Abstract
Rationale and objectives
Major depressive disorder (MDD) is associated with an increased risk for cardiovascular disease (CVD). Apart from biological and life style factors, the use of antidepressants and their potentially adverse effects might contribute to the increased CVD risk. Therefore, we compared cardiovascular risk profiles between relatively young depressed patients without CVD with and without antidepressant medication and healthy participants.
Methods
We investigated 44 depressed patients (with antidepressants N = 20 (13 women), mean age 43.2 years; without antidepressants N = 24 (15 women), mean age 40.0) and 41 healthy participants (matched for sex, age, education). As markers of CVD risk, blood pressure, body mass index (BMI), and plasma levels of fasting glucose, cholesterol, low density lipoprotein (LDL), high density lipoprotein (HDL), and high sensitivity C-reactive protein (h-CRP) were measured.
Results
We found significant differences between groups for BMI (p < .01), systolic (p = .02) and diastolic blood pressure (p < .01), and glucose (p < .001). Post hoc analyses indicated differences between both patient groups compared to the healthy control group, but not between patients groups. Further controlling for BMI diminished the effect of diagnosis on blood pressure; however, this was not the case for glucose level. There were no between-group differences in cholesterol, LDL, HDL, and h-CRP.
Conclusions
We found a clearly increased CVD risk in this group of rather young depressed patients. Importantly, there was no significant difference in CVD risk between patients with vs. without antidepressants. This suggests that major depression per se and not antidepressant medication is associated with increased CVD risk.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Major depression is associated with an increased risk for cardiovascular diseases (CVDs) such as myocardial infarction (Yusuf et al. 2004) or stroke (O’Donnell et al. 2010; Pan et al. 2011). The magnitude of the increased risk due to depression is comparable to well-established cardiovascular risk factors such as obesity, metabolic syndrome, low high density lipoprotein (HDL), high cholesterol, or C-reactive protein (CRP) (Nicholson et al. 2006; Penninx et al. 2013; Seldenrijk et al. 2015). Major depression increases also the probability of CVD risk factors such as hypertension (Meng et al. 2012), type 2 diabetes (Mezuk et al. 2008), and obesity (Luppino et al. 2011). Furthermore, depressive symptoms predict mortality after an acute myocardial infarct (Frasure-Smith et al. 1993), heart failure (Fan et al. 2014), or in case of a preexisting CVD (Nicholson et al. 2006).
Biological and life style factors associated with major depression might contribute to the increased cardiovascular risk (Penninx et al. 2013; Whooley and Wong 2013). Furthermore, the use of antidepressants and its potentially adverse effects might also contribute to the increased CVD risk. However, due to equivocal findings, this is still a matter of debate. Some studies have reported adverse cardiovascular effects for tricyclic antidepressants (TCAs) (Cohen et al. 2000; Hamer et al. 2011; Jiang and Davidson 2005; Licht et al. 2015; Serodio et al. 2014; Smoller et al. 2009; Zimmermann-Viehoff et al. 2014) or serotonin reuptake inhibitors (SSRIs) (Licht et al. 2015; Rieckmann et al. 2013; Smoller et al. 2009; Weeke et al. 2012; Xiong et al. 2006). There is also evidence suggesting an unfavorable impact of antidepressant medication on prognostic cardiac markers such as heart rate variability (Kemp et al. 2014; Licht et al. 2010). Importantly, a partly recovery of heart rate variability was found when patients had stopped their medication (Licht et al. 2010). Another study reports an association of antidepressant use and increased risk for hypertension while a major depressive disorder without medication was rather associated with reduced blood pressure (Licht et al. 2009). Significant associations of a severe depressive episode and the use of TCAs with obesity have been reported (van Reedt Dortland et al. 2010). However, on the other hand, it is important to note that other studies did not find an increased morbidity or mortality risk or even found a reduced mortality risk for antidepressant medication with TCAs (Pratt et al. 1996; Rahman et al. 2013; Serodio et al. 2014) or SSRIs (Baumeister et al. 2011; Cohen et al. 2000; Glassman et al. 2002; Hamer et al. 2011; Lesperance et al. 2007; O’Connor et al. 2010; Rieckmann et al. 2013; Serodio et al. 2014; Weeke et al. 2012).
