Abstract
The trophic ecology of two dominant intertidal caprellids, Caprella penantis and Caprella grandimana, was studied by examining gut contents. Samples were collected from Tarifa Island (Punta Marroquí, 36°00′00.7″N 5°36′37.5″W) bimonthly from December 2005 to December 2006 to explore seasonal and sex-related differences in diet. The gut contents of the two species included detritus, metazoan prey (crustaceans, polychaetes, hydroids, nematodes, and turbellarians), macroalgae, microalgae, and dinoflagellates. The dominant component was detritus, followed by crustaceans, mainly copepods, and macroalgal tissues. A three-way analysis of variance was conducted using the factors “species” (C. penantis vs. C. grandimana), “sex” (males vs. females), and “month” (seven dates, December 2005–December 2006). Males of both species had a significantly higher proportion of prey and a lower proportion of detritus in their guts than females. The larger body and gnathopod 2 of males appeared to be related to a more carnivorous diet. Both species fed more on crustaceans and macroalgae during the summer (April, June, and August), probably owing to fluctuations in prey availability and seasonal variations in algal abundance. Moreover, C. penantis was significantly more predatory than C. grandimana, but both species had a diet based mainly on detritus and crustaceans. Caprella penantis dwells at the lower intertidal and C. grandimana at the mid-tide level, which may reduce competition between the species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Amphipods are among the most diverse benthic fauna (Dauby et al. 2001; Lourido et al. 2008), constituting a dominant group in soft bottom and marine rocky habitats (de-la-Ossa-Carretero et al. 2010). They represent the most diverse group of crustaceans with respect to lifestyle, habitat, and size (De Broyer and Jazdzewski 1996). The trophic diversity of amphipods at the interspecific level indicates that they have species-specific functional roles as mediators in trophic pathways (Jeong et al. 2012); hence, they play a key role in energy flow through food webs (Vazquez-Luis et al. 2013). Despite the importance of amphipods in marine ecosystems, little is known about the feeding habits of these crustaceans; indeed, functional types have been studied in fewer than 10 % of amphipod species (Guerra-García et al. 2014).
Caprellids, also called skeleton shrimps, are small marine amphipods common to many littoral habitats that inhabit algae, hydroids, ascidians, anthozoans, bryozoans, sponges, and seagrasses. They cling to artificial (buoys, ropes, and litter) and natural (macroalgae) floating material, which facilitates their dispersal. Caprellids are also useful bioindicators of marine pollution and environmental stress (Guerra-García and García-Gómez 2001; Guerra-García and Koonjul 2005), as they have the capacity to accumulate trace metals (Guerra-García et al. 2009) and are extremely sensitive to tributyltin (Takeuchi et al. 2004; Aono and Takeuchi 2008). These amphipods form an important trophic link between primary producers and higher trophic levels (Woods 2009) by feeding on a wide variety of organisms, such as copepods, gammarids, hydroids, sponges, polychaetes, macroalgae, and diatoms (see Guerra-García and Tierno de Figueroa 2009; Alarcón-Ortega et al. 2012; Baeza-Rojano et al. 2014). Moreover, it has been concluded that these amphipods are detritivores (see Guerra-García et al. 2014), but it remains controversial whether they should be considered omnivores. Some recent studies have contributed to knowledge of Caprellidae from the Iberian Peninsula and nearby areas, particularly the Strait of Gibraltar, where there is a very high proportion of endemic caprellid species, contributing 30.8 % to the endemism of the Mediterranean Sea (Guerra-García et al. 2002). Although some research has considered the feeding habits of peracarids in the intertidal zone of the Strait of Gibraltar (Torrecilla-Roca and Guerra-García 2012), seasonal dietary variations of caprellids and intraspecific differences have not been explored.
We chose Caprella penantis and Caprella grandimana to conduct a diet analysis because they are the most common species in the calcareous macroalgae Corallina elongata, which is dominant in the shallow waters of the Mediterranean (Baeza-Rojano et al. 2011). Previous studies in this area reported that C. penantis and C. grandimana are the dominant taxa in the intertidal rocky ecosystems of the Strait of Gibraltar and are associated with Gelidium corneum and Corallinaceae algae (C. elongata and Jania rubens, respectively; see Guerra-García et al. 2011). Caprella penantis requires unperturbed areas subjected to exposure and strong currents that reduce sedimentation and suspended organic matter and solids (Guerra-García and García-Gómez 2001). Caprella penantis has also been proposed as a suitable bioindicator, as it accumulates trace metals at higher concentrations than other amphipods (Guerra-García et al. 2009). The taxonomy of C. penantis is considered one of the most problematic of caprellids worldwide, as it shows wide intraspecific morphological variations due to multihabitat preferences. Cabezas et al. (2013) confirmed cryptic speciation and at least four distinct monophyletic mitochondrial lineages. Only individuals from Tarifa Island were sampled in the present study, which all belonged to the same species of C. penantis sensu stricto. Caprella grandimana is also common throughout the Mediterranean Sea (Krapp-Schickel 1993) and along the Atlantic African coast (Bellan-Santini and Ruffo 1998). This species is mainly associated with the algae C. elongata and J. rubens in the intertidal zones of the Strait of Gibraltar, and it reproduces year around. This species grows and reproduces in captivity on a diet of diatoms alone (Baeza-Rojano et al. 2011), and it is a potential aquaculture resource because of its higher omega-3 fatty acid and polar lipid contents compared with gammarids (Baeza-Rojano et al. 2014).
