Abstract
Magnification of visual feedback (VF) impairs force control in older adults. In this study, we aimed to determine whether the age-associated increase in force variability with magnification of visual feedback is a consequence of increased amplitude or speed of visual feedback. Seventeen young and 18 older adults performed a constant isometric force task with the index finger at 5% of MVC. We manipulated the vertical (force gain) and horizontal (time gain) aspect of the visual feedback so participants performed the task with the following VF conditions: (1) high amplitude-fast speed; (2) low amplitude-slow speed; (3) high amplitude-slow speed. Changing the visual feedback from low amplitude-slow speed to high amplitude-fast speed increased force variability in older adults but decreased it in young adults (P < 0.01). Changing the visual feedback from low amplitude-slow speed to high amplitude-slow speed did not alter force variability in older adults (P > 0.2), but decreased it in young adults (P < 0.01). Changing the visual feedback from high amplitude-slow speed to high amplitude-fast speed increased force variability in older adults (P < 0.01) but did not alter force variability in young adults (P > 0.2). In summary, increased force variability in older adults with magnification of visual feedback was evident only when the speed of visual feedback increased. Thus, we conclude that in older adults deficits in the rate of processing visual information and not deficits in the processing of more visual information impair force control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In motor control experiments, visual feedback of the force output is a tool to enhance force control. However, a consistent finding is that magnification of visual feedback impairs force control in older adults. This is demonstrated as an increase in force variability with magnification of the force visual feedback (Baweja et al. 2012; Fox et al. 2013; Kennedy and Christou 2011; Sosnoff and Newell 2006; Tracy et al. 2007; Welsh et al. 2007a, b). Although strategies used to magnify force visual feedback increase both the amplitude and speed of the feedback, their relative contribution to the exacerbation of force variability in older adults remains unclear. Here, we attempt to understand the relative contribution of amplitude and speed of visual feedback to the age-associated increase in force variability with magnification of visual feedback.
Deficits in visual information processing could explain the increase in force variability with magnification of visual feedback in older adults. The first evidence in the literature likely comes from Sosnoff and Newell (Sosnoff and Newell 2006). They compared finger force variability of young and older adults during a constant force task at various levels of visual feedback magnification (250-fold range). Age-associated differences in force variability increased as a function of visual feedback magnification. The authors concluded that these findings indicate impaired visual information processing in older adults relative to young adults. Several follow-up studies supported the notion that the age-associated differences in force variability increase with magnification of visual feedback (Baweja et al. 2010; Fox et al. 2013; Kennedy and Christou 2011; Sosnoff and Newell 2006; Welsh et al. 2007a). Further support for an age-associated deficit in visual information processing comes from reaction time and reaching task studies (Temprado et al. 2013; Van Halewyck et al. 2015; Welsh et al. 2007b). Specifically, older adults exhibit slower pre-motor time (processing time) during reaction time tasks (Anderson et al. 1997; Curran et al. 2001), which is exacerbated with the addition of visual stimuli or increased cognitive load (Hahn et al. 2011, 2006; Huxhold et al. 2006; Vaportzis et al. 2014; Voelcker-Rehage and Alberts 2007). Furthermore, the impaired reaching accuracy in older adults is associated with a deficit in visual information processing rather than to a deficit in programming the movement (Temprado et al. 2013; Van Halewyck et al. 2015; Welsh et al. 2007b). The age-associated visual information processing deficits are likely related to a functional decline and volume loss of the visual cortex (Andersen 2012; Mendelson and Wells 2002; Park et al. 2004) and prefrontal cortex (Raz et al. 1997), which impairs visual information transmission and integration with the execution of motor output. Thus, age-associated deficits in visual information processing exacerbate force variability with magnification of visual feedback.
Interestingly, magnification of visual feedback increases both the amplitude and speed of visual feedback. This is important because increased amplitude of visual feedback requires a greater amount of visual information processing, whereas a faster speed of visual feedback requires a higher rate of visual information processing. Nonetheless, it remains unclear whether the impaired force variability with magnification of visual feedback in older adults relates to the increased amplitude or speed of visual feedback. In this study, we aimed to determine whether the age-associated increase in force variability with magnification of visual feedback is a consequence of increased amplitude or speed of visual feedback. To accomplish this, we manipulated force visual feedback amplitude and speed individually or concomitantly by changing the vertical (force gain) and horizontal (time gain) aspect of the visual feedback. We hypothesize that the decline in force control with magnification of visual feedback is a consequence of increased speed of visual feedback.
