Abstract
Rationale
Withdrawal symptoms stand as a core feature of alcohol dependence. Our previous results have shown that inhibition of phosphodiesterase-4 (PDE4) decreased ethanol seeking and drinking in alcohol-preferring rodents. However, little is known about whether PDE4 is involved in ethanol abstinence-related behavior.
Objective
The objective of this study was to characterize the role of PDE4 in the development of anxiety- and depressive-like behavior induced by abstinence from ethanol exposure in different animal models.
Methods
Using three rodent models of ethanol abstinence, we examined the effects of rolipram, a prototypical, selective PDE4 inhibitor, on (1) anxiety-like behavior induced by repeated ethanol abstinence in the elevated plus maze test in fawn-hooded (FH/Wjd) rats, (2) anxiety-like behavior in the open-field test and light-dark transition test following acute ethanol abstinence in C57BL/6J mice, and (3) anxiety- and depressive-like behavior induced by protracted ethanol abstinence in the elevated plus maze, forced-swim, and tail-suspension tests in C57BL/6J mice.
Results
Pretreatment with rolipram (0.1 or 0.2 mg/kg) significantly increased entries and time spent in the open arms of the elevated plus maze test in rats with repeated ethanol abstinence. Similarly, in mice with acute ethanol abstinence, administration of rolipram (0.25 or 0.5 mg/kg) dose-dependently increased the crossings in the central zone of the open-field test and duration and transitions on the light side of the light-dark transition test, suggesting anxiolytic-like effects of rolipram. Consistent with these, chronic treatment with rolipram (0.1, 0.3, or 1.0 mg/kg) increased entries in the open arms of the elevated plus maze test; it also reduced the increased duration of immobility in both the forced-swim and tail-suspension tests in mice after protracted ethanol abstinence, suggesting antidepressant-like effects of rolipram.
Conclusions
These results provide the first demonstration for that PDE4 plays a role in modulating the development of negative emotional reactions associated with ethanol abstinence, including anxiety and depression. PDE4 inhibitors may be a novel class of drugs for treatment of alcoholism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Withdrawal symptoms, in response to the reduction or termination of alcohol exposure, are considered as the core feature of alcohol dependence (American Psychiatric Association 2013; De Witte et al. 2003). The negative affective states associated with alcohol withdrawal, such as anxiety and depression, represent motivating factors for excessive alcohol consumption and may account for the relapse of alcohol use and abuse (Koob 2003; Pandey 2003). In preclinical studies, animals exhibited similar alcohol abstinence symptoms with those observed in human. The anxiety- and depressive-like behavior in rodent models mimics human dysphoric emotion related to alcohol abstinence and may aid in testing the therapeutic potential of promising agents for alcoholism (Kim et al. 2017; Pandey et al. 2003).
Phosphodiesterase 4 (PDE4), an enzyme belonging to the PDE superfamily (PDE1–11), specifically catalyzes the hydrolysis of cyclic AMP (cAMP) and plays a critical role in controlling its intracellular levels. PDE4 has been previously reported by our laboratories to modulate ethanol-drinking and ethanol-seeking behavior in alcohol-preferring rodents (Hu et al. 2011; Wen et al. 2012). Systemic administration of the selective PDE4 inhibitor rolipram or Ro 20–1724 decreases ethanol intake in C57BL/6J (C57) mice and reduces both ethanol consumption and oral self-administration in fawn-hooded (FH/Wjd) rats. Consistent with our studies, other laboratories also have demonstrated that, in addition to rolipram, other PDE4-specific inhibitors, namely piclamilast, CDP840, and mesopram, are all able to suppress ethanol intake in C57 mice (Blednov et al. 2014). In addition, the nonspecific PDE inhibitor ibudilast, which inhibits PDE4 and other PDEs such as PDE3, PDE10, and PDE11, reduces ethanol consumption in C57 mice, P rats, and HAD rats (Bell et al. 2015). However, compared to the role of PDE4 in ethanol-drinking and ethanol-seeking behaviors, little is known about the regulation of PDE4 in ethanol abstinence-induced behaviors, including anxiety- and depressive-like behavior.
In earlier studies, PDE4 inhibitors have been demonstrated for potential treatment for depressive disorders for more than 3 decades (Wachtel 1983; Zhang 2009). The antidepressant efficacy of rolipram has been confirmed in both preclinical researches (Mizokawa et al. 1988; Wachtel 1983) and clinical trials (Fleischhacker et al. 1992; Hebenstreit et al. 1989; Zeller et al. 1984). Besides, inhibition of PDE4 is also shown to produce anxiolytic-like effects in rodents (Li et al. 2009; Silvestre et al. 1999). To some extent, the antidepressant- and anxiolytic-like effects of PDE4 inhibitors may share similar neurobiological mechanisms (Li et al. 2009).
