Abstract
There is an intense interest in long-term trends of species abundance that may reflect, for example, climate change or conservation actions. Less well studied are patterns in the magnitude of inter-annual variability in abundance across large spatial scales. We collated abundance time-series for 133 nesting sites across the globe of the seven sea turtle species. Inter-annual variability in nest numbers was lowest in loggerhead turtles (Caretta caretta), Kemp’s ridley (Lepidochelys kempii) and flatback turtle (Natator depressus) and highest in green turtles (Chelonia mydas), likely reflecting their lower trophic position compared to other species and hence tighter coupling of food availability to environmental conditions each year. The annual number of nests in green turtles could vary by 60-fold between successive years. We identified regional patterns in the magnitude of inter-annual variability in green turtle nest numbers, variability being highest for nesting beaches around Australia and lowest in the western Indian Ocean and equatorial Atlantic. These regional patterns are likely linked to corresponding patterns of environmental variability with, for example, areas subjected environmental extremes as part of the El Nino Southern Oscillation (ENSO) showing high inter-annual variability in nest numbers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Across biological systems there is intense interest in the findings from long-time series for measures such as species abundance, health, range and the seasonal timing of events (phenology), such as breeding and migration (Poloczanska et al. 2016; Pinsky et al. 2020). These long-time series have been particularly important for assessing how biological systems are changing over time, for example in response to conservation efforts, harvesting or climate change, thereby informing policy and management (Edwards et al. 2010). In addition to species abundance, the body condition (e.g. weight to length ratio) of breeding individuals may also vary across years with profound demographic consequences (Gardner et al. 2018; Ozgul et al. 2010). However, the extent of this inter-annual variation in body condition has rarely been considered over large spatial scales, such as across ocean basins or globally. Here we address this question by using a group, sea turtles, that do not generally breed every year, in common with some other animals (Jouventin and Dobson 2002), and hence whose annual breeding numbers indicate that subset of the adult population that has attained suitable body condition to breed (Hays 2000).
In a seminal study, it was suggested that the extent of inter-annual variability in nesting numbers in sea turtles varies with trophic position, with the working hypothesis that for species that forage lower down the food chain there will be greater inter-annual variability in both their forage availability and also hence their nest numbers (Broderick et al. 2001). Initial examination of data from 45 nesting sites supported this hypothesis (Broderick et al. 2001). Here we conduct the most extensive examination of this hypothesis and examine three a priori predictions. First, the inter-species patterns reported in Broderick et al. (2001) with a relatively small sample size, should be maintained when a much larger number of data-sets that have emerged in the interim are examined. Second, when all seven species of sea turtles are examined, compared to the four species examined in Broderick et al. (2001), the patterns on inter-annual variability in nest numbers should be explainable by likely variability in forage conditions. Third, it might be expected that regional patterns will exist in the extent of inter-annual variability in nesting numbers, given that the extent of inter-annual variability in foraging conditions is likely to vary regionally. Here we examine these predictions using the largest data-set assembled to date on inter-annual variability in nesting numbers. Any regional patterns in variability in nesting numbers may have important consequences for the survey effort needed to detect changes in population size in sea turtles (Mazaris et al. 2017) which in turn feed into IUCN conservation assessments of species conservation status. As such, an improved understanding of inter-annual variability in nesting numbers may help in conservation assessments for sea turtles.
