganachaud et al. 2010). Recent records using satellite telemetry showed frequent and deep diving behaviour of reef manta rays in New Caledonia (Lassauce et al. 2020) suggesting foraging activity on demersal zooplankton (Couturier et al. 2013). More research using acoustic or satellite telemetry to identify important feeding grounds and environmental factors that influence visitation patterns would help understand the spatial ecology of the species and characterise the actual range pattern around each studied site. This information is essential to assess critical habitat and address potential localised threats, and thus, inform by conservation projects.
Connectivity
Reef manta rays of New Caledonia seem to demonstrate site fidelity and little connectivity is observed between the studied sites. We reported only 5.4% of individuals at more than one location from 2003 to 2020, reflecting limited movements between known aggregation sites. These records were also insufficient to detect any movement patterns. Individual movements may depend on local food availability in the absence of a significant anthropogenic influence (Dewar et al. 2008; Anderson et al. 2011a, b; Armstrong et al. 2016; Jaine et al. 2014; Couturier et al. 2018). In fact, occurrence and movements of reef manta rays has been linked with zooplankton productivity. The archipelago of New Caledonia undergoes slight variations in environmental conditions with temperature varying from 22 to 24 °C in peak winter months and from 26 to 28 °C in peak summer months (IFREMER 2020) with little seasonal variation in chlorophyll a concentration (used a proxy for zooplankton abundance, Burgess 2017) (Dupouy 1990). In addition, all studied sites are in proximity of deep waters (> 2000 m-deep), potential source of demersal zooplankton that may constitute an important part of the diet of the reef manta ray (Couturier et al. 2013). These parameters coupled with our findings suggest that reef manta rays seem to have sufficient food resource year-round at each of their respective aggregation sites. In other part of the world, seasonal presence and movements of manta rays was explained by monsoonal shift in Indonesia in the Komodo Marine Park (Dewar et al. 2008) and Raja Ampat (Setyawan et al. 2018), in Western Australia (Sleeman et al. 2007) and in the Maldives (Anderson et al. 2011a). Jaine et al. (2012) and Couturier et al. (2018) attributed the increase in abundance in winter to optimal oceanographic conditions for zooplankton productivity at Lady Elliot Island, East coast of Australia.
While only few movements were recorded, these connections (N = 30) link all parts of New Caledonia, between sites around the Main Island and Ouvea Islands, indicating that movements along continuous coastline (up to 325 km) and between island chain (up to 260 km) exist. Large scale movements have been documented in many locations in the Indo-pacific region with the largest recorded distance along a continuous coastline being 1150 km (East Australia, Armstrong et al. 2019) and 450 km across archipelagos (Germanov & Marshall 2014). This study recorded a majority of movement along a continuous coastline but also showed the capacity of reef manta rays to connect between sites separated by deep water channel. These findings bring further evidence that deep-water channels do not seem to constitute a complete barrier as suggested by Deakos et al. 2011, 2023) and verified furthermore in New Caledonia (Lassauce et al. 2022, 2023). Different resighting rates between studied sites also indicate potential nuances in this supposition. Higher fidelity rates at a given site may reflect more optimal condition year-round than at other locations. In addition, further sampling effort to neighbouring sites may highlight additional connections, and thus, extend the range of occupancy around each of the known aggregation sites. In this sense, we found that almost a third of the manta rays that were observed at more than one site were sighted at the Isle of Pines where a total of 27 sightings were recorded in only 15 days of sampling. These connections were in majority with Noumea (approximatively 100 km away), suggesting that more movements potentially exist between these adjacent locations. Surveys at aggregation sites along the west coast of the Main Island may show more connectivity with Noumea or same potential connections between Touho and other sites of the east coast or Ouvea with other Loyalty Islands.