Based on these findings, we hypothesized that PDE4 may play a role in the negative emotional states related to abstinence from ethanol drinking. To test this hypothesis, we examined the effects of rolipram on anxiety- and depressive-like responses to cessation of chronic ethanol drinking in alcohol-preferring FH/Wjd rats and C57 mice, which are successfully used in behavioral and neurochemical studies of alcohol dependence (Overstreet et al. 2007; Rezvani et al. 2007; Yoneyama et al. 2008). The whole study consisted of three experiments. First, effects of rolipram pretreatment were assessed on anxiety-like behavior induced by repeated ethanol abstinence in FH/Wjd rats (experiment 1). Second, acute rolipram administration was tested in anxiety-like behavior induced by acute ethanol abstinence in C57 mice (experiment 2). Finally, chronic rolipram treatment was examined on anxiety- and depressive-like behavior induced by protracted ethanol abstinence in C57 mice (experiment 3). It was found that rolipram attenuated anxiety- and depressive-like behavior induced by ethanol abstinence in both rats and mice. This was the first demonstration for the role of PDE4 in the mediation of ethanol abstinence symptoms.
Methods
Animals
Male FH/Wjd rats as described previously (Wen et al. 2012) were used at 3 months of age (220–300 g) in experiment 1; adult male C57BL/6J mice (20–25 g) purchased from Laboratory Animal Center of Southern Medical University (Guangzhou, China) and Animal Center of Chinese Academy of Sciences (Shanghai, China) were used in experiment 2 and experiment 3, respectively. Male rodents were simply used in all the experiments in order to avoid the potential influence of menstrual cycles on behavior in female animals and to make the data comparable to our previous studies (Hu et al. 2011; Wen et al. 2012). All the animals were housed in local animal facilities at a controlled temperature (22 ± 1 °C) and a relative humidity (50 ± 10%) with a 12:12 light/dark cycle (lights on from 08:00 AM to 08:00 PM). They were allowed to acclimatize to the housing environment for at least 5 days prior to experiments. Water and food were available ad libitum in their home cages. All experiments were conducted in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals (NIH Publication No. 80-23, revised 1996) and were approved by local committees on animal care and use.
Drugs
Ethanol solutions (v/v) were prepared with water using anhydrous ethanol (Beijing Chemicals Works, Beijing, China). Rolipram was purchased from Sigma-Aldrich (St. Louis, MO) and dissolved in saline (NS) containing 5% dimethyl sulfoxide (DMSO) freshly prepared before each testing session. Rolipram or vehicle was administered via subcutaneous (s.c.) or intraperitoneal (i.p.) injections for FH/Wjd rats and C57 mice, respectively.
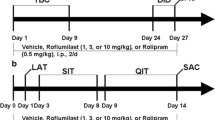
Experiment 1: effects of rolipram on anxiety-like behavior following repeated ethanol abstinence in FH/Wjd rats
Ethanol and drug treatment procedures
Repeated ethanol abstinence has been shown to sensitize anxiety-like behavior in rats (Wills et al. 2009; Knapp et al. 2007). In previous experiments, we found that FH/Wjd rats with prior ethanol abstinence experience exhibited anxiety-like behavior with a shorter duration than rats withdrawn from ethanol for the first time. Thus, repeated ethanol abstinence procedures were applied in this study to determine the role of PDE4 in the regulation of anxiety-like behavior in response to ethanol abstinence. FH/Wjd rats were individually housed in hyaline plastic cages and randomly divided into three groups: control, ethanol non-abstinent, and ethanol abstinent. Rats in the control group had only access to water, while rats in the ethanol-non-abstinent and -abstinent groups were given access to both 5% (v/v) ethanol and water via two-bottle free-choice paradigm. The positions of the two bottles were switched daily to control for side preference. Ethanol and water consumption was measured and calculated for a daily average per week. After 3-week ethanol drinking, the ethanol bottles were removed on day 22 for 24 h in the ethanol-abstinent group of rats and placed back on day 23 for another week. On day 29, abstinence was induced in the ethanol-abstinent group by removing the ethanol bottles again at the onset of the dark cycle (08:00 PM). Liquids provided in the other groups remained unchanged. At the same time, vehicle or rolipram (0.2 mg/kg for the control and ethanol groups; 0.1 and 0.2 mg/kg for the ethanol-abstinent group) was administered to rats in each group. The dose of rolipram was selected based on our previous results (Wen et al. 2012). Three hours later, anxiety-like behaviors were analyzed in the elevated plus maze (EPM) test.
EPM test
The EPM apparatus was made of hard plastic material and elevated to a height of 70 cm. It consisted of two open arms (50 × 10 cm) and two enclosed arms (50 × 10 cm with 40-cm-high walls) arranged opposite to each other. The junction of the four arms formed a central square area (10 × 10 cm). At the beginning of each session, the test rat was placed in the center of the maze facing an open arm. The number of entries into and time spent in both open and enclosed arms were recorded by the SMART system (Panlab Harvard Apparatus, Spain) for 5 min. The anxiety level of rats was measured by the mean percentage of open-arm entries and time spent in open arms as previously described (Pandey et al. 2005). The general motor activity of each rat was determined by calculating the total number of arm entries (open- plus enclosed-arm entries) in the EPM.