Methods
We assembled time-series of sea turtle nesting numbers from the literature following procedures outlined in Mazaris et al. (2017). Each nesting site was allocated to a particular Regional Management Unit (RMU) based on the definition of RMUs in Wallace et al. (2010). We considered data from all species of sea turtles and time-series with a minimum of 6 years of data. We used data-sets from Mazaris et al. (2017) with the additional of more recent time-series (published before November 2021) located using the same literature searching procedures outlined in Mazaris et al. (2017). To avoid any confounding effects in tiny nesting populations, studies were only included if the minimum and mean number of annual nests exceeded 25 and 40 respectively. These cut-off points were selected pragmatically to exclude minor sites and the minimum of 25 was also based on the upper bound of the lowest class in the hierarchical scheme of nesting abundance provided by the SWOT database for describing colony size (http://seamap.env.duke.edu/swot). Further, we excluded nesting sites where the long-term trend exceeded a three-fold increase/decrease in nest numbers within a five-year period. This trend was calculated by regression analysis for any 5-year period. This filter was used simply to exclude cases (n = 17) where an extreme trend (upwards or downwards) in nesting numbers, generated a spuriously high value for the index of inter-annual variability. If the abundance in the first year of monitoring was close to zero, it was assumed to reflect incomplete sampling at the start of a monitoring project and was not included. In a small proportion of cases the original publication reported the number of nesting females. These values were multiplied by three, i.e. a value that has been assumed as the mean number of nests per female for sea turtles (Mazaris et al. 2017). To assess the extent of inter-annual variability in nesting numbers, we calculated an inter-annual variability index using the raw annual nest numbers. Specifically, between successive monitoring years, the larger value for nest numbers was divided by the smaller value. This process was repeated for every pair of successive years in the time-series and then the mean value calculated. For example, consider 6 years with annual counts of 520, 260, 780, 390, 3900, 650. So for each pair of successive years the inter-annual variability index values are 2, 3, 2, 10 and 6, and then the mean is 4.6, i.e. the mean difference in abundance between successive years is 4.6-fold.
Results
We assessed patterns of inter-annual variability for 133 nesting sites across the globe, including all seven sea turtle species: Chelonia mydas (green turtle), Dermochelys coriacea (leatherback turtle), Lepidochelys olivacea (olive ridley), Eretmochelys imbricata (hawksbill turtle), Caretta caretta (loggerhead turtle), Lepidochelys kempii (Kemp’s ridley), Natator depressus (flatback turtle) (Fig. 1a) (Table S1). Across species, the maximum number of nesting sites for which inter-annual variability was assessed was for green turtles (n = 52 sites) and lowest for Kemp’s ridley turtles (n = 1 site). Across nesting sites, there could be large differences in the magnitude of inter-annual variability in nesting numbers (Fig. 1b and c).
Global assessment of the magnitude of inter-annual variability in nesting numbers. a Sites around the world where levels of inter-annual variability were assessed for different sea turtle species. Numbers reflect the RMU for green turtle nesting areas that are mentioned in the text. b and c For green turtles nesting on Raine Island, Australia (RMU 39, Pacific Southwest) and Jiwani beach, Pakistan (RMU 43, Indian Northwest), time-series of nesting numbers showing examples of high and low inter-annual variability respectively
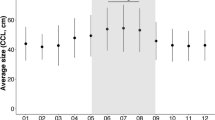
There were significant differences in the extent of inter-annual variability across species (Fig. 2) being highest for green turtles and lowest for loggerhead turtles (ANOVA, F5,126 = 5.0, P < 0.001) (Fig. 2a). Significant differences across species remained even when green turtles were excluded from the analysis (ANOVA, F4,75 = 6.1, P < 0.001), i.e. there were significant differences between the indices of the non-green turtle species. For green turtles, there was sufficient data across RMUs to examine regional patterns of inter-annual variability. For green turtles there were 12 RMUs within which nesting numbers were recorded for multiple nesting sites. There were significant differences in the extent of inter-annual variability across these RMUs (ANOVA, F11,37 = 14.1, P < 0.001)(Fig. 2b). Inter-annual variability was highest for RMUs 39 (Pacific Southwest) which included data from four Australian nesting sites: Raine Island, Heron Island, Wreck Island, and Northwest Island, with the maximum ratio in nesting numbers between successive years for these sites being 55, 60, 27, and 32 respectively. So, for example, for Heron Island (Australia) there was sometimes a 60-fold difference between the number of nests in year n versus n + 1. Furthermore, there was also very high inter-annual variability for green turtle nesting sites in RMU 40 (Indian ocean southeast), which included nesting sites in Western Australia at Ningaloo and Northwest Cape. Taken together inter-annual variability in nesting numbers was generally high for green turtle nesting sites in Australia. This finding was reiterated by the pattern of generally increasing variability moving eastwards across the Indian Ocean to Australia, with inter-annual variability in nest numbers tending to be lowest for green turtles in the western Indian Ocean (Fig. 2c). When we repeated this type analysis for green turtle nesting sites in the Atlantic, again significant patterns of inter-annual variability in nesting numbers were evident, with variability decreasing with latitude in the western and central Atlantic (Fig. 2d). For example, the mean inter-annual variability index values were 4.7 and 3.4 on the Yucatan (Mexico) and Archie Carr National Wildlife Refuge (Florida, USA) respectively, but decreased to 1.8 and 1.9 at Ascension Island (UK) and Atol das Rocas (Brazil) respectively.