Our analysis did not find a sex bias in the individuals that recorded these movements. This contradicts previous research that documented sex-based difference in habitat use (movements and site fidelity) induced by reproductive behaviour in populations of reef manta rays (Deakos et al. 2011; Stevens 2016; Germanov et al. 2019). This observation is also true for many other marine species including other elasmobranchs such as the white shark (Carcharodon carcharias, Anderson et al. 2011a, b), scalloped hammerhead shark (Sphyrna lewini, Daly-Engel et al. 2012) and blacktip reef shark (Carcharhinus melanopterus, Mourier & Planes 2013), and also sea turtles (Lee et al. 2007) and cetaceans (Engelhaupt et al. 2009). In fact, studies predict the consistency in resources availability may prevent reef manta rays to travel to other islands to forage and thus could strongly increase site fidelity among islands (Whitney et al. 2023). Stevens (2016) also showed that females tend to reside longer in a site with sufficient food resources while males tend to move between sites in search for mating opportunities. In New Caledonia, genetic analysis showed limited gene flow and genetic differentiation between these study areas sites: Noumea, Touho and Ouvea (Lassauce et al. 2022). This suggests little reproductive interactions between individuals from each site, indicating that the recorded movements might not be associated with reproductive behaviour but rather be related to searches for foraging grounds. It is also possible that our analysis does not reflect the actual sex distribution of manta rays that were sighted at more than one site since based on only few observations (N = 21). Further monitoring efforts would be needed to better comprehend this behavioural pattern.
Injuries and attempted predation rate
In New Caledonia, 44.8% of reef manta rays had noticeable wounds or injuries. Most of the injuries recorded in the population were caused by natural attempted predation (67.4%) and only few marks were of anthropogenic origin (9.8%). This study shows that the reef manta rays in New Caledonia do not seem to be highly impacted by anthropogenic stressors as recorded in other parts of the world where manta rays are also not directly targeted by fisheries. For example, in French Polynesia, Carpentier et al. (2019) reported 85% and 53% of injuries caused by fishing lines or boat propellers at Bora Bora (N = 107) and Maupiti (N = 51), respectively. In western Australia (N = 140), 13.1% of the population had injuries caused by vessel strikes (McGregor et al. 2019). Anthropogenic stressors include boat/propeller strikes, cuts due to fishing hooks or entanglement in fishing lines/nets and anchor lines (Deakos et al. 2011; Carpentier et al. 2019). These sub-lethal wounds can have a severe impact on the population growth and distribution (Heithaus 2001; Deakos et al. 2011). Reproductive behaviour can be altered as the injured animal would spend more time and energy recovering, inducing a delayed sexual maturity (Harris 1989), or even causing early abortion in elasmobranch species (Adams et al. 2018). Manta rays have been recorded to spend more time at cleaning stations when injured to enhance the healing process, preventing them from foraging or mating (Marshall et al. 2011). In New Caledonia, inshore fishing remains relatively low (0.26 t/km2/year) compared to other countries in the world, but the fast-growing population and the socio-economic rapid changes may result in a large increase in fishing pressure (Guillemot et al. 2009). In addition, the current pressure is not homogeneous since certain parts, including Noumea, are highly exploited and close to overfishing (Guillemot et al. 2009). In this context, further monitoring could be required to assess sites where potential high fishing pressure overlaps with the regular presence of manta rays.
The proportion of individuals bearing bite-related injuries (29.7%) was close to those reported in Hawaii (24%, Deakos et al. 2011) or east Australia (23%, Couturier et al. 2014). The highest attempted predation rate was documented in Mozambique, representing 75% of the population (Marshall & Bennett 2010a) and the lowest was in French Polynesia (only few individuals, Carpentier et al. 2019). Locally, we recorded a higher rate in Ouvea (43.1%) which may suggest a higher abundance of predators at this location compared to all other sites in New Caledonia. The proportion of manta rays that die from natural predation remains unknown although such bite-induced wounds have the same impact on the individual (and the population) as with any other injuries (as aforementioned).