Experiment 2: effects of rolipram on anxiety-like behavior induced by acute ethanol abstinence in C57 mice
Ethanol and drug treatment procedures
C57 mice were individually housed and randomly assigned into two groups: the control group had only access to water, and the ethanol group received two-bottle choice drinking for both 10% (v/v) ethanol and water. The positions of the two bottles were switched daily to prevent side preference. After 3-week ethanol drinking, the ethanol-abstinent group of mice was withdrawn from ethanol for 12 h. Rolipram (0.25, 0.5 mg/kg) or vehicle was administered 30 min before open-field and light-dark transition tests. The dose and injecting time of rolipram were selected based on the previous report in our laboratory (Hu et al. 2011).
Open-field test
Locomotor activity of C57 mice was examined as described previously (Zhang et al. 2014). Line crossings (with all four paws into a new square) were recorded for 5 min in the 40 × 40 × 15-cm test chamber with the floor divided into 5 × 5-cm identical squares. The central 16 squares were defined as the central zone. Crossings into the central zone were used to evaluate the level of anxiety of animals, while total crossings were considered as general activity.
Light-dark transition test
Each mouse was placed in the dark compartment (15 × 23 cm) of the light-dark chamber. The latency to cross through the hole (8 × 6 cm) into the light compartment (30 × 23 cm; illuminated with a 60-W bulb positioned 50 cm above), the time spent in the light side, and transitions from the dark to light compartments were recorded for 5 min as described previously (Zhang et al. 2008).
Experiment 3: effects of rolipram on behavior following protracted ethanol abstinence in C57 mice
Ethanol and drug treatment procedures
Mice were individually housed and randomly assigned into the control and ethanol-abstinent groups. The control group of mice had only access to water, and the ethanol-abstinent group received three-bottle choice drinking for water, 5% (v/v) ethanol, and 20% (v/v) ethanol. The positions of the bottles (100-ml plastic centrifuge tubes) were changed daily to exclude position bias. The daily intake of three types of liquid was measured to calculate the percentage of preference as the volume of each type liquid divided by total volume of liquid intake. After 6-week ethanol drinking, mice were withdrawn from ethanol for 14 consecutive days. Rolipram (0.1, 0.3, or 1.0 mg/kg) or vehicle was administered daily from the beginning of ethanol abstinence. EPM, forced-swim, and tail-suspension tests were performed on days 1, 13, and 14 after abstinence from ethanol exposure, respectively.
EPM test
Mice were placed individually in the center of the maze (40 cm above the floor) facing an open arm. The number of entries and time spent in both open arms (30 × 5 cm) and enclosed arms (30 × 5 cm with 15-cm-high walls) were recorded for 5 min as previously described (Zhang et al., 2008). The percentages of entries and time spent in open arms were calculated as open-arm entries and time divided by total arm entries and total time, respectively.
Forced-swim test
The test was carried out as described previously (Porsolt et al. 1978; Zhang et al. 2002). Mice were placed individually in a plastic cylinder (45 cm high × 20 cm diameter) filled with water (23–24 °C; 28 cm in depth), allowing for free swimming. The duration of immobility, which was defined as floating in an upright position without additional movement other than that necessary for the animal to keep its head above the water, was recorded during the last 4 min of the 6-min test period.
Tail-suspension test
Each mouse was suspended 40 cm above the floor using adhesive tape placed 1 cm from the tip of the tail, as described previously (Steru et al. 1985; Zhang et al. 2002). The duration of immobility was recorded for 6 min. Mice were considered immobile only when they hung motionless.
Statistical analysis
Data presented are expressed as means ± SEM. All data were analyzed using one-way analysis of variance (ANOVA) followed by Bonferroni’s post hoc analysis. An independent sample t test was used where it was appropriate in experiment 3. A significance level of p < 0.05 was used throughout the tests.
Results
Effects of rolipram on anxiety-like behavior induced by repeated ethanol abstinence in FH/Wjd rats
To determine whether PDE4 is involved in the development of ethanol abstinence-related anxiety-like behavior, we first examined the effects of rolipram on the performance of FH/Wjd rats with repeated ethanol abstinence in EPM test. The rats exhibited high levels of ethanol intake (>3.9 mg/kg) and preference (>80%) during the 4-week two-bottle drinking paradigm (Fig. 1a). On day 29, rats in the ethanol-abstinent group were withdrawn from ethanol for 3 h after the prior ethanol abstinence on day 22 for 24 h. In the EPM test, ethanol-abstinent rats showed a significant reduction in the percentage of open-arms entries (F (4, 25) = 4.656, p < 0.01; Fig. 1b), and the percentage of time spent in the open arms (F (4, 25) = 10.051, p < 0.001; Fig. 1c), compared to the control and ethanol-non-abstinent rats. Rolipram (0.1, 0.2 mg/kg, s.c.) administered at the beginning of abstinence from ethanol significantly and dose-dependently inhibited the anxiogenic-like responses in ethanol-abstinent rats, as revealed by increased number of entries and time spent in open arms compared to ethanol-abstinent rats that received vehicle administration (Fig. 1b, c). Rolipram (0.2 mg/kg) had no effect on behavior in the EPM in the control and ethanol-non-abstinent rats (data not shown). Moreover, neither rolipram treatment nor ethanol abstinence altered total arm entries in these experiments (data not shown), indicating the general motor activity of the rats remained unaffected. These results suggest that PDE4 may play a role in anxiety-like behavior induced by abstinence from ethanol exposure.
