Abstract
The interface between the sample and the transducer surface is critical to the performance of a biosensor. In this work, we compared different strategies for covalent self-assembly of antibodies onto bare gold substrates by introducing disulfide groups into the immunoglobulin structure, which acted as anchor molecules able to chemisorb spontaneously onto clean gold surfaces. The disulfide moieties were chemically introduced to the antibody via the primary amines, carboxylic acids, and carbohydrates present in its structure. The site-directed modification via the carbohydrate chains exhibited the best performance in terms of analyte response using a model system for the detection of the stroke marker neuron-specific enolase. SPR measurements clearly showed the potential for creating biologically active densely packed self-assembled monolayers (SAMs) in a one-step protocol compared to both mixed SAMs of alkanethiol compounds and commercial immobilization layers. The ability of the carbohydrate strategy to construct an electrochemical immunosensor was investigated using electrochemical impedance spectroscopy (EIS) and differential pulse voltammetry (DPV) transduction.

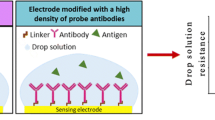
Left: Functionalization strategies of bare gold substrates via direct bio-SAM using disulfide-containing antibody chemically modified via their primary amines (A), carbohydrates (B) and carboxylic acids (C). Right: Dependence of the peak height with NSE concentration at NSE21-CHO modified electrochemical immunosensor. Inset: Logarithmic calibration plot
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Immunosensors have been developed for a plethora of applications, including medical diagnostics and environmental analysis, and have great potential for commercialization but still face a challenge in achieving a simple, robust, and inexpensive surface functionalization method compatible with mass-manufacturing techniques. Generally, functionalization processes require the biochemical modification of either the sensor surface or the antibody or both, and this usually involves several steps that in some cases are time consuming, costly, and difficult to implement in large-scale production process.
The immobilization of antibodies on solid support transducers is a critical issue for the sensor performance, as the affinity, orientation, and stability of the antibodies are affected by the immobilization strategy selected [1]. A variety of surface chemistry methods have been reported with those introducing orientation and maintaining native antibody conformation being of greatest relevance [2, 3]. Specific orientation should expose free antigen-binding regions of the antibody following surface anchoring, resulting in increased analyte binding and improved sensitivity. Passive adsorption of biomolecules on solid substrates has been widely explored [4] due to its simplicity but results in unstable adsorption with no control of orientation, and often results in protein unfolding [5, 6]. Alternatively, the use of self-assembled monolayers (SAMs), with two terminal functional groups, one enables binding to the sensor surface and the other allows coupling of the biocomponent, has been widely used [7–11], due to the high diversity of functionalized SAMs and chemical cross linkers available, and provides a strong and stable attachment. Nevertheless, it needs a previous functionalization of the surface and the molecules attached to the SAMs can be randomly oriented [12]. To minimize random orientation and uniformly orientate the antibodies on the surface, several strategies have been developed, including the use of receptors that bind the Fc portion of the antibody (e.g., proteins A, G, L, anti-Fc), which improves sensitivity [1, 13–15] but also requires an initial surface modification. Immobilization via antibody fragments through sulfhydryl groups also improves the sensitivity [16–19] but may form a very compact layer, giving rise to significant steric hindrance effects [20] and the potential loss of biological activity of the antibody fragments [21]. Oxidized oligosaccharide moieties of the antibodies coupled to amine- or hydrazine-modified solid supports have also been provided to providing great sensitivity [22–24] but again require a surface prefunctionalization step.
A single immobilization step can be achieved via the introduction of sulfur-containing molecules into the bioelement structure prior to its chemisorption onto gold [23, 25] (Scheme 1). Antibody molecules possess a number of functional groups suitable for modification including lysine ε-amine, N-terminal α-amine groups, and C-terminal aspartic acid and glutamic acid residues. Chemical conjugation reactions with antibody molecules are generally more successful at preserving activity if the functional groups utilized are present in limiting quantities and only at discrete sites on the molecule. In one approach, the disulfide in the hinge region of the antibody that holds the heavy chains together can be selectively cleaved with a reducing agent such as 2-mercaptoethylamine to create two half-antibody molecules, each containing an antigen-binding site [26]. An alternate method is based on the modification of antibodies via their carbohydrates, which are located in the CH 2 domain within the Fc region. Mild oxidation of the polysaccharide sugar residues with sodium periodate generates aldehyde groups, which can then be used for coupling to another molecule [26]. By proper selection of the conjugation reaction and knowledge of antibody structure, antibodies can be oriented so that their bivalent binding potential for antigen remains available.