Sex ratio
The sex ratio (M: F) of the population of reef manta rays was equitable (1.0:1.15) which corresponds to previous findings in the Maldives (Stevens 2016), Hawaii (Deakos et al. 2011) and Japan (Kashiwagi 2014). Other studies reported female-biased distributions for populations in Mozambique (1.0:3.55, Marshall et al. 2011), the Maldives (1.0:1.8, Kitchen-Wheeler et al. 2012), east Australia (1.0:1.3) Couturier et al. 2014), Raja Ampat (1.0–1.58, Setyawan et al. 2020) west Australia (1.0:1.26, Armstrong et al. 2020)and only in Nusa Penida, Indonesia, was a male-biased ratio found (1.4:1.0, Germanov et al. 2019). Sex distribution in reef manta rays might also depend on the use of the site. Most sites investigated in this study are cleaning stations. Sightings at these specific habitats have been described to be strongly skewed towards females and databases with a majority of sightings at cleaning stations, ours included, tend to underestimate the proportion of male in the population (Stevens 2016). In New Caledonia, a female-biased ratio was only found in Noumea (1.0:1.47). A potential explanation for this female-biased ratio could result from less bold attitude and smaller size of males that may diminish the likelihood of identification (Stevens 2016). Following this hypothesis, Noumea is by far the most visited aggregation area by diving operators, which could be an important restricting factor for unadventurous male to frequent the area. In this scenario, males might occur in the deeper and outer part of the reef where they are less likely to be detected. Sites adjacent to potential nursing ground might be frequented by more mature females than male (Marshall & Bennett 2010b) while males (smaller in size) may favor shallow coastal areas offering more protection from predator (McCauley et al. 2014, Stevens 2016; Germanov et al. 2019). There is no evidence for any nearby nursery area since only few juveniles were sighted at all the sites we investigated. Thus, further data collection recording the size, using standardized measurement techniques (Deakos 2010), and expanding the sampling area in search for breeding and nursery ground would help provide a better understanding of the population structure.
Melanism
The population of reef manta rays of New Caledonia has the highest proportion of melanistic individual reported to date (43.0%) with locally 50.1% in Ouvea. Processes affecting intraspecific colour polymorphisms, especially in the marine environment, remain unclear. Our results add more perspective into the global documentation of this trait and may contribute to a better understanding of the underlying evolutionary origin. Melanistic individuals appear to be in minority with large differences in frequencies among populations of reef manta rays around the world (Venables et al. 2019). The second largest proportion for melanistic individuals was recorded in Raja Ampat with 40.7% while other studied locations recorded less than ten percent including Hawaii and the Maldives with no record of melanistic individuals (Venables et al. 2019). The present study is the only published estimation of melanism within the southern Pacific region while the population of Eastern Australia approximate 10% of melanistic manta rays (Armstrong, pers. comm.). The differences in proportion of melanistic manta rays among populations seem to be due to neutral genetic processes alone since no evidence was found to support selection induced by predation (Venables et al. 2019). To date, no molecular studies investigated the genetic basis of melanism in manta rays.
Conclusion
This study is the first to describe the characteristics of the population of reef manta rays in New Caledonia. The use of photo-identification combined to field surveys allowed the collection of reef manta ray sightings over more than two decades and provided a valuable insight on the distribution and the connectivity. Through this approach, we were also able to investigate site fidelity and residency of the species as well as estimate the size of the population at given aggregation areas in New Caledonian waters. These findings on reef manta rays will be used as a baseline for long-term monitoring of the population and to evaluate the potential impact of eventual future threat to the species. However, due to the spatially and temporally limited dataset, these fundings need to be considered as a complement to other approaches already used such as satellite telemetry (Lassauce et al. 2023) and genetics (Lassauce et al. 2022) to have a comprehensive understanding of the spatial ecology and structure of this population. In regard to these previous results that suggested site fidelity and low connectivity that induced fine scale genetic structure, the present findings bring further indications of long-term site fidelity over decades for relatively small groups of reef manta rays. The study also highlighted the need for extended field efforts in areas that were not yet investigated, and especially remote reefs where the species could be even more isolated.
Data availability
We have presented extensive data generated by this study in the tables and Supplementary Materials. Contact the corresponding author for further requests.
