Abstract
Rationale
Opioid receptors are implicated in the regulation of motivation and emotion. However, animal studies show that activation of κ opioid receptor produces contrasting mood-altering effects in models of anxiety-like and depressive-like behaviors, and consequently, the role of κ receptor in mood control remains unsettled. The effect of κ/μ opioid combination in emotion regulation was unexplored.
Objectives
The aim of the study was to investigate the effects of (-)-3-N-ethylaminothiazolo [5,4-b]-N-cyclopropylmethylmorphinan hydrochloride (ATPM-ET), a novel κ agonist and μ partial agonist, in regulating emotional responses.
Methods
The emotional responses of ATPM-ET were detected in the elevated plus maze (EPM), open field test (OFT), forced swim test (FST), and tail suspension test (TST). Selective κ antagonist nor-binaltorphimine (nor-BNI) and μ antagonist β-funaltrexamine (β-FNA) were applied to determine the type of receptor involved. The conditioned place aversion model was used to evaluate the effects on aversive emotion.
Results
In the EPM and OFT, ATPM-ET (1 and 2 mg/kg, s.c.) significantly increased the time spent in the open arm and in the central area, respectively. In the FST and TST, ATPM-ET (0.5 and 1 mg/kg, s.c.) significantly reduced the duration of immobility. These effects were prevented by nor-BNI (10 mg/kg, i.p., −24 h), but not by β-FNA (10 and20 mg/kg, i.p., −24 h) pretreatment. At the dose of 2 mg/kg, ATPM-ET did not induce conditioned place aversion.
Conclusions
ATPM-ET, at doses from 0.5 to 2 mg/kg, produced anxiolytic- and antidepressant-like effects without inducing aversive emotion. These effects were more closely mediated by activation of κ receptor than μ receptor.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
κ, μ, and δ opioid receptors, belonging to the rhodopsin subfamily of the G protein-coupled receptor (GPCR) family, are broadly expressed throughout central and peripheral nervous system. In addition to pain modulation and addiction, these three opioid receptors and their endogenous opioid peptides also have been implicated in the regulation of emotional states (Lutz and Kieffer 2013). Studies using δ agonists and δ receptor knockout mice demonstrated that activation of δ receptor produced anxiolytic- and antidepressant-like effects (Filliol et al. 2000; Jutkiewicz et al. 2005, 2006; Naidu et al. 2007; Saitoh et al. 2004; Vergura et al. 2008). The role of μ receptor in mood was paradoxical, since pharmacological activation of μ receptor produced antidepressant-like behaviors in rodents (Berrocoso et al. 2013; Rojas-Corrales et al. 2002; Tejedor-Real et al. 1995), whereas μ receptor knockout mice also showed antidepressant- and anxiolytic-like effects (Filliol et al. 2000; Yoo et al. 2004). Accumulating evidence has indicated that κ receptor and the endogenous ligand dynorphin are involved in the regulation of emotion (Bruchas et al. 2010; Knoll and Carlezon 2010; Lutz and Kieffer 2013; Van’t Veer and Carlezon 2013). Activation of κ receptor has been shown to induce depressant-, antidepressant-, anxiogenic-, and anxiolytic-like effects in different animal behavioral models (Braida et al. 2009; Femenía et al. 2011; Hang et al. 2015; Kudryavtseva et al. 2004, 2005; Kuzmin et al. 2006; Smith et al. 2012; Valdez and Harshberger 2012; Wittmann et al. 2009), whereas κ receptor antagonists exerted consistent anxiolytic- and antidepressant-like effects (Beardsley et al. 2005; Bruchas et al. 2009; Carr and Lucki 2010; Knoll et al. 2007, 2011; Mague et al. 2003; Peters et al. 2011; Rogala et al. 2012; Shirayama et al. 2004). Early research demonstrated that κ/μ opioid combination showed promise for the treatment of drug abuse with less addiction potential and less undesirable side effects (Archer et al. 1996), but the effects in emotion regulation was unclear. Buprenorphine, a κ antagonist and μ partial agonist, has been shown to produce antidepressant effects in nonopioid-dependent treatment-resistant patients (Bodkin et al. 1995; Karp et al. 2014; Nyhuis et al. 2008). Animal behavioral studies supported this clinical evidence demonstrating that buprenorphine produced antidepressant- and anxiolytic-like effects in mice (Falcon et al. 2015).