Inter-specific and regional patterns in the magnitude of inter-annual variability. a The magnitude of inter-annual variability in nest numbers across species. Each symbol reflects a different nesting site, with the horizontal line showing each species mean. Cm green turtle, Dc leatherback, Lo olive ridley, Ei hawksbill turtle, Cc loggerhead turtle, Lk Kemp’s ridley, Nd flatback turtle. The number refers to the sample size. b For green turtles the magnitude of inter-annual variability in nest numbers differed across RMUs. Each point represents a different nesting site (ANOVA, F11,37 = 14.1, P < 0.001). Highest levels of inter-annual variability were recorded in RMUs 39 and 40 (Pacific Southwest and Indian Ocean Southeast respectively). c For green turtles the magnitude of inter-annual variability in nesting numbers changed from the western Indian Ocean across to Australasia. Variability was linked to longitude in this region with lowest variability in the western Indian Ocean. Inter-annual variability index = 0.077 longitude (°W)—1.75. (F1,14 = 41.9, r2 = 0.75, P < 0.001). d In the western and central Atlantic region, for green turtles inter-annual variability in nest numbers decreased with latitude, being higher for nesting sites in the Gulf of Mexico and Florida and lower for nesting sites in the Caribbean, Suriname and Ascension Island. Inter-annual variability index = 0.06 latitude (°N) + 1.96. (F1,9 = 9.8, r2 = 0.52, P = 0.012). For the other species for which there were data from at least 2 nesting sites for multiple RMUs, there were no significant differences in the magnitude of inter-annual variability across RMUs: for leatherback turtles, n = 16 nesting sites across two RMUs (t6 = 1.33, P = 0.23); for hawksbill turtles n = 14 nesting sites across three RMUs (ANOVA, F2,23 = 2.7, P = 0.11); for loggerhead turtles, n = 33 nesting sites across five RMUs (ANOVA, F4,25 = 1.8, P = 0.17). The data for green turtles are from Mazaris et al. (2017), Okuyama et al. (2020), Ferreira et al. (2019), Mohd Salleh et al. (2020), Mortimer et al. (2020), Omeyer et al. (2021), Olendo et al. (2019), Seminoff et al. (2018), Shimada et al. (2021). For loggerhead turtles from Mazaris et al. (2017), Willson et al. (2020), Sönmez et al. (2021), Omeyer et al. (2021). For leatherbacks from Mazaris et al. (2017), Whiting et al. (2020). For hawksbills from Mazaris et al. (2017), Gane et al. (2020), Mortimer et al. (2020), Balladares and Quintero‐Torres (2019). For Kemp’s ridleys from Mazaris et al. (2017). For olive ridleys from Mazaris et al. (2017), Ariano-Sanchez et al. (2020), Sosa-Cornejo et al. (2021), Ossman (2019), Kurniawan & Gitayan (2020). For flatbacks from Mazaris et al. (2017). See Supplementary Information for further details
Discussion
Our results extend the findings of Broderick et al. (2001) by showing that the extent of inter-annual variability in breeding numbers of turtles may vary regionally and in accord with a species’ trophic position. For the first time we identify clear regional patterns in the magnitude of this inter-annual variability for green turtles. There are clearly some caveats to these data comparisons, such as the wide variation in the start and end years for the various nesting number time-series, which might potentially blur any real differences in the extent of inter-annual variability between nesting sites. Nevertheless, a previous examination of 45 nesting sites across four species very clearly showed that for sea turtles the highest inter-annual variability in nesting numbers tended to occur in green turtles (Broderick et al. 2001). Our results from 133 nesting sites both confirm and extend these previous conclusions.