Functionalization strategies of bare gold substrates via direct bio-SAM using disulfide-containing antibody chemically modified via their primary amines (A), carbohydrates (B), and carboxylic acids (C) and via classic SAM (D) using unmodified antibodies attached on a long chain of alkanethiols previously self-assembled onto a bare gold substrate
In this work, we developed a simple one-step surface functionalization method based on the covalent coupling of antibodies by chemical introduction of sulfur-derivative molecules into the antibody structure prior to its adsorption onto gold via the primary amines, carboxylic acids, and carbohydrates present in its structure. Additionally, a classic SAM approach was also evaluated using unmodified antibodies attached to a long chain of alkanethiols previously self-assembled onto a bare gold substrate. The model antibody-antigen system applied to carry out this work consisted of two monoclonal antibodies, a capture anti-NSE21 antibody and a reporter anti-NSE17 antibody, against neuron-specific enolase (NSE), a dimeric isoenzyme of the glycolytic enzyme enolase and derives from neuronal cytoplasm and neuroendocrine cells [27]. Several studies have demonstrated significantly lower NSE concentrations in serum in healthy subjects, with levels lower than 12.5 ng/mL, than in patients with acute ischemic stroke [28], and have thus been identified as a possible biological marker for the diagnosis of ischemic stroke [29] and used in an electrochemical sensor, achieving a detection limit of 0.18 ng/mL [30]. A Biacore® 3000 surface plasmon resonance (SPR) system was employed to characterize the different immobilization techniques and monitor the antibody-antigen interactions and the optimal antibody modification applied in an immunosensor, achieving a clinically relevant detection limit of 4.6 ng/mL.
Experimental
Chemicals and materials
NSE, anti-NSE21 monoclonal antibody (MAb), and anti-PSA66 (prostate-specific antigen) MAb were kindly supplied by Fuijirebio Diagnostics (Gothenburg, Sweden). Human carcinoembryonic antigen (CEA) was purchased from SCIPAC (Sittingbourne, UK). Both anti-PSA66 MAb and human CEA were employed for nonspecific binding studies as nonspecific ligand and as nonspecific marker, respectively. Bare gold substrates (SIA-kit), gold substrates mounted in chip (Au chip) dextran-coated gold substrates (CM5 chip), and HEPES buffered saline (HBS) (10 mM 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid, 150 mM NaCl, 3.4 mM ethylenediaminetetraacetate, and 0.005 % Tween 20 (pH 7.4)) were purchased from Biacore (GE Healthcare, Barcelona). Thiolated polyethylenglycol 1-(mercaptoundec-11-yl)-tetra(ethylene glycol) (PEG) was supplied by SensoPath Technologies (Bozeman, USA). Cystamine dihydrochloride, 1-ethyl-3-(3-dimethylamino-propyl)carbodiimide (EDC), N-hydroxysuccinimide (NHS), sulfo-NHS, dithiopropionic acid succinimidyl ester (DTPS), 16-mercapto-1-hexadecanoic acid (16-MHA), 11-mercapto-1-undecanol (11-MUOH), and carbonate-bicarbonate capsules for preparation of carbonate buffer (0.05 M, pH 9.6) were purchased from Sigma (Barcelona, Spain). Ethanol, acetone, dimethyl sulfoxide (DMSO), sodium di-hydrogen phosphate (NaH2PO4), and di-sodium hydrogen phosphate (Na2HPO4) were obtained from Panreac Química (Barcelona, Spain). Sodium chloride (NaCl), sodium hydroxide (NaOH), potassium chloride (KCl), sodium acetate, acetic acid, and sodium periodate were supplied by Scharlau (Barcelona, Spain). Centrifugal filter membranes of 100 molecular weight cutoff (MWCO) and 0.2 μm membrane filters were supplied by Whatman GmbH (Dassel, Germany). Deionized water was produced using a Milli-Q RG system (Millipore Ibérica, Madrid, Spain). The concentration of the chemically modified antibodies was determined using a Cary 100 UV-Vis spectrophotometer supplied by Varian (Barcelona, Spain). To evaluate the different immobilization techniques, a Biacore® 3000 surface plasmon resonance (SPR) system was used.
Chemical modification of biocomponents
Disulfide groups were covalently introduced into the structure of both anti-NSE21 and anti-PSA66 via its primary amines, carboxylic acids, and carbohydrates, whereas the anti-NSE17 was used in its unmodified state as a second primary antibody for sandwich assay experiments.
Chemical introduction of disulfides in anti-NSE21 antibody using −NH2 residues
Introduction of disulfides by this synthetic route was performed by reacting terminal primary amines and lysine residues of IgG with a disulfide-containing active ester, giving rise to a covalent attachment between disulfide groups and IgG. Anti-NSE21 (3.6 × 10−9 mol) diluted in 0.5 mL of 0.01 M carbonate buffer pH 9.5 was mixed with 0.07 mg of DTPS (1.8 × 10−7 mol) prepared in 0.05 mL of DMSO. The mixture was allowed to react in dark conditions for 5 h at room temperature under vigorous stirring. The excess of DTPS was removed by ultrafiltration (100 kDa MWCO membranes) and the modified antibody was recollected in PBS buffer pH 7.4. The concentration of the modified antibody was determined by UV-Vis spectrophotometry at 280 nm using an extinction coefficient of 1.38 mL/mg cm for 1 mg/mL IgG solutions in a 1-cm path length [31].
