Abstract
In a recent study, we showed that tactile perception can be enhanced by applying a placebo manipulation consisting of verbal suggestion and conditioning (Fiorio et al., Neuroscience 217:96–104, 2012). Whether this change in perception is related to a better tactile functioning is still unknown. Aim of this study is to investigate whether placebo-induced enhancement of tactile perception results in better somatosensory temporal discrimination threshold (STDT), as a proxy of tactile acuity. To this purpose, a group of subjects (experimental group) was verbally influenced and conditioned about the effect of an inert cream in enhancing tactile perception, while a control group was informed about the real nature of the cream. In both groups, we measured STDT before and after cream application, by means of pairs of electrical stimuli delivered on the index fingertip and separated by ascending inter-stimulus intervals. STDT was defined as the shortest time interval at which the two stimuli were perceived as separated. Results revealed an increase in subjective perception of stimulus intensity and a reduction of STDT only in the experimental group. This study proves that a placebo procedure, consisting of verbal suggestion and a short conditioning, can reduce the temporal discrimination threshold.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Somatosensory perception, like perception in other sensory systems, depends not only on the integrity of the peripheral receptors and afferent pathways, but also on the functioning of central brain regions. The latter can be influenced by different factors, like cognitive functions, training, and brain stimulation. For example, reward can modulate the hemodynamic response in the primary somatosensory cortex and affect somatosensory judgments (Pleger et al. 2008). In addition, attention can facilitate tactile perception, both in the spatial (knowledge of where a stimulus is likely to occur) and in the temporal domain (knowledge of when this stimulus is likely to occur) (Nobre 2001; van Ede et al. 2011). Moreover, a sensory training with tactile tasks improves tactile spatial acuity and this improvement can transfer even to untrained fingers (Harris et al. 2001; Wong et al. 2013; Harrar et al. 2014). Related to this, it was demonstrated that a training with Braille reading induces plastic changes in the primary somatosensory cortex (Debowska et al. 2016) and reduces the somatosensory spatial threshold (Wong et al. 2011; Zeuner et al. 2002). Somatosensory temporal processing, as well, is improved in blind Braille readers, thus supporting again the link between tactile training and somatosensory functional changes (Bhattacharjee et al. 2010). More recently, it was observed that somatosensory spatial acuity can be improved even without external stimulation or training (Philipp et al. 2015). In particular, meditation can induce a reduction of the spatial discrimination threshold when focused on the same body district that will be tested. This suggests that endogenous, top–down processes like mental imagery or meditation can induce plastic changes that result in better perceptual ability.
Recent evidence suggests that also placebo procedures can change somatic perception. More precisely, placebo can induce de-novo somatic sensations (Beissner et al. 2015), convert a tactile stimulus into pain (Colloca et al. 2008) and enhance the perceived intensity of tactile stimuli (Fiorio et al. 2012). In a previous study, we found that after a placebo procedure, participants perceived the same tactile stimulus as more intense than before and that this modulation was associated to changes in the late components of the somatosensory evoked potentials, reflecting higher order cortical elaboration of sensory information (Fiorio et al. 2012). What is still unknown is whether, in addition to perceptual and neurophysiological changes, a placebo procedure in the tactile modality can also influence the processing of tactile information. To address this question, in the current study, we induced expectation of enhanced tactile perception through a placebo procedure and tested the psychophysical effects on the somatosensory temporal discrimination threshold (STDT) as a proxy of tactile function. STDT is defined as the shortest time interval at which two tactile stimuli are perceived as separate and represents a fundamental ability of the somatosensory system, that is to elaborate the timing of sensory events. Different studies converge in indicating that STDT computed with the method of limits is reproducible in time both in healthy individuals and in pathological populations, suggesting that it has fair-to-good reliability (Morgante et al. 2011; Conte et al. 2012; Ramos et al. 2016). Moreover, stimulation of the primary somatosensory cortex with theta-burst TMS improves STDT (Conte et al. 2012; Rocchi et al. 2016), suggesting that STDT may well represent somatosensory functions. The prediction in our study is that the placebo procedure should result in a decrease of the STDT.