Methods
Participants
Seventeen young adults (20.5 ± 2.1 years, nine females) and 18 older adults (72.7 ± 6.4 years, nine females) volunteered to participate in this study. All participants reported being healthy without any known orthopedic or neurological problems, and were right handed according to a standardized survey (Oldfield 1971). The Institutional Review Board at the University of Florida approved all the procedures for this study, and the experiment was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki. Participants signed a written consent form before participating in this study.
Experimental protocol and procedures
Participants attended a single 1-h experimental session. Each participant began with familiarization of the experimental procedures. The familiarization period included a verbal explanation of the index finger constant isometric force task and ten practice trials. Following the familiarization, each participant performed the following: (1) maximum voluntary contraction (MVC) force with abduction of the index finger; (2) constant isometric force task with the index finger at 5% MVC. For the constant isometric force task, each participant performed 6 trials at three different visual feedback conditions. The order of the visual feedback conditions was random. Each trial lasted 20 s and participants received 30 s of rest between trials, and 1 min of rest between visual feedback conditions.
Experimental arrangement
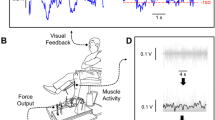
Experimental setup and apparatus Participants were seated comfortably in an upright position and faced a 32-inch monitor (Sync MasterTM 320MP-2, Samsung Electronics America, NJ, USA) located 1.65 m away at eye level. They placed their left arm on a custom-built metal plate that secured the arm on the table (Fig. 1). They abducted the left shoulder at 45° and flexed the elbow at 90°. We secured the pronated left forearm of the participants on the metal plate and restrained all fingers from movement with Velcro straps except the index finger. This arrangement allowed the abduction of the index finger about the metacarpophalangeal joint in the horizontal plane.
Force measurement We recorded the constant isometric force produced by the abduction of the index finger with a one-dimensional force transducer (Futek LRF400—FSH00261, capacity: 1 lb, Futek Advanced Sensor Technology Inc. CA, USA). The axis of the force transducer was aligned perpendicular to the proximal inter-phalangeal (PIP) joint. The force signal was sampled at 1 kHz with a NI-DAQ card (Model USB6210, National Instruments, Austin, TX, USA) and stored on a personal computer.
MVC task Participants abducted their index finger from baseline to maximum force over a 2-s period and maintained the maximum force for 4–7 s. We repeated MVC trials until two trials were within 5% of each other. We gave participants 1-min rest between MVC trials. We quantified the MVC force as the average force over 3–6 s (constant part) around the peak force. This procedure permitted the identification of a more conservative MVC, which reflected the person’s capability to perform constant isometric contractions (Baweja et al. 2010; Kennedy and Christou 2011).
Constant isometric force task The goal of the participants was to accurately match the target force (5% MVC) line with abduction of the index finger. The target force line (red) and the force they exerted (blue) simultaneously displayed on the monitor (Fig. 2). Participants gradually pushed against the force transducer and increased their force to match the target force within 3 s. After they reached the target, they maintained their force on the target line as accurately and as consistently as possible for 17 s.
Task and visual feedback manipulation. Participants performed a constant isometric contraction exerting 5% of MVC for 20 s with three different visual feedback conditions (low amplitude-slow speed, high amplitude-fast speed, and high amplitude-slow speed). a We compared the low amplitude-slow speed and high amplitude-fast speed conditions, which manipulated both the amplitude (0.06°–1.2°) and speed of visual feedback (3.64–5.94 cm/s). b We compared the low amplitude-slow speed and high amplitude-slow speed conditions, which manipulated only the amplitude of visual feedback (0.06°–1.2°). c We compared the high amplitude-slow speed and high amplitude-fast speed conditions, which manipulated only the speed of visual feedback (3.97–5.94 cm/s)
Visual feedback manipulation We manipulated visual feedback with a custom-written program in Matlab® (Math Works™ Inc., Natick, Massachusetts, USA) by changing the vertical (force gain) and horizontal (time gain) axis scale on the monitor. The horizontal feedback varied by changing the horizontal size of the display from 14 to 70 cm (full length of the monitor). The vertical feedback varied by changing the visual angle of force from 0.06° to 1.2°. We applied the following formula to manipulate the visual angle of force, as we have done previously (Baweja et al. 2010, 2012; Fox et al. 2013; Kennedy and Christou 2011):
where VAForce is the visual angle of force, h1 is the half height of force amplitude displayed on the monitor, and d is the distance of the eye to the center of the monitor. For each participant, the distance from the eye to the center of the monitor (d) was held constant (1.65 m) and participants were monitored to ensure that they maintained their position in the chair. To alter the visual angle of force, we manipulated the amplitude of force fluctuations (h1) by changing the y-axis scale (zooming in or out of the force display). The amplitude of force fluctuations was estimated to be 3% of targeted force (CV of mean force).