(−)-3-N-Ethylaminothiazolo [5,4-b]-N-cyclopropylmethylmorphinan hydrochloride (ATPM-ET) was a novel κ agonist and μ partial agonist. We previously found that ATPM-ET produced potent antinociception and significantly inhibited morphine-induced physical dependence and behavior sensitization with less sedative side effects (Sun et al. 2010). We also found that ATPM-ET significantly inhibited morphine-induced acquisition of conditioned place preference, heroin-reinforced self-administration, and heroin-primed reinstatement of drug-seeking behaviors (Sun et al. 2014).
The aim of the present work was to explore the effects ATPM-ET on emotional responses in mice. The elevated plus maze (EPM) paradigm and the open field test (OFT) were used for evaluating anxiety-related behaviors, and the forced swim test (FST) and the tail suspension test (TST) were used for evaluating depressant-like behaviors. The conditioned place aversion model was used for detecting the aversive emotion. The selective κ receptor antagonist nor-binaltorphimine (nor-BNI) and μ receptor antagonist β-FNA were applied to investigate which type of opioid receptor was involved in the behavioral changes induced by ATPM-ET.
Materials and methods
Animals
Kunming mice (Shanghai Experimental Animal Center of Chinese Academy of Sciences, Shanghai, China) (18–22 g) were housed in cages (five per cage) in a temperature-controlled room (24 ± 2 °C) under a 12-h light/12-h dark cycle (lights on at 7:00 AM). Animals were allowed free access to food and water throughout the experiments. Experiments were carried out in accordance with the National Institutes of Health Guide for Care and Use of Laboratory Animals and were approved by the Bioethics Committee of the Shanghai Institute of Materia Medica (Shanghai, China). Mice were moved to the testing room and allowed to the new environment for at least 3 h before testing.
Drugs
Fluoxetine hydrochloride (FLX) and U50,488H were purchased from Sigma-Aldrich. Nor-BNI and β-funaltrexamine hydrochloride (β-FNA) were purchased from Tocris Bioscience. Diazepam (DIA) was purchased from Shanghai Xu Dong Hai Pu Pharmaceutical Co., Ltd. ATPM-ET was synthesized by Prof. Ao Zhang.
Elevated plus maze (EPM)
EPM test was carried out as described previously (Braida et al. 2009). The apparatus (65 × 65 cm) was made of black plastic, consisting of two closed and two open arms, and it was elevated 0.5 m above the floor. Each arm was 5 × 30 cm (width × length), and the intersection of the arms was 5 × 5 cm. The experiments were performed between 10:00 and 16:00 h under a light (approximately 15 lux). At the beginning of the test, the mice were placed in the center with its head facing the open arm and allowed free to move for 5 min. Animal behavior was recorded, and the time spent in the open arms was analyzed (all four paws expose to the arm). The maze was cleaned with 70 % ethanol and H2O between trials.
Open field test (OFT)
OFT was performed according to Bahi (2013). The chamber was 40 × 40 cm. The center 20 × 20 cm area was defined as “center area,” and the rest was defined as “peripheral area.” The illumination was set to 150 lux. Animals were placed in the center area and given 5 min to explore it freely. Animal behavior was recorded, and the total time spent in the center was analyzed.
Forced swim test (FST)
FST was performed according to our published work (Zan et al. 2015). Mice were placed in a cylinder of water (23–25 °C; 12 cm in diameter; 25 cm in height) for 6 min. The water depth was high enough to keep animals away from the bottom and the wall with hind limbs. Animal behavior was captured by camera on the side. The immobility time of each animal during the last 4 min of total 6 min was obtained by a trained observer who remained unaware of the treatments. Immobility was defined as the time spent by animals remaining motionless or making only movements necessary to keep their head above water.
Tail suspension test (TST)
TST was performed according to the method described by Steru et al. (1985). Briefly, animals were suspended individually on a paper adhesive tape, with 35 cm above the table top. The tape was placed 1 cm from the tip of the tail. Animal was suspended for 6 min, and the last 4-min immobility time was counted by a trained observer blind of the animal treatment. Animals were classified as immobility only if they hung passively and completely motionless. The animals were excluded from further analysis, if they climbed their tails during the test.