Materials and methods
Subjects
Forty-six healthy right-handed subjects participated in the experiment and were randomly assigned to two groups: 25 subjects (9 females; mean age 24.48 ± 3.9 years) have been exposed to a placebo-like procedure (experimental group) and other 21 subjects (10 females; mean age 25.95 ± 5.1 years) served as control (control group). By self-declaration, no subject reported any pathological condition and none was under medication. At the time of enrolment, the subjects were informed that we were collaborating with a pharmaceutical company to study the effects of a new developed cream. Written informed consent was obtained from all participants prior to the study. After study completion, participants were fully informed about the real aims of the experiments. The study was approved by the local ethical committee of the Department of Neurosciences, Biomedicine and Movement Sciences of the University of Verona.
Temporal discrimination threshold
Subjects’ right hand was comfortably positioned on a table. To measure the somatosensory temporal discrimination threshold (STDT), we delivered pairs of tactile stimuli by means of a constant current stimulator (STM 140, HTL, Udine, Italy) through a surface skin electrode (1 mm of diameter) applied to the right index fingertip. The anode was located 0.5 cm distally from the cathode. The tactile stimulus consisted of a train of four square wave electrical pulses of 0.5 ms each separated by 1 ms interval (for a total of 5 ms stimulus duration). The intensity of stimulation was determined for each subject, by delivering series of stimuli with increasing intensity from 0 mA in steps of 0.5 mA. The lowest intensity at which the electric stimulus was perceived in 10 out of 10 stimuli was considered as the sensory threshold (ST) and was used in the experimental procedure. During the temporal discrimination task, pairs of tactile stimuli were delivered always at the same intensity (corresponding to the ST) with ascending inter-stimulus intervals (ISI) in four consecutive blocks. In the first trial of each block, one stimulus was delivered (ISI = 0 ms) and in the two subsequent trials stimuli were separated in steps of 10 ms (trial 2, ISI = 10 ms and trial 3, ISI = 20 ms). This was done to reduce the number of stimulations below the range of ISIs that typically characterizes the STDT in healthy participants (30–50 ms) to avoid habituation of the somatosensory system. Starting from trial number 3 (ISI = 20 ms), the procedure became more fine-tuned and ISIs were progressively increased in steps of 2 ms in the subsequent trials. At each trial, subjects were required to report whether they perceived one or two stimuli. When the participant responded “two” for three consecutive trials, the block was ended and the next block could start. The first out of three consecutive ISIs at which subjects recognized two asynchronous stimuli was considered as threshold of the block. The average of the four values (one for each block) was considered as STDT and was entered in the data analysis. The choice of applying 4 repetitions was based on the previous studies in which a similar procedure was adopted and the reliability of STDT was demonstrated (Conte et al. 2012; Rocchi et al. 2016). During the ascending series, 3 catch trials with ISI of 0 ms were randomly delivered to control whether the subjects could correctly differentiate between one or two stimuli throughout the procedure.
Placebo procedure
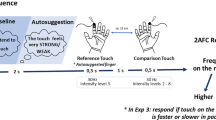
The procedure consisted of three sessions: baseline, manipulation and final (Fig. 1). In the baseline session, we measured the STDT for each subject, as described above. Each STDT testing lasted about 10 min. At the end of the baseline stimulation session, subjects were required to judge the perceived intensity of electrical stimulation on a Number Rating Scale (NRS) ranging from 0 (no sensation at all) to 10 (very strong sensation).