Based on a combination of the horizontal and vertical aspects of the display we created the following three distinct types of visual feedback conditions:
-
1.
The horizontal length of the display was 70 cm and the visual angle of force was 0.06°. We termed this visual feedback low amplitude-slow speed.
-
2.
The horizontal length of the display was 14 cm and the visual angle of force was 1.2°. We termed this visual feedback high amplitude-slow speed.
-
3.
The horizontal length of the display was 70 cm and the visual angle of force was 1.2°. We termed this visual feedback high amplitude-fast speed.
By changing the visual feedback from one condition to another we were able to manipulate the amplitude and speed of visual feedback. We performed the following three comparisons:
Manipulation of visual feedback amplitude and speed We changed the visual feedback condition from low amplitude-slow speed to high amplitude-fast speed (Fig. 2a) by manipulating the force gain (0.06°–1.2°) while maintaining the time gain (70 cm). This manipulation resulted in increased amplitude of the force signal on the y-axis (20 times) and increased speed of visual feedback (3.64–5.94 cm/s). The comparison of these two conditions reflects the traditional manipulation of visual feedback in the literature (Baweja et al. 2010, 2012; Fox et al. 2013; Kennedy and Christou 2011).
Manipulation of visual feedback amplitude We changed the visual feedback condition from low amplitude-slow speed to high amplitude-slow speed (Fig. 2b) by manipulating both the force gain (0.06°–1.2°) and time gain (x-axis length: 70–14 cm). This manipulation resulted in increased amplitude of the force signal on the y-axis (20 times), but no significant change in the speed of visual feedback (3.64–3.97 cm/s).
Manipulation of speed of visual feedback We changed the visual feedback condition from high amplitude-slow speed to high amplitude-fast speed (Fig. 2c) by manipulating the time gain (x-axis length: 14–70 cm) while maintaining the force gain (1.2°). This manipulation resulted in increased speed of visual feedback (3.97–5.94 cm/s), but no change in the amplitude of visual feedback.
Data analysis
Data were analyzed offline using custom-written programs in Matlab® (Math Works™ Inc., Natick, Massachusetts, USA). We quantified the finger force performance from a 15 s segment (from 4 to 19 s of each trial). We selected this time segment for analysis to exclude the initial adjustments (from 0 to 4 s) and end-of-trial force drop (from 19 to 20 s).
Force error We quantified force error with the root mean square error (RMSE) from targeted force.
Force variability We quantified force variability with the standard deviation (SD) of the processed force signal. Processing of the force signal included: (1) the raw force signal was low-pass filtered at 10 Hz with a second order Butterworth filter; (2) the filtered force signal was detrended. Detrending the force signal eliminated drifting of force that may be caused by the reduction of visual feedback. We use force variability and SD of force interchangeably throughout the paper.
Statistical analysis
The independent variables were the age of the group (young adults and older adults) and the visual feedback conditions (low amplitude-slow speed, high amplitude-fast speed, high amplitude-slow speed). The major dependent variables were: (1) MVC force; (2) mean force; (3) RMSE; and (4) SD of force. We compared the MVC force between the two age groups with an independent t test. To compare the effect of visual feedback in young and older adults on mean force, RMSE, and SD of force we used a two-way mixed Analysis of Variance (ANOVA) [2 age groups × 2 visual feedback conditions] with repeated measures on visual feedback conditions. Following significant main effects and interactions from the ANOVA models we performed appropriate post hoc analyses. We performed all statistical analyses with IBM SPSS Statistics (IBM Corp. Version 22.0. Armonk, NY: IBM Corp. SPSS, Inc., Chicago, IL). The alpha level for all statistical tests was 0.05 and was adjusted for multiple comparisons with Bonferroni corrections. Data are reported as mean ± SD within the text and as mean ± standard error of the mean (SEM) in the figures. Only the significant main effects and interactions are presented, unless otherwise noted.