References
Adams KR, Fetterplace LC, Davis AR, Taylor MD, Knott NA (2018) Sharks, rays and abortion: the prevalence of capture-induced parturition in elasmobranchs. Biol Cons 217:11–27
Akçakaya HR, Mills G, Doncaster CP (2007) The role of metapopulations in conservation. Key Top Conserv Biol 1:64–84
Anderson RC, Adam MS, Goes JI (2011a) From monsoons to mantas: seasonal distribution of Manta alfredi in the Maldives. Fish Oceanogr 20(2):104–113
Anderson SD, Chapple TK, Jorgensen SJ, Klimley AP, Block BA (2011b) Long-term individual identification and site fidelity of white sharks, Carcharodon carcharias, off California using dorsal fins. Mar Biol 158:1233–1237
Armstrong AO, Armstrong AJ, Jaine FR, Couturier LI, Fiora K, Uribe-Palomino J, Richardson AJ (2016) Prey density threshold and tidal influence on reef manta ray foraging at an aggregation site on the Great Barrier Reef. PLoS One 11(5):e0153393
Armstrong AO, Armstrong AJ, Bennett MB, Richardson AJ, Townsend KA, Dudgeon CL (2019) Photographic identification and citizen science combine to reveal long distance movements of individual reef manta rays Mobula alfredi along Australia’s east coast. Mar Biodivers Rec 12(1):14. https://doi.org/10.1186/s41200-019-0173-6
Armstrong AJ, Armstrong AO, McGregor F, Richardson AJ, Bennett MB, Townsend KA, Hays GC, van Keulen M, Smith J, Dudgeon CL (2020) Satellite tagging and photographic identification reveal connectivity between two UNESCO World Heritage Areas for reef manta rays. Front Mar Sci 7:725. https://doi.org/10.3389/fmars.2020.00725
Baker JD, Harting AL, Johanos TC (2006) Use of discovery curves to assess abundance of Hawaiian monk seals. Mar Mamm Sci 22(4):847–861
Bansemer CS, Bennett MB (2009) Reproductive periodicity, localised movements and behavioural segregation of pregnant Carcharias taurus at Wolf Rock, southeast Queensland, Australia. Mar Ecol Prog Ser 374:215–227
Bansemer CS, Bennett MB (2011) Sex-and maturity-based differences in movement and migration patterns of grey nurse shark, Carcharias taurus, along the eastern coast of Australia. Mar Freshw Res 62(6):596–606
Barr Y, Abelson A (2019) Feeding–cleaning trade-off: Manta ray “decision-making” as a conservation tool. Front Mar Sci 6:88
Beale CS, Stewart JD, Setyawan E, Sianipar AB, Erdmann MV (2019) Population dynamics of oceanic manta rays (Mobula birostris) in the Raja Ampat Archipelago, West Papua, Indonesia, and the impacts of the El Niño-Southern Oscillation on their movement ecology. Divers Distrib 25(9):1472–1487
Bessudo S, Soler GA, Klimley AP, Ketchum JT, Hearn A, Arauz R (2011) Residency of the scalloped hammerhead shark (Sphyrna lewini) at Malpelo Island and evidence of migration to other islands in the Eastern Tropical Pacific. Environ Biol Fish 91:165–176
Bowen BW, Gaither MR, DiBattista JD, Iacchei M, Andrews KR, Grant WS, Briggs JC (2016) Comparative phylogeography of the ocean planet. Proc Natl Acad Sci 113(29):7962–7969
Braun CD, Skomal GB, Thorrold SR, Berumen ML (2015) Movements of the reef manta ray (Manta alfredi) in the Red Sea using satellite and acoustic telemetry. Mar Biol 162:2351–2362
Burgess K (2017) Feeding ecology and habitat use of the giant manta ray Manta birostris at a key aggregation site off mainland Ecuador.
Carpentier AS, Berthe C, Ender I, Jaine FR, Mourier J, Stevens G, Clua E (2019) Preliminary insights into the population characteristics and distribution of reef (Mobula alfredi) and oceanic (M. birostris) manta rays in French Polynesia. Coral Reefs 38(6):1197–1210
Chabanne DB, Finn H, Bejder L (2017) Identifying the relevant local population for Environmental Impact Assessments of mobile marine fauna. Front Mar Sci 4:148
Chapman DD, Feldheim KA, Papastamatiou YP, Hueter RE (2015) There and back again: a review of residency and return migrations in sharks, with implications for population structure and management. Ann Rev Mar Sci 7:547–570
Clark TB (2010) Abundance, home range, and movement patterns of manta rays (Manta alfredi, M. birostris) in Hawaiʻi. Doctoral dissertation, [Honolulu]:[University of Hawaii at Manoa], (December 2010).