Chemical introduction of disulfides in anti-NSE21 antibody using carbohydrates
Polysaccharide residues were oxidized to aldehydes that react with primary amine groups of cystamine via the formation of Schiff bases. Initially, 0.5 mg of anti-NSE21 was diluted in 100 μL of 0.01 M acetate buffer pH 5.0, and then a 5-mM solution of sodium periodate was added to the antibody solution. The mixture was left to react in the dark and under stirring conditions for 1 h at room temperature. The oxidized antibody solution was slowly added to 900 μL of a 0.1-M cystamine solution diluted in 0.05 M carbonate buffer (pH 9.5) and left to react for 3 h at room temperature. In the next step, unstable imines were reduced to amine bonds by dropping 10 mM cyanoborohydride into the solution. Finally, the antibody solution was purified from the large excess of cystamine by filtration using 100 kDa MWCO membranes, and the modified antibody subsequently recollected in PBS buffer pH 7.4.
Chemical introduction of disulfides in anti-NSE21 antibody using −COOH residues
Disulfides were covalently attached to the IgG structure through the terminal carboxylic acids and the glutamic acid residues. Firstly, 0.5 mg of anti-NSE21 antibody (3.6 × 10−9 mol), 7.8 mg of sulfo-NHS (3.6 × 10−5 mol), and 6.9 mg of EDC (3.6 × 10−5 mol) were mixed and allowed to react for 10 min under stirring conditions at room temperature in 100 μL of 10 mM acetate buffer pH 4.5. Subsequently, this mixture was added to 900 μL of 50 mM HEPES buffer pH 8.5 containing 20 mg of cystamine (9 × 10−5 mol). Nucleophilic substitution took place during 2 h at room temperature under vigorous stirring conditions. Excess reagents and by-products were removed by filtration (100 kDa MWCO membranes) and the antibodies were dissolved in PBS buffer pH 7.4.
Biological functionalization of bare gold substrates
Different biological functionalization strategies of bare gold substrates were characterized using a Biacore® 3000 SPR system [32]. Surface functionalization was exploited using two different approaches: (i) direct bio-SAM using the chemically modified antibodies containing disulfide groups and (ii) classic SAM using unmodified antibodies attached to a long chain of alkanethiols previously self-assembled onto a bare gold substrate (Scheme 1). Additionally, the efficiency of these two approaches was compared with the performance of both a commercial carboxymethylated dextran surface (CM5 chip) and the immobilization of the unmodified antibody directly on gold substrates.
Direct bio-SAM approach
Bare gold substrates were rinsed with acetone and incubated for 15 min in an UV/O3 chamber to remove all organic contaminants on gold substrates [33]. Covalent immobilization of chemically modified antibodies containing disulfide groups was achieved by injecting 100 μL of the modified antibody solution (100 μg/mL in PBS buffer pH 7.4), followed by injection of 35 μL of a 1-mM PEG solution in order to block remaining free sites on the surface. A continuous flow of HBS (10 mM 4-(2-hydroxyethyl) piperazine-1-ethanesulfonic acid, 150 mM NaCl, 3.4 mM ethylenediaminetetraacetate, and 0.005 % v/v Tween 20 (pH 7.4)) at 5 μL/min was maintained during the immobilization step. To test the nonspecific adsorption of NSE on the PEG blocking layer, a control experiment was performed by injecting 100 μL of the nonspecific PSA66 antibody (100 μg/mL in PBS buffer pH 7.4) previously modified with disulfides via its primary amines, carboxylic acids, and carbohydrates as described above. Subsequently, the surface was backfilled by an injection of 35 μL of a 1-mM PEG solution. NSE detection experiments were performed by injecting serial dilutions of NSE (3.1–200 ng/mL) prepared in HBS buffer over the antibody-immobilized surface at a flow rate of 20 μL/min. After 6 min of association, the sample solution was replaced by a HBS buffer flow for 7 min, allowing the complex to dissociate. Regeneration of the surface was performed by injecting two pulses of 10 mM glycine (pH 2.2) between each analyte injection. The recognition experiments were carried out at 25 °C. Specificity of the recognition layer for NSE was tested by injecting two concentrations of human CEA (1 and 10 μg/mL) as a nonspecific marker.
In addition to the NSE recognition experiments, sandwich assays were also performed to test the specificity of the NSE binding. These assays consisted of a NSE recognition step by injecting both 100 and 200 ng/mL of NSE for 6 min, followed by a 10-μg/mL solution of unmodified NSE17 for a further 6 min. Subsequent to association, the sample solution was replaced by a HBS buffer flow for 7 min at 20 μL/min.
Classic SAM approach
Immediately after cleaning, the gold substrates were immersed in a mixture of 5 % 1 mM 16-MHA and 95 % 1 mM 11-MUOH in ethanol. Mixed SAMs were prepared in glass recipients cleaned with 2-M NaOH for at least 1 h. After 3 h of SAM deposition, the substrates were thoroughly rinsed with ethanol and dried under a stream of nitrogen, producing stable and fully covered SAMs on gold [34]. The mixed SAM used is considered as optimal for obtaining a high degree of antibody immobilization with maximal elimination of nonspecific adsorption due to the high content of thiols with OH end groups [35].