Results
MVC and mean force during task
Young and older adults exhibited similar MVC (Young: 25.32 ± 6.08 N; Older: 23.11 ± 7.89 N; F1,35 = 0.948, P = 0.34), therefore, age-associated differences during the various visual feedback conditions were not due to strength differences. Mean forces were not different between young and older adults and between different visual feedback conditions (no main effects or interactions; P > 0.4). Therefore, we used the SD of force as our measure of force variability.
Manipulation of amplitude and speed of visual feedback
Changing the visual feedback from low amplitude-slow speed to high amplitude-fast speed (Fig. 3a) increased the amplitude (0.06°–1.2°) and the speed of visual feedback (3.64–5.94 cm/s). This manipulation resulted in the following:
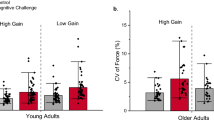
Force error and force variability. When both the amplitude and speed of visual feedback increased with our visual feedback manipulation (a from low amplitude-slow speed to high amplitude-fast speed), force error decreased for both age groups (b). In contrast, variability of force increased for older adults and decreased for young adults (c). When only the amplitude of visual feedback increased with our manipulation (d from low amplitude-slow speed to high amplitude-slow speed), force error decreased for both age groups (e). Force variability for young adults decreased, while older adults did not change (f). When only the speed of visual feedback increased with our manipulation (g from high amplitude-slow speed to high amplitude-fast speed), force error did not change for both groups (h). Force variability for older adults increased, while young adults did not change (i). In summary, these results demonstrate that increased speed of visual feedback increases force variability in older adults, whereas increased amplitude of visual feedback decreases force error in both age groups. +, visual feedback main effect; ○, age main effect; *, visual feedback differences; and #, age differences
Force error There was a significant age group main effect (F1,33 = 10.625, P < 0.001; Fig. 3b) and a significant visual feedback main effect (F1,33 = 16.326, P < 0.001; Fig. 3b). The age main effect indicates that older adults exhibited greater force error than young adults regardless of the visual feedback.
Force variability There was a significant age group × visual feedback interaction (F1,33 = 15.255, P < 0.001; Fig. 3c). Post hoc analyses demonstrated that older adults showed a significant increase in force variability from low amplitude-slow speed to high amplitude-fast speed (0.064 ± 0.022–0.080 ± 0.03 N, t = 2.315, P = 0.033), while young adults showed a significant decrease from low amplitude-slow speed to high amplitude-fast speed (0.056 ± 0.02–0.041 ± 0.019 N, t = 4.458, P < 0.001). In addition, the age-associated difference in force variability was significant only during high amplitude-fast speed condition (young: 0.041 ± 0.019 N, older: 0.080 ± 0.03 N, t = 4.284, P < 0.001), but not during low amplitude-slow speed condition (young: 0.056 ± 0.02 N, older: 0.064 ± 0.022 N, t = 1.174, P = 0.249).
These findings suggest that increasing the amplitude (0.06°–1.2°) and the speed of visual feedback (3.64–5.94 cm/s) decreases force error for both groups. In contrast, this manipulation results in a decrease in force variability for young adults and a significant increase in force variability for older adults.
Manipulation of visual feedback amplitude
Changing the visual feedback from low amplitude-slow speed to high amplitude-slow speed (Fig. 3d) increases the amplitude of visual feedback (0.06°–1.2°) but not the speed of visual feedback (3.97 and 3.64 cm/s). This manipulation resulted in the following:
Force error There was a significant age (F1,33 = 13.354, P = 0.001) and visual feedback main effect (F1,33 = 20.857, P < 0.001; Fig. 3e). These two main effects demonstrated that older adults exhibited greater force error than young adults regardless of the visual feedback (Young: 0.058 ± 0.021 N, Older: 0.092 ± 0.032 N), and that force error decreased for both groups from low amplitude-slow speed to high amplitude-slow speed (0.088 ± 0.038–0.064 ± 0.032 N).
Force variability There was a significant age main effect (F1,33 = 6.317, P = 0.017) and a significant age group × visual feedback interaction (F1,33 = 7.550, P = 0.01; Fig. 3f). Post hoc analyses demonstrated that young adults exhibited significant decrease in force variability from low-amplitude-slow speed to high amplitude-slow speed (0.056 ± 0.02–0.42 ± 0.023 N, t = 3.615, P = 0.002), while older adults did not change (t = 0.723, P = 0.48). In addition, the age-associated difference in force variability was significant only during high amplitude-slow speed condition (young: 0.042 ± 0.023 N, older: 0.068 ± 0.022 N, t = 3.359, P = 0.002), but not during low amplitude-slow speed condition (young: 0.056 ± 0.02 N, older: 0.064 ± 0.022 N, t = 1.176, P = 0.25).