Our findings are consistent with the hypothesis that for herbivorous species there is likely more inter-annual variability in their food availability and so higher variability in annual nesting numbers. So, for example, green turtles are largely herbivorous, feeding on seagrass and macroalgae (Esteban et al. 2020) the abundance and quality of which can change profoundly between years linked to environmental variability (Nowicki et al. 2019). Leatherback turtles had the next highest levels of interannual variability in nest numbers, presumably because, although individuals range very broadly to forage across ocean basins (Bailey et al. 2012), there can be marked inter-annual variability in abundance of their jellyfish prey (Lilley et al. 2009). In contrast, hawksbill and loggerhead turtles tended to show much less variability in annual nesting numbers, consistent with expectations about the inter-annual variability in their food availability. Hawksbill turtles tend to feed on sponges and loggerhead turtles on bivalve molluscs and other benthic invertebrates (Bjorndal 1997). These prey groups are long-lived and show continuous growth so that their abundance likely reflects the integration of environmental conditions across many years and, hence, less inter-annual variability. For some species there is very limited information on diet, such as for flatback turtles (Bjorndal 1997). It is notable that this species showed relatively little variation in annual nesting numbers implying that, like hawksbill and loggerhead turtles, they feed at a relatively high trophic level on prey whose abundance is relatively constant across years. Hence we highlight how inter-annual variability in nesting numbers in sea turtles likely conveys useful information about the consistency of their food resource availability across years supporting the conclusions of others (Saba et al. 2007; Suryan et al. 2009; Van Houtan and Halley 2011).
We showed major differences across and between ocean basins in the magnitude of the inter-annual variability in green turtle nesting numbers, which implies that the stability of their foraging conditions changes systematically over these same spatial scales. While focal studies have examined foraging conditions for green turtles at particular sites (Nowicki et al. 2019), ours is one of the first to draw conclusions about inter-annual variability in foraging conditions for sea turtles across the globe. For green turtles we showed highest inter-annual variability in nesting numbers for nesting sites in Australia including the largest green turtle rookery in the world at Raine Island in the northern Great Barrier Reef. The El Nino Southern Oscillation (ENSO) conditions may drive the forage availability for green turtles in Australasia (Limpus and Nicholls 1988) and hence underpin the large inter-annual variability in nest numbers. ENSO is well known to cause massive perturbations in the weather and so impact a range of broad range of habitats in the Pacific and beyond (Balance et al. 2006). Conditions on the west coast of Australia may also change markedly across years due to ENSO events impacting sea temperatures and also variable flows on the warm south-flowing Leeuwin Current (Feng et al. 2003). In some years, for example, oceanographic conditions may drive marine heatwaves and associated sea grass die-off along the west coast of Australia (Arias-Ortiz et al. 2018) and so have a major negative impact of green turtle foraging conditions (Nowicki et al. 2019). ENSO may also impact green turtle foraging conditions in the eastern Pacific and help drive inter-annual variability in reproductive output in that region (Bruno et al. 2020; Ramírez et al. 2021). It has been well established that, across their adult life-spans, sea turtles tend to have tight fidelity to individual foraging sites (Shimada et al. 2020). So generally their strategy appears to be to largely endure years where forage availability is poor, for example due to environmental forcing, rather than relocate to other more suitable areas. Taken together our results point to high variability in both environmental and foraging conditions for green turtles that nest in Australia. This finding has wider implications for censuses to detect changes in population size, which is central to assessing the population status of sea turtles (Whiting et al. 2020). While it is well understood that long-term data are often needed to assess population trends in sea turtles (e.g. Mazaris et al. 2017), in addition, as the extent of inter-annual variability in nest numbers increases, so it will generally take more years of monitoring to detect any changes in population size.