Schematic representation of the experimental procedure. Temporal discrimination thresholds (STDT) were measured in the baseline and final sessions. After each session, subjective intensity judgements (NRS) were also collected. In the baseline and final sessions’ intensity of stimulation was set at sensory threshold (ST), as measured before starting the whole experimental procedure. Between the baseline and final session there was the experimental manipulation, with the application of a cream on the right index finger. The experimental and control groups received different verbal information in regards to the effects of the cream. Moreover, the experimental group went through a short conditioning procedure consisting of 4 stimuli applied at 2 × ST
During the manipulation session, an experimenter wearing gloves and a white coat applied a transparent and jelly Vaseline cream at room temperature on the participant’s right index fingertip by means of a cotton swap. The cream was carefully removed after 5 min. The experimental group was told that the cream was a new active treatment capable of increasing tactile sensation and under experimental investigation for potential clinical use. This procedure was similar to that of the previous studies (Fiorio et al. 2012, 2014) and was used to induce expectation of increased tactile sensation. Participants of the control group were overtly told that they had been assigned to a control group in which an inert vaseline cream was to be applied as required by the protocol to compare their data with those of an experimental group. In the previous studies, we demonstrated that placebo effect in the tactile modality can be obtained if verbal suggestion is accompanied by a conditioning phase in which participants can experience an increase in sensation (Fiorio et al. 2012, 2014). Therefore, in the current study, after having removed the cream, the electrode was positioned again and a very short conditioning procedure was applied, in which the intensity of stimulation was surreptitiously increased two times above the ST. Four stimuli were delivered with this enhanced intensity by asking participants to report whether they felt the stimulation. Participants were only told that we wanted to be sure that they were feeling the stimulus, but actually, this was a manoeuvre to convince them that the cream had increased their sensation. In the control group, the same procedure was applied, in which four stimuli were applied without increasing the intensity of stimulation. We decided not to increase stimulus intensity in the control group to limit as much as possible any further reasoning or speculation by particularly sceptical participants on the potential effects of the inert cream. After this short procedure, the intensity of stimulation was set again to the baseline value and the final session started to measure STDT. At the end, subjective intensity judgements were measured again with the NRS.
To control for the investigator’s bias, the STDT testing (stimulus delivery and response collection) was automatized by means of a PC, thus limiting as much as possible the interaction between the investigator (who was not blind to the participant’s group membership) and the participant.
Statistical analysis
The absolute ST of the experimental and control groups was compared by means of t test for independent samples. STDT and subjective intensity judgements (NRS) were analysed by means of mixed-model ANOVA with Group (experimental vs. control) as between–subjects factor and Session (baseline vs. final) as within-subjects factor. Post-hoc analyses were carried out by means of paired or independent samples t tests, by applying the Bonferroni correction for multiple comparisons where necessary. The magnitude of the placebo effect was computed as the difference between the final and the baseline sessions in STDT values and NRS scores in the two groups. The difference was analysed by comparing the two groups with t test for independent samples. Finally, to check whether changes in discrimination thresholds were associated to changes in subjective judgements, we correlated the difference in STDT values with the difference in NRS scores for each group, by means of Spearman’s coefficient of correlation. The level of significance was set at p < 0.050.
Additional experiment
To check for an eventual effect of verbal suggestion alone in influencing tactile perception and STDT, we ran an additional experiment on 11 different participants (6 females, mean age: 25.3 ± 2.7) in which the same inert Vaseline cream was applied to the right index fingertip together with verbal information on its effects in enhancing tactile sensation. In this case, however, we did not apply the conditioning procedure, and therefore, the electrical stimulation was not increased after the treatment application. Before and after the treatment, participants were tested for the STDT and tactile perception (NRS) and data were analysed with t test for paired samples. The level of significance was set at p < 0.050.
Results
Age did not differ between groups (t(44) = − 1.11, p = 0.272). Analysis of the absolute ST did not show any significant difference between groups (t(44) = 0.442, p = 0.661), suggesting that intensity of stimulation was not different between the experimental (mean ± SEM, 3 ± 0.33 mA) and the control group (2.81 ± 0.26 mA).
