Abstract
Background
It has been known that anesthetic adjuvants such as dexamethasone or ketamine might change mood. This study aimed to investigate the effects of a single dose of each drug individually along with their combined usage on postoperative mood changes in patients undergoing gynecologic surgery.
Methods
Two hundred ninety-seven patients randomly allocated were divided into three groups. Group K (n = 99) received a single dose of ketamine (0.5 mg/kg iv); group D (n = 99) received a single dose of dexamethasone (0.1 mg/kg iv), and group KD (n = 99) received both ketamine (0.5 mg/kg iv) and dexamethasone (0.1 mg/kg iv) at 5 min after the induction of anesthesia. A change in the patient health questionnaire (PHQ)-9 scores on the first and third day after surgery, the duration of anesthesia, the postoperative visual analog scale (VAS) for pain, and the patient controlled analgesia (PCA) consumption were evaluated.
Results
Groups K and KD showed a significant reduction in PHQ-9 score on both the first and third day after surgery compared with those recorded preoperatively and in group D (P < 0.01). There were no differences in the group D PHQ-9 scores pre- and post-operatively. The VAS for pain 24 h after surgery and the PCA consumption in group KD decreased significantly compared to the other two groups (P < 0.05).
Conclusions
A single dose of ketamine (0.5 mg/kg) with or without combination with dexamethasone (0.1 mg/kg) give iv 5 min after induction of general anesthetic produced significant improvement in the postoperative mood scores. A single intravenous dose of dexamethasone (0.1 mg/kg) alone did not change postoperative mood scores. The VAS for pain 24 h after surgery and the PCA consumption was significantly lower in patients who received combination of both drugs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Many studies have reported that ketamine or dexamethasone used as anesthetic adjuvants might not only be effective analgesics, but may also treat mood disorders such as depression (Safavi et al. 2012; Dinan et al. 1997; Arana et al. 1995; Garay et al. 2017; Jiang et al. 2016). In case of dexamethasone, it is well known as a potent antiemetic.
Most of the drugs used to treat depression act on the monoamine systems of the brain. These drugs tend to be slow to elicit anti-depressant effects (Ponto et al. 1977). However, a single intravenous sub-anesthetic dose of ketamine, an N-methyl-D-aspartate (NMDA) antagonist, exerts rapid anti-depressant effects in patients with treatment-resistant mood disorders (Garay et al. 2017; Salvadore and Singh 2013). A high dose of dexamethasone, the synthetic glucocorticoid, for a short duration was effective in treating refractory major depression (Arana et al. 1995; Dinan et al. 1997).
As many as 20% of hospitalized surgical patients are found to have a mood somewhat more negative after surgery compared to the average healthy population who have never had surgery (Urban-Baeza et al. 2015). Previous studies have reported that depressive symptoms during the perioperative period are associated with poor outcomes following surgery (Pakarinen et al. 2016; Tharin et al. 2012).
The PHQ-9 is a multipurpose instrument given to patients in a primary care setting to screen for the presence and to monitor the severity of depression and response to treatment. It can, however, be used to make a tentative diagnosis of depression in at-risk populations (Cameron et al. 2008; Haddad et al. 2013).
We hypothesized that ketamine or dexamethasone might change postoperative mood, and the combination of two drugs enhanced more than the individual drug on post-operative mood scores with PHQ-9 in patients undergoing laparoscopically assisted-gynecologic surgery.
Materials and methods
Ethical approval for this study (Registration No. 3031) was provided by the Institutional Review Board (IRB) in April 2016. Written informed consent was obtained from all participants. The study was performed at University Hospital from August 2017 to January 2018. Patients who were scheduled for laparoscopic gynecologic surgery were enrolled in this study. The trial was registered at clinicaltrials.gov (NCT03194594).
Patients with hepatic and renal diseases, history of allergy to the drugs being studied, previous gastric ulcer, diabetes mellitus, exposure to analgesics, antiemetics, antihistamines, steroids, or psychiatric drugs within 24 h before surgery were excluded from the study.