Pretreatment with rolipram suppressed anxiety-like behavior induced by repeated ethanol abstinence in FH/Wjd rats. The rats exhibited stable ethanol consumption and preference (a) during 4-week ethanol two-bottle choice drinking. Rolipram (0.1, 0.2 mg/kg) significantly reversed the decreased percentages of entries (b) into and time (c) spent in the open arms in the EPM test 3 h after abstinence from ethanol. The values are expressed as means ± SEM. * p < 0.05, *** p < 0.001 compared to control; $ p < 0.05, $$$ p < 0.001 compared to ethanol; # p < 0.05, ## p < 0.01, ### p < 0.001 compared to abstinence; n = 6–7
Effects of rolipram on anxiety-like behavior induced by acute ethanol abstinence in C57 mice
To further clarify whether rolipram had an effect on established anxiety-like behavior, we used C57 mice with 3-week ethanol exposure using two-bottle choice. As there was no difference in the measurement of anxiety-like behavior between the control and ethanol-non-abstinent groups of rats in experiment 1, we used mice with access to water as the only control group in the following experiments. Mice with 12-h ethanol abstinence exhibited significant decreases in crossings into the central zone of the open-field test compared to the control (F (3,35) = 13.167, p < 0.001; Fig. 2a). Rolipram (0.25, 0.5 mg/kg) administered 30 min before behavioral testing dose-dependently attenuated the decreased crossings in the central zone with statistical significance at the dose of 0.5 mg/kg (p < 0.01). The general activity was similar among different treatment groups as there was no difference in total horizontal crossings among these mice (data not shown).
Acute administration of rolipram decreased anxiety-like behavior induced by acute ethanol abstinence in C57 mice. Rolipram (0.5 mg/kg) significantly attenuated the decreased percentage of crossings in the central zone in the open-field test (a). It also reversed the decreased duration (b) and transitions (c) in the light compartment in the light-dark transition test 12 h after abstinence from ethanol exposure. Values shown are means ± SEM. * p < 0.05, *** p < 0.001 compared to control (simply water, without ethanol exposure); # p < 0.05, ## p < 0.01 compared to the vehicle-treated ethanol-abstinent group; n = 8–10
In the light-dark transition test, mice with acute ethanol abstinence showed significant decreases in the time spent in the illuminated chamber (F (3,35) = 3.143, p < 0.05; Fig. 2b) and the transitions from the dark compartment to the light side (F (3,35) = 5.362, p < 0.01; Fig. 2c), but the latency to the light side remained unaffected (data not shown). These data further support that PDE4 is involved in the expression of ethanol abstinence-induced anxiety-like behavior.
Effects of rolipram on depressive- and also anxiety-like behavior induced by protracted ethanol abstinence
To determine whether PDE4 is involved in depressive-like behavior induced by abstinence from ethanol drinking, we tested the antidepressant-like effects of rolipram in C57 mice using a protracted ethanol abstinence protocol. Mice with 6-week access to ethanol in three-bottle choice exhibited preference to ethanol at the higher concentration (20%) during the 4 consecutive days before abstinence from ethanol (data not shown). The anxiety- and depressive-like behavior was assessed using EPM, forced-swim test (FST), and tail-suspension test (TST) on days 1, 13, and 14, respectively, after the beginning of ethanol abstinence. After 24-h ethanol abstinence, mice exhibited decreases the percentages of entries and time spent in open arms in the EPM test (Fig. 3a, b). Rolipram (0.1, 0.3, 1.0 mg/kg) produced significant increases in the former and a tendency to increase the time spent in open arms although no statistical significance was observed for the latter compared to the vehicle-treated ethanol-abstinent mice. These data confirmed the results in experiments 1 and 2 and further proved the role of PDE4 in anxiety-like behavior related to different patterns of ethanol abstinence.
Chronic rolipram administration decreased anxiety- and depressive-like behavior induced by protracted abstinence from ethanol drinking in the three-bottle (water, 5% ethanol, and 20% ethanol) choice test in C57 mice. Rolipram (0.5 mg/kg) attenuated the decreased entries (a) into and time (b) spent in open arms in the EPM on day 1 and the increased duration of immobility in the FST (c) and TST (d) on days 13 and 14, respectively, after abstinence from ethanol drinking. Values shown are means ± SEM. * p < 0.05, ** p < 0.01 compared to the control without ethanol exposure; # p < 0.05 compared to the vehicle-treated ethanol-abstinent group; n = 9–13
On day 13 after abstinence from ethanol, abstinent mice showed significant increases in duration of immobility in the FST compared to the control group of mice (F (4, 43) = 3.172, p < 0.05; Fig. 3c); this was attenuated by rolipram (0.1, 0.3, 1.0 mg/kg) in a dose-dependent manner compared to the vehicle-treated ethanol-abstinent mice.