Analysis of the STDT revealed a significant effect of Session (F(1,44) = 15.08, p < 0.001) and Group × Session interaction (F(1,44) = 4.79, p = 0.034), whereas Group was not significant (p = 0.536) (Fig. 2a). Post-hoc comparisons revealed that the experimental group had lower STDT values in the final (48.95 ± 0.81 ms) compared to the baseline session (54.03 ± 1.32 ms) (p = 0.001), whereas in control subjects STDT remained stable throughout the sessions (baseline: 51.37 ± 1.26 ms, final: 49.95 ± 0.96 ms) (p = 0.177). Moreover, the two groups did not differ neither in the baseline nor in the final session (for both comparisons, p > 0.156). These results indicate that the experimental procedure induced a significant reduction of the temporal discrimination threshold only in the experimental group. The STDT change between the final and baseline sessions was significantly different between the experimental (− 5.08 ± 1.27) and the control group (− 1.42 ± 1.01) (t(44) = − 2.19, p = 0.034) (Fig. 3a).
a Tactile temporal discrimination thresholds (STDT, ms) of the two groups (experimental, red and control, green). In the experimental group STDT was reduced in the final (striped bars) compared to the baseline session. b Subjective intensity judgements at the NRS in the two groups (experimental, red and control, green). Subjects of the experimental group perceived the tactile stimulus stronger in the final (striped bars) compared to the baseline session, despite the intensity of stimulation being exactly the same in the two sessions. Data are expressed as mean values and standard errors. *p < 0.050
a Amount of change computed as absolute difference between baseline and final session in STDT (ms) of the two groups (experimental, red and control, green). In the experimental group, change in STDT was higher than in the control group. b Amount of change computed as difference between the final and baseline session in subjective intensity judgements at the NRS in the two groups (experimental, red and control, green). Subjects of the experimental group display a bigger change in perception compared to the control group. Data are expressed as mean values and standard errors. *p < 0.050
Analysis of intensity judgements at the NRS revealed a significant effect of Session (F(1,44) = 15.63, p < 0.001) and Group × Session interaction (F(1,44) = 5.23, p = 0.027), whereas Group was not significant (p = 0.202) (Fig. 2b). Post-hoc comparisons revealed that the experimental group had higher judgements in the final (5.44 ± 0.4) compared to the baseline session (4.46 ± 0.41) (p = 0.001), whereas in control subjects perception judgements remained stable throughout the sessions (baseline: 4.14 ± 0.33, final: 4.41 ± 0.38) (p = 0.157). The two groups did not differ neither in the baseline nor in the final session (for both comparisons, p > 0.068). The difference between the final and baseline sessions in NRS scores was significantly higher in the experimental group (0.98 ± 0.25) compared to the control group (0.26 ± 0.18) (t(44) = 2.29, p = 0.027) (Fig. 3b). These results indicate that, despite the intensity of stimulation being identical in the two sessions, the experimental group perceived the stimulus as being stronger in the final than in the baseline session.
No correlation was found between difference in NRS scores and difference in STDT values (for both groups, Spearman’s rho < − 0.431, p > 0.051).
Additional experiment
Analysis of the STDT revealed no significant difference between the final (51.18 ± 1.61 ms) and the baseline session (50.45 ± 1.76 ms) (t(10) = − 0.312, p = 0.761). This result indicates that verbal suggestion alone did not induce a change in the temporal discrimination threshold (Fig. 4a). Analysis of the intensity judgements at the NRS revealed higher scores in the final (6.0 ± 0.63) compared to the baseline session (4.5 ± 0.56) (t(10) = − 5.75, p < 0.001), indicating that the procedure could induce an increase in the perception of stimulus intensity (Fig. 4b).