Two hundred ninety-seven patients between the ages of 18 and 70 years, classified into classes I and II according to the American Society of Anesthesiologists (ASA) were enrolled. Following this, the patients were randomly (sealed envelopes) categorized into the three treatment groups: group K (n = 99) received a single dose of ketamine (0.5 mg/kg iv) plus 2 ml normal saline; group D (n = 99) received dexamethasone (0.1 mg/kg iv, with a maximum dose of 8 mg) plus 2 ml saline, and group KD (n = 99) received ketamine (0.5 mg/kg iv) and dexamethasone (0.1 mg/kg iv, with a maximum dose of 8 mg), 5 min after the induction of anesthesia in a double-blinded method.
On the day before surgery, patients were taught how to use the visual analog scale (VAS) and the patient-controlled analgesia (PCA) device. They were instructed to deliver analgesia on their own whenever they experienced pain.
All patients were not pre-medicated before arriving in the operating room. The patients were placed on pulse oximeter, automated blood pressure (BP) cuff, electrocardiogram (EKG), and end-tidal CO2 (ETCO2) devices. In addition, urinary catheters were attached as part of the patient’s management. Tympanic temperature was measured immediately before induction of anesthesia and again just before extubation.
The induction of anesthesia began with a slow (30–60 s) iv bolus dose of propofol (2 mg/kg). Tracheal intubation was facilitated with rocuronium (0.9 mg/kg) in all groups. Anesthesia was maintained with desflurane and a mixture of air and 50% oxygen. When desflurane was required, the administration was started at the end-tidal concentration of 1 minimum alveolar concentration (MAC), and the concentration was adjusted by a 1% stepwise titration according to acceptable hemodynamic limits (mean arterial blood pressure between − 30 and + 15% and heart rate between − 40 and + 15%) and to a target bispectral index (BIS) between 40 and 60.
Upon completion of the surgery, neuromuscular blockade was reversed with pyridostigmine (0.2 mg/kg) and glycopyrrolate (0.008 mg/kg) when the train-of-four (TOF) ratio had returned to 25%. When BIS values reached 80 and spontaneous breathing was achieved, extubation was performed.
An anesthesiologist performed the assessment of perioperative depressed mood change using the Patient Health Questionnaire-9 (PHQ-9). The PHQ-9 is the depression module, which scores each of the nine DSM-IV criteria as “0” (not at all) to “3” (nearly every day) with a total score ranging from 0 to 27, 0–4 none, 5–9 mild, 10–14 moderate, 15–19 moderately severe, and 20–27 severe depressed mood (Table 1).
Each patient was administered analgesics using a PCA pump containing morphine (60 mg), ketorolac (150 mg), and ramosetron (0.6 mg) in a total volume of 100 ml of saline. This device was set to deliver a basal infusion of 2 ml/h and bolus doses of 0.5 ml with a 15-min lockout period. Postoperative pain intensity was documented using a 100-mm linear visual analog scale (VAS). The VAS consisted of a straight line with the left end of the line representing no pain and the right end of the line representing the worst pain. Patients were asked to mark the position on the line corresponding to their perception of pain. The VAS score for pain was measured at intervals of 1, 6, 24, and 48 h after surgery.
The change in the PHQ-9 scores on the first day after surgery was the primary outcome in this study. The secondary outcomes included the PHQ-9 score on the third day after surgery, duration of anesthesia, postoperative visual analog scale (VAS) for pain, and patient-controlled analgesia (PCA) consumption in the 24-h-period following surgery, side effects of the study drugs. Nausea or vomiting were treated with IV ondansetron (4 mg).