Locomotor activity
Locomotor activity was measured as described previously (Dulawa et al. 2004) with slight modification. Briefly, mice were placed in the open field chamber (Shanghai Jiliang Software Technology, China) with infrared video recorder, and the activity was monitored for 60 min. The total distance traveled (mm) was defined as the general locomotor activity.
Conditioned place aversion
The conditioned place aversion apparatus (32 cm length × 16 cm width × 38 cm height) is divided into two equal-sized compartments separated by a removable board (6 × 6 cm), which allows mice free access to each compartment. Two compartments are distinguished by visual and tactile cues: the one with black wall and smooth floor, whereas the other with white wall and textured floor. The distinctive visual and tactile stimuli serve as the conditioning cues.
Conditioned place aversion procedure has been described previously (Zan et al. 2015). Briefly, the procedure includes three phases: preconditioning, conditioning, and testing. In the preconditioning phase, animals were allowed to free explore the entire apparatus for 15 min, and the time spent in each compartment was recorded. The conditioning phase took place over the next 4 days. In the morning, animals were given saline and assigned to the unfavored compartment for 45 min. Four hours later, animals were injected with saline, ATPM-ET or U50,488H and assigned to the preferred compartment for 45 min. This compartment will be referred to as the “drug treatment-paired compartment.” Testing phase took place 24 h after the conditioning trial, and all animals were allowed to freely explore the entire apparatus for 15 min, and the time spent in each compartment was recorded. Conditioned place aversion score represents the time spent in the drug treatment-paired compartment during the testing phase minus that during the preconditioning phase.
Statistical analysis
The data are presented as the mean ± SEM. One-way ANOVA, followed by Newman-Keuls comparison test was used for the statistical evaluation. Differences with p < 0.05 were considered statistically significant.
Results
The anxiolytic-like effects of ATPM-ET in the EPM and OFT in mice
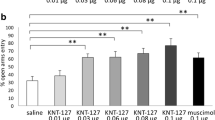
The effects of ATPM-ET on anxiety-related behaviors were evaluated in the EPM in mice. As shown in Fig. 1a, mice receiving 1 and 2 mg/kg ATPM-ET significantly spent more time exploring the unprotected open arms. The influence of ATPM-ET on the OFT was also examined. As shown in Fig. 1b, mice receiving 1 and 2 mg/kg ATPM-ET significantly spent more time in the central area. DIA was used as a positive control drug. As shown in Fig. 1a, b, DIA, at the dose of 1 mg/kg, significantly increased time spent in the open arms and in the central area in the EPM and OFT, respectively, which was consistent with previous studies (Gupta et al. 2015; Kurhe et al. 2014; Mesfin et al. 2014; Wilson et al. 2004), indicating that DIA produced anxiolytic-like effects in mice.
Effects of ATPM-ET and diazepam (DIA) on anxiety-related behaviors tested in the mice EPM (a) and OFT (b). Mice were injected with ATPM-ET (0.5, 1, and 2 mg/kg, s.c.) for 15 min or DIA (1 mg/kg, s.c.) for 30 min, and then, the behavioral effects were measured in the EPM and OFT as described in the “Materials and methods.” Value presents as mean ± SEM of data obtained from at least ten mice. *p < 0.05, **p < 0.01, ***p < 0.001 compared with saline group
The effects of pretreatment with κ antagonist nor-BNI and μ antagonist β-FNA, alone or in combination with ATPM-ET (2 mg/kg), were evaluated in the EPM. As shown in Fig. 2a, nor-BNI (10 mg/kg, i.p.) pretreatment significantly suppressed ATPM-ET-induced increased time spent in the open arm. In contrast to the actions of nor-BNI, pretreatment with β-FNA (10, 20 mg/kg, i.p.) did not affect ATPM-ET’s effects (Fig. 2b).