Colwell RK, Mao CX, Chang J (2004) Interpolating, extrapolating, and comparing incidence-based species accumulation curves. Ecology 85(10):2717–2727
Couturier LI, Jaine FR, Townsend KA, Weeks SJ, Richardson AJ, Bennett MB (2011) Distribution, site affinity and regional movements of the manta ray, Manta alfredi (Krefft, 1868), along the east coast of Australia. Mar Freshw Res 62(6):628–637
Couturier LIE, Marshall AD, Jaine FRA, Kashiwagi T, Pierce SJ, Townsend KA, Richardson AJ (2012) Biology, ecology and conservation of the Mobulidae. J Fish Biol 80(5):1075–1119
Couturier LI, Rohner CA, Richardson AJ, Marshall AD, Jaine FR, Bennett MB, Nichols PD (2013) Stable isotope and signature fatty acid analyses suggest reef manta rays feed on demersal zooplankton. PLoS One 8(10):e77152
Couturier LI, Dudgeon CL, Pollock KH, Jaine FRA, Bennett MB, Townsend KA, Richardson AJ (2014) Population dynamics of the reef manta ray Manta alfredi in eastern Australia. Coral Reefs 33:329–342
Couturier LIE, Newman P, Jaine FRA, Bennett MB, Venables WN, Cagua EF, Richardson AJ (2018) Variation in occupancy and habitat use of Mobula alfredi at a major aggregation site. Mar Ecol Prog Ser 599:125–145
Daly-Engel TS, Seraphin KD, Holland KN, Coffey JP, Nance HA, Toonen RJ, Bowen BW (2012) Global phylogeography with mixed-marker analysis reveals male-mediated dispersal in the endangered scalloped hammerhead shark (Sphyrna lewini). PLoS One 7(1):e29986
Deakos MH (2010) Paired-laser photogrammetry as a simple and accurate system for measuring the body size of free-ranging manta rays Manta alfredi. Aquat Biol 10(1):1–10
Deakos MH, Baker JD, Bejder L (2011) Characteristics of a manta ray Manta alfredi population off Maui, Hawaii, and implications for management. Mar Ecol Prog Ser 429:245–260
Dewar H, Mous P, Domeier M, Muljadi A, Pet J, Whitty J (2008) Movements and site fidelity of the giant manta ray, Manta birostris, in the Komodo Marine Park, Indonesia. Mar Biol 155:121–133
Dupouy C (1990) La chlorophylle de surface observée par le satellite NIMBUS-7 dans une zone d’archipel (Nouvelle-Calédonie et Vanuatu): une première analyse. Halieutique, océanographie et télédétection: contribution française aux colloques francojaponais: thème: télédétection, edited by: Petit, M. and Stretta, J.-M., Bulletin de l’Institut Océanographique de Monaco,(spécial 6), 125–148.
Engelhaupt D, Rus Hoelzel A, Nicholson C, Frantzis A, Mesnick S, Gero S, Mignucci-Giannoni AA (2009) Female philopatry in coastal basins and male dispersion across the North Atlantic in a highly mobile marine species, the sperm whale (Physeter macrocephalus). Mol Ecol 18(20):4193–4205
Ganachaud A, Vega A, Rodier M, Dupouy C, Maes C, Marchesiello P, Le Borgne R (2010) Observed impact of upwelling events on water properties and biological activity off the southwest coast of New Caledonia. Mar Pollut Bull 61(7–12):449–464
Germanov ES, Marshall AD (2014) Running the gauntlet: regional movement patterns of Manta alfredi through a complex of parks and fisheries. PLoS One 9(10):e110071
Germanov ES, Bejder L, Chabanne DB, Dharmadi D, Hendrawan IG, Marshall AD, Loneragan NR (2019) Contrasting habitat use and population dynamics of reef manta rays within the Nusa Penida marine protected area, Indonesia. Front Mar Sci. https://doi.org/10.3389/fmars.2019.00215
GNC (2013) Arrêté n° 2013–1007/GNC du 23 avril 2013 relatif à l'exploitation des requins dans l'espace maritime de la Nouvelle-Calédonie, p. 3776. https://juridoc.gouv.nc/JuriDoc/JdJonc.nsf/0/0D7814D4B4BB6D6B4B257B6A00195792/%24File/8903.pdf
Guillemot N, Léopold M, Cuif M, Chabanet P (2009) Characterization and management of informal fisheries confronted with socio-economic changes in New Caledonia (South Pacific). Fish Res 98(1–3):51–61
Hammer O (2001) PAST: paleontological statistics software package for education and data analysis palaeontologia electronica. https://cir.nii.ac.jp/crid/1570009750883534464. Accessed Sept 2023