The sample size in the present study was calculated by using PASS 2008 (NCSS, LLC. Kaysville, Utah, USA) software. A preliminary investigation showed that hypothesized means of the three treatment groups for postoperative mood change after surgery as a primary outcome were 3.10, 3.50, and 2.95 respectively, with 0.67 standard deviation (SD) between subjects and effect size 0.19. Thus, a sample size of 90 patients per group was required to demonstrate a significant difference with a power of 80% and an α-coefficient of 0.05. Assuming a 10% dropout rate, the final sample size was determined to be 99 patients per group. The statistical analysis was performed using SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA). The results are presented as mean ± standard deviation (SD) or percentage of patients. Comparisons of age, body weight, anesthetic time, VAS for pain intensity, PHQ-9, and the PCA consumption for 24 h after surgery among the groups were conducted using one-way analysis of variance. Post hoc comparisons were performed with Bonferroni correction of the significance level. Analysis of the pre- and post-operative PHQ-9 change was conducted using paired t test. Chi-square test was used to analyze postoperative PONV and the side effects of the drugs. P < 0.05 was considered statistically significant.
Results
A total of 297 patients were assessed for eligibility and received study medication after randomization. Sixteen patients were initially enrolled but withdrawn due to a conversion to open surgery, lost to follow-up, re-exploration for postoperative bleeding, or refusal to postoperative assessment (Fig. 1).
There were no significant differences between the three treatment groups with respect to age, weight, duration of anesthesia, and side effects (hallucination or genital pruritus; Table 2).
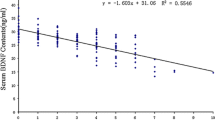
The PHQ-9 score was significantly reduced in groups K and KD on the first and third day after surgery compared with pre-operation and group D scores (P < 0.01). There was no significant differences in the PHQ-9 score between groups K and KD. There were no differences in the pre- and post-operative PHQ-9 scores of group D (Fig. 2).
The VAS for pain for the 24 h following surgery and the PCA consumption in group KD decreased significantly compared to the other two groups (P < 0.05). There were no significant differences in PONV incidence among the three treatment groups (Table 2).
Discussion
This present study showed that the PHQ-9 score in groups K and KD who received ketamine was significantly reduced on the first and third day after surgery compared with pre-operation and group D who received dexamethasone. There were no differences in the pre- and post-operative PHQ-9 scores of group D. The PHQ-9 score between groups K and KD were not significantly different. Therefore, there was no synergic effect of combining the two drugs on mood change.
Several studies show ketamine’s ability to produce rapid and robust antidepressant effects in patients with mood and anxiety disorders that were previously resistant to treatment (Abdallah et al. 2015; Hu et al. 2016; Wan et al. 2015) Preclinical studies have provided compelling insights into the mechanisms underlying the rapid-acting antidepressant effects of ketamine, which are mediated by a glutamate surge that leads to a cascade of events resulting in synaptogenesis and the reversal of the negative effects of chronic stress and depression, particularly within the prefrontal cortex (PFC) (Mjellem et al. 1993; Paul et al. 1994).
The mechanism by which the corticosteroid induces antidepressant effect is not clear. Some available evidences suggest the direct action of steroids on central monoaminergic neurons such as noradrenergic (NA) and 5-hydroxytryptaminergic (5-HT) neurons. The administration of corticosteroid is associated with decreased levels of corticotrophin, norepinephrine, and beta-endorphin in the cerebrospinal fluid (Brown 2009; Bhatt et al. 2016; Ciriaco et al. 2013).
The present study showed that the combination of two drugs tended to reduce the antidepressant effect of ketamine than synergic effect as shown Fig. 2, although not statistically significant. Corticosteroids induce an increased release of glutamate that induces neuronal toxicity due to accumulation effect (Benyamin et al. 2008). It may explain for the reduction of dexamethasone on ketamine-induced antidepressant effect in the present study.