a Tactile temporal discrimination thresholds (STDT, ms) in the additional experiment in which only verbal suggestion was applied. STDT was stable in the final (striped bars) compared to the baseline session. b Subjective intensity judgements at the NRS in the additional experiment were higher in the final (striped bars) compared to the baseline session. Data are expressed as mean values and standard errors. *p < 0.050
Discussion
This study shows for the first time that somatosensory temporal discrimination thresholds can be improved by a placebo procedure in the tactile modality. More precisely, after the placebo procedure STDT was lower than before only in the experimental group and a greater amount of STDT reduction across sessions was found in the experimental group compared to the control group. A slight change of STDT was qualitatively observable also in the control group; nonetheless, this effect was not relevant, as evidenced by the lack of significant difference across sessions. The experimental group in our study also presented with higher intensity judgements at the NRS after the placebo procedure than before, despite the intensity of stimulation was the same in the two sessions. This finding confirms the reliability of the protocol in inducing a tactile placebo response, in line with a previous study by our group (Fiorio et al. 2012). The lack of correlation between NRS scores and STDT values suggests that the two measures could be related to different mechanisms and complexity levels. Of note, NRS scores could have been biased by subject’s compliance to the experimenter’s information about the effects of the cream in enhancing tactile sensation. Conversely, STDT was reasonably less biased by compliance, since we did not tell participants that the cream would have changed their ability to discriminate the stimuli and we measured STDT in an automatized way, thus limiting any investigator bias. Moreover, explicit judgements of perception at the NRS were recorded only once, after each experimental session (baseline and final), whereas the procedure to obtain the STDT was repeated four times in each session. Finally, the NRS score was given on a 0–10 scale that could have been not sensitive enough to unveil subtle effects. Conversely, the STDT was measured with the method of limits in steps of 2 ms, and could, therefore, represent a more fine-tuned measure of tactile function. These profound differences between the two measures could explain why the magnitude of the two phenomena was not quantitatively correlated.
The additional experiment revealed that verbal suggestion alone induced an increase in perception of stimulus intensity measured with the NRS, but did not impact on the STDT. This finding suggests that a combination of verbal suggestion and conditioning induces stronger placebo effects than verbal suggestion alone and it is in line with the previous studies in the tactile modality (Fiorio et al. 2012, 2014) and in pain (Colloca et al. 2008).
It could be argued that the increased stimulation intensity used to condition participants of the experimental group was the main factor contributing to the reduction of the STDT, rather than the placebo procedure. An experimental way to tackle this issue would be to apply only a conditioning procedure without any verbal suggestion. It should be noticed, however, that stimulus intensity per se does not seem to affect the STDT (Conte et al. 2016; Leodori et al. 2017). Hence, we could reasonably suggest that the placebo procedure consisting of a combination of verbal suggestion and conditioning was the most important factor influencing the STDT.
A possible theoretical framework to explain our findings is the predictive coding model of brain functioning (Friston 2010). In this frame of reference, the placebo procedure could have induced a peculiar brain’s state in the experimental group that resulted in a modified subjective interpretation of the stimulus characteristics, similarly to what already described for vision (Summerfield and Egner 2009). Consequently, the cognitive processing of tactile inputs would have been facilitated, thus resulting in a shorter time interval needed to discriminate two stimuli as separated.
The neural correlates of this effect could rely on a cerebral circuit linking brain regions involved in placebo-induced expectation and anticipation of sensory events with those involved in temporal processing of sensory stimuli. Based on the previous neuroimaging (Wager et al. 2004) and neurophysiological studies (Fiorio et al. 2012), we could hypothesize that prefrontal brain regions, like the anterior cingulate and dorsolateral prefrontal cortex, act as possible anatomical origin of the cognitive influence of expectation on tactile perception. Brain areas involved in STDT are the pre-supplementary motor area, the anterior cingulate cortex and the basal ganglia (Harrington et al. 1998; Pastor et al. 2004, 2006), although the primary somatosensory cortex seems to play a putative role (Hannula et al. 2008; Bolognini et al. 2010; Conte et al. 2012; Rai et al. 2012; Rocchi et al. 2016). Of note, studies also showed changes of STDT induced by stimulating the primary somatosensory cortex with theta-burst stimulation (Conte et al. 2012; Rocchi et al. 2016). Taken all this evidence into account, the lower STDT after the placebo procedure may hint at a top–down strengthening in the connections between prefrontal brain areas (activated by expectation) and the circuit subtending the temporal elaboration of tactile stimuli (mainly the primary somatosensory cortex), thus resulting in a fine-tuned tactile timing function.