To further clarify the role of antidepressant medication for the increased CVD risk in depressed patients, we compared cardiovascular risk profiles between depressed patients with and without antidepressant medication and a healthy control group. Blood pressure, body mass index (BMI), as well as plasma levels of glucose, cholesterol, low density lipoprotein (LDL), high density lipoprotein (HDL), and high sensitivity C-reactive protein (h-CRP) were measured as markers of CVD risk. We hypothesized that depressed patients with antidepressant medication would show the most pronounced adverse CVD risk profile, followed by depressed patients without medication, and with healthy controls exhibiting the most favorable CVD risk profile.
Material and methods
Participants
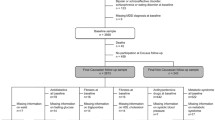
We recruited 44 depressed patients (without antidepressant medication, N = 24; with antidepressant medication, N = 20) from specialized depression clinics at the Department of Psychiatry and Psychotherapy and the Department of Psychosomatic Medicine, University Medical Center Hamburg, Germany, and 41 healthy subjects by public postings. Healthy subjects were matched for sex, age, and education.
Depression was assessed according to DSM-IV criteria using the Mini-International Neuropsychiatric Interview (MINI) conducted by two experienced psychiatrists (C.M and K.H.). To assess the severity of the depressive episode, two clinical depression interviews (17-Item Hamilton Depression Scale (HAMD-17; Hamilton 1960) and Montgomery-Asberg Depression Rating Scale (MADRS; Montgomery and Asberg 1979)) and a self-rating questionnaire (Beck’s Depression Inventory (BDI; Beck et al. 1961)) were conducted.
Criteria for exclusion were dementia, schizophrenia spectrum disorder, bipolar disorder, substance dependence, serious medical conditions associated with adrenal dysfunction, steroid use, pregnancy, and nursing.
All participants were examined by a physical exam, a blood count, and a clinical interview. In the clinical interview, also the use of medication (pharmaceutical agent, dosage, duration, and adherence), the number of past depressive episodes, and the length of the current episode were assessed. The examinations did not show an indication of a CVD neither in the patients nor in the healthy participants. All examinations were conducted by C.M. and K.H.
Twenty-four patients (15 women, 9 men) were free of psychotropic medication. One of these patients used medication of the group of AT blockers.
Twenty patients (13 women, 7 men) were treated with antidepressant medication: selective serotonin reuptake inhibitors (N = 8), selective norepinephrine reuptake inhibitor (N = 2), selective serotonin norepinephrine reuptake inhibitor (N = 3), mirtazapine (N = 3), agomelatine (N = 2), amitriptyline (N = 2), St. John’s wort (N = 2), tranylcypromine (N = 1), and opipramol (N = 1). The mean duration time of antidepressant use was 226 days (SD 345). All of the antidepressant medication was in the recommended dosage range. Six of these patients used medication of the group of α- or ß-blockers (N = 3), ACE-/AT-blockers (N = 3), or diuretics (N = 1).
Healthy participants (26 women, 15 men) were free of former and present DSM-IV axis I disorders as assessed by the MINI, had no physical illness, and were free of medication apart from two cases using medication of the group of ß-blockers (N = 2), ACE-/AT blockers (N = 1), statins (N = 1), or diuretics (N = 1).
The study was carried out in accordance with the latest version of the Declaration of Helsinki and approved by the responsible local ethics committee (ethics committee of the Medical Association of Hamburg). Informed consent of the participants was obtained after the nature of the procedures had been fully explained.
Markers of CVD risk
Blood samples for measurement of glucose, cholesterol, LDL, HDL, and h-CRP levels were taken under fasting conditions between 8 and 9:30 a.m. at the University Medical Center Hamburg by medical doctors. Additionally, weight and height for BMI calculations were assessed and blood pressure was recorded after 5 min of rest. All participants were requested to avoid physical activity such as biking before examination. All blood samples were analyzed as part of the routine assessment in the central laboratory of the University Medical Center Hamburg (Germany).