Amphipod feeding preferences have been traditionally assessed using in situ and laboratory observations, feeding experiments, gut content analyses, and studies of the functional morphology of feeding appendages (Legeżyńska et al. 2012). Knowledge of amphipod feeding ecology has recently expanded owing to the use of biomarkers, such as fatty acid biochemical profiles (Jaschinski et al. 2011; McLeod et al. 2013; Baeza-Rojano et al. 2014) and stable nitrogen (δ15N) and carbon (δ13C) isotopes (Olabarria et al. 2009; Mancinelli 2012; Jeong et al. 2012). However, these techniques are indirect indicators, requiring fresh material and preventing the use of specimens fixed in ethanol or formalin. They also depend on an abundance of available material (particularly in small species) to have sufficient quantities for chemical analyses. In contrast, a gut content examination is a direct indicator, which is an advantage. Traditional studies that directly examine digestive contents are limited in number because they are time-consuming (Guerra-García et al. 2014); therefore, we used the methodology proposed by Bello and Cabrera (1999) to study gut contents. This method makes the specimens transparent so gut contents can be observed.
The objective of this study was to gain insight into the feeding ecology of Caprellidae in the rocky intertidal areas of a relatively pristine ecosystem (Tarifa Island). The fact that the two caprellid species dwell at different heights in the intertidal associated with different macroalgal species could affect their role in the trophic chain of this ecosystem. A series of anatomic features, such as the gnathopod 2 and swimming setae on antenna 2, have been used previously to define the feeding habits of different caprellid species. Moreover, it has been demonstrated that larger crustaceans of the same species with stronger feeding appendages tend to consume more prey (Kapiris et al. 2010); therefore, sex-related differences in carnivorous behavior are expected as a reflection of the sexual dimorphism in C. penantis and C. grandimana. Finally, seasonal variations are a major feature in the dynamics of the rocky intertidal community and have been investigated in other amphipods, such as mesograzer species in an eelgrass community (Jaschinski et al. 2011) or Talitridae (Olabarria et al. 2009). Some caprellid species are known to be opportunists that exploit different organic matter sources throughout the year. The feeding plasticity of the species in our study was assessed as we examined seasonal shifts in their diet.
Materials and methods
Study area and sampling
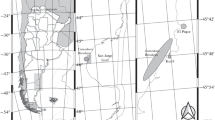
This study was conducted in the Strait of Gibraltar, an important biogeographic zone, in which fauna of the Mediterranean Sea and the Atlantic Ocean along one axis and fauna of Europe and Africa along the other overlap. The Spanish side of the Strait is protected under the marine terrestrial Strait Natural Park (Parque Natural del Estrecho), which was declared a protected area in 2003. Tarifa Island is a marine reserve inside the park with a unique biogeographic position in which long-term military access restrictions have contributed to maintain the richest rocky shore intertidal ecosystems in southern Spain (Guerra-García and García-Gómez 2000). This study was conducted at the most southern point of Tarifa Island (Punta Marroquí, 36°00′00.7″N 5°36′37.5″W). The tidal range at this location is approximately 250 cm (Guerra-García et al. 2011). We considered five levels to establish the zonation of these two species (level 1: from zero tidal level to 0.5 m; level 2: 0.5–1 m; level 3: 1–1.5 m; level 4: 1.5–2 m; and level 5: 2–2.5 m). Previous studies on vertical distribution of peracarid fauna in this area revealed that caprellids are the dominant taxa in levels 1 and 3 (C. penantis and C. grandimana, respectively; Guerra-García et al. 2011). Three replicates (20 × 20-cm quadrats) were sampled every 2 months (from December 2005 to December 2006). The surface was scraped at each level, and all macroalgae and associated fauna were collected.
All samples were fixed immediately after collection in 80 % ethanol to avoid excretion of fecal pellets. The samples were washed over a 0.5-mm mesh sieve in the laboratory, and the amphipods were separated from the algae. The caprellids from the three sampling replicates were sexed using a stereomicroscope and analyzed following the methods of Bello and Cabrera (1999) with slight variations. Individuals with an empty gut were discarded. We examined the digestive contents of 560 specimens (280 C. penantis and 280 C. grandimana or 20 specimens of each sex/month). Specimens of each sex and species were placed in vials containing Hertwig’s liquid (270 g chloral hydrate, 19 ml 1 N HCl, 150 ml distilled water, and 60 ml glycerin) and heated in an oven at 65 °C for 2–5 h depending on cuticle thickness of the specimens. This treatment dissolved the fat in the tissue so the specimens became transparent and their gut contents could be observed. Then, the specimens were mounted on slides for study under the microscope. The percentage of absolute gut content (%Abs) (at 40× or 100×), as the total area occupied in the entire digestive tract, and relative gut content (at 100× or 400×), as the area occupied by each component within all gut contents, were estimated using a microscope equipped with an ocular micrometer.
Data analysis
Mean and SE were calculated to determine the percentages of absolute gut contents and each component of the diet throughout the year. Univariate and multivariate analyses were used to identify potential differences among species and sexes, as well as seasonal fluctuations in the diet, based on the null hypothesis of no change in the diet.