Harris RN (1989) Nonlethal injury to organisms as a mechanism of population regulation. Am Nat 134(6):835–847
Harty K, Guerrero M, Knochel AM, Stevens GM, Marshall A, Burgess K, Stewart JD (2022) Demographics and dynamics of the world’s largest known population of oceanic manta rays Mobula birostris in coastal Ecuador. Mar Ecol Prog Ser 700:145–159
Harris JL, McGregor PK, Oates Y, Stevens GM (2020) Gone with the wind: seasonal distribution and habitat use by the reef manta ray (Mobula alfredi) in the Maldives, implications for conservation. Aquat Conserv: Mar Freshwat Ecosyst 30(8):1649–1664. https://doi.org/10.1002/aqc.3350
Hearn A, Ketchum J, Klimley AP, Espinoza E, Penaherrera C (2010) Hotspots within hotspots? Hammerhead shark movements around wolf island, galapagos marine reserve. Mar Biol 157:1899–1915
Heithaus MR (2001) Shark attacks on bottlenose dolphins (Tursiops aduncus) in Shark Bay, Western Australia: attack rate, bite scar frequencies, and attack seasonality. Mar Mamm Sci 17(3):526–539
Hueter RE, Heupel MR, Heist EJ, Keeney DB (2004) The implications of philopatry in sharks for the management of shark fisheries. J Northwest Atl Fish Sci 35:239–247
IFREMER. (n.d.) Retrieved December 26, 2020, Moyennes mensuelles des températures de surface depuis 2003. https://nouvelle-caledonie.ifremer.fr/Outils-pour-la-gestion/Donnees/SST-Moyennes-mensuelles
Jaine FR, Couturier LI, Weeks SJ, Townsend KA, Bennett MB, Fiora K, Richardson AJ (2012) When giants turn up: sighting trends, environmental influences and habitat use of the manta ray Manta alfredi at a coral reef. PLoS One. https://doi.org/10.1371/journal.pone.0046170
Jaine FRA, Rohner CA, Weeks SJ, Couturier LIE, Bennett MB, Townsend KA, Richardson AJ (2014) Movements and habitat use of reef manta rays off eastern Australia: offshore excursions, deep diving and eddy affinity revealed by satellite telemetry. Mar Ecol Prog Ser 510:73–86
Kashiwagi T (2014) Conservation biology and genetics of the largest living rays: manta rays. PhD. Thesis thesis, University of Queensland, St Lucia, QLD
Kitchen-Wheeler AM, Ari C, Edwards AJ (2012) Population estimates of Alfred mantas (Manta alfredi) in central Maldives atolls: North Male, Ari and Baa. Environ Biol Fishes 93:557–575
Knochel AM, Cochran JE, Kattan A, Stevens GM, Bojanowksi E, Berumen ML (2022) Crowdsourced data reveal multinational connectivity, population demographics, and possible nursery ground of endangered oceanic manta rays in the Red Sea. Aquat Conserv Mar Freshwat Ecosyst 32(11):1774–1786
Krefft G (1868) Deratoptera alfredi (Prince Alfred’s ray). Illustrated Sydney News 5(50):1–16
Laran S, Doremus G, Mannocci L, Van Canneyt O, Watremez P, Cadinouche A & Rildoux V (2016) Progress of the REMMOA aerial surveys conducted in the French EEZ and adjacent waters: contrasted cetacean habitats in the southwest Indian Ocean.
Lassauce H, Chateau O, Erdmann MV, Wantiez L (2020) Diving behavior of the reef manta ray (Mobula alfredi) in New Caledonia: More frequent and deeper night-time diving to 672 meters. PLoS One 15(3):e0228815
Lassauce H, Dudgeon CL, Armstrong AJ, Wantiez L, Carroll EL (2022) Evidence of fine-scale genetic structure for reef manta rays Mobula alfredi in New Caledonia. Endanger Spec Res 47:249–264
Lassauce H, Chateau O, Wantiez L (2023) Spatial ecology of the population of reef manta rays, Mobula alfredi (Krefft, 1868), in New Caledonia using satellite telemetry 1–horizontal behaviour. Fishes 8(6):328
Lee PL, Luschi P, Hays GC (2007) Detecting female precise natal philopatry in green turtles using assignment methods. Mol Ecol 16(1):61–74
Mannocci L, Boustany AM, Roberts JJ, Palacios DM, Dunn DC, Halpin PN, Winship AJ (2017) Temporal resolutions in species distribution models of highly mobile marine animals: Recommendations for ecologists and managers. Divers Distrib 23(10):1098–1109
Marshall AD (2008) Biology and population ecology of Manta birostris in southern Mozambique. Ph.D. Thesis, The University of Queensland, Brisbane.