Statistical analysis
Differences in demographic characteristics between both patient groups and healthy participants were compared using one-way analyses of variance (ANOVAs) for continuous variables and χ 2 tests for dichotomous variables.
One-way analyses of covariance (ANCOVAs) were used to calculate the effect of group (patients without medication, patients with medication, healthy participants) for single risk CVD factors (systolic and diastolic blood pressure, BMI, levels of glucose, cholesterol, LDL, HDL, h-CRP). Post hoc tests (Bonferroni) were used to compare differences between groups. In a second step, for those variables that differed between groups, BMI was included as a covariate in one-way ANCOVAs of group (patients without medication, patients with medication, healthy participants) × blood pressure or glucose level, respectively, to disentangle the influence of BMI and depression on these variables.
A p value smaller than .05 was considered to indicate statistical significance.
Results
For most demographic characteristics (age (years), sex (female/male), education (length of education in years)), we found no significant differences between groups, but for “current smoking” (yes/no; χ 2 = 6.82, p = .03). Thus, “current smoking” was included as control variable in all ANCOVAs.
The mean age in the group of depressed patients without use of antidepressants was 40.0 years (SD 11.8), 43.1 years (SD 9.9) in the group of depressive patients using antidepressants, and 41.2 years (SD 11.6) in the group of healthy participants.
Variables related to psychopathology (first vs. recurrent depressive episode, number of past depressive episodes, mean BDI score, mean HAMD-17 score, and mean MADRS score) did not differ significantly between the two patient groups.
In the group of patients without use of antidepressants, the mean number of past depressive episodes was 2.2 (SD 2.5); for seven patients, the current episode was the first one. The length of the current episode was 12.4 months (SD 12.1).This patient group had a mean BDI score of 33.5 (SD 10.6), a mean HAMD score of 22.8 (SD 4.5), and a mean MADRS score of 29.5 (SD 5.8).
In the group of patients using antidepressants, the mean number of past depressive episodes was 2.5 (SD 5.1); for nine patients, the current episode was the first one. The length of the current episode was 11.4 months (SD 13.1).This patient group had a mean BDI score of 30.7 (SD 8.1), a mean HAMD score of 21.2 (SD 3.9), and a mean MADRS score of 30.7 (SD 5.9).
See Table 1 for demographic and psychopathological characteristics.
Univariate ANCOVAs of group (patients without AD medication, patients with AD medication, healthy participants) × CVD risk factor, including smoking as a covariate, revealed a significant effect of group (F 2,78 = 4.30; p = .02) for systolic blood pressure. Post hoc tests revealed significant differences between patients with AD medication and healthy participants (p = .04) and at trend between patients without AD medication and healthy participants (p = .09), but no significant differences between both patient groups (see Fig. 1a).
For diastolic blood pressure, a significant effect of group (F 2,78 = 5.13; p < .01) was found. Post hoc tests revealed significant differences between patients with AD medication and healthy participants (p = .02) and at trend between patients without AD medication and healthy participants (p = .06), but no significant differences between both patient groups (see Fig. 1b).
For plasma glucose levels, a significant effect of group (F 2,77 = 9.56; p < .001) was found. Post hoc tests revealed significant differences between patients with AD medication and healthy participants (p < .01) and between patients without AD medication and healthy participants (p < .06), but no significant differences between both patient groups (see Fig. 2). For cholesterol, LDL, HDL, and h-CRP levels, there were no significant effects of group.
For BMI, a significant effect of group (F 2,80 = 5.71; p < .01) was found. Post hoc tests revealed significant differences between patients with AD medication and healthy participants (p = .02) and between patients without AD medication and healthy participants (p < .05), but no significant differences between both patient groups.
See Table 2 for an overview, including means of each CVD risk factor according to group.