In the western and central Atlantic, inter-annual variability in green turtle nesting numbers tended to decrease between the Gulf of Mexico/Florida and more equatorial nesting sites. As with the gradient in variability in nesting numbers between the western Indian Ocean and Australasia, this pattern likely reflects a gradient of inter-annual variability in foraging conditions. For example, the Gulf of Mexico region is subjected to marked inter-annual variability in environmental conditions, with environmental perturbations, such as storms, causing major inter-annual changes in the extent of seagrass beds, upon which green turtles forage in the region (Esteban et al. 2020; Rodemann et al. 2021). In contrast to green turtles, regional differences in the extent of inter-annual variability in nest numbers were less evident in other species, consistent with their higher trophic position which likely means that their food availability will tend to show less variability across years (Broderick et al. 2001). Continued assessment of inter-annual variability in sea turtle nest numbers may provide further insights into the impacts of climate change on the foraging ecology of this iconic group, including predicted increases in environmental variability (van Hooidonk et al. 2016).
Data availability
The dataset supporting this study is stored in the electronic supplementary material and the main text.
References
Ariano-Sanchez D, Muccio C, Rosell F, Reinhardt S (2020) Are trends in olive ridley sea turtle (Lepidochelys olivacea) nesting abundance affected by El Nino Southern Oscillation (ENSO) variability ? Sixteen years of monitoring on the Pacific coast of northern Central America. Glob Ecol Conserv 24:e01339. https://doi.org/10.1016/j.gecco.2020.e01339
Arias-Ortiz A, Serrano O, Masqué P, Lavery PS, Mueller U, Kendrick GA, Rozaimi M, Esteban A, Fourqurean JW, Marbà N, Mateo MA, Murray K, Rule MJ, Duarte CM (2018) A marine heatwave drives massive losses from the world’s largest seagrass carbon stocks. Nat Clim Change 8:338–344. https://doi.org/10.1038/s41558-018-0096-y
Bailey H, Fossette S, Bograd SJ, Shillinger GL, Swithenbank AM, Georges J-Y, Gaspar P, Patrik Stromberg KH, Paladino FV, Spotila JR, Block BA, Hays GC (2012) Movement patterns for a critically endangered species, the leatherback turtle (Dermochelys coriacea), linked to foraging success and population status. PLoS ONE 7:e36401. https://doi.org/10.1371/journal.pone.0036401
Balance LT, Pitman RL, Fiedler PC (2006) Oceanographic influences on seabirds and cetaceans of the eastern tropical Pacific: a review. Prog Oceanogr 69:360–390. https://doi.org/10.1016/j.pocean.2006.03.013
Balladares C, Quintero-Torres E (2019) Is a small sea turtles rookery doomed to local extinction? Decreasing nesting trends at the Paria Gulf. Venezuela Mar Ecol 40:e12562. https://doi.org/10.1111/maec
Bjorndal KA (1997) Foraging ecology and nutrition of sea turtles. In: Lutz PL, Musick JA (eds) The biology of sea turtles. CRC Press, Boca Raton, pp 199–231
Broderick AC, Godley BJ, Hays GC (2001) Trophic status drives inter-annual variability in nesting numbers of marine turtles. Proc R Soc Lond B 268:1481–1487. https://doi.org/10.1098/rspb.2001.1695
Bruno RS, Restrepo JA, Valverde R (2020) Effects of El Niño southern oscillation and local ocean temperature on the reproductive output of green turtles (Chelonia mydas) nesting at Tortuguero. Costa Rica Mar Biol 167:128. https://doi.org/10.1007/s00227-020-03749-z