However, the effect of corticosteroids on psychiatric symptoms including mood change depends on dosage, and often occur early in treatment. Wolkowitz et al. (Mohtadi et al. 2014) reported that healthy 12 volunteers 80 mg of prednisone daily for 5 days showed some symptoms including depressed or elevated mood. Mohtadi et al. (Mohtadi et al. 2014) reported perioperative single dose systemic dexamethasone (0.1 mg/kg) improved mood as secondary outcomes. The potential beneficial effects of short-term dexamethasone therapy remain to be seen. Therefore, we investigated the effect of dexamethasone on mood change as primary outcome. A single dose schedule of dexamethasone showed unstable therapeutic effect in our results.
In the present study, the combination of ketamine and dexamethasone reduced postoperative pain and analgesic consumption more effectively than each drug individually. The anti-inflammatory effect of dexamethasone, which decreases local edema and pain (Benyamin et al. 2008; Mohtadi et al. 2014), and the analgesic effect of ketamine may contribute to the results obtained in this study.
A previous study reported that the combination of dexamethasone and ketamine did not seem to offer any advantages, including pain relief, during the postoperative interval (Brown 2009). These inconsistent findings might result from the variability in the type of surgery, the dose and timing of dexamethasone, the anesthetic regimen, and the type of postoperative rescue analgesic used.
Our study had some limitations. We only recorded total PCA consumption for the first 24 h following surgery. Therefore, our results cannot document whether the opioid-sparing effect of the combined use of dexamethasone with ketamine was prolonged beyond the expected duration of action or whether it was limited merely to the early postoperative period.
Another limitation of the current study was that we did not repeat the dexamethasone-ketamine combination in the postoperative period. Therefore, the effects of continuing the medication could not be evaluated.
In conclusion, there is great interest in developing novel and rapid antidepressants with greater target specificity and/or decreased adverse effects. Ketamine alone and in combination with dexamethasone showed satisfactory effects on mood change after surgery. Future research into the pharmacotherapy of mood change in response to single dose of dexamethasone in patients undergoing surgery is needed.
References
Abdallah CG, Sanacora G, Duman RS, Krystal JH (2015) Ketamine and rapid-acting antidepressants: a window into a new neurobiology for mood disorder therapeutics. Annu Rev Med 66:509–523
Arana GW, Santos AB, Laraia MT, McLeod-Bryant S, Beale MD, Rames LJ, Roberts JM, Dias JK, Molloy M (1995) Dexamethasone for the treatment of depression: a randomized, placebo-controlled, double-blind trial. Am J Psychiatry 152:265–267
Benyamin RM, Vallejo R, Kramer J, Rafeyan R (2008) Corticosteroid induced psychosis in the pain management setting. Pain Physician 11:917–920
Bhatt S, Shukla P, Raval J, Goswami S (2016) Role of aspirin and dexamethasone against experimentally induced depression in rats. Basic Clin Pharmacol Toxicol 119:10–18
Brown ES (2009) Effects of glucocorticoids on mood, memory, and the hippocampus. Treatment and preventive therapy. Ann N Y Acad Sci 1179:41–55
Cameron IM, Crawford JR, Lawton K, Reid IC (2008) Psychometric comparison of PHQ-9 and HADS for measuring depression severity in primary care. Br J Gen Pract 58:32–36
Ciriaco M, Ventrice P, Russo G, Scicchitano M, Mazzitello G, Scicchitano F, Russo E (2013) Corticosteroid-related central nervous system side effects. J Pharmacol Pharmacother 4:S94–S98
Dinan TG, Lavelle E, Cooney J, Burnett F, Scott L, Dash A, Thakore J, Berti C (1997) Dexamethasone augmentation in treatment-resistant depression. Acta Psychiatr Scand 95:58–61