When BMI was included as an additional covariate, results for systolic blood pressure revealed a highly significant effect of BMI (F 1,77 = 14.03; p < .001). The effect of group was not significant in this ANCOVA. For diastolic blood pressure, a similar pattern was found: there was a significant effect of BMI (F 1,77 = 14.03; p < .01), but not for group. However, for plasma glucose levels, BMI had no significant effect as a covariate (as well as smoking), but the effect of group remained significant in this analysis (F 2,75 = 6.87; p < .01).
Discussion
In this study, we compared cardiovascular risk profiles among depressed patients with and without antidepressant medication and healthy individuals. Both patient groups differed, at least at trend level, from the healthy control group regarding systolic and diastolic blood pressure, plasma glucose levels, and BMI. In contrast, there were no significant differences between the two patient groups. These results clearly indicate an increased cardiovascular risk even in this group of relatively young depressed patients as assessed by the well-established CVD risk factors higher blood pressure, higher glucose levels, and higher BMI (Bogers et al. 2007; Mottillo et al. 2010). Interestingly, further analyses of the results for blood pressure revealed a highly significant effect of BMI as a covariate, which diminished the effect of group. However, the effect of MDD diagnosis on glucose was independent of BMI.
Our results confirm evidence from the literature indicating an increased cardiovascular risk in patients with depressive symptoms (Nicholson et al. 2006; Seldenrijk et al. 2015). The rather young age of the sample, being in their early 40s, further underlines the alarming impact of depression as a cardiovascular risk factor (Penninx et al. 2013).
Interestingly, there was no significant difference according to antidepressant medication regarding the CVD risk markers. This suggests that antidepressant medication did not play a prominent role for the cardiovascular risk in this sample. One reason for this finding might be the fact that only 2 out of 20 patients in our sample received a tricyclic antidepressant. Previous studies that examined the role of antidepressant medication as a cardiovascular risk factor in depression are heterogenous (Cohen et al. 2000; Glassman et al. 2002; Hamer et al. 2011; Jiang and Davidson 2005; Lesperance et al. 2007; Licht et al. 2015; O’Connor et al. 2010; Pratt et al. 1996; Rahman et al. 2013; Rieckmann et al. 2013; Serodio et al. 2014; Smoller et al. 2009; Weeke et al. 2012; Xiong et al. 2006; Zimmermann-Viehoff et al. 2014). However, in sum it appears from the literature that the use of SSRIs might be safer regarding the cardiovascular risk compared to TCA use, while other groups of antidepressants are not investigated sufficiently until now (Hare et al. 2014; Ho et al. 2014; Jiang and Davidson 2005; Schramm et al. 2014). Still, we cannot draw firm conclusions concerning this aspect from our sample since the size of the depressed group with antidepressants was too small to be divided into different classes of antidepressants. Furthermore, it is also important to note differences in study populations of depressed patients with regard to age or gender in previous studies that examined the role of antidepressants (Ho et al. 2014; Licht et al. 2015; Smoller et al. 2009) and preexisting cardiovascular risk (Glassman et al. 2002; Jiang and Davidson 2005; Lesperance et al. 2007; O’Connor et al. 2010; Rieckmann et al. 2013; Zimmermann-Viehoff et al. 2014).
Our results clearly indicate that the depressed patients with and without antidepressant medication exhibit an increased cardiovascular risk with no significant difference between both patient groups. That emphasizes the importance of biological factors increasing the cardiovascular risk which are associated with depression and rather independent from medication. In our study, especially sympathetic and metabolic factors seem to be important. Also, unfavorable life style factors associated with depressive symptoms (Bonnet et al. 2005; de Wit et al. 2010; Whooley et al. 2008; Ziegelstein et al. 2000) such as inactivity and unbalanced nutrition might play an important role and could explain the results.