Edwards M, Beaugrand G, Hays GC, Koslow JA, Richardson AJ (2010) Multi-decadal oceanic ecological datasets and their application in marine policy and management. Trends in Ecol Evol 25:602–610. https://doi.org/10.1016/j.tree.2010.07.007
Esteban N, Mortimer JA, Stokes HJ, Laloë J-O, Unsworth RKF, Hays GC (2020) A global review of green turtle diet: sea surface temperature as a potential driver of omnivory levels. Mar Biol 167:183. https://doi.org/10.1007/s00227-020-03786-8
Feng M, Meyers G, Pearce A, Wijffels S (2003) Annual and interannual variations of the Leeuwin Current at 32S. J Geophys Res 108:3355. https://doi.org/10.1029/2002JC001763
Ferreira RL, Martins HR, Bolten AB (2019) Hawksbill (Eretmochelys imbricata) and green turtle (Chelonia mydas) nesting and beach selection at Príncipe Island, West Africa. Arquipel Life Mar Sci 36:61–77. https://doi.org/10.25752/arq.19680
Gane J, Downs CT, Olivier I, Brown M (2020) Nesting ecology and hatching success of the hawksbill turtle (2004–2014) on Cousine Island, Seychelles. Afr J Mar Sci 42:53–65. https://doi.org/10.2989/1814232X.2020.1727952
Gardner JL, Rowley E, de Rebeira P, de Rebeira A, Brouwer L (2018) Associations between changing climate and body condition over decades in two southern hemisphere passerine birds. Clim Chang Responses 5:2. https://doi.org/10.1186/s40665-018-0038-y
Hays GC (2000) The implications of variable remigration intervals for the assessment of population size in marine turtles. J Theor Biol 206:221–227. https://doi.org/10.1006/jtbi.2000.2116
Jouventin P, Dobson FS (2002) Why breed every other year? The case of albatrosses. Proc R Soc Lond B 269:1955–1961. https://doi.org/10.1098/rspb.2002.2080
Kurniawan N, Gitayan A (2020) Why did the population of the Olive Ridley Turtle Lepidochelys olivacea (Eschscholtz, 1829) increase in Alas Purwo National Park’s Beach, East Java, Indonesia? Russ J Mar Biol 46:338–345. https://doi.org/10.1134/S1063074020050065
Lilley MKS, Houghton JDR, Hays GC (2009) A review of distribution, extent of inter-annual variability and diet of the bloom-forming jellyfish Rhizostoma in European waters. J Mar Biol Assoc UK 89:39–48. https://doi.org/10.1017/S0025315408002439
Limpus CJ, Nicholls N (1988) The southern oscillation regulates the annual numbers of green turtles (Chelonia mydas) breeding around northern Australia. Aust J Wildlife Res 15:157–161. https://doi.org/10.1071/WR9880157
Mazaris AD, Schofield G, Gkazinou C, Almpanidou V, Hays GC (2017) Global sea turtle conservation successes. Sci Adv 3:e1600730. https://doi.org/10.1126/sciadv.1600730
Mohd Salleh S, Nishizawa H, Mohd Sah SA, Khan Chowdhury AJ (2020) Reproductive seasonality and environmental effects in green turtle (Chelonia mydas) nesting at Penang Island, Malaysia. J Mar Biol Assoc UK 100:645–650. https://doi.org/10.1017/S0025315420000399
Mortimer J, Esteban N, Guzman AN, Hays GC (2020) Estimates of marine turtle nesting populations in the south-west Indian ocean indicate the importance of the Chagos Archipelago. Oryx 54:332–343. https://doi.org/10.1017/S0030605319001108
Nowicki R, Heithaus M, Thomson J, Burkholder D, Gastrich K, Wirsing A (2019) Indirect legacy effects of an extreme climatic event on a marine megafaunal community. Ecol Mono 83:e01365. https://doi.org/10.1002/ecm.1365