We considered the percentages of detritus and prey to test whether the diet was similar for the sexes of C. penantis and C. grandimana throughout the year. To determine the quantity of prey in the diet, we pooled the percentages of crustaceans, polychaetes, turbellarians, nematodes, and hydroids. We used analysis of variance (ANOVA) with the following factors: “species,” a fixed factor with two levels, C. penantis and C. grandimana; “sex,” a fixed factor with two levels, males and females; and “month,” a fixed factor with seven levels, December 2005, February 2006, April 2006, June 2006, August 2006, October 2006, and December 2006. All factors were orthogonal. Twenty replicates were used. Prior to the ANOVA, heterogeneity of variance was tested with Cochran’s C test. Variances remained heterogeneous, even when the data were transformed. Therefore, untransformed data were analyzed, as ANOVA is a robust statistical test and is relatively unaffected by heterogeneity of variances, particularly in balanced experiments with a relatively large number of replicates (Underwood 1997). However, to reduce type I error, the level of significance was reduced to <0.01. When ANOVA indicated a significant difference, the source of the difference was identified using the Student–Newman–Keuls (SNK) test. Univariate analyses were conducted with GMAV5 (Underwood et al. 2002).
Nonparametric multidimensional scaling (MDS) was used as the ordination method to explore differences in the caprellid diets (Clarke and Warwick 1994). Data were square root transformed, and the similarity matrix was calculated using the Bray–Curtis index.
Results
We examined the digestive contents of 560 specimens (280 C. penantis and 280 C. grandimana). The average area occupied by the contents of the entire digestive tract ranged from 42 to 84 % in C. penantis and from 25 to 73 % in C. grandimana (Tables 1, 2). Gut contents of the caprellid species included detritus, metazoan prey (crustaceans, polychaetes, hydroids, nematodes, and turbellarians), macroalgae, microalgae, and dinoflagellates. The dominant component was detritus, which represented 87.6 ± 1.6 % (n = 280) [mean ± SE, n] of the C. penantis diet and 90.1 ± 1.2 % (n = 280) of the C. grandimana diet, followed by crustaceans, mainly copepods, and macroalgae. Crustaceans represented 10.4 ± 1.6 % (n = 280) in C. penantis and 6.27 ± 1.37 % (n = 280) in C. grandimana. Macroalgal tissues represented 1.23 ± 0.43 % (n = 280) in C. penantis and 2.42 ± 0.65 % (n = 280) in C. grandimana. Diatoms and polychaetes were present occasionally. Dinoflagellates, together with the remaining prey, such as hydroids, turbellarians, and nematodes, were scarcely represented, with percentages of 0.03 % (hydroids) to 0.07 % (turbellarians) in C. penantis and 0.01 % (hydroids and dinoflagellates) to 0.04 % (turbellarians) in C. grandimana. Polychaetes were detected by the presence of their setae, and hydroids were detected by the presence of their cnidocytes. No nematodes were found in C. penantis (Tables 1, 2). Therefore, these two caprellids species fed mainly on detritus, but they complete their diet with a number of different prey and vegetal tissues.
The percentages of prey in the diets of C. penantis and C. grandimana differed (significant effect of species, F = 20.77, P < 0.001), and they were consistent over time and between sexes (no significant species × month interaction, F = 1.56, P > 0.01; or species × sex interaction, F = 0.79, P > 0.01) (Table 3). Caprella penantis had a higher percentage of prey (10.7 ± 0.8 %, n = 280) than that of C. grandimana (6.63 ± 0.64 %, n = 280). The pattern for the percentage of detritus was the opposite: C. penantis had a significantly lower percentage of detritus (87.6 ± 1.6 %, n = 280) than that of C. grandimana (90.6 ± 1.2 %, n = 280) (significant effect of species, F = 8.54, P < 0.01), and these differences were consistent over time and between sexes (no significant species × month interaction, F = 2.03, P > 0.01; or species × sex interaction, F = 0.01, P > 0.01) (Table 4).
Values for the same variables also differed throughout the year (significant effect of month, F = 6.26, P < 0.001 for prey and F = 6.66, P < 0.001 for detritus), with a similar pattern for both species that was consistent in both sexes (no significant month × sex interaction, F = 2.24, P > 0.01 for prey and F = 2.76, P > 0.01 for detritus) (Tables 3, 4). Lower percentages of prey and macroalgal tissues tended to be found in the diet during cold months and a higher percentage during warmer months. October showed an intermediate value (Fig. 1). December 2005 and February 2006 showed significantly lower prey values (5.13 ± 1.13 %, n = 80 and 5.04 ± 0.95 %, n = 80, respectively) than April (9.69 ± 1.51 %, n = 80) (SNK, P < 0.05), June (9.31 ± 1.38 %, n = 80) (SNK, P < 0.05 but not significant for February 2006), and August (12.9 ± 1.7 %, n = 80) (SNK, P < 0.01). As expected, the amount of detritus in the diet also fluctuated during the year. The caprellids seemed to feed more on detritus during cold months, but this percentage decreased during summer. The percentages of detritus from December 2005 (93.2 ± 1.3 %, n = 80) and February 2006 (94.1 ± 0.9 %, n = 80) were significantly higher than those for April (86.2 ± 1.7 %, n = 80) (SNK, P < 0.01), June (88.1 ± 1.4 %, n = 80) (SNK, P < 0.05), and August (85.2 ± 1.7 %, n = 80) (SNK, P < 0.01). Moreover, the percentages of polychaetes, turbellarians, nematodes, hydroids, and dinoflagellates remained <1 % except in February 2006 for C. penantis and December 2005 for C. grandimana. Detritus was the dominant component throughout the year in both species, followed by crustaceans.