Unfortunately, we have no data available regarding life style factors such as activities or nutrition, so that we cannot draw any firm conclusion about the impact of these factors. Furthermore, due to the sample size of the study, the depressed group with antidepressant medication cannot further be divided into different classes of antidepressants. Since many studies found a difference, especially between TCAs and SSRIs, it would be interesting to further clarify if the effects were more pronounced in one group of medication. Due to the sample size of the study, we cannot further divide the groups of depressed patients into subtypes of depression either. By now, there is accumulating evidence that the subtypes “melancholic” vs. “atypical” depression are associated with different risk profiles (Penninx et al. 2013). Especially for the atypical subtype, an increased risk for metabolic syndrome, obesity-related disturbances (Lamers et al. 2010; Seppala et al. 2012), and higher inflammation levels (Kaestner et al. 2005; Lamers et al. 2013; Yoon et al. 2012) was observed, whereas hypercortisolism is more often observed in the melancholic type (Kaestner et al. 2005; Karlovic et al. 2012; Lamers et al. 2013). Especially, the metabolic dysregulations appear plausible since weight gain is a cardinal symptom of atypical depression. Another important aspect might be the reasons why some depressed patients get a prescription of antidepressants and others do not. It is possible that patients with an already existing cardiovascular disease are less likely to get a prescription of antidepressants. However, since we did not find differences regarding the CVD risk factors between both patient groups (with vs. without antidepressant medication), this seems not a likely explanation in our sample.
Of note, also the inverse relationship of a CVD increasing the risk for depression has been reported (Lippi et al. 2009). Some mechanisms, such as subclinical inflammation, could have bidirectional effects and might enhance the risk for depression as well as for cardiovascular diseases. In addition, shared genetic effects, childhood maltreatment, or low socio-economic status could be a risk factor for both, depression and somatic diseases (Penninx et al. 2013). Because of the cross-sectional design of the study, we cannot draw any conclusion about the direction of the results.
Besides these limitations, an important strength of this study is the fact that the potentially confounding factor of medication was examined in the association between depression and CVD risk whereas many studies do not differentiate between the role of depression and antidepressant medication. Further strengths include the exact matching of the three groups and the rather young age of the sample.
In summary, our results confirm a significantly increased cardiovascular risk for depressed patients. Our results further point out that also rather young patients suffering from a major depression are exposed to an increased risk. This risk is also increased for patients without antidepressant medication.
References
Baumeister H, Hutter N, Bengel J (2011) Psychological and pharmacological interventions for depression in patients with coronary artery disease. Cochrane Database Syst Rev 9:CD008012. doi:10.1002/14651858.CD008012.pub3
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J (1961) An inventory for measuring depression. Arch Gen Psychiatry 4:561–571
Bogers RP, Bemelmans WJ, Hoogenveen RT, Boshuizen HC, Woodward M, Knekt P, Shipley MJ (2007) Association of overweight with increased risk of coronary heart disease partly independent of blood pressure and cholesterol levels: a meta-analysis of 21 cohort studies including more than 300 000 persons. Arch Intern Med 167(16):1720–1728. doi:10.1001/archinte.167.16.1720