On day 14 after ethanol abstinence, similar to the FST, abstinent mice showed significant increases in duration of immobility in the TST compared to the control group of mice (F (4, 39) = 3.555, p < 0.05; Fig. 3d); this was attenuated by rolipram (0.1, 0.3, 1.0 mg/kg), although no statistical significance was observed compared to the vehicle-treated ethanol-abstinent mice. The results in the FST and TST suggest that PDE4 may also play a role in the depressive-like behavior in response to protracted ethanol abstinence.
Discussion
In the present study, we demonstrated that inhibition of PDE4 via acute or chronic rolipram administration attenuated anxiety- and depressive-like behavior in different animal models of ethanol abstinence, suggesting that PDE4 contributes to the development of the negative affective status associated with cessation of chronic ethanol exposure. The results provide a promising demonstration for the important role of PDE4 in the mediation of alcoholism. To our knowledge, this is the first study investigating the role of PDEs in regulating the development of ethanol abstinence-related behavior.
Using the repeated ethanol abstinence protocol, we aimed to determine the potential role of PDE4 in the induction phase of ethanol abstinence-related anxiety-like behavior; this was achieved via rolipram administration at the beginning of abstinence from ethanol drinking. The repeated abstinence procedure we used was a little different from that previously published (Overstreet et al. 2005; 2002), but was also effective and efficient in inducing anxiety-like behavior in FH/Wjd rats. Since rolipram produces sedation in the first 1 h after administration as shown in our previous studies (Hu et al. 2011), we performed the EPM test 3 h after administration of rolipram and the beginning of ethanol abstinence in order to avoid the potential influence of the sedative effect of rolipram on its anxiolytic activity. At this time point, rolipram selectively decreased anxiety-like behavior of ethanol-abstinent rats without altering their general motor activity.
To verify the anxiolytic-like effect of rolipram in FH/Wjd rats with repeated abstinence from ethanol drinking, acute ethanol-abstinent C57 mice were used in experiment 2. Similar to the observations in rats, rolipram suppressed anxiety-like behavior produced 12 h after abstinence from ethanol exposure, as evidenced by reversing ethanol abstinence-induced decreases in crossings into the central zone in the open-field test and duration in and transitions into the illuminated compartment in the light-dark transition test. It was noted that rolipram failed to produce a significant effect on the time spent in the central zone in the open-field test (data not shown), which occurred likely due to the behavioral variation of mice in response to rolipram at the testing doses. This could be clarified in future studies using optimized doses of rolipram or modified ethanol abstinence procedures.
The anxiolytic-like effects of rolipram were verified in C57 mice with abstinence from ethanol using three-bottle choice. Similar to the two-bottle choice paradigm, ethanol abstinence also produced anxiety-like behavior in mice under ethanol three-bottle choice, as evidenced by decreased percentages of entries and time spent in the open arms of the EPM, both of which were attenuated by rolipram, although the latter was not statistically significant. Since depressive-like behavior occurs 2 weeks after abstinence from ethanol (Stevenson et al. 2009), we used the protracted ethanol abstinence protocol in C57 mice in order to identify the role of PDE4 in ethanol abstinence-related depressive-like behavior. This cannot be achieved using FH/Wjd rats given their innately highly depressive properties (Rezvani et al. 2002). Ethanol abstinence-induced depressive-like behavior was attenuated by rolipram in both the FST and TST, although the latter lacked statistical significance. While it is not clear what caused the different responses to the FST and TST, it has been noted that the TST appears not as sensitive as the FST in testing the responses of mice to inhibition of PDE4, in particular PDE4B, which is involved in anxiety-like behavior (Zhang et al. 2008) and most likely contributes to ethanol-drinking behavior based on its predominant expression in the striatum and amygdala (Cherry and Davis 1999; Pérez-Torres et al. 2000). In addition, it was noted that a higher dose (1.0 mg/kg) of rolipram was required to block ethanol abstinence-induced anxiety- and depressive-like behaviors probably due to the involvement of neurogenesis. Abstinence from ethanol decreases hippocampal neurogenesis while causing depressive-like effects (Stevenson et al. 2009). Rolipram at a dose of above 1.0 mg/kg produces antidepressant-like effects via increased adult neurogenesis (Li et al. 2009).