Covalent immobilization of unmodified anti-NSE21 monoclonal antibody was accomplished via coupling to their primary amines. A continuous flow of HBS at 5 μL/min was maintained during the immobilization step. The carboxylic groups of the mixed SAM were activated by injection of 50 μL of a 1:1 mixture of 400 mM EDC and 100 mM NHS in deionized water. Subsequently, 100 μL of the antibody solution (100 μg/mL in 10 mM acetate buffer pH 5.0) was injected, followed by injection of 35 μL of ethanolamine (1.0 M in deionized water, pH 8.5) in order to block the remaining NHS ester active groups, followed by two 10-μL injections of 10 mM glycine (pH 2.2) in order to remove nonspecifically bound molecules from the surface. To test the nonspecific binding of NSE, a control experiment was performed by injecting 100 μL of unmodified anti-PSA66 solution (100 μg/mL in 10 mM acetate buffer pH 5.0) as control antibody. The experiments were carried out at 25 °C.
Biofunctionalization of CM5
A commercial carboxymethylated dextran surface (CM5 chip) was used to immobilize the unmodified anti-NSE21. Immobilization was achieved via coupling to their primary amines to the previously activated carboxylic groups of the dextran matrix. Immobilization was performed in the same manner as described above for the classic SAM approach.
Direct immobilization on gold substrates of unmodified anti-NSE21
Immobilization of unmodified anti-NSE21 was carried out by injecting 100 μL of the antibody solution (100 μg/mL in PBS buffer pH 7.4), followed by an injection of 35 μL of a 1-mM thiolated PEG solution.
Electrochemical instrumentation
Electrochemical measurements were performed on a PC-controlled PGSTAT12 Autolab potentiostat (EcoChemie, the Netherlands) with an in-built frequency response analyzer FRA2 module. Electrochemical impedance measurements were performed using a standard three-electrode configuration (reference electrode: Ag/AgCl(sat), counter electrode Pt wire) in 1 mM Fe(CN)6 3−/4− in 0.1 M KCl, as previously described [36].
Electrochemical characterization of SAM formation of anti-NSE21-CHO-modified electrodes
For the impedimetric study of SAM formation, clean gold electrodes were functionalized by immersion in a freshly prepared 1-μg/mL solution of disulfide-modified anti-NSE21 in PBS for fixed times followed by rinsing with copious amounts of PBS-Tween. After each modification and washing, Faradaic EIS were recorded [36]; the electrodes were then washed with Milli-Q water and argon dried.
Electrochemical immunosensor construction and optimization
The incubation time of the specific recognition of NSE was optimized by immersion of the antibody-modified electrode in a 100-ng/mL solution of NSE in PBS at different incubation times (0–60 min) and recording the change in R ct of the Faradaic response 1 mM Fe(CN)6 3−/4− in 0.1 M KCl.
Electrochemical detection of NSE
A SAM of disulfide-modified antibody was formed by immersion of a clean electrode in a 1-μg/mL solution of NSE-CHO in PBS for 3 h followed by blocking with 1 mM 1-(mercaptoundec-11-yl)-tetra(ethylene glycol) in PBS for 30 min. The electrodes were then exposed to different concentrations of NSE antigen in PBS for 30 min, rinsed with PBS-Tween, and further incubated with 10 μg/mL of anti-NSE17-HRP conjugate in PBS for 10 min at room temperature. The differential pulse voltammetry (DPV) response was recorded in triplicate in the potential range 0.2 to −0.4 V versus Ag/AgCl using a modulation amplitude of 25 mV, a step potential of 5 mV, and a scan rate of 50 mV s−1 after 5 min of addition of a mixture of hydroquinone (1 mM) and hydrogen peroxide (1 mM) in PBS pH 6.
Results and discussion
Immobilization efficiency of modified and unmodified antibodies
Immobilization efficiency of the different functionalization strategies was evaluated by monitoring the coupling level of the antibodies on the gold substrates. For all immobilization strategies, PEG or ethanolamine, depending on the surface, was used to backfill the remaining free sites on the surface and to cap any remaining activated groups. Immobilization levels were measured after the addition of the backfiller or capping agent depending on the experiment (Fig. 1).
SPR signals for immobilization of unmodified NSE21 antibody on bare gold substrate; disulfide-containing antibody chemically modified via their primary amines (−NH2), carbohydrates (−CHO), and carboxylic acids (−COOH); unmodified antibody via mixed SAM; and unmodified antibody on a commercial layer (CM5 chip)
Based on the linear regression of the response and the amount of protein coated on the sensor surface, the theoretical surface concentrations of the antibodies were determined assuming that for matrix surfaces, an SPR signal of 1000 RU corresponds to 1 ng/mm2 of protein [37, 38]. For planar surfaces, however, this assumption needs to be corrected by a factor 3, so that 3000 RU equals 1 ng/mm2 [39, 40]. Additionally, each sensor’s maximum antigen-binding capacity (R max) was also evaluated for each immobilization strategy by using the following relation (1):
where MwAnalyte and MwLigand are the molecular weights of the analyte and ligand, respectively; R Ligand is the SPR response due to the ligand immobilization; and V Ligand is the valency of the ligand (amount of binding sites). The valency for IgGs is considered to be equal to 2 (Table 1).