Bonnet F, Irving K, Terra JL, Nony P, Berthezene F, Moulin P (2005) Anxiety and depression are associated with unhealthy lifestyle in patients at risk of cardiovascular disease. Atherosclerosis 178(2):339–344
Cohen HW, Gibson G, Alderman MH (2000) Excess risk of myocardial infarction in patients treated with antidepressant medications: association with use of tricyclic agents. Am J Med 108(1):2–8
de Wit LM, Fokkema M, van Straten A, Lamers F, Cuijpers P, Penninx BW (2010) Depressive and anxiety disorders and the association with obesity, physical, and social activities. Depress Anxiety 27(11):1057–1065. doi:10.1002/da.20738
Fan H, Yu W, Zhang Q, Cao H, Li J, Wang J, Hu X (2014) Depression after heart failure and risk of cardiovascular and all-cause mortality: a meta-analysis. Prev Med 63:36–42. doi:10.1016/j.ypmed.2014.03.007
Frasure-Smith N, Lesperance F, Talajic M (1993) Depression following myocardial infarction. Impact on 6-month survival. JAMA 270(15):1819–1825
Glassman AH, O’Connor CM, Califf RM, Swedberg K, Schwartz P, Bigger JT Jr, McLvor M (2002) Sertraline treatment of major depression in patients with acute MI or unstable angina. Jama 288(6):701–709
Hamer M, Batty GD, Seldenrijk A, Kivimaki M (2011) Antidepressant medication use and future risk of cardiovascular disease: the Scottish Health Survey. Eur Heart J 32(4):437–442. doi:10.1093/eurheartj/ehq438
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 23:56–62
Hare DL, Toukhsati SR, Johansson P, Jaarsma T (2014) Depression and cardiovascular disease: a clinical review. Eur Heart J 35(21):1365–1372. doi:10.1093/eurheartj/eht462
Ho JM, Gomes T, Straus SE, Austin PC, Mamdani M, Juurlink DN (2014) Adverse cardiac events in older patients receiving venlafaxine: a population-based study. J Clin Psychiatry 75(6):e552–e558. doi:10.4088/JCP.13m08508
Jiang W, Davidson JR (2005) Antidepressant therapy in patients with ischemic heart disease. Am Heart J 150(5):871–881. doi:10.1016/j.ahj.2005.01.041
Kaestner F, Hettich M, Peters M, Sibrowski W, Hetzel G, Ponath G, Rothermundt M (2005) Different activation patterns of proinflammatory cytokines in melancholic and non-melancholic major depression are associated with HPA axis activity. J Affect Disord 87(2–3):305–311. doi:10.1016/j.jad.2005.03.012
Karlovic D, Serretti A, Vrkic N, Martinac M, Marcinko D (2012) Serum concentrations of CRP, IL-6, TNF-alpha and cortisol in major depressive disorder with melancholic or atypical features. Psychiatry Res 198(1):74–80. doi:10.1016/j.psychres.2011.12.007
Kemp AH, Brunoni AR, Santos IS, Nunes MA, Dantas EM, Carvalho de Figueiredo R, Lotufo PA (2014) Effects of depression, anxiety, comorbidity, and antidepressants on resting-state heart rate and its variability: an ELSA-Brasil cohort baseline study. Am J Psychiatry. doi:10.1176/appi.ajp.2014.13121605
Lamers F, de Jonge P, Nolen WA, Smit JH, Zitman FG, Beekman AT, Penninx BW (2010) Identifying depressive subtypes in a large cohort study: results from the Netherlands Study of Depression and Anxiety (NESDA). J Clin Psychiatry 71(12):1582–1589. doi:10.4088/JCP.09m05398blu
Lamers F, Vogelzangs N, Merikangas KR, de Jonge P, Beekman AT, Penninx BW (2013) Evidence for a differential role of HPA-axis function, inflammation and metabolic syndrome in melancholic versus atypical depression. Mol Psychiatry 18(6):692–699. doi:10.1038/mp.2012.144
Lesperance F, Frasure-Smith N, Koszycki D, Laliberte MA, van Zyl LT, Baker B, Guertin MC (2007) Effects of citalopram and interpersonal psychotherapy on depression in patients with coronary artery disease: the Canadian Cardiac Randomized Evaluation of Antidepressant and Psychotherapy Efficacy (CREATE) trial. JAMA 297(4):367–379
Licht CM, de Geus EJ, Seldenrijk A, van Hout HP, Zitman FG, van Dyck R, Penninx BW (2009) Depression is associated with decreased blood pressure, but antidepressant use increases the risk for hypertension. Hypertension 53(4):631–638. doi:10.1161/hypertensionaha.108.126698