These findings suggest that solely increasing the amplitude of visual feedback (0.06°–1.2°) decreases force error for both groups. In contrast, this manipulation results in a decrease in force variability for young adults but in no significant change in force variability for older adults.
Manipulation of speed of visual feedback
Changing the visual feedback from high amplitude-slow speed to high amplitude-fast speed (Fig. 3g) increased the speed of visual feedback (3.97–5.94 cm/s), but not the amplitude of visual feedback (1.2° for both). This manipulation resulted in the following:
Force error There was a significant age main effect (F1,33 = 23.620, P < 0.001; Fig. 3h). This main effect demonstrated that older adults exhibited greater force error than young adults regardless of the visual feedback.
Force variability There was a significant age main effect (F1,33 = 17.691, P < 0.001; Fig. 3i) and an age × visual feedback interaction (F1,33 = 4.190, P = 0.049; Fig. 3i). Post hoc analyses demonstrated that older adults exhibited significantly greater force variability than young adults during both high amplitude-slow speed (older: 0.068 ± 0.022 N, young: 0.042 ± 0.023 N, t = 3.359, P = 0.002) and high amplitude-fast speed (older: 0.080 ± 0.03 N, young: 0.041 ± 0.019 N, t = 4.345, P < 0.001) conditions. However, the difference in force variability was greater at high amplitude-fast speed visual feedback condition. In addition, older adults showed a significant increase in force variability from high amplitude-slow speed to high amplitude-fast speed (0.068 ± 0.022–0.080 ± 0.03 N, t = 2.131, P = 0.048), whereas the force variability did not change for young adults (0.042 ± 0.023 and 0.041 ± 0.019 N, t = 0.397, P = 0.7, Fig. 3i).
These findings suggest that solely increasing the speed of visual feedback (3.97–5.94 cm/s) does not influence force error for both groups. In contrast, this manipulation results in an increase in force variability for older adults but no significant change in force variability for young adults.
Discussion
The finding that magnification of visual feedback impairs motor control in older adults has been widely accepted in the literature. However, it remains unclear whether this impairment relates to the magnification-induced increase in amplitude or speed of visual feedback. This is important because the amplitude of visual feedback relates to the amount of visual information that must be processed, whereas the speed of visual feedback relates to the rate by which visual information must be processed. Using a series of experimental conditions that uniquely manipulated the amplitude and speed of visual feedback; we provide novel evidence that the increase in speed, but not the increase in amplitude of visual feedback impaired force variability in older adults. We conclude that for older adults, the motor control impairment observed with magnification of visual feedback is likely related to a deficit in processing visual information fast.
Visual feedback and force variability
Magnification of visual feedback increases force variability in older adults, but not in young adults. This has been demonstrated with manipulation of visual feedback from very low-gain to high-gain with contractions of the upper (Baweja et al. 2009; Fox et al. 2013; Kennedy and Christou 2011; Sosnoff and Newell 2006) and lower limb (Baweja et al. 2012; Tracy et al. 2007; Welsh et al. 2007a). Our current results support this finding. Specifically, we show that magnifying the visual feedback 20 times (increased the visual angle from 0.06° to 1.2°), led to an increase in force variability in older adults and a decrease in young adults. Magnifying the visual feedback also increased the speed of visual feedback ~ 2 times (3.64–5.94 cm/s). Thus, this raises the following question: “Does magnification of visual feedback increase force variability in older adults because of increased amplitude or speed of visual feedback?”