Effects of nor-BNI (a) and β-FNA (b) on ATPM-ET-induced anxiolytic effects tested in the mice EPM. Mice were pretreated with nor-BNI (10 mg/kg, i.p.) for 24 h, or β-FNA (10 and 20 mg/kg, i.p.) for 24 h, and then injected with ATPM-ET (2 mg/kg, s.c.). After 15 min, the behavioral effects were tested in the mice EPM. Value presents as mean ± SEM of data obtained from at least 10 mice. **p < 0.01, ***p < 0.001 when comparisons are within saline + saline group and saline + ATPM-ET group. $ p < 0.05 when comparisons are within saline + ATPM-ET and nor-BNI + ATPM-ET group
The antidepressant-like effects of ATPM-ET in the FST and TST in mice
The antidepressant-like effects of ATPM-ET were further evaluated in the FST and TST in mice. As shown in Fig. 3a, ATPM-ET (0.5 and 1 mg/kg) significantly decreased the duration of immobility in the FST. ATPM-ET (0.05, 0.5, and 1 mg/kg) also significantly decreased immobility time detected in the TST. FLX, given as a reference drug, decreased the duration of immobility in both TST and TST, which was consistent with previous studies (Gupta et al. 2014; Kordjazy et al. 2015), indicating that FLX produced antidepressant-like effects in mice.
Effects of ATPM-ET and fluoxetine (FLX) on depression-related behaviors tested in the mice FST (a) and TST (b). Mice were injected with ATPM-ET (0.05, 0.5, and 1 mg/kg, s.c.) for 15 min or FLX (10 mg/kg, s.c.) for 30 min, and then, the behavioral effects were measured in the FST and TST as described in the “Materials and methods.” Value presents as mean ± SEM of data obtained from at least ten mice. *p < 0.05, **p < 0.01, ***p < 0.001 compared with saline group
The effects of nor-BNI and β-FNA on ATPM-ET-induced antidepressant effects were evaluated in the mice FST. As shown in Fig. 4a, b, pretreatment with nor-BNI, but not β-FNA, completely prevented ATPM-ET-induced decreased duration of immobility.
Effects of nor-BNI (a) and β-FNA (b) on ATPM-ET-induced antidepressant effects tested in the mice FST. Mice were pretreated with nor-BNI (10 mg/kg, i.p.) for 24 h or β-FNA (10 mg/kg, 20 mg/kg, i.p.) for 24 h, and then injected with ATPM-ET (0.5 mg/kg, s.c.). After 15 min, the behavioral effects were tested in the mice FST. Value presents as mean ± SEM of data obtained from at least ten mice. **p < 0.01, ***p < 0.001 when comparisons are within saline + saline group and saline + ATPM-ET group. $$$ p < 0.001 when comparisons are within saline + ATPM-ET and nor-BNI+ATPM-ET group
The effects of ATPM-ET on locomotor activity
The effects of ATPM-ET on mice general locomotor were measured in the open filed chamber. As shown in Fig. 5, ATPM-ET, at doses from 0.05 to 2 mg/kg, did not affect mice locomotor activity.
The effects of ATPM-ET on conditioned place aversion
The design of behavioral experiment is shown in Fig. 6a. As shown in Fig. 6b, there was a trend that mice pretreated with 2 mg/kg ATPM-ET spent less time in the drug-paired compartment, but it did not reach significant difference, indicating that 2 mg/kg ATPM-ET did not induce aversive emotion in mice. To ensure that our mice condition place aversion model is well established, U50,488H (5 mg/kg) as a positive control was used. As shown in Fig. 6b, mice pretreated with 5 mg/kg U50,488H significantly spent less time in the drug-pair compartment.
Effects of ATPM-ET on conditioned place aversion. a Timeline for conditioned place aversion experiments. b Conditioned place aversion score. After preconditioning, animals were conditioned for 4 days using saline, U50,488H (5 mg/kg, s.c.), or ATPM-ET (2 mg/kg, s.c.). Testing (15 min) occurred 24 h after conditioning. Value presents as mean ± SEM of data obtained from at least ten mice in each group. *p < 0.05 compared with saline-treated group
Discussion
The present study demonstrated that the novel κ agonist and μ partial agonist ATPM-ET produced anxiolytic- and antidepressant-like effects in mice. These effects were more closely mediated by activation of κ opioid receptor than μ opioid receptor.