Licht CM, de Geus EJ, van Dyck R, Penninx BW (2010) Longitudinal evidence for unfavorable effects of antidepressants on heart rate variability. Biol Psychiatry 68(9):861–868
Licht CM, Naarding P, Penninx BW, van der Mast RC, de Geus EJ, Comijs H (2015) The association between depressive disorder and cardiac autonomic control in adults 60 years and older. Psychosom Med 77(3):279–291. doi:10.1097/psy.0000000000000165
Lippi G, Montagnana M, Favaloro EJ, Franchini M (2009) Mental depression and cardiovascular disease: a multifaceted, bidirectional association. Semin Thromb Hemost 35(3):325–336
Luppino FS, van Reedt Dortland AK, Wardenaar KJ, Bouvy PF, Giltay EJ, Zitman FG, Penninx BW (2011) Symptom dimensions of depression and anxiety and the metabolic syndrome. Psychosom Med 73(3):257–264. doi:10.1097/PSY.0b013e31820a59c0
Meng L, Chen D, Yang Y, Zheng Y, Hui R (2012) Depression increases the risk of hypertension incidence: a meta-analysis of prospective cohort studies. J Hypertens 30(5):842–851. doi:10.1097/HJH.0b013e32835080b7
Mezuk B, Eaton WW, Albrecht S, Golden SH (2008) Depression and type 2 diabetes over the lifespan: a meta-analysis. Diabetes Care 31(12):2383–2390. doi:10.2337/dc08-0985
Montgomery SA, Asberg M (1979) A new depression scale designed to be sensitive to change. Br J Psychiatry 134:382–389
Mottillo S, Filion KB, Genest J, Joseph L, Pilote L, Poirier P, Eisenberg MJ (2010) The metabolic syndrome and cardiovascular risk a systematic review and meta-analysis. J Am Coll Cardiol 56(14):1113–1132. doi:10.1016/j.jacc.2010.05.034
Nicholson A, Kuper H, Hemingway H (2006) Depression as an aetiologic and prognostic factor in coronary heart disease: a meta-analysis of 6362 events among 146 538 participants in 54 observational studies. Eur Heart J 27(23):2763–2774
O’Connor CM, Jiang W, Kuchibhatla M, Silva SG, Cuffe MS, Callwood DD, Krishnan R (2010) Safety and efficacy of sertraline for depression in patients with heart failure: results of the SADHART-CHF (Sertraline Against Depression and Heart Disease in Chronic Heart Failure) trial. J Am Coll Cardiol 56(9):692–699. doi:10.1016/j.jacc.2010.03.068
O’Donnell MJ, Xavier D, Liu L, Zhang H, Chin SL, Rao-Melacini P, Yusuf S (2010) Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case–control study. Lancet 376(9735):112–123. doi:10.1016/s0140-6736(10)60834-3
Pan A, Sun Q, Okereke OI, Rexrode KM, Hu FB (2011) Depression and risk of stroke morbidity and mortality: a meta-analysis and systematic review. JAMA 306(11):1241–1249. doi:10.1001/jama.2011.1282
Penninx BW, Milaneschi Y, Lamers F, Vogelzangs N (2013) Understanding the somatic consequences of depression: biological mechanisms and the role of depression symptom profile. BMC Med 11:129. doi:10.1186/1741-7015-11-129
Pratt LA, Ford DE, Crum RM, Armenian HK, Gallo JJ, Eaton WW (1996) Depression, psychotropic medication, and risk of myocardial infarction. Prospective data from the Baltimore ECA follow-up. Circulation 94(12):3123–3129
Rahman I, Humphreys K, Bennet AM, Ingelsson E, Pedersen NL, Magnusson PK (2013) Clinical depression, antidepressant use and risk of future cardiovascular disease. Eur J Epidemiol 28(7):589–595. doi:10.1007/s10654-013-9821-z
Rieckmann N, Kronish IM, Shapiro PA, Whang W, Davidson KW (2013) Serotonin reuptake inhibitor use, depression, and long-term outcomes after an acute coronary syndrome: a prospective cohort study. JAMA Intern Med 173(12):1150–1151. doi:10.1001/jamainternmed.2013.910
Schramm PJ, Poland RE, Rao U (2014) Bupropion response on sleep quality in patients with depression: implications for increased cardiovascular disease risk. Eur Neuropsychopharmacol 24(2):207–214. doi:10.1016/j.euroneuro.2013.09.007
Seldenrijk A, Vogelzangs N, Batelaan NM, Wieman I, van Schaik DJ, Penninx BJ (2015) Depression, anxiety and 6-year risk of cardiovascular disease. J Psychosom Res 78(2):123–129. doi:10.1016/j.jpsychores.2014.10.007