Binding efficiency of the different functionalization strategies: NSE detection
The affinity of the anti-NSE antibody immobilized using different strategies was evaluated by capture assay by injecting serial dilutions of NSE (3.1–200 ng/mL) prepared in HBS buffer over the antibody-immobilized surfaces for an association time of 6 min. A zero analyte concentration was also included to obtain measurements for system-related bias. SPR signals for NSE recognition levels of gold substrates functionalized with both disulfide-containing and unmodified IgGs are depicted in Figs. 2 and 3, respectively. The degree of binding was calculated by measuring the response signal at the end of the dissociation phase in three replicate experiments subtracted by the signal from the control surface (nonspecific PSA66 MAb). In the case of the disulfide-containing antibodies (Fig. 2), only the antibodies modified via their carbohydrate moiety exhibited a typical sigmoidal response. Very low responses to different NSE concentrations were obtained for the IgGs modified via their amine or carboxylate groups. The carbohydrate chains are attached to the CH2 domain within the Fc region of the IgGs. This site-directed conjugation orientates the attached molecule away from the antigen-binding regions, preventing blockage of these sites and thus preserving activity [26]. Both amine and carboxylate groups within the 3D structure of an antibody are nearly uniform throughout the surface topology [26], and conjugation procedures that utilize these groups randomly cross link to many parts of the antibody molecule, leading to a random orientation of the antibody within the conjugate structure, often blocking the antigen-binding sites, resulting in a decrease in antigen-binding activity. The level of nonspecific adsorption of CEA on the surfaces containing the modified IgGs was lower than 9 RU and therefore insignificant.
SPR signals for NSE binding levels (capture assay) of gold substrates functionalized with disulfide-containing anti-NSE21 antibodies modified via their primary amines, carbohydrates, and carboxylic acids and immobilized on a bare gold substrate. Inset: NSE binding levels obtained with the anti-NSE21 modified via their primary amines and carboxylic acids. Error bars represent n = 3
Figure 3 illustrates the target response levels for the unmodified anti-NSE21 immobilized on a CM5 chip, a mixed SAM, and a bare gold substrate. NSE was only detected using the CM5 and mixed SAM functionalized surfaces. Target binding levels on these two immobilization strategies are very similar up to a concentration of 100 ng/mL, and at higher antigen concentrations, the mixed SAM surface starts to become saturated, as most of the antibodies have been associated with the target. On the other hand, the response on the CM5 keeps increasing linearly. This indicates the capacity of the CM5 chip to accommodate more antigen molecules, as was expected taking into account the high immobilization level of antibodies obtained for this 3D surface. The level of nonspecific adsorption of CEA on the surfaces functionalized with unmodified IgGs was lower than 5 RU for the both the CM5 and the surface containing unmodified IgG, whereas for the mixed SAM, the levels were lower than 11 RU. NSE detection using antibodies immobilized via the carbohydrate modification, on CM5 or via mixed SAMs, was also evaluated by measuring assay critical parameters such as the limit of detection (LOD), sensitivity, EC50 (concentration of the target needed to obtain 50 % of the maximum signal), and dynamic range. The sensitivity, EC50, and dynamic range were not determined for the CM5 chip because the top plateau was not defined by the experimental data, and thus, these parameters would lack accuracy. The LOD was taken as the mean concentration value obtained for three blanks plus three times the standard deviation of the blank. The low LOD (6.8 ± 2.1 ng/mL) obtained for the −CHO modification compared well with the other two strategies: classic SAM (1.8 ± 0.3 ng/mL) and commercial layer (3.3 ± 1.3 ng/mL) (Table 2).
SPR was also used to determine the strength of the antibody-antigen binding by calculating the dissociation constant (K D) using the BIAevaluation® software from Biacore. Experimental kinetic data was fitted to the 1:1 binding model and the goodness of fit was assessed by calculating the chi-square (χ 2). The K D for the −CHO, CM5, and mixed SAM strategies was of 4.4 × 10−11 M (χ 2 = 2.3), 4.8 × 10−12 M (χ 2 = 2.2), and 1.9 × 10−9 M (χ 2 = 5.1) respectively. Besides the low chi-square values obtained that suggested the good fitting to the ideal 1:1 binding model, differences in the K D of 1 and 2 orders of magnitude were exhibited among the different strategies. These results clearly indicate that binding kinetics were affected by the immobilization approaches, mass transfer limitations, and surface heterogeneity due to the covalent immobilization procedures. The 3D surface of the CM5 chip is prone to mass transport limitations, and therefore, the kinetic parameter is underestimated, probably due to (i) the slower transport step of the antigens to the immobilized anti-NSE21 because of the 3D dextran structure and (ii) the high binding capacity of ligands of this chip. Immobilization procedures on both the CM5 and mixed SAM are achieved via coupling primary amines of the antibodies, and this can modify the antigen-binding sites, resulting in a decrease in affinity. On the other hand, the −CHO strategy offers a 2D surface, less subjected to mass transfer limitations, where the antibody binding sites have been unaffected by chemical reactions, and thus, this surface is more reliable for binding kinetic calculations.