The downstream targets of cAMP signaling, which is critically regulated by PDE4, have been well proven to act as molecular substrates for anxiety-like behavior related to ethanol abstinence (Pandey et al. 2003) as well as the development and maintenance of ethanol-dependent process (Pandey 2004). Decreased phosphorylation of cAMP-response element binding (CREB) protein in the amygdala is associated with innate or ethanol abstinence-induced anxiety-like behavior in rodents (Pandey et al. 2003; Pandey et al. 2005). Normalization of the decreased phosphorylated CREB (pCREB) levels reduces ethanol intake (Pandey et al. 2005) and prevents anxiety-like behavior induced by abstinence from ethanol (Pandey et al. 2003), while downregulation of pCREB in normal animals provokes anxiety-like behavior and increases ethanol intake and preference (Pandey et al. 2003; Pandey et al. 2005). Thus, it is possible that rolipram decreased anxiety- and depressive-like behavior in ethanol-abstinent rodents via increasing CREB phosphorylation in brain regions closely related to negative emotional reactions and ethanol dependence, such as the amygdala, although the precise mechanisms underlying the anxiolytic- and antidepressant-like effects of rolipram remain to be further elucidated. This is supported by our previous studies showing that rolipram produces anxiolytic- and antidepressant-like effects via increased pCREB in the brain (Li et al. 2009).
In addition to modulating ethanol-dependent behavior, PDE4 has been demonstrated to play a role in the dependent process of other abused drugs in earlier studies. Inhibition of PDE4 by rolipram prevents methamphetamine- and cocaine-induced behavioral sensitization (Iyo et al. 1996; Iyo et al. 1995; Janes et al. 2009), suppresses self-administration to cocaine and heroin (Knapp et al. 1999; Lai et al. 2014), inhibits morphine- and cocaine-induced conditional place preference (Liddie et al. 2012; Thompson et al. 2004; Zhong et al. 2012), and attenuates morphine withdrawal symptoms (Gonzalez-Cuello et al. 2007; Hamdy et al. 2001; Nunez et al. 2009) in rodents. These results indicate that PDE4 may be involved in a common route by which drugs of abuse exert their rewarding/reinforcing actions and trigger neuro-adaptational changes in the brain.
While it is categorized as a legal drug in most countries, alcohol is among the most commonly abused substances and considered more harmful than heroin and cocaine (Lee and Forsythe 2011; Nutt et al. 2010). The development of alcohol dependence can be attributable not only to the rewarding and reinforcing properties of alcohol but also to the negative affective states related to pre-existing conditions or alcohol withdrawal (Koob and Le Moal 1997; Moonat et al. 2010; Pandey 2004). Based on our previous findings, we hereby expanded the modulatory role of PDE to negative emotional reactions to ethanol abstinence, which may act as a negative reinforce for alcohol abuse. Inhibition of PDE4 provides a novel approach to relieving the negative symptoms following ethanol abstinence. PDE4 inhibitors with less side effects may represent a novel and promising class of drugs for the treatment of alcoholism.
References
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders: DSM-5., 5th ed, American Psychiatric Association,
Bell RL, Lopez MF, Cui C, Egli M, Johnson KW, Franklin KM, Becker HC (2015) Ibudilast reduces alcohol drinking in multiple animal models of alcohol dependence. Addict Biol 20:38–42
Blednov YA, Benavidez JM, Black M, Harris RA (2014) Inhibition of phosphodiesterase 4 reduces ethanol intake and preference in C57BL/6J mice. Front Neurosci 8:129
Cherry JA, Davis RL (1999) Cyclic AMP phosphodiesterases are localized in regions of the mouse brain associated with reinforcement, movement, and affect. J Comp Neurol 407:287–301
De Witte P, Pinto E, Ansseau M, Verbanck P (2003) Alcohol and withdrawal: from animal research to clinical issues. Neurosci Biobehav Rev 27:189–197
Fleischhacker WW, Hinterhuber H, Bauer H, Pflug B, Berner P, Simhandl C, Wolf R, Gerlach W, Jaklitsch H, Sastre-y-Hernandez M, Et A (1992) A multicenter double-blind study of three different doses of the new cAMP-phosphodiesterase inhibitor rolipram in patients with major depressive disorder. Neuropsychobiology 26:59–64
Gonzalez-Cuello A, Sanchez L, Hernandez J, Teresa CM, Victoria MM, Laorden ML (2007) Phosphodiesterase 4 inhibitors, rolipram and diazepam block the adaptive changes observed during morphine withdrawal in the heart. Eur J Pharmacol 570:1–9