One limitation of the psychophysical method adopted in this study is that it does not allow to clearly disambiguate whether the STDT improvement in the experimental group was caused by an actual improvement in the sensory discrimination function or by a change in the participants’ subjective response criterion (greater tendency to respond “two”). To this aim, other psychophysical approaches could be more suitable, for instance by inserting a higher number of trials with a single stimulus to obtain the false alarm rate. Here we can only speculate that since the STDT has a fair-to-good reliability and is not affected by the interval of time between tests, it is likely not influenced by decision processes (Ramos et al. 2016) and it may represent genuine sensory abilities. Moreover, the evidence that STDT relies more on brain areas that process somatosensory information (like the primary somatosensory cortex) rather than on brain areas that process decision-making (like the dorsolateral prefrontal cortex) (Conte et al. 2012; Rocchi et al. 2016) further supports the idea that it reflects sensory discrimination functions. However, being the placebo effect a complex phenomenon caution should be used in interpreting our data and future investigations adopting other psychophysical methods together with neurophysiological recordings (like somatosensory evoked potentials, SEP) should clarify the nature of the somatosensory improvement. With regard to this, using a similar protocol we previously recorded SEPs before and after the placebo-induced enhancement of tactile perception and found an increase in the amplitude of the late SEP components (N140 and P200) after the placebo procedure (Fiorio et al. 2012). This finding was interpreted as a cognitive top–down modulation of somatosensory processing due to the placebo procedure (Fiorio et al. 2012).
In the present explorative study, we decided to measure the STDT with the method of limits, because this method lends itself easily to clinical applications, thus favoring future translational research in clinical populations. Namely, this method has been proved to be suitable to investigate sensory processing in different neurological diseases (Fiorio et al. 2003, 2007, 2008a, b; Tinazzi et al. 2004; Bradley et al. 2012; Scontrini et al. 2009; Conte et al. 2010). Hence, our findings could acquire significant importance in inspiring lines of research addressing all those pathological conditions that comprise deficits in the temporal elaboration of tactile stimuli, as for example dystonia (Tinazzi et al. 2009) and Parkinson’s disease (Artieda et al. 1992; Fiorio et al. 2008b).
References
Artieda J, Pastor MA, Lacruz F, Obeso JA (1992) Temporal discrimination is abnormal in Parkinson’s disease. Brain 115:199–210
Beissner F, Brünner F, Fink M, Meissner K, Kaptchuk TJ, Napadow V (2015) Placebo-induced somatic sensations: a multi-modal study of three different placebo interventions. PLoS One 10(4):e0124808. https://doi.org/10.1371/journal.pone.0124808
Bhattacharjee A, Ye AJ, Lisak JA, Vargas MG, Goldreich D (2010) Vibrotactile masking experiments reveal accelerated somatosensory processing in congenitally blind Braille readers. J Neurosci 30:14288–14298
Bolognini N, Papagno C, Moroni D, Maravita A (2010) Tactile temporal processing in the auditory cortex. J Cogn Neurosci 22(6):1201–1211
Bradley D, Whelan R, Kimmich O, O’Riordan S, Mulrooney N, Brady P, Walsh R, Reilly RB, Hutchinson S, Molloy F, Hutchinson M (2012) Temporal discrimination thresholds in adult-onset primary torsion dystonia: an analysis by task type and by dystonia phenotype. J Neurol 259(1):77–82