The largest differences appeared when values were compared between sexes (significant effect of sex, F = 37.77, P < 0.001 for detritus and F = 81.56, P < 0.001 for prey). Males of C. penantis and C. grandimana were clearly more carnivorous than females (Fig. 2). Males fed more on prey (12.8 ± 0.8 %, n = 280) and less on detritus (86.1 ± 1.2 %, n = 280) than females (4.60 ± 0.47 %, n = 280 and 92.2 ± 1.2 %, n = 280, respectively). In addition, males and females were clearly separated in the two-dimensional MDS plot (Fig. 3). Consequently, the sex of the specimens had an effect regardless of species and time of year when all components of the diet were considered.
Discussion
Our results revealed that both species feed mainly on detritus (>85 % in C. penantis and 90 % in C. grandimana) throughout the year and supplement their diets with crustaceans (>10 % in C. penantis and >6 % in C. grandimana). Macroalgal tissues (>1 % in C. penantis and >2 % in C. grandimana) and diatoms were less represented, suggesting that these species do not selectively eat microalgae under natural conditions. Dinoflagellates and a wide range of metazoan prey (polychaetes, nematodes, turbellarians, and hydroids) were also found but contributed very little to the diet. A similar pattern was reported for these two species in a previous dietary analysis (Guerra-García and Tierno de Figueroa 2009).
Detritus was always the greatest contributor to the diets of these caprellids, which was consistent with several studies (Jeong et al. 2012; Vazquez-Luis et al. 2013; Guerra-García et al. 2014). Detritus plays a key role in marine invertebrate food webs and trophic pathways in marine ecosystems (Valiela 1995). More energy and materials flow through detrital food webs than through grazer food webs in most freshwater, estuarine, and marine ecosystems, and many animals use detritus directly because it is highly nutritious after a short period of microbial colonization (Mann 1998). In addition, amphipod species richness and total abundance increase with an increase in detrital content (Vazquez-Luis et al. 2012). However, whether caprellids should be considered detritivores or omnivores has been widely debated, as the limits for distinguishing between these two strategies are difficult to establish based on percentages of each item. Many authors consider that >95 % detritus in the diet is indicative of a detritivore (Guerra-García et al. 2014), while amphipods that feed alternatively on different trophic resources, such as detritus, small invertebrates, and meiobenthos, are considered omnivores (Fanelli et al. 2009; Navarro-Barranco et al. 2013). We suggest that C. penantis and C. grandimana should be considered omnivores based on these definitions. Vazquez-Luis et al. (2013) considered C. grandimana to be an omnivore and Guerra-García and Tierno de Figueroa (2009) referred to both species as opportunistic.
The second most abundant component after detritus was crustaceans (mainly copepods and some gammarids), which were detected by their mouthparts, broken antennae and gnathopods, or other appendages. Both species live and graze on macroalgae, but this component can have a relatively high content of indigestible fiber and low nitrogen content (Mann 1998). Animal matter is more easily assimilated. Caprellids display cannibalism when food is inadequate, but we found no evidence of cannibalism in the guts we examined. We considered the percentage of particular prey in the gut as an indication of predation by these species. In fact, prey densities are important when investigating variations in dietary composition of benthic amphipods (Yu et al. 2003).
It is common for opportunistic amphipods to change their feeding strategy if trophic resources change owing to environmental factors. Consequently, it seems logical that prey availability can greatly influence feeding habits (Jeong et al. 2012; Ros et al. 2014) and metabolic rate, allowing for highly efficient energy utilization (Doyle et al. 2012). Thus, seasonal peaks in macroalgae and copepod abundance would coincide with greater consumption of these components by C. penantis and C. grandimana. Several studies in the intertidal areas of Tarifa Island during the same year reported seasonal fluctuations in seaweed (Guerra-García et al. 2011), which are related to cyclic variations in environmental factors, such as temperature, day length, and wave action (Neto 2000). The macroalgal contribution to the diet increased in both species from April to August, when maximum seaweed biomass occurs, whereas both species decreased their feeding on macroalgae during cold months when seaweed abundance was at its lowest. A similar seasonal abundance pattern has been confirmed for copepods in a number of freshwater (Iskaros and El-Otify 2012) and marine ecosystems worldwide (Jaschinski et al. 2011; Kurt and Polat 2012; Rajkumar et al. 2013). Therefore, the higher percentage of prey consumed during summer can be explained by an increase in crustacean availability from April to August. The ability to vary feeding preference in response to prey availability is quite common in marine vertebrates (Varo et al. 2011; Griffin et al. 2012; Matić-Skoko et al. 2014) and in several invertebrate phyla, such as gorgonians (Leal et al. 2014), nemerteans (Caplins et al. 2012), and echinoderms (Chiantore et al. 2002). Soler-Membrives et al. (2011) found a very similar trend to ours in the trophodynamics of the pycnogonid Ammothella longipes, which appears to be carnivorous during spring and early summer, but prefers detritus when prey availability diminishes during winter. Opportunistic feeding has been reported in amphipods on several occasions (Werner and Auel 2005; Nunez-Pons et al. 2012), as well as in herbivorous species, such as some talitrids (Bessa et al. 2014). Opportunistic feeding can be a key factor in the success of an invasive species (Maazouzi et al. 2007; Cook et al. 2009). Moreover, some opportunistic, Arctic, under-ice amphipod species switch from a predominantly herbivorous diet in summer to metazoan prey, including planktonic copepods in winter, when ice algae are scarce and they must rely on stored lipids as an energy source (Werner and Auel 2005).