Marshall AD, Bennett MB (2010a) The frequency and effect of shark-inflicted bite injuries to the reef manta ray Manta alfredi. Afr J Mar Sci 32(3):573–580
Marshall AD, Bennett MB (2010b) Reproductive ecology of the reef manta ray Manta alfredi in southern Mozambique. J Fish Biol 77(1):169–190
Marshall AD, Dudgeon CL, Bennett MB (2011) Size and structure of a photographically identified population of manta rays Manta alfredi in southern Mozambique. Mar Biol 158:1111–1124
Marshall A, Barreto R, Carlson J, Fernando D, Fordham S, Francis MP, Herman K, Jabado RW, Liu KM, Pacoureau N, Rigby CL, Romanov E, Sherley RB (2022) Mobula alfredi (amended version of 2019 assessment). IUCN Red List Threat Spec. https://doi.org/10.2305/IUCN.UK.2022-1.RLTS.T195459A214395983.en
McCauley DJ, DeSalles PA, Young HS, Papastamatiou YP, Caselle JE, Deakos MH, Micheli F (2014) Reliance of mobile species on sensitive habitats: a case study of manta rays (Manta alfredi) and lagoons. Mar Biol 161:1987–1998
McCoy E, Burce R, David D, Aca EQ, Hardy J, Labaja J, Araujo G (2018) Long-term photo-identification reveals the population dynamics and strong site fidelity of adult whale sharks to the coastal waters of Donsol, Philippines. Front Mar Sci 5:271
McGregor F, Richardson AJ, Armstrong AJ, Armstrong AO, Dudgeon CL (2019) Rapid wound healing in a reef manta ray masks the extent of vessel strike. PLoS One 14(12):e0225681
Mourier J, Planes S (2013) Direct genetic evidence for reproductive philopatry and associated fine-scale migrations in female blacktip reef sharks (Carcharhinus melanopterus) in French Polynesia. Mol Ecol 22(1):201–214
O’Malley MP, Townsend KA, Hilton P, Heinrichs S, Stewart JD (2017) Characterization of the trade in manta and devil ray gill plates in China and South-east Asia through trader surveys. Aquat Conserv Mar Freshwat Ecosyst 27(2):394–413
Palumbi SR (1994) Genetic divergence, reproductive isolation, and marine speciation. Annu Rev Ecol Syst 25(1):547–572
Peel LR, Stevens GM, Daly R, Keating Daly CA, Collin SP, Nogués J, Meekan MG (2020) Regional movements of reef manta rays (Mobula alfredi) in Seychelles waters. Front Mar Sci 7:558
Ramírez-Macías D, Vázquez-Haikin A, Vázquez-Juárez R (2012) Whale shark Rhincodon typus populations along the west coast of the Gulf of California and implications for management. Endanger Spec Res 18(2):115–128
Robinson DP, Jaidah MY, Bach S, Lee K, Jabado RW, Rohner CA, Pierce SJ (2016) Population structure, abundance and movement of whale sharks in the Arabian Gulf and the Gulf of Oman. PLoS One 11(6):e0158593
Setyawan E, Stevenson BC, Erdmann MV, Hasan AW, Sianipar AB, Mofu I, Constantine R (2022) Population estimates of photo-identified individuals using a modified POPAN model reveal that Raja Ampat’s reef manta rays are thriving. Front Mar Sci 9:1014791
Setyawan E, Sianipar AB, Erdmann M, Fischer AM, Haddy JA, Beale CS, Lewis SA and Mambrasar R (2018) Site fidelity and movement patterns of reef manta rays (Mobula alfredi: Mobulidae) using passive acoustic telemetry in northern Raja Ampat, Indonesia.
Setyawan E, Erdmann MV, Lewis SA, Mambrasar R, Hasan AW, Templeton S, Beale 9CS, Sianipar AB, Shidqi R, Heuschkel H & Ambafen O (2020) Natural history of manta rays in the Bird's Head Seascape, Indonesia, with an analysis of the demography and spatial ecology of Mobula alfredi (Elasmobranchii: Mobulidae).