Colloca L, Sigaudo M, Benedetti F (2008) The role of learning in nocebo and placebo effects. Pain 136:211–218
Conte A, Modugno N, Lena F, Dispenza S, Gandolfi B, Iezzi E, Fabbrini G, Berardelli A (2010) Subthalamic nucleus stimulation and somatosensory temporal discrimination in Parkinson’s disease. Brain 133(9):2656–2663
Conte A, Rocchi L, Nardella A, Dispenza S, Scontrini A, Khan N, Berardelli A (2012) Theta-burst stimulation-induced plasticity over primary somatosensory cortex changes somatosensory temporal discrimination in healthy humans. PLoS One 7(3): e32979. https://doi.org/10.1371/journal.pone.0032979
Conte A, Belvisi D, Manzo N, Bologna M, Barone F, Tartaglia M, Upadhyay N, Berardelli A (2016) Understanding the link between somatosensory temporal discrimination and movement execution in healthy subjects. Physiol Rep 4(18): e12899. https://doi.org/10.14814/phy2.12899
Debowska W, Wolak T, Nowicka A, Kozak A, Szwed M, Kossut M (2016) Functional and structural neuroplasticity induced by short-term tactile training based on Braille reading. Front Neurosci 10:460. https://doi.org/10.3389/fnins.2016.00460
Fiorio M, Tinazzi M, Bertolasi L, Aglioti SM (2003) Temporal processing of visuotactile and tactile stimuli in writer’s cramp. Ann Neurol 53(5):630–635
Fiorio M, Gambarin M, Valente EM, Liberini P, Loi M, Cossu G, Moretto G, Bhatia KP, Defazio G, Aglioti SM, Fiaschi A, Tinazzi M (2007) Defective temporal processing of sensory stimuli in DYT1 mutation carriers: a new endophenotype of dystonia? Brain 130(1):134–142
Fiorio M, Tinazzi M, Scontrini A, Stanzani C, Gambarin M, Fiaschi A, Moretto G, Fabbrini G, Berardelli A (2008a) Tactile temporal discrimination in patients with blepharospasm. J Neurol Neurosurg Psychiatry 79(7):796–798
Fiorio M, Valente EM, Gambarin M, Bentivoglio AR, Ialongo T, Albanese A, Barone P, Pellecchia MT, Brancati F, Moretto G, Fiaschi A, Tinazzi M (2008b) Subclinical sensory abnormalities in unaffected PINK1 heterozygotes. J Neurol 255(9):1372–1377
Fiorio M, Recchia S, Corrà F, Simonetto S, Garcia-Larrea L, Tinazzi M (2012) Enhancing non-noxious perception: behavioural and neurophysiological correlates of a placebo-like manipulation. Neuroscience 217:96–104
Fiorio M, Recchia S, Corrà F, Tinazzi M (2014) Behavioral and neurophysiological investigation of the influence of verbal suggestion on tactile perception. Neuroscience 258:332–339
Friston K (2010) The free-energy principle: a unified brain theory? Nat Rev Neurosci 11(2):127–138
Hannula H, Neuvonen T, Savolainen P, Tukiainen T, Salonen O, Carlson S, Pertovaara A (2008) Navigated transcranial magnetic stimulation of the primary somatosensory cortex impairs perceptual processing of tactile temporal discrimination. Neurosci Lett 437(2):144–147
Harrar V, Spence C, Makin TR (2014) Topographic generalization of tactile perceptual learning. J Exp Psychol Hum Percept Perform 40:15–23
Harrington DL, Haaland KY, Knight RT (1998) Cortical networks underlying mechanisms of time perception. J Neurosci 18:1085–1095
Harris JA, Harris IM, Diamond ME (2001) The topography of tactile learning in humans. J Neurosci 21:1056–1061
Leodori G, Formica A, Zhu X, Conte A, Belvisi D, Cruccu G, Hallett M, Berardelli A (2017) The third-stimulus temporal discrimination threshold: focusing on the temporal processing of sensory input within primary somatosensory cortex. J Neurophysiol 118(4):2311–2317