It is important to address this question because the amplitude and speed of visual feedback challenge different neural processes. Greater amplitude is associated with the amount of visual information that must be processed, whereas faster speed challenges the rate by which visual information must be processed. Although there is strong evidence that visual information processing is impaired in older adults (Briggs et al. 1999; Burns and Nettelbeck 2001; Hertzog et al. 1986; Nettelbeck and Rabbitt 1992; Plude and Hoyer 1986; Salthouse 1996, 2000; van der Lubbe and Verleger 2002). It remains unknown whether the deficits in older adults relate to increased amplitude or speed of visual feedback. Previous studies that magnified visual feedback were limited in addressing this question because they magnified the vertical part of the visual display and thus changed both the amplitude and speed of visual feedback concurrently. Here, we compared visual feedback conditions that uniquely manipulated the amplitude and speed of the visual feedback. When we increased the amplitude 20 times, but not the speed of the visual feedback (low amplitude-slow speed to high amplitude-slow speed; Fig. 2b), force variability did not change in older adults despite the significant increase in the amplitude of visual feedback. In contrast, when we increased the speed two times but not the amplitude of the visual feedback (high amplitude-slow speed to high amplitude-fast speed; Fig. 2c), force variability increased in older adults despite the fact that the amplitude of visual feedback did not change. Older adults consistently increased their force variability only when the speed of visual feedback increased (Fig. 3c, i). Thus, we conclude that the impairment in force control with magnification of visual feedback for older adults relates to a deficit in processing visual information fast rather than to deficits in processing a greater amount of visual information (Baweja et al. 2010; Kennedy and Christou 2011) or fine motor control (Christou 2011; Enoka et al. 2003; Tracy and Enoka 2002).
Our finding suggests that the generally accepted notion that the increased motor output variability in older adults during magnified visual feedback (high-gain) is contributed by an inability to process visual information fast enough. Age-associated changes in the central nervous system can explain the visual information processing rate deficits, which contributes to motor control deficits in older adults. It is well documented that age-associated visual information processing speed deficits are related to various changes in central nervous system such as decreased density in visual cortex (Andersen 2012; Mendelson and Wells 2002), and reduced white matter integrity in corticostriatal tracks (Bogacz et al. 2010; Forstmann et al. 2011). Specifically, selective degeneration of the dorsolateral prefrontal cortex delays visual information transmission to the primary motor cortex which results to impaired integration of visual information with the execution of motor output (Raz et al. 1997). In addition, a selective loss of functional specialization of the neurons in the ventral visual cortex with aging (Park et al. 2004) contributes to impaired visual information transmission with magnification of visual gain (Coombes et al. 2010). In a closed loop visuomotor task where continuous visual information processing is required, impaired and delayed information processing in the central nervous system can lead to a wrong or late motor execution. Thus, the inability of older adults to process visual information at a fast rate (delayed visual information processing) could be due to age-associated structural changes at the brain and consequently lead to an increased motor output variability with magnified visual gain.
Our findings demonstrate that young adults decrease force variability with magnification of visual feedback. Young adults decreased force variability when the amplitude of visual feedback increased (Fig. 3c, f). Thus, we conclude that young adults are able to use the amplitude of visual feedback and enhance their visuomotor corrections and reduce force variability. These results support previous findings in the literature (Baweja et al. 2010; Coombes et al. 2010; Slifkin et al. 2000; Vaillancourt et al. 2006). However, our study provides the novel finding that in young adults, increasing the speed of visual feedback has no influence on their force variability (Fig. 3i).
Visual feedback and force error
A secondary interest of our study was to determine the effect of visual feedback magnification on force error. Similar to previous studies that compared low-gain and high-gain visual feedback, we show that force error decreased (accuracy increased) with the high-gain visual feedback for both young and older adults. Our findings demonstrate that both young and older adults decrease force error when the amplitude of visual feedback increased (Fig. 3b, e). Thus, we conclude that both young and older adults are able to efficiently use the amplitude of visual feedback and associated visuomotor corrections to reduce the distance of their force output relative to the targeted force. These results support previous findings in the literature (Beuter et al. 1995; Coombes et al. 2010, 2011; Poon et al. 2013). However, this study provides the novel finding that increasing the speed of visual feedback had no influence on the force error of young or older adults (Fig. 3h).
In this study, we aimed to determine whether the increase in force variability with magnification of visual feedback in older adults is related to the increase in amplitude or speed of visual feedback. We provide novel evidence that the increase in speed but not the increase in amplitude of visual feedback increases force variability in older adults. We conclude that the exacerbation of force control with magnification of visual feedback in older adults is likely related to a deficit in processing visual information fast. These results can be useful in rehabilitation protocols developed for older adults to improve motor control.