Diet composition varied between the species we studied. These differences were also observed in fatty acid-based nutritional analyses by Guerra-García et al. (2004) and Baeza-Rojano et al. (2014). Caprella grandimana consumed more diatoms and macroalgae than C. penantis, which is more carnivorous. Caine (1977) reported that a combination of weak swimming setae on antenna 2 and a long basis of gnathopod 2 suggests a predatory mode of life, but our results suggest that C. grandimana, which bears these characteristics, has a less predatory mode of life than C. penantis. We support the conclusions of Guerra-García and Tierno de Figueroa (2009) that there are not always clear relationships between morphological characters and digestive content. The higher percentage of crustaceans found in C. penantis than that in C. grandimana may be related to body size differences. Poltermann (2001) reported that crustaceans are present in considerably lower proportions in the guts of smaller species and young or small individuals than those of larger species of Arctic amphipods. Jaschinski et al. (2011) reached the same conclusion regarding significantly higher δ15N values in larger amphipods, and Felten et al. (2008) also suggested that greater predatory behavior by freshwater amphipods is related to body size. Despite these variations, we found that both species fed mainly on detritus and crustaceans throughout the year. Nelson (1979) also reported an overlap in the diets of several amphipod species and argued that food competition does not play a large role in the structure of the amphipod community in an eelgrass ecosystem where great quantities of detritus are present year around. However, rocky intertidal habitats constitute steep vertical gradients, composed of a patchy mosaic of assemblages in which competition for a limiting resource is important. Therefore, these environmental characteristics can strongly affect community composition (Crain and Bertness 2006; Masterson et al. 2008). The presence of C. penantis in the lower intertidal (mainly associated with G. corneum) and C. grandimana at intermediate levels (mainly associated with C. elongata and J. rubens) avoids interspecific competition. Finally, anatomic features may also be why adult C. penantis and C. grandimana males were more carnivorous than adult females. Significant intraspecific differences have been reported as fairly common among marine organisms (Bearhop et al. 2006; Hoeinghaus and Davis 2007; Olabarria et al. 2009). The structures of crustacean feeding appendages have been suggested to influence the diet, as they determine handling ability and strength (Kolts et al. 2013). The larger gnathopod 2 in males than females is probably related to a more predatory strategy, as it allows the male to catch and manipulate a greater number of prey.
The gut examination methodology used in this study has been applied previously by entomologists (Tierno de Figueroa et al. 2006) and was successfully used for amphipods (Navarro-Barranco et al. 2013; Vazquez-Luis et al. 2013; Guerra-García et al. 2014). Gut content analyses are an accurate direct indicator, which is an advantage compared with indirect indicators, such as stable isotope and fatty acid analyses. However, a combination of these methods, together with mouthpart morphological studies and behavioral observations, is needed to obtain complete data on caprellid feeding habits and food web interactions in ecosystems where they dominate (Vazquez-Luis et al. 2013), particularly when studying seasonal variations. Although our understanding of amphipod feeding habits has increased during the last few years, several authors have pointed out that qualitative and quantitative characterizations of the amphipod species-specific trophic ecology are needed to better understand their potential role in the trophic dynamics, structure, and nutrient fluxes in marine ecosystems (Graeve et al. 2001; Jeong et al. 2012). In this sense, our study represents the first comprehensive dietary analysis of Caprellidae to investigate seasonal and intraspecific variations. Further research is necessary to establish trophic preferences of other amphipods and their roles in the food web of the highly variable intertidal zone.