Morgante F, Tinazzi M, Squintani G, Martino D, Defazio G, Romito L, Albanese A, Di Matteo A, Quartarone A, Girlanda P, Fiorio M, Berardelli A (2011) Abnormal tactile temporal discrimination in psychogenic dystonia. Neurology 77(12):1191–1197
Nobre AC (2001) Orienting attention to instants in time. Neuropsychologia 39:1317–1328
Pastor MA, Day BL, Macaluso E, Friston KJ, Frackowiak RSJ (2004) The functional neuroanatomy of temporal discrimination. J Neurosci 24:2585–2591
Pastor MA, Macaluso E, Day BL, Frackowiak RS (2006) The neural basis of temporal auditory discrimination. Neuroimage 30:512–520
Philipp ST, Kalisch T, Wachtler T, Dinse HR (2015) Enhanced tactile acuity through mental states. Sci Rep 5:13549. https://doi.org/10.1038/srep13549
Pleger B, Blankenburg F, Ruff CC, Driver J, Dolan RJ (2008) Reward facilitates tactile judgments and modulates hemodynamic responses in human primary somatosensory cortex. J Neurosci 28:8161–8168
Rai N, Premji A, Tommerdahl M, Nelson AJ (2012) Continuous theta-burst rTMS over primary somatosensory cortex modulates tactile perception on the hand. Clin Neurophysiol 123(6):1226–1233
Ramos VF, Esquenazi A, Villegas MA, Wu T, Hallett M (2016) Temporal discrimination threshold with healthy aging. Neurobiol Aging 43:174–179
Rocchi L, Casula E, Tocco P, Berardelli A, Rothwell J (2016) Somatosensory temporal discrimination threshold involves inhibitory mechanisms in the primary somatosensory area. J Neurosci 36(2):325–335
Scontrini A, Conte A, Defazio G, Fiorio M, Fabbrini G, Suppa A, Tinazzi M, Berardelli A (2009) Somatosensory temporal discrimination in patients with primary focal dystonia. J Neurol Neurosurg Psychiatry 80(12):1315–1319
Summerfield C, Egner T (2009) Expectation (and attention) in visual cognition. Trends Cogn Sci 13(9):403–409
Tinazzi M, Fiorio M, Bertolasi L, Aglioti SM (2004) Timing of tactile and visuo-tactile events is impaired in patients with cervical dystonia. J Neurol 251(1):85–90
Tinazzi M, Fiorio M, Fiaschi A, Rothwell JC, Bhatia KP (2009) Sensory functions in dystonia: insights from behavioral studies. Mov Disord 24(10):1427–1436
van Ede F, de Lange F, Jensen O, Maris E (2011) Orienting attention to an upcoming tactile event involves a spatially and temporally specific modulation of sensorimotor alpha- and beta-band oscillations. J Neurosci 31(6):2016–2024
Wager TD, Rilling JK, Smith EE, Sokolik A, Casey KL, Davidson RJ, Kosslyn SM, Rose RM, Cohen JD (2004) Placebo-induced changes in FMRI in the anticipation and experience of pain. Science 303(5661):1162–1167
Wong M, Gnanakumaran V, Goldreich D (2011) Tactile spatial acuity enhancement in blindness: evidence for experience-dependent mechanisms. J Neurosci 31(19):7028–7037
Wong M, Peters RM, Goldreich D (2013) A physical constraint on perceptual learning: tactile spatial acuity improves with training to a limit set by finger size. J Neurosci 33:9345–9352
Zeuner KE, Bara-Jimenez W, Noguchi PS, Goldstein SR, Dambrosia JM, Hallett M (2002) Sensory training in patients with focal hand dystonia. Ann Neurol 51(5):593–598
Acknowledgements
We thank Marialuisa Maiorano and Alessandro Zizza for help in data collection.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Fiorio, M., Emadi Andani, M., Recchia, S. et al. The somatosensory temporal discrimination threshold changes after a placebo procedure. Exp Brain Res 236, 2983–2990 (2018). https://doi.org/10.1007/s00221-018-5357-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-018-5357-5