Garay RP, Zarate CA Jr, Charpeaud T, Citrome L, Correll CU, Hameg A, Llorca PM (2017) Investigational drugs in recent clinical trials for treatment-resistant depression. Expert Rev Neurother 17:593–609
Haddad M, Walters P, Phillips R, Tsakok J, Williams P, Mann A, Tylee A (2013) Detecting depression in patients with coronary heart disease: a diagnostic evaluation of the PHQ-9 and HADS-D in primary care, findings from the UPBEAT-UK study. PLoS One 8:e78493
Hu YD, Xiang YT, Fang JX, Zu S, Sha S, Shi H, Ungvari GS, Correll CU, Chiu HFK, Xue Y, Tian TF, Wu AS, Ma X, Wang G (2016) Single iv ketamine augmentation of newly initiated escitalopram for major depression: results from a randomized, placebo-controlled 4-week study. Psychol Med 46:623–635
Jiang M, Wang MH, Wang XB, Liu L, Wu JL, Yang XL, Liu XR, Zhang CX (2016) Effect of intraoperative application of ketamine on postoperative depressed mood in patients undergoing elective orthopedic surgery. J Anesth 30:232–237
Mjellem N, Lund A, Hole K (1993) Reduction of NMDA-induced behaviour after acute and chronic administration of desipramine in mice. Neuropharmacology 32:591–595
Mohtadi A, Nesioonpour S, Salari A, Akhondzadeh R, Masood Rad B, Aslani SM (2014) The effect of single-dose administration of dexamethasone on postoperative pain in patients undergoing laparoscopic cholecystectomy. Anesth Pain Med 4:e17872
Pakarinen M, Tuomainen I, Koivumaa-Honkanen H, Sinikallio S, Lehto SM, Airaksinen O, Viinamäki H, Aalto T (2016) Life dissatisfaction is associated with depression and poorer surgical outcomes among lumbar spinal stenosis patients: a 10-year follow-up study. Int J Rehabil Res 39:291–295
Paul IA, Nowak G, Layer RT, Popik P, Skolnick P (1994) Adaptation of the N-methyl-D-aspartate receptor complex following chronic antidepressant treatments. J Pharmacol Exp Ther 269:95–102
Ponto LB, Perry PJ, Liskow BI, Seaba HH (1977) Drug therapy reviews: tricyclic antidepressant and monoamine oxidase inhibitor combination therapy. Am J Hosp Pharm 34:954–961
Safavi M, Honarmand A, Habibabady MR, Baraty S, Aghadavoudi O (2012) Assessing intravenous ketamine and intravenous dexamethasone separately and in combination for early oral intake, vomiting and postoperative pain relief in children following tonsillectomy. Med Arh 66:111–115
Salvadore G, Singh JB (2013) Ketamine as a fast acting antidepressant: current knowledge and open questions. CNS Neurosci Ther 19:428–436
Tharin S, Mayer E, Krishnaney A (2012) Lumbar microdiscectomy and lumbar decompression improve functional outcomes and depression scores. Evid Based Spine Care J 3:65–66
Urban-Baeza A, Zárate-Kalfópulos B, Romero-Vargas S, Obil-Chavarría C, Brenes-Rojas L, Reyes-Sánchez A (2015) Influence of depression symptoms on patient expectations and clinical outcomes in the surgical management of spinal stenosis. J Neurosurg Spine 22:75–79
Wan LB, Levitch CF, Perez AM, Brallier JW, Iosifescu DV, Chang LC, Foulkes A, Mathew SJ, Charney DS, Murrough JW (2015) Ketamine safety and tolerability in clinical trials for treatment-resistant depression. J Clin Psychiatry 76:247–252
Funding
This study was supported by Wonkwang University in 2018.
Author information
Authors and Affiliations
Contributions
C Lee: study design, data collection, data analysis, patient recruitment, revising the article critically for important intellectual content, and final approval of the version to be submitted.
J Lee: data collection, patient recruitment, and final approval of the version to be submitted.
J Hwang: study design, data analysis, revising the article critically for important intellectual content, and final approval of the version to be submitted.
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, C., Lee, J., Lee, G. et al. The effects of a combination of intravenous dexamethasone and ketamine on postoperative mood in patients undergoing laparoscopically assisted-gynecologic surgery. Psychopharmacology 235, 2417–2422 (2018). https://doi.org/10.1007/s00213-018-4939-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4939-z