References
Alarcón-Ortega LC, Guerra-García J, Sánchez-Moyano JE, Cupul-Magaña FG (2012) Feeding habits of caprellids (Crustacea: Amphipoda) from the west coast of Mexico. Do they feed on their hosting substrates? Zool Baetica 23:11–20
Aono A, Takeuchi I (2008) Effects of tributyltin at concentrations below ambient levels in seawater on Caprella danilevskii (Cruastacea: Amphipoda: Caprellidae). Mar Poll Bull 57:515–523
Baeza-Rojano E, Guerra-García JM, Cabezas MP, Pacios I (2011) Life history of Caprella grandimana (Crustacea: Amphipoda) reared under laboratory conditions. Mar Biol Res 7:85–92
Baeza-Rojano E, Hachero-Cruzado I, Guerra-García JM (2014) Nutritional analysis of freshwater and marine amphipods from the Strait of Gibraltar and potential aquaculture applications. J Sea Res 85:29–36
Bearhop S, Phillips RA, McGill R, Cherel Y, Dawson DA, Croxall JP (2006) Stable isotopes indicate sex-specific and long-term individual foraging specialization in diving seabirds. Mar Ecol Prog Ser 311:157–164
Bellan-Santini D, Ruffo S (1998) Faunistics and Zoogreography. In: Ruffo S (ed) The Amphipoda of the Mediterranean, Musée Oceanographique, Monaco 13, pp 895–911
Bello CL, Cabrera MI (1999) Uso de la técnica microhistológica de Cavender y Hansen en la identificación de insectos acuáticos. Bol Entom Venez 14:77–79
Bessa F, Baeta A, Marques JC (2014) Niche segregation amongst sympatric species at exposed Sandy shores with contrasting wrack availabilities illustrated by stable isotopic analysis. Ecol Indic 36:694–702
Cabezas MP, Cabezas P, Machordom A, Guerra-García JM (2013) Hidden diversity and cryptic speciation refute cosmopolitan distribution in Caprella penantis (Crustacea: Amphipoda: Caprellidae). J Zoolog Syst Evol Res 51:85–99
Caine EA (1977) Feeding mechanisms and possible resource partitioning of the Caprellidae (Crustacea: Amphipoda) from Puget Sound, USA. Mar Biol 42:331–336
Caplins S, Penna-Díaz MA, Godoy E, Valdivia N, Turbeville JM, Thiel M (2012) Activity patterns and predatory behavior of an intertidal nemertean from rocky shores: Prosorhochmus nelsoni (Hoplonemertea) from the Southeast Pacific. Mar Biol 159:1363–1374
Chiantore M, Cattaneo-Vietti R, Elia L, Guidetti M, Antonini M (2002) Reproduction and condition of the scallop Adamussium colbechki (Smith 1902), the sea-urchin Sterechinus neumayeri (Meissner1900) and the sea-star Odontaster validus Koehler 1911 at Terra Nova Bay (Ross Sea): different strategies related to…. Polar Biol 25:251–255
Clarke KR, Warwick RM (1994) Changes in marine communities: an approach to statistical analysis and interpretation. Natural Environment Research Council: Plymouth Marine Laboratory, Plymouth
Cook EJ, Shucksmith R, Orr H, Ashton GV, Berge J (2009) Fatty acid composition as a dietary indicator of the invasive caprellid, Caprella mutica (Crustacea: Amphipoda). Mar Biol 157:19–27
Crain CM, Bertness MD (2006) Ecosystem engineering across environmental gradients: implications for conservation and management. Bioscience 56:211–218
Dauby P, Scailteur Y, De Broyer C (2001) Trophic type diversity within the eastern Weddell Sea amphipod community. Hydrobiologia 443:69–86
De Broyer C, Jazdzewski K (1996) Biodiversity of the Southern Ocean: towards a new synthesis for the Amphipoda (Crustacea). Bool Mus iv Stor Nat Verona 20:547–568
De-la-Ossa-Carretero JA, Dauvin JC, Del-Pilar-Ruso Y, Giménez-Casalduero F, Sánchez-Lizaso JL (2010) Inventory of benthic amphipods from fine sand community of the Iberian Peninsula east coast (Spain), western Mediterranean, with new records. Mar Biodivers Records. doi:10.1017/S1755267210001065
Doyle SR, Momo FR, Brêthes JC, Ferreyra GA (2012) Metabolic rate and food availability of the Antarctic amphipod Gondogeneia antarctica (Chevreux 1906): seasonal variation in allometric scaling and temperature dependence. Polar Biol 35:413–424
Fanelli E, Cartes JE, Badalamenti F, Rumolo P, Sprovieri M (2009) Trophodynamics of suprabenthic fauna on coastal muddy bottoms of the southern Tyrrhenian Sea (western Mediterranean). J Sea Res 61:174–187
Felten V, Tixier G, Guerold F, De Billy VDC, Dangles O (2008) Quantification of diet variability in a stream amphipod: implications for ecosystem functioning. Fund Appl Limnol 170:303–313
Graeve M, Dauby P, Scailteur Y (2001) Combined lipid, fatty acid and digestive tract content analyses: a penetrating approach to estimate feeding modes of Antarctic amphipods. Polar Biol 24:853–862
Griffin R, Pearce B, Handy RD (2012) Dietary preference and feeding selectivity of common dragonet Callionyus lyra in UK. J Fish Biol 81:1019–1031
Guerra-García JM, García-Gómez (2000) La fauna submarina de la Isla de las Palomas (Tarifa, Cádiz). Temas de flora, fauna y ecología del Campo de Gibraltar. Cuadernos del Instituto, Campo de Gibraltar, Cádiz, Spain, pp 7–17. II