Hamdy MM, Mamiya T, Noda Y, Sayed M, Assi AA, Gomaa A, Yamada K, Nabeshima T (2001) A selective phosphodiesterase IV inhibitor, rolipram blocks both withdrawal behavioral manifestations, and c-Fos protein expression in morphine dependent mice. Behav Brain Res 118:85–93
Hebenstreit GF, Fellerer K, Fichte K, Fischer G, Geyer N, Meya U, Sastre-y-Hernandez M, Schony W, Schratzer M, Soukop W, Et A (1989) Rolipram in major depressive disorder: results of a double-blind comparative study with imipramine. Pharmacopsychiatry 22:156–160
Hu W, Lu T, Chen A, Huang Y, Hansen R, Chandler LJ, Zhang HT (2011) Inhibition of phosphodiesterase-4 decreases ethanol intake in mice. Psychopharmacology 218:331–339
Iyo M, Bi Y, Hashimoto K, Inada T, Fukui S (1996) Prevention of methamphetamine-induced behavioral sensitization in rats by a cyclic AMP phosphodiesterase inhibitor, rolipram. Eur J Pharmacol 312:163–170
Iyo M, Maeda Y, Inada T, Kitao Y, Sasaki H, Fukui S (1995) The effects of a selective cAMP phosphodiesterase inhibitor, rolipram, on methamphetamine-induced behavior. Neuropsychopharmacology 13:33–39
Janes AC, Kantak KM, Cherry JA (2009) The involvement of type IV phosphodiesterases in cocaine-induced sensitization and subsequent pERK expression in the mouse nucleus accumbens. Psychopharmacology 206:177–185
Kim HJ, Park SD, Lee RM, Lee BH, Choi SH, Hwang SH, Rhim H, Kim HC, Nah SY (2017) Gintonin attenuates depressive-like behaviors associated with alcohol withdrawal in mice. J Affect Disord 215:23–29
Knapp CM, Foye MM, Ciraulo DA, Kornetsky C (1999) The type IV phosphodiesterase inhibitors, Ro 20-1724 and rolipram, block the initiation of cocaine self-administration. Pharmacol Biochem Behav 62:151–158
Knapp DJ, Overstreet DH, Breese GR (2007) Baclofen blocks expression and sensitization of anxiety-like behavior in an animal model of repeated stress and ethanol withdrawal. Alcohol Clin Exp Res 31:582–595
Koob GF (2003) Alcoholism: allostasis and beyond. Alcohol Clin Exp Res 27:232–243
Koob GF, Le Moal M (1997) Drug abuse: hedonic homeostatic dysregulation. Science 278:52–58
Lai M, Zhu H, Sun A, Zhuang D, Fu D, Chen W, Zhang HT, Zhou W (2014) The phosphodiesterase-4 inhibitor rolipram attenuates heroin-seeking behavior induced by cues or heroin priming in rats. Int J Neuropsychopharmacol 17:1397–1407
Lee GA, Forsythe M (2011) Is alcohol more dangerous than heroin? The physical, social and financial costs of alcohol. Int Emerg Nurs 19:141–145
Li YF, Huang Y, Amsdell SL, Xiao L, O'Donnell JM, Zhang HT (2009) Antidepressant- and anxiolytic-like effects of the phosphodiesterase-4 inhibitor rolipram on behavior depend on cyclic AMP response element binding protein-mediated neurogenesis in the hippocampus. Neuropsychopharmacology 34:2404–2419
Liddie S, Anderson KL, Paz A, Itzhak Y (2012) The effect of phosphodiesterase inhibitors on the extinction of cocaine-induced conditioned place preference in mice. J Psychopharmacol 26:1375–1382
Mizokawa T, Kimura K, Ikoma Y, Hara K, Oshino N, Yamamoto T, Ueki S (1988) The effect of a selective phosphodiesterase inhibitor, rolipram, on muricide in olfactory bulbectomized rats. Jpn J Pharmacol 48:357–364
Moonat S, Starkman BG, Sakharkar A, Pandey SC (2010) Neuroscience of alcoholism: molecular and cellular mechanisms. Cell Mol Life Sci 67:73–88
Nunez C, Gonzalez-Cuello A, Sanchez L, Vargas ML, Milanes MV, Laorden ML (2009) Effects of rolipram and diazepam on the adaptive changes induced by morphine withdrawal in the hypothalamic paraventricular nucleus. Eur J Pharmacol 620:1–8
Nutt DJ, King LA, Phillips LD (2010) Drug harms in the UK: a multicriteria decision analysis. Lancet 376:1558–1565
Overstreet DH, Knapp DJ, Breese GR (2002) Accentuated decrease in social interaction in rats subjected to repeated ethanol withdrawals. Alcohol Clin Exp Res 26:1259–1268
Overstreet DH, Knapp DJ, Breese GR (2005) Pharmacological modulation of repeated ethanol withdrawal-induced anxiety-like behavior differs in alcohol-preferring P and Sprague-Dawley rats. Pharmacol Biochem Behav 81:122–130
Overstreet DH, Rezvani AH, Djouma E, Parsian A, Lawrence AJ (2007) Depressive-like behavior and high alcohol drinking co-occur in the FH/WJD rat but appear to be under independent genetic control. Neurosci Biobehav Rev 31:103–114
Pandey SC (2003) Anxiety and alcohol abuse disorders: a common role for CREB and its target, the neuropeptide Y gene. Trends Pharmacol Sci 24:456–460
Pandey SC (2004) The gene transcription factor cyclic AMP-responsive element binding protein: role in positive and negative affective states of alcohol addiction. Pharmacol Ther 104:47–58