References
Andersen GJ (2012) Aging and vision: changes in function and performance from optics to perception Wiley. Interdiscip Rev Cogn Sci 3:403–410. https://doi.org/10.1002/wcs.1167
Anderson M, Nettelbeck T, Barlow J (1997) Reaction time measures of speed of processing: speed of response selection increases with age but speed of stimulus categorization does not British. J Dev Psychol 15:145–157
Baweja HS, Patel BK, Martinkewiz JD, Vu J, Christou EA (2009) Removal of visual feedback alters muscle activity and reduces force variability during constant isometric contractions. Exp Brain Res 197:35–47. https://doi.org/10.1007/s00221-009-1883-5
Baweja HS, Kennedy DM, Vu J, Vaillancourt DE, Christou EA (2010) Greater amount of visual feedback decreases force variability by reducing force oscillations from 0 to 1 and 3–7. Hz Eur J Appl Physiol 108:935–943. https://doi.org/10.1007/s00421-009-1301-5
Baweja HS, Kwon M, Christou EA (2012) Magnified visual feedback exacerbates positional variability in older adults due to altered modulation of the primary agonist muscle. Exp Brain Res 222:355–364. https://doi.org/10.1007/s00221-012-3219-0
Beuter A, Haverkamp H, Glass L, Carriere L (1995) Effect of manipulating visual feedback parameters on eye and finger movements. Int J Neurosci 83:281–294
Bogacz R, Wagenmakers EJ, Forstmann BU, Nieuwenhuis S (2010) The neural basis of the speed-accuracy tradeoff. Trends Neurosci 33:10–16. https://doi.org/10.1016/j.tins.2009.09.002
Briggs SD, Raz N, Marks W (1999) Age-related deficits in generation and manipulation of mental images: I. The role of sensorimotor speed and working memory. Psychol Aging 14:427–435
Burns NR, Nettelbeck T (2001) Speed of information processing and cognitive abilities Australian. J Psychol 53:141–142
Christou EA (2011) Aging and variability of voluntary contractions Exerc. Sport Sci Rev 39:77–84. https://doi.org/10.1097/JES.0b013e31820b85ab
Coombes SA, Corcos DM, Sprute L, Vaillancourt DE (2010) Selective regions of the visuomotor system are related to gain-induced changes in force error. J Neurophysiol 103:2114–2123. https://doi.org/10.1152/jn.00920.2009
Coombes SA, Corcos DM, Vaillancourt DE (2011) Spatiotemporal tuning of brain activity and force performance. Neuroimage 54:2226–2236. https://doi.org/10.1016/j.neuroimage.2010.10.003
Curran T, Hills A, Patterson MB, Strauss ME (2001) Effects of aging on visuospatial attention: an ERP study. Neuropsychologia 39:288–301
Enoka RM, Christou EA, Hunter SK, Kornatz KW, Semmler JG, Taylor AM, Tracy BL (2003) Mechanisms that contribute to differences in motor performance between young and old adults. J Electromyogr Kinesiol 13:1–12
Forstmann BU, Tittgemeyer M, Wagenmakers EJ, Derrfuss J, Imperati D, Brown S (2011) The speed-accuracy tradeoff in the elderly brain: a structural model-based approach. J Neurosci 31:17242–17249. https://doi.org/10.1523/JNEUROSCI.0309-11.2011
Fox EJ, Baweja HS, Kim C, Kennedy DM, Vaillancourt DE, Christou EA (2013) Modulation of force below 1 Hz: age-associated differences and the effect of magnified visual feedback. PLoS One 8:e55970. https://doi.org/10.1371/journal.pone.0055970
Hahn S, Carlson C, Singer S, Gronlund SD (2006) Aging and visual search: automatic and controlled attentional bias to threat faces. Acta Psychol (Amst) 123:312–336. https://doi.org/10.1016/j.actpsy.2006.01.008
Hahn M, Wild-Wall N, Falkenstein M (2011) Age-related differences in performance and stimulus processing in dual task situation. Brain Res 1414:66–76. https://doi.org/10.1016/j.brainres.2011.07.051
Hertzog C, Raskind CL, Cannon CJ (1986) Age-related slowing in semantic information processing speed: an individual differences analysis. J Gerontol 41:500–502
Huxhold O, Li SC, Schmiedek F, Lindenberger U (2006) Dual-tasking postural control: aging and the effects of cognitive demand in conjunction with focus of attention. Brain Res Bull 69:294–305. https://doi.org/10.1016/j.brainresbull.2006.01.002
Kennedy DM, Christou EA (2011) Greater amount of visual information exacerbates force control in older adults during constant isometric contractions. Exp Brain Res 213:351–361. https://doi.org/10.1007/s00221-011-2777-x