Guerra-García JM, Koonjul MS (2005) Metaprotella sandalensis (Crustacea: Amphipoda: Caprellidae): a bioindicator of nutrient enrichment on coral reefs? A preliminary study at Mauritius Island. Environ Monit Assess 104:353–367
Guerra-García JM, Tierno de Figueroa JM (2009) What do caprellids (Crustacea: Amphipoda) feed on? Mar Biol 156:1881–1890
Guerra-García JM, García-Gómez JC (2001) The spatial distribution of Caprellidea (Crustacea: Amphipoda): a stress bioindicator in Ceuta (North Africa, Gibraltar area). Mar Ecol 22:357–367
Guerra-García JM, Corzo J, García-Gómez JC (2002) Clinging behaviour of the Caprellidae (Amphipoda) from the Strait of Gibraltar. Crustaceana 75:41–50
Guerra-García JM, Martínez-Pita I, Pita ML (2004) Fatty acid composition of the Caprellidae (Crustacea: Amphipoda) from the Strait of Gibraltar. Sci Mar 68:501–510
Guerra-García JM, Baeza-Rojano JM, Cabezas MP, Díaz-Pavón JJ, Pacios I, García-Gómez JC (2009) The amphipods Caprella penantis and Hyale schmidtii as biomonitors of trace metal contamination in intertidal ecosystems of Algeciras Bay, Southern Spain. Mar Poll Bull 58:765–786
Guerra-García JM, Baeza-Rojano E, Cabezas MP, García-Gómez JC (2011) Vertical distribution and seasonality of peracarid crustaceans associated with intertidal macroalgae. J Sea Res 65:256–264
Guerra-García JM, Tierno de Figueroa JM, Navarro-Barranco C, Ros M, Sánchez-Moyano JE, Moreira J (2014) Dietary analysis of the marine Amphipoda (Crustacea: Peracarida) from the Iberian Peninsula. J Sea Res 85:508–517
Hoeinghaus DJ, Davis SE III (2007) Size-based trophic shifts of saltmarsh dwelling blue crabs elucidated by dual stable C and N isotope analyses. Mar Ecol-Prog Ser 334:199–204
Iskaros IA, El-Otify AM (2012) Seasonal periodicity of plankton and benthic fauna community structure and diversity in a small North African reservoir. Water Environ J 27:561–574
Jaschinski S, Brepohl DC, Sommer U (2011) Seasonal variation in carbon sources of mesograzers and small predators in an eelgrass community: stable isotope and fatty acid analyses. Mar Ecol Prog Ser 431:69–82
Jeong SJ, Suh HL, Kang CK (2012) Trophic diversity in amphipods within a temperate eelgrass ecosystem as determined by gut contents and C and N isotope analysis. Mar Biol 159:1943–1954
Kapiris K, Thessalou-Legaki M, Petrakis G, Conides A (2010) Ontogenetic shifts and temporal changes in the trophic patterns of the deep-sea red shrimp, Aristaeomorpha foliacea (Decapods: Aristeidae), in the Eastern Ionian Sea (Eastern Mediterranean). Mar Ecol 31:341–354
Kolts JM, Lovvron JR, North CA, Grebmeier JM, Cooper LW (2013) Effects of body size, gender, and prey availability on diets of snow crabs in the northern Bering Sea. Mar Ecol Prog Ser 483:209–220
Krapp-Schickel T (1993) Suborder Caprellidae. In: Ruffo S (ed). The Amphipoda of the Mediterranean, Musée Oceanographique, Monaco 13, pp 773–809
Kurt TT, Polat S (2012) Seasonal distribution of coastal mesozooplankton community in relation to the environmental factors in İskenderun Bay (north-east Levantine, Mediterranean Sea). J Mar Biol Assoc UK 93:1163–1174
Leal MC, Berger SA, Ferrier-Pagès C, Calado R, Brandes J, Frischer ME, Nejstgaard JC (2014) Temporal changes in the trophic ecology of the symbiotic gorgonian Leptogorgia virgulata. Mar Biol 11:2191–2197
Legeżyńska J, Kędra M, Walkusz W (2012) When season does not matter: summer and winter trophic ecology of Arctic amphipods. Hydrobiologia 684:189–214
Lourido A, Moreira H, Troncoso JS (2008) Assemblages of peracarid crustaceans in subtidal sediments from the Ria de Aldán (Galicia, NW Spain). Helgol Mar Res 62:289–301
Maazouzi C, Masson G, Izquierdo MS, Pihan JC (2007) Fatty acid composition of the amphipod Dikerogammarus villosus: feeding strategies and trophic links. Comp Biochem Phys A 147:868–875
Mancinelli G (2012) On the trophic ecology of Gammaridea (Crustacea: Amphipoda) in coastal waters: a European-scale analysis of stable isotopes data. Est Cst Shelf Sci 114:130–139
Mann KH (1998) Production and use of detritus in various freshwater, estuarine and coastal marine ecosystems. Limnol Oceanogr 33:910–930
Masterson P, Arenas FA, Thompson RC, Jenkins SR (2008) Interaction of top down and bottom up factors in intertidal rockpools: Effects on early successional macroalgal community composition, abundance and productivity. J Exp Mar Biol Ecol 363:12–20
Matić-Skoko S, Tutman P, Bojanić Varezić D, Skaramuca D, Đikić D, Lisičić D, Skaramuca B (2014) Food preferences of the Mediterranean moray eel, Muraena helena (Pisces: Muraenidae), in the southern Adriatic Sea. Mar Biol Res 10:807–815
McLeod RJ, Hyndes GA, Hurd CL, Frew RD (2013) Unexpected shifts in fatty acid composition in response to diet in a common littoral amphipod. Mar Ecol Prog Ser 479:1–12