Seppala J, Vanhala M, Kautiainen H, Eriksson J, Kampman O, Mantyselka P, Koponen H (2012) Prevalence of metabolic syndrome in subjects with melancholic and non-melancholic depressive symptoms. A Finnish population-based study. J Affect Disord 136(3):543–549. doi:10.1016/j.jad.2011.10.032
Serodio KJ, Ardern CI, Rotondi MA, Kuk JL (2014) Tricyclic and SSRI usage influences the association between BMI and health risk factors. Clin Obes 4(6):296–302. doi:10.1111/cob.12067
Smoller JW, Allison M, Cochrane BB, Curb JD, Perlis RH, Robinson JG, Wassertheil-Smoller S (2009) Antidepressant use and risk of incident cardiovascular morbidity and mortality among postmenopausal women in the Women’s Health Initiative study. Arch Intern Med 169(22):2128–2139. doi:10.1001/archinternmed.2009.436
van Reedt Dortland AK, Giltay EJ, van Veen T, Zitman FG, Penninx BW (2010) Metabolic syndrome abnormalities are associated with severity of anxiety and depression and with tricyclic antidepressant use. Acta Psychiatr Scand 122(1):30–39. doi:10.1111/j.1600-0447.2010.01565.x
Weeke P, Jensen A, Folke F, Gislason GH, Olesen JB, Andersson C, Torp-Pedersen C (2012) Antidepressant use and risk of out-of-hospital cardiac arrest: a nationwide case-time-control study. Clin Pharmacol Ther 92(1):72–79. doi:10.1038/clpt.2011.368
Whooley MA, Wong JM (2013) Depression and cardiovascular disorders. Annu Rev Clin Psychol 9:327–354. doi:10.1146/annurev-clinpsy-050212-185526
Whooley MA, de Jonge P, Vittinghoff E, Otte C, Moos R, Carney RM, Browner WS (2008) Depressive symptoms, health behaviors, and risk of cardiovascular events in patients with coronary heart disease. JAMA 300(20):2379–2388. doi:10.1001/jama.2008.711
Xiong GL, Jiang W, Clare R, Shaw LK, Smith PK, Mahaffey KW, Newby LK (2006) Prognosis of patients taking selective serotonin reuptake inhibitors before coronary artery bypass grafting. Am J Cardiol 98(1):42–47. doi:10.1016/j.amjcard.2006.01.051
Yoon HK, Kim YK, Lee HJ, Kwon DY, Kim L (2012) Role of cytokines in atypical depression. Nord J Psychiatry 66(3):183–188. doi:10.3109/08039488.2011.611894
Yusuf S, Hawken S, Ounpuu S, Dans T, Avezum A, Lanas F, Lisheng L (2004) Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case–control study. Lancet 364(9438):937–952. doi:10.1016/s0140-6736(04)17018-9
Ziegelstein RC, Fauerbach JA, Stevens SS, Romanelli J, Richter DP, Bush DE (2000) Patients with depression are less likely to follow recommendations to reduce cardiac risk during recovery from a myocardial infarction. Arch Intern Med 160(12):1818–1823
Zimmermann-Viehoff F, Kuehl LK, Danker-Hopfe H, Whooley MA, Otte C (2014) Antidepressants, autonomic function and mortality in patients with coronary heart disease: data from the Heart and Soul Study. Psychol Med 44(14):2975–2984. doi:10.1017/s003329171400066x
Acknowledgments
The authors received no funding. Dr. LK, Dr. CM, Dr. KH, Dr. LD, Dr. KW, and Dr. CS report no conflict of interest. Dr. CO has received honoria fees for lectures from Lundbeck and Servier.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was carried out in accordance with the latest version of the Declaration of Helsinki and approved by the responsible local ethics committee (ethics committee of the Medical Association of Hamburg). Informed consent of the participants was obtained after the nature of the procedures had been fully explained.
Additional information
Linn K. Kuehl and Christoph Muhtz contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kuehl, L.K., Muhtz, C., Hinkelmann, K. et al. Association between major depression and cardiovascular risk: the role of antidepressant medication. Psychopharmacology 233, 3289–3295 (2016). https://doi.org/10.1007/s00213-016-4361-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4361-3