Okuyama J, Ishii H, Tanizaki S, Suzuki T, Abe O, Nishizawa H, Yano A, Tsujimura M, Ishigaki T, Kobayashi M, Yanagida H (2020) Quarter-century (1993–2018) nesting trends in the peripheral populations of three sea turtle species at Ishigakijima Island, Japan. Chelonian Conserv Biol 19:101–110. https://doi.org/10.2744/CCB-1428.1
Olendo MI, Okemwa GM, Munga CN, Mulupi LK, Mwasi LD, Mohamed HB, Sibanda M, Ong’anda H (2019) The value of long-term, community-based monitoring of marine turtle nesting: a study in the Lamu archipelago, Kenya. Oryx 53:71–80. https://doi.org/10.1017/S0030605317000771
Omeyer LCM, Stokes KL, Beton D, Cicek BA, Davey S, Fuller WJ, Godley BJ, Sherley RB, Snape RTE, Broderick AC (2021) Investigating differences in population recovery rates of two sympatrically nesting sea turtle species. Anim Conserv 24:832–846. https://doi.org/10.1111/acv.12689
Ossmann M (2019) Sea turtle nesting trends from 2011–2017 on the Osa Peninsula. Masters project, Duke University, North Carolina USA, Costa Rica
Ozgul A, Childs DZ, Oli MK, Armitage KB, Blumstein DT, Olson LE, Tuljapurkar S, Coulson T (2010) Coupled dynamics of body mass and population growth in response to environmental change. Nature 466:482–485. https://doi.org/10.1038/nature09210
Pinsky ML, Selden RL, Kitchel ZJ (2020) Climate-driven shifts in marine species ranges: scaling from organisms to communities. Ann Rev Mar Sci 12:153–179. https://doi.org/10.1146/annurev-marine-010419-010916
Poloczanska ES, Burrows MT, Brown CJ, García Molinos J, Halpern BS, Hoegh-Guldberg O, Kappel CV, Moore PJ, Richardson AJ, Schoeman DS, Sydeman WJ (2016) Responses of marine organisms to climate change across oceans. Front Mar Sci 3:66. https://doi.org/10.3389/fmars.2016.00062
Ramírez H, Valverde-Cantillo V, Santidrián Tomillo P (2021) El Niño events and chlorophyll levels affect the reproductive frequency but not the seasonal reproductive output of east Pacific green turtles. Mar Ecol Prog Ser 659:237–246. https://doi.org/10.3354/meps13587
Rodemann JR, James WR, Santos RO, Furman BT, Fratto ZW, Bautista V, Lara Hernandez J, Viadero NM, Linenfelser JO, Lacy LA, Hall MO, Kelble CR, Kavanagh C, Rehage JS (2021) Impact of extreme disturbances on suspended sediment in western Florida Bay: implications for seagrass resilience. Front Mar Sci 8:633240. https://doi.org/10.3389/fmars.2021.633240
Saba VS, Santidrian P, Reina RD, Spotila JR, Musick JA, Evans DA, Paladino FV (2007) The effect of the El niño southern oscillation on the reproductive frequency of eastern Pacific leatherback turtles. J Appl Ecol 44:395–404. https://doi.org/10.1111/j.1365-2664.2007.01276.x
Seminoff J, Bustos L, Quiñones J, Espinoza E (2018) Inter-American convention for the protection and conservation of sea turtles green turtle (Chelonia mydas) nesting trends in the eastern Pacific Ocean: Status update and conservation priorities. Technical Document CIT-CC15–2018-Tec.15\, p 10
Shimada T, Limpus CJ, Hamann M, Bell I, Esteban N, Groom R, Hays GC (2020) Fidelity to foraging sites after long migrations. J Anim Ecol 89:1008–1016. https://doi.org/10.1111/1365-2656.13157
Shimada T, Meekan MG, Baldwin R, Al-Suwailem AM, Clarke C, Santillan AS, Duarte CM (2021) Distribution and temporal trends in the abundance of nesting sea turtles in the Red Sea. Biol Cons 261:109235. https://doi.org/10.1016/j.biocon.2021.109235