Navarro-Barranco C, Tierno de Figueroa JM, Guerra-García JM, Sánchez-Tocino L, García-Gómez JC (2013) Feeding habits of amphipods (Crustacea: Malacostraca) from shallow soft bottom communities: comparison between marine caves and open habitats. J Sea Res 78:1–7
Nelson WG (1979) An analysis of structural pattern in an eelgrass (Zostera marina L.) amphipod community. J Exp Mar Biol Ecol 39:231–264
Neto AI (2000) Observations on the biology and ecology of selected macroalgae from the littoral of São Miguel (Azores). Bot Mar 43:483–498
Nunez-Pons L, Rodríguez-Arias M, Gómez-Garreta A, Ribera-Siguan A, Avila C (2012) Feeding deterrency in Antarctic marine organisms: bioassays with the omnivore amphipod Cheirimedon femoratus. Mar Ecol Prog Ser 462:163–174
Olabarria C, Incera M, Garrido J, Rofil IF, Rossi F (2009) Intraspecific diet shift in Talitrus saltator inhabiting exposed sandy beaches. Est Cst Shelf Sci 84:282–288
Poltermann M (2001) Arctic sea ice as feeding ground for amphipods-food sources and strategies. Polar Biol 24:89–96
Rajkumar M, Sun J, Jenkinson IR, Rahman MM (2013) Seasonal variations in the structure of copepod assemblages in tropical marine and estuarine waters, Coleroon, south-east India. J Mar Biol Assoc UK 94:521–535
Ros M, Tiernos de Figueroa JM, Guerra-García JM, Navarro-Barranco C, Lacerda MB, Vázquez-Luis M, Masunari S (2014) Exploring trophic strategies of exotic caprellids (Crustacea: Amphipoda): Comparison between habitat types and native vs. introduced distribution ranges. Est Cst Shelf Sci 139:88–98
Soler-Membrives A, Rossi S, Munilla T (2011) Feeding ecology of Ammothella longipes (Arthropoda: Pycnogonida) in the Mediterranean Sea: A fatty acid biomarker approach. Est Cst Shelf Sci 92:588–597
Takeuchi I, Takahashi S, Tanabe S, Miyazaki N (2004) Butylin concentrations along the Japanese coast from 1997 to 1999 monitored by Caprella spp. (Crustacea: Amphipoda). Mar Environ Res 57:397–414
Tierno de Figueroa JM, Vera A, López-Rodríguez MJ (2006) Adult and nymphal feeding in the stonefly species Antarctoperla michaelseni and Limnoperla jaffueli from Central Chile (Plecoptera: Gripopterygidae). Entomol Gen 29:39–45
Torrecilla-Roca I, Guerra-García JM (2012) Feeding habits of the peracarid crustaceans associated to the algae Fucus spiralis in Tarifa Island, Cádiz (Southern Spain). Zool Baetica 23:39–47
Underwood AJ (1997) Experiments in Ecology: their logical design and interpretation using analysis of variance. Cambridge University Press, Cambridge
Underwood AJ, Chapman MG, Richards SA (2002) GMAV-5 for Windows. An analysis of variance programme. Centre for research on ecological impacts of coastal cities. Marine Ecology Laboratories, University of Sydney, Australia
Valiela I (1995) Marine ecological processes. Springer, New York
Varo N, Green AJ, Sánchez MI, Ramo C, Gómez J, Amat JA (2011) Behavioural and population responses to changing availability of Artemia prey by moulting black-necked grebes, Podiceps nigricollis. Hydrobiologia 664:163–171
Vazquez-Luis M, Borg JA, Sánchez-Jerez P, Bayle-Sempere JT (2012) Habitat colonisation by amphipods: Comparison between native and alien algae. J Exp Mar Biol Ecol 432–433:162–170
Vazquez-Luis M, Sánchez-Jerez P, Bayle-Sempere JT (2013) Does the invasion of Caulerpa racemosa var.cylindracea affect the feeding habits of amphipods (Crustacea: Amphipoda). J Mar Biol Assoc UK 93:87–94
Werner I, Auel H (2005) Seasonal variability in abundance, respiration and lipid composition of Arctic under-ice amphipods. Mar Ecol Prog Ser 292:251–262
Woods CMC (2009) Caprellid amphipods: an overlooked marine finfish aquaculture resource? Aquaculture 289:199–211
Yu OH, Suh H-L, Shirayama Y (2003) Feeding ecology of three amphipod species Synchelidium lenorostralum, S. trioostegitum and Gitanopsis japonica in the surf zone of a sandy shore. Mar Ecol-Prog Ser 258:189–199
Acknowledgments
Special thanks to the Director of the Parque Natural del Estrecho and to the Comandancia General de la Guardia Civil for providing authorization and facilitating access to the Isla de Tarifa marine reserve. Thanks are also due to M.M. López, M.J. Jiménez, D. Vázquez, I. Pacios, A. García, D. González, and J.J. Díaz for field assistance and sample sorting. Financial support for this study was provided by the Ministerio de Economía y Competitividad (Project CGL2011-22474, internal reference 2011-707) co-financed by FEDER funds of the European Union and by the Consejería de Economía, Innovación, Ciencia y Empleo, Junta de Andalucía (Project P11-RNM-7041).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: J. Grassle.
Reviewed by undisclosed experts.
Rights and permissions
About this article
Cite this article
Martínez-Laiz, G., Guerra-García, J.M. Dietary analysis of caprellids Caprella penantis and Caprella grandimana (Crustacea: Amphipoda) in southern Spain. Mar Biol 162, 2057–2066 (2015). https://doi.org/10.1007/s00227-015-2733-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-015-2733-6