Sleeman JC, Meekan MG, Wilson SG, Jenner CK, Jenner MN, Boggs GS, Bradshaw CJ (2007) Biophysical correlates of relative abundances of marine megafauna at Ningaloo Reef, Western Australia. Mar Freshw Res 58(7):608–623
Stevens, GMW (2016) Conservation and population ecology of manta rays in the Maldives. Doctoral dissertation, University of York.
Stewart JD (2018) Ecology and conservation of manta and devil rays. University of California, San Diego
Switzer PV (1993) Site fidelity in predictable and unpredictable habitats. Evol Ecol 7:533–555
Venables SK, Marshall AD, Germanov ES, Perryman RJ, Tapilatu RF, Hendrawan IG, Kennington WJ (2019) It’s not all black and white: investigating colour polymorphism in manta rays across Indo-Pacific populations. Proc R Soc B 286(1912):20191879
Venables SK, Marshall AD, Armstrong AJ, Tomkins JL, Kennington WJ (2021) Genome-wide SNPs detect no evidence of genetic population structure for reef manta rays (Mobula alfredi) in southern Mozambique. Heredity 126(2):308–319
Ward-Paige CA, Mora C, Lotze HK, Pattengill-Semmens C, McClenachan L, Arias-Castro E, Myers RA (2010) Large-scale absence of sharks on reefs in the greater-Caribbean: a footprint of human pressures. PLoS One 5(8):e11968
Whitehead H (2001) Analysis of animal movement using opportunistic individual identifications: application to sperm whales. Ecology 82(5):1417–1432
Whitehead HAL (2007) Selection of models of lagged identification rates and lagged association rates using AIC and QAIC. Commun Stat Simul Comput ® 36(6):1233–1246
Whitehead H (2009) SOCPROG programs: analysing animal social structures. Behav Ecol Sociobiol 63:765–778
Whitney JL, Coleman RR, Deakos MH (2023) Genomic evidence indicates small island-resident populations and sex-biased behaviors of Hawaiian reef Manta Rays. BMC Ecol Evol 23(1):31
Acknowledgements
We express particular thanks to the Keidanren Nature Conservation Fund (KNCF) who funded the study through the SATO YAMA UMI Project and Conservation International. We thank the Southern Province of New Caledonia for their financial support in the form of a scholarship to H.L. We thank Conservation International, the Aquarium des Lagons and The Manta Trust for their overall support for the study. We thank all dive clubs and divers that largely contributed to the collection of photo-identifications, especially Sandro Iannuzzella who originally started the data collection in Touho and Luc Bourdil who has contributed the most to build the database. Special thanks to Franck Bouilleret, Hugues Gossuin, Mael Imirizaldu, and Thomas Auger for their priceless assistance in field operations, as well as Jean-Christophe Lefeuvre and François Tron for their support. We also thank the Hô-üt Association and its members as well as all those involved in the field operations for their valuable help (Pierre Kaouma, Marino Tiaou, Jean-Baptiste Badiou, Ludovic Mazens and the team of divers from the Aquarium des Lagons). We finally gratefully acknowledge the customary representatives of Ouvéa and Touho, and the three Provinces for allowing such operations.
Funding
We express particular thanks to the Keidanren Nature Conservation Fund (KNCF) who funded the study through the SATO YAMA UMI Project and Conservation International. We thank the Southern Province of New Caledonia for their financial support in the form of a scholarship to HL. Conservation International played a role in the study design and the data collection but had no role in analysis, decision to publish, or preparation of the manuscript. The remaining funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization, H.L., L.W. and O.C.; methodology, H.L. and L.W.; validation, H.L., L.W. and O.C.; formal analysis, H.L. and L.W.; investigation, H.L.; resources, H.L., L.W. and O.C.; data curation, H.L.; writing—original draft preparation, H.L.; writing—review and editing, H.L., L.W. and O.C.; visualization, H.L., L.W. and O.C.; supervision, L.W. and O.C.; project administration, H.L. and L.W.; funding acquisition, H.L., L.W. and O.C. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Author Hugo Lassauce, Laurent Wantiez and Olivier Chateau have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: J. Carlson .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lassauce, H., Chateau, O. & Wantiez, L. Characteristics, residency and site fidelity of photo-identified reef manta rays (Mobula alfredi) population in New Caledonia. Mar Biol 171, 153 (2024). https://doi.org/10.1007/s00227-024-04470-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04470-x