Sönmez B, Elginöz E, Ilgaz M, Altınkaya H (2021) Nesting activity of loggerhead turtles (2013–2020) and 20 years abundance trend (2001–2020) on Çıralı Beach. Turk Reg Stud Mar Sci 44:101758. https://doi.org/10.1016/j.rsma.2021.101758
Sosa-Cornejo I, Martín-del-Campo R, Contreras-Aguilar HR, Enciso-Saracho F, González-Camacho ZB, Guardado-González JI, Campista-Leon S, Peinado- Guevara LI (2021) Nesting trends of olive ridley sea turtles Lepidochelys olivacea (Testudinata: Cheloniidae) on two beaches in northwestern Mexico after 30 and 40 years of conservation. Revista Biol Trop 69:1124–1137. https://doi.org/10.15517/rbt.v69i3.46490
Suryan RM, Saba VS, Wallace BP, Hatch SA, Frederiksen M, Wanless S (2009) Environmental forcing on life history strategies: evidence for multi-trophic level responses at ocean basin scales. Prog Oceanogr 81:214–222. https://doi.org/10.1016/j.pocean.2009.04.012
Van Hooidonk R, Maynard J, Tamelander J, Gove J, Ahmadia G, Raymundo L, Williams G, Heron SF, Planes S (2016) Local-scale projections of coral reef futures and implications of the Paris agreement. Sci Rep 6:39666. https://doi.org/10.1038/srep39666
Van Houtan KS, Halley JM (2011) Long-term climate forcing in loggerhead sea turtle nesting. PLoS ONE 6:e19043. https://doi.org/10.1371/journal.pone.0019043
Wallace BP, DiMatteo AD, Hurley BJ, Finkbeiner EM, Bolten AB, Chaloupka MY, Hutchinson BJ, Abreu-Grobois FA, Amorocho D, Bjorndal KA, Bourjea J, Bowen BW, Dueñas RP, Casale P, Choudhury BC, Costa A, Dutton PH, Fallabrino A, Girard A, Girondot M, Godfrey MH, Hamann M, López-Mendilaharsu M, Marcovaldi M, Mortimer JA, Musick JA, Nel R, Pilcher NJ, Seminoff JA, Troëng S, Witherington B, Mast RB (2010) Regional management units for marine turtles: a novel framework for prioritizing conservation and research across multiple scales. PLoS ONE 5:e15465. https://doi.org/10.1371/journal.pone.0015465
Whiting AU, Chaloupka M, Limpus CJ (2020) Sampling nesting sea turtles: impact of survey error on trend detection. Mar Ecol Prog Ser 634:213–223. https://doi.org/10.3354/meps13202
Willson A, Witherington B, Baldwin R, Tiwari M, Al Sariri T, Al Kiyumi A, Al Harthi S, Willson MS, Al Bulushi A, Al Farsi G, Al Humaidy J, Al Araimi J, Daar LA, Schroeder B, Ross JP, Possardt E (2020) Evaluating the long-term trend and management of a globally important loggerhead population nesting on Masirah Island, Sultanate of Oman. Front Mar Sci 7:666. https://doi.org/10.3389/fmars.2020.00666
Acknowledgements
We are grateful to Alex Rattray for preparing the map in Fig 1a.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This work was supported by the Bertarelli Foundation as part of the Bertarelli Programme in Marine Science (BPMS-2017-4).
Author information
Authors and Affiliations
Contributions
GCH: conceived the study, GS and AM: assembled the data, GS and GCH: analysed the data with support from AM. GCH: led the writing with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
No conflicts of interest or competing interests to declare.
Ethical approval
The work only used data extracted from the literature.
Additional information
Responsible Editor: L. Avens.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hays, G.C., Mazaris, A.D. & Schofield, G. Inter-annual variability in breeding census data across species and regions. Mar Biol 169, 54 (2022). https://doi.org/10.1007/s00227-022-04042-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-022-04042-x