NSE recognition levels did not reach the theoretical maximum antigen-binding capacity (R max) for any of the immobilization techniques. This revealed that all immobilized ligand molecules were not fully accessible or functional, or that the NSE concentration assayed was not high enough to interact with all ligand molecules. The stoichiometry of the binding of the NSE to antibody was calculated using the molecular mass values for the anti-NSE MAb (150 kDa) and NSE (78 kDa) and their immobilization degree and response level. To this end, experimental stoichiometries of anti-NSE21 were 0.11, 0.20, and 0.03 for carbohydrate modification, mixed SAM, and CM5 chip, respectively.
For the recognition experiments, the flow rate was increased to 20 μL/min to minimize rebinding and to reduce mass transport limitations, allowing rapid diffusion of the analyte from the bulk solution to the surface. Sensor chips were regenerated by selective dissociation of the analyte from the covalently immobilized ligand. Conditions were chosen to achieve complete dissociation of the analyte without affecting the binding characteristics of the ligand. Regeneration of the surfaces was achieved by injecting two pulses of 10 mM glycine (pH 2.2) between each analyte injection [41]. Regeneration efficiencies were higher than the 90 % for all experiments. Sandwich assays were performed to test the specificity of the NSE binding, consisting of a NSE recognition step by injecting 100 and 200 ng/mL of NSE, followed by injection of 10 μg/mL of unmodified NSE17 (Fig. 4). Binding responses obtained for the addition of the second primary antibody demonstrated the higher presence of analyte molecules bound on the surface prepared using the −CHO modification, mixed SAM, and CM5 chip, confirming the results obtained for the direct capture of NSE. Both carbohydrate modification and mixed SAM displayed the highest responses for 100 ng/mL of NSE, while for an antigen concentration of 200 ng/mL the best surface was the −CHO modified. While high binding levels were obtained for the direct capture of NSE in a CM5 chip, low binding levels of NSE17 were observed, which can be attributed to a highly packed surface too dense to accommodate the subsequent binding of the second antibody.
SPR signals for anti-NSE17 binding levels (sandwich assay) for a gold substrate functionalized with anti-NSE21. Unmodified antibodies were immobilized on a CM5 chip, mixed SAM, and bare gold substrate. Disulfide-containing antibodies were modified via their primary amines, carbohydrates, and carboxylic acids immobilized on a bare gold substrate. Error bars represent n = 3
Electrochemical detection of NSE
The immobilization of CHO-modified anti-NSE on gold electrodes used as an electrochemical immunosensor was explored. Impedance changes following different antibody immobilization times were monitored. Charge transfer resistance (R ct) values, indicative of the opposition of the interface to the passage of electrical current from an electroactive probe present in solution, were obtained from simulation of the equivalent circuit shown in Scheme 2, where C dl is the double layer capacitance and R s is the solution resistance of the circuit. The R ct values increased steadily with time (Fig. 5), reaching saturation after 3 h. This impedance increase does not account for multilayer formation since each point represents the constant impedance value obtained after repeated washings to remove physically adsorbed molecules. The maximum deposition time for NSE-CHO is considerably lower than that observed for the formation of SAMs of alkanethiols, which usually require an overnight exposure of the electrodes to the modifying solution in order to form a compact monolayer, highlighting the advantage of the direct attachment of modified antibodies on surfaces in immunosensor construction. The same procedure was employed in order to optimize the time required for the specific recognition of NSE (Fig. 5, inset). The impedance response increased up to saturation after 30 min of interaction and this time was thus used in the detection experiments. Electrochemical determination of NSE was carried out using anti-NSE17-HRP conjugate as reporter antibody with differential pulse voltammetric detection. Figure 6a shows the DPV responses of the immunosensor with increasing concentrations of NSE in the potential range 0.1 to −0.4 V. As can be seen from Fig. 6b, the peak height increased with NSE concentration and showed a linear relationship with the logarithm of the NSE concentration. The limit of detection (4.6 ng/mL) was calculated with a linear relationship between 0 and 25 ng/mL.
Conclusions
Different strategies have been developed for antibody immobilization, based on the chemical modification of their functional groups with disulfide “anchors” able to spontaneously chemisorb onto gold, with no need for surface prefunctionalization. Among the three chemical routes investigated, the site-directed conjugation of antibodies via their carbohydrate chains exhibited a good analyte response in both capture and sandwich assays using SPR. Surfaces prepared with this approach also compared well with both the classic two-step SAM scenario and the 3D-CM5 chip in terms of analyte response, LOD, and sensitivity, suggesting that the immobilization of carbohydrate-modified antibodies driven by chemisorption of their disulfide moieties represents a successful approach for creating biologically active dense monolayers on gold devices due to an optimization of orientation of the capture (primary) antibody. The chemical specificity of the reaction toward carbohydrate residues opens up an attractive option for oriented antibody immobilization since their sugar moieties are specifically located on the constant region of the immunoglobulins. Finally, in terms of simplicity, required time, and minimal use of reagents, the use of modified carbohydrate residues presents an extremely effective approach for antibody immobilization with application in electrochemical, optical, and gravimetric immunosensors.
References
Suárez G, Jackson RJ, Spoors JA, McNeil CJ. Chemical introduction of disulfide groups on glycoproteins: a direct protein anchoring scenario. Anal Chem. 2007;79:1961–9. doi:10.1021/ac0613030.