Pandey SC, Roy A, Zhang H (2003) The decreased phosphorylation of cyclic adenosine monophosphate (cAMP) response element binding (CREB) protein in the central amygdala acts as a molecular substrate for anxiety related to ethanol withdrawal in rats. Alcohol Clin Exp Res 27:396–409
Pandey SC, Zhang H, Roy A, Xu T (2005) Deficits in amygdaloid cAMP-responsive element-binding protein signaling play a role in genetic predisposition to anxiety and alcoholism. J Clin Invest 115:2762–2773
Pérez-Torres S, Miró X, Palacios JM, Cortés R, Puigdoménech P, Mengod G (2000) Phosphodiesterase type 4 isozymes expression in human brain examined by in situ hybridization histochemistry and[3H]rolipram binding autoradiography. Comparison with monkey and rat brain. J Chem Neuroanat 20(3-4):349–374
Porsolt RD, Anton G, Blavet N, Jalfre M (1978) Behavioural despair in rats: a new model sensitive to antidepressant treatments. Eur J Pharmacol 47:379–391
Rezvani AH, Overstreet DH, Cleves M, Parsian A (2007) Further genetic characterization of the fawn-hooded (FH/Wjd) rat, an animal model of comorbid depression and alcoholism. Psychiatr Genet 17:77–83
Rezvani AH, Parsian A, Overstreet DH (2002) The fawn-hooded (FH/Wjd) rat: a genetic animal model of comorbid depression and alcoholism. Psychiatr Genet 12:1–16
Silvestre JS, Fernandez AG, Palacios JM (1999) Effects of rolipram on the elevated plus-maze test in rats: a preliminary study. J Psychopharmacol 13:274–277
Steru L, Chermat R, Thierry B, Simon P (1985) The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology 85:367–370
Stevenson JR, Schroeder JP, Nixon K, Besheer J, Crews FT, Hodge CW (2009) Abstinence following alcohol drinking produces depression-like behavior and reduced hippocampal neurogenesis in mice. Neuropsychopharmacology 34:1209–1222
Thompson BE, Sachs BD, Kantak KM, Cherry JA (2004) The type IV phosphodiesterase inhibitor rolipram interferes with drug-induced conditioned place preference but not immediate early gene induction in mice. Eur J Neurosci 19:2561–2568
Wachtel H (1983) Potential antidepressant activity of rolipram and other selective cyclic adenosine 3′,5′-monophosphate phosphodiesterase inhibitors. Neuropharmacology 22:267–272
Wen RT, Zhang M, Qin WJ, Liu Q, Wang WP, Lawrence AJ, Zhang HT, Liang JH (2012) The phosphodiesterase-4 (PDE4) inhibitor rolipram decreases ethanol seeking and consumption in alcohol-preferring fawn-hooded rats. Alcohol Clin Exp Res 36:2157–2167
Wills TA, Knapp DJ, Overstreet DH, Breese GR (2009) Sensitization, duration, and pharmacological blockade of anxiety-like behavior following repeated ethanol withdrawal in adolescent and adult rats. Alcohol Clin Exp Res 33:455–463
Yoneyama N, Crabbe JC, Ford MM, Murillo A, Finn DA (2008) Voluntary ethanol consumption in 22 inbred mouse strains. Alcohol 42:149–160
Zeller E, Stief HJ, Pflug B, Sastre-y-Hernandez M (1984) Results of a phase II study of the antidepressant effect of rolipram. Pharmacopsychiatry 17:188–190
Zhang C, Cheng Y, Wang H, Wang C, Wilson SP, Xu J, Zhang HT (2014) RNA interference-mediated knockdown of long-form phosphodiesterase-4D (PDE4D) enzyme reverses amyloid-beta42-induced memory deficits in mice. J Alzheimers Dis 38:269–280
Zhang HT (2009) Cyclic AMP-specific phosphodiesterase-4 as a target for the development of antidepressant drugs. Curr Pharm Des 15:1688–1698
Zhang HT, Huang Y, Jin SL, Frith SA, Suvarna N, Conti M, O'Donnell JM (2002) Antidepressant-like profile and reduced sensitivity to rolipram in mice deficient in the PDE4D phosphodiesterase enzyme. Neuropsychopharmacology 27:587–595
Zhang HT, Huang Y, Masood A, Stolinski LR, Li Y, Zhang L, Dlaboga D, Jin SL, Conti M, O'Donnell JM (2008) Anxiogenic-like behavioral phenotype of mice deficient in phosphodiesterase 4B (PDE4B). Neuropsychopharmacology 33:1611–1623
Zhong P, Wang W, Yu F, Nazari M, Liu X, Liu QS (2012) Phosphodiesterase 4 inhibition impairs cocaine-induced inhibitory synaptic plasticity and conditioned place preference. Neuropsychopharmacology 37:2377–2387
Acknowledgements
This work was supported by research grants from the National Nature Science Foundation of China (81373384, to J-PX; 81503050, to R-TW; and 30870894, to J-HL), the Natural Science Foundation of Zhejiang Province, China (LY14H310012; to J-CP), the Foundation of Overseas Distinguished Taishan Scholars of Shandong Province (to H-TZ), and China and US NIH/NIAAA (AA020042; to H-TZ).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Gong, MF., Wen, RT., Xu, Y. et al. Attenuation of ethanol abstinence-induced anxiety- and depressive-like behavior by the phosphodiesterase-4 inhibitor rolipram in rodents. Psychopharmacology 234, 3143–3151 (2017). https://doi.org/10.1007/s00213-017-4697-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4697-3