Mendelson JR, Wells EF (2002) Age-related changes in the visual cortex. Vision Res 42:695–703
Nettelbeck T, Rabbitt PMA (1992) Aging, cognitive performance and mental speed. Intelligence 16:189–205 doi. https://doi.org/10.1016/0160-2896(92)90004-B
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Park DC, Polk TA, Park R, Minear M, Savage A, Smith MR (2004) Aging reduces neural specialization in ventral visual cortex. Proc Natl Acad Sci USA 101:13091–13095. https://doi.org/10.1073/pnas.0405148101
Plude DJ, Hoyer WJ (1986) Age and the selectivity of visual information processing. Psychol Aging 1:4–10
Poon C, Coombes SA, Corcos DM, Christou EA, Vaillancourt DE (2013) Transient shifts in frontal and parietal circuits scale with enhanced visual feedback and changes in force variability and error. J Neurophysiol 109:2205–2215. https://doi.org/10.1152/jn.00969.2012
Raz N et al (1997) Selective aging of the human cerebral cortex observed in vivo: differential vulnerability of the prefrontal gray matter. Cereb Cortex 7:268–282
Salthouse TA (1996) The processing-speed theory of adult age differences in cognition. Psychol Rev 103:403–428 doi. https://doi.org/10.1037/0033-295x.103.3.403
Salthouse TA (2000) Aging and measures of processing speed. Biol Psychol 54:35–54 doi. https://doi.org/10.1016/S0301-0511(00)00052-1
Slifkin AB, Vaillancourt DE, Newell KM (2000) Intermittency in the control of continuous force production. J Neurophysiol 84:1708–1718
Sosnoff JJ, Newell KM (2006) Information processing limitations with aging in the visual scaling of isometric force. Exp Brain Res 170:423–432. https://doi.org/10.1007/s00221-005-0225-5
Temprado JJ, Sleimen-Malkoun R, Lemaire P, Rey-Robert B, Retornaz F, Berton E (2013) Aging of sensorimotor processes: a systematic study in Fitts’ task. Exp Brain Res 228:105–116. https://doi.org/10.1007/s00221-013-3542-0
Tracy BL, Enoka RM (2002) Older adults are less steady during submaximal isometric contractions with the knee extensor muscles. J Appl Physiol (1985) 92:1004–1012. https://doi.org/10.1152/japplphysiol.00954.2001
Tracy BL, Dinenno DV, Jorgensen B, Welsh SJ (2007) Aging, visuomotor correction, and force fluctuations in large muscles. Med Sci Sports Exerc 39:469–479. https://doi.org/10.1249/mss.0b013e31802d3ad3
Vaillancourt DE, Haibach PS, Newell KM (2006) Visual angle is the critical variable mediating gain-related effects in manual control. Exp Brain Res 173:742–750. https://doi.org/10.1007/s00221-006-0454-2
Van Halewyck F, Lavrysen A, Levin O, Boisgontier MP, Elliott D, Helsen WF (2015) Factors underlying age-related changes in discrete aiming. Exp Brain Res 233:1733–1744. https://doi.org/10.1007/s00221-015-4247-3
van der Lubbe RH, Verleger R (2002) Aging and the Simon task. Psychophysiology 39:100–110. https://doi.org/10.1017/S0048577202001221
Vaportzis E, Georgiou-Karistianis N, Stout JC (2014) Age and task difficulty differences in dual tasking using circle tracing and serial subtraction tasks. Aging Clin Exp Res 26:201–211. https://doi.org/10.1007/s40520-013-0151-5
Voelcker-Rehage C, Alberts JL (2007) Effect of motor practice on dual-task performance in older adults. J Gerontol Ser B-Psychol Sci Soc Sci 62:P141–P148
Welsh SJ, Dinenno DV, Tracy BL (2007a) Variability of quadriceps femoris motor neuron discharge and muscle force in human aging. Exp Brain Res 179:219–233. https://doi.org/10.1007/s00221-006-0785-z
Welsh TN, Higgins L, Elliott D (2007b) Are there age-related differences in learning to optimize speed, accuracy, and energy expenditure? Hum Movement Sci 26:892–912. https://doi.org/10.1016/j.humov.2007.04.004
Acknowledgements
This study was supported by R01 AG031769 to Evangelos A. Christou.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, C., Yacoubi, B. & Christou, E.A. Speed but not amplitude of visual feedback exacerbates force variability in older adults. Exp Brain Res 236, 2563–2571 (2018). https://doi.org/10.1007/s00221-018-5317-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-018-5317-0