Baniukevic J, Kirlyte J, Ramanavicius A, Ramanaviciene A. Application of oriented and random antibody immobilization methods in immunosensor design. Sensors Actuators B Chem. 2013;189:217–23. doi:10.1016/j.snb.2013.03.126.
Baniukevic J, Hakki Boyaci I, Goktug Bozkurt A, Tamer U, Ramanavicius A, Ramanaviciene A. Magnetic gold nanoparticles in SERS-based sandwich immunoassay for antigen detection by well oriented antibodies. Biosens Bioelectron. 2013;43:281–8. doi:10.1016/j.bios.2012.12.014.
Vashist SK, Dixit CK, MacCraith BD, O’Kennedy R. Effect of antibody immobilization strategies on the analytical performance of a surface plasmon resonance-based immunoassay. Analyst. 2011;136:4431. doi:10.1039/c1an15325k.
Makaraviciute A, Ramanaviciene A. Site-directed antibody immobilization techniques for immunosensors. Biosens Bioelectron. 2013;50:460–71. doi:10.1016/j.bios.2013.06.060.
Trilling AK, Beekwilder J, Zuilhof H. Antibody orientation on biosensor surfaces: a minireview. Analyst. 2013;138:1619–27. doi:10.1039/c2an36787d.
Castner DG, Ratner BD. Biomedical surface science: foundations to frontiers. Surf Sci. 2002;500:28–60. doi:10.1016/S0039-6028(01)01587-4.
Borges J, Campiña JM, Silva AF. Chitosan biopolymer-F(ab′)2 immunoconjugate films for enhanced antigen recognition. J Mater Chem B. 2013;1:500–11. doi:10.1039/c2tb00115b.
Baniukevic J, Kirlyte J, Ramanavicius A, Ramanaviciene A. Comparison of oriented and random antibody immobilization techniques on the efficiency of immunosensor. Proc Eng. 2012;47:837–40. doi:10.1016/j.proeng.2012.09.277.
Niu Y, Matos AI, Abrantes LM, Viana AS, Jin G. Antibody oriented immobilization on gold using the reaction between carbon disulfide and amine groups and its application in immunosensing. Langmuir. 2012;28:17718–25. doi:10.1021/la303032f.
Ferreira NS, Sales MGF. Disposable immunosensor using a simple method for oriented antibody immobilization for label-free real-time detection of an oxidative stress biomarker implicated in cancer diseases. Biosens Bioelectron. 2014;53:193–9. doi:10.1016/j.bios.2013.09.056.
Perez JB, Tyagi D, Mo Y, Calvo L, Perez R, Moreno E, et al. Predicting the right spacing between protein immobilization sites on self-assembled monolayers to optimize ligand binding. Anal Biochem. 2015;484:133–5. doi:10.1016/j.ab.2015.05.005.
Luna DMN, Avelino KYPS, Cordeiro MT, Andrade CAS, Oliveira MDL. Electrochemical immunosensor for dengue virus serotypes based on 4-mercaptobenzoic acid modified gold nanoparticles on self-assembled cysteine monolayers. Sensors Actuators B Chem. 2015;220:565–72. doi:10.1016/j.snb.2015.05.067.
Hazarika P, Behrendt JM, Petersson L, Wingren C, Turner ML. Photopatterning of self assembled monolayers on oxide surfaces for the selective attachment of biomolecules. Biosens Bioelectron. 2014;53:82–9. doi:10.1016/j.bios.2013.09.001.
Kausaite-Minkstimiene A, Ramanaviciene A, Kirlyte J, Ramanavicius A. Comparative study of random and oriented antibody immobilization techniques on the binding capacity of immunosensor. Anal Chem. 2010;82:6401–8. doi:10.1021/ac100468k.
De Juan-Franco E, Caruz A, Pedrajas JR, Lechuga LM. Site-directed antibody immobilization using a protein A-gold binding domain fusion protein for enhanced SPR immunosensing. Analyst. 2013;138:2023–31. doi:10.1039/c3an36498d.
Miyao H, Ikeda Y, Shiraishi A, Kawakami Y, Sueda S. Immobilization of immunoglobulin-G-binding domain of protein A on a gold surface modified with biotin ligase. Anal Biochem. 2015;484:113–21. doi:10.1016/j.ab.2015.05.010.
Tajima N, Takai M, Ishihara K. Significance of antibody orientation unraveled: well-oriented antibodies recorded high binding affinity. Anal Chem. 2011;83:1969–76. doi:10.1021/ac1026786.
Crivianu-Gaita V, Thompson M. Immobilization of Fab′ fragments onto substrate surfaces: a survey of methods and applications. Biosens Bioelectron. 2015;70:167–80. doi:10.1016/j.bios.2015.03.032.
Trilling AK, Hesselink T, Van Houwelingen A, Cordewener JHG, Jongsma MA, Schoffelen S, et al. Orientation of llama antibodies strongly increases sensitivity of biosensors. Biosens Bioelectron. 2014;60:130–6. doi:10.1016/j.bios.2014.04.017.
Balevicius Z, Ramanaviciene A, Baleviciute I, Makaraviciute A, Mikoliunaite L, Ramanavicius A. Evaluation of intact- and fragmented-antibody based immunosensors by total internal reflection ellipsometry. Sensors Actuators B Chem. 2011;160:555–62. doi:10.1016/j.snb.2011.08.029.
Nassef HM, Civit L, Fragoso A, O’Sullivan CK. Amperometric immunosensor for detection of celiac disease toxic gliadin based on Fab fragments. Anal Chem. 2009;81:5299–307. doi:10.1021/ac9005342.
Vikholm I. Self-assembly of antibody fragments and polymers onto gold for immunosensing. Sensors Actuators B Chem. 2005;106:311–6. doi:10.1016/j.snb.2004.07.034.
Bonroy K, Frederix F, Reekmans G, Dewolf E, De Palma R, Borghs G, et al. Comparison of random and oriented immobilisation of antibody fragments on mixed self-assembled monolayers. J Immunol Methods. 2006;312:167–81. doi:10.1016/j.jim.2006.03.007.
Prieto-Simón B, Saint C, Voelcker NH. Electrochemical biosensors featuring oriented antibody immobilization via electrografted and self-assembled hydrazide chemistry. Anal Chem. 2014;86:1422–9. doi:10.1021/ac401747j.
Sorci M, Dassa B, Liu H, Anand G, Dutta AK, Pietrokovski S, et al. Oriented covalent immobilization of antibodies for measurement of intermolecular binding forces between zipper-like contact surfaces of split inteins. Anal Chem. 2013;85:6080–8. doi:10.1021/ac400949t.
Ho J-AA, Hsu W-L, Liao W-C, Chiu J-K, Chen M-L, Chang H-C, et al. Ultrasensitive electrochemical detection of biotin using electrically addressable site-oriented antibody immobilization approach via aminophenyl boronic acid. Biosens Bioelectron. 2010;26:1021–7. doi:10.1016/j.bios.2010.08.048.
Hermanson GT. Bioconjugate techniques. Rockford: Thermo Fischer Scientific; 2013. doi: 10.1016/B978-0-12-382239-0.00026-1.
Barone FC, Clark RK, Price WJ, White RF, Feuerstein GZ, Storer BL, et al. Neuron-specific enolase increases in cerebral and systemic circulation following focal ischemia. Brain Res. 1993;623:77–82.
Zhong Z, Peng N, Qing Y, Shan J, Li M, Guan W, et al. An electrochemical immunosensor for simultaneous multiplexed detection of neuron-specific enolase and pro-gastrin-releasing peptide using liposomes as enhancer. Electrochim Acta. 2011;56:5624–9.
Johnstone A, Thorpe R. Immunochemistry in practice. 2nd ed. Oxford: Blackwell Scientific; 1988.
Jönsson U, Fägerstam L, Ivarsson B, Johnsson B, Karlsson R, Lundh K, et al. Real-time biospecific interaction analysis using surface plasmon resonance and a sensor chip technology. Biotechniques. 1991;11:620–7.
Moon DW, Kurokawa A, Ichimura S, Lee HW, Jeon IC. Ultraviolet-ozone jet cleaning process of organic surface contamination layers. J Vac Sci Technol A Vacuum, Surfaces, Film 1999;17:150. doi: 10.1116/1.581565
Frederix F, Bonroy K, Reekmans G, Laureyn W, Campitelli A, Abramov MA, et al. Reduced nonspecific adsorption on covalently immobilized protein surfaces using poly(ethylene oxide) containing blocking agents. J Biochem Biophys Methods. 2004;58:67–74.
Frederix F, Bonroy K, Laureyn W, Reekmans G, Campitelli A, Dehaen W, et al. Enhanced performance of an affinity biosensor interface based on mixed self-assembled monolayers of thiols on gold. Langmuir. 2003;19:4351–7. doi:10.1021/la026908f.
Fragoso A, Laboria N, Latta D, O’Sullivan CK. Electron permeable self-assembled monolayers of dithiolated aromatic scaffolds on gold for biosensor applications. Anal Chem. 2008;80:2556–63.
Biacore sensor surface handbook. Uppsala: Biacore AB; 2003.
Stenberg E, Persson B, Roos H, Urbaniczky C. Quantitative determination of surface concentration of protein with surface plasmon resonance using radiolabeled proteins. J Colloid Interface Sci. 1991;143:513–26. doi:10.1016/0021-9797(91)90284-F.
Gizeli E, Lowe CR. Biomolecular sensors. London: Taylor & Francis; 2002.
Schasfoort RBM, Tudos AJ. Handbook of surface plasmon resonance. Cambridge: RSC Pub; 2008.
Andersson K, Areskoug D, Hardenborg E. Exploring buffer space for molecular interactions. J Mol Recognit. 1999;12:310–5.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Acero Sánchez, J.L., Fragoso, A., Joda, H. et al. Site-directed introduction of disulfide groups on antibodies for highly sensitive immunosensors. Anal Bioanal Chem 408, 5337–5346 (2016). https://doi.org/10.1007/s00216-016-9630-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9630-9