The anxiolytic-like effects of ATPM-ET were supported by the observations showing that ATPM-ET significantly increased time spent in the open arm in the EPM test and also significantly increased time spent in the central area in the OFT. The antidepressant-like effects of ATPM-ET were supported by the behavioral data, which showed that ATPM-ET significantly decreased immobility time in the both FST and TST. Our mice EPM, OFT, FST, and TST models were actually capable of detecting anxiety- and depressant-related behaviors, since by using these tests, we found that the reference drugs DIA and FLX produced anxiolytic-like and antidepressant-like effects, respectively, effects that were similar to those of established work (Gupta et al. 2014, 2015; Kordjazy et al. 2015; Mesfin et al. 2014; Wilson et al. 2004).
The mechanism that mediated the anxiolytic- and antidepressant-like effects of ATPM-ET was still unclear; however, the fact that the selective κ antagonist nor-BNI but not the selective μ antagonist β-FNA prevented these effects led to the idea that ATPM-ET’s anxiolytic- and antidepressant-like effects were more closely associated with its actions as a κ agonist than a μ agonist. Because the most potent effects of ATPM-ET involve mixed activities as κ agonist and μ partial agonist, it was not surprising to observe ATPM-ET acted via the activation of κ receptor. A growing body of evidence has indicated that the dynorphin/κ opioid receptor system play a critical role in modulating mood and emotional behaviors. Our findings agree with previously published studies showing that κ agonist salvinorin A, U50488H and U69593, through selective action of κ receptor, produce anxiolytic-like and/or antidepressant-like effects in rodents (Braida et al. 2009). These results, together with previous work showing that deletion of prodynorphin gene increased anxiety-like behaviors in mice, support an anxiolytic role for κ receptor activation. However, our findings also appear to contradict previously published studies conducted in rodents, indicating that activation of κ receptor produces anxiety-like behaviors (Knoll and Carlezon 2010; Smith et al. 2012; Van’t Veer and Carlezon 2013; Valdez and Harshberger 2012; Wittmann et al. 2009). Interestingly, there was evidence demonstrating that salvinorin A, a potent κ agonist, produced antidepressant effects at low doses (Braida et al. 2009), whereas produced prodepressant effects at high doses (Carlezon et al. 2006). Nevertheless, this dose-dependent manner was not involved in ATPM-ET-induced anxiolytic effects, because when the dose was decreased to 0.05 mg/kg, it still did not affect emotional responses (data not shown). It was surprising to observe that ATPM-ET’s μ agonist activity was not attributed to its action on emotional responses, since existing literature has identified a role for each of the opioid receptors in the regulation of emotional responses (Lutz and Kieffer 2013). Glover and Davis (2008) reported that buprenorphine, a κ antagonist and μ partial agonist, produced anxiolytic-like effects in rat due to its μ agonist activity.
κ agonists have been shown to cause dysphoria in human (Pfeiffer et al. 1986) and aversive effects in animals (Shippenberg and Herz 1986). To well define the effects of ATPM-ET in emotional responses, conditioned place aversion model was used. Since in the present work, the effective doses of ATPM-ET to produce anxiolytic-like and antidepressant effects were from 0.05 to 2 mg/kg, we chose highest dose 2 mg/kg of ATPM-ET to evaluate the effects on aversion emotion. We found that 2 mg/kg ATPM-ET did not induce aversive emotion in mice; however, the positive control 5 mg/kg U50,488 did. It is worth pointing out that we cannot exclude the possibility that administration of higher dose ATPM-ET (>2 mg/kg) produces aversive reactions.
In summary, the present study demonstrated that ATPM-ET, as a mixed κ agonist and μ partial agonist, produced anxiolytic- and antidepressant-like effects in mice. ATPM-ET’s action as an agonist at κ opioid receptor contributes to its anxiolytic- and antidepressant-like effects. Since chronic pain and addiction are always comorbid with mood disorders including depression and anxiety, the present findings, together with previous work demonstrating that ATPM-ET was capable of producing potent antinociception and effectively treating heroin addiction with less abuse potential and less undesirable side effects, suggest that compounds with mixed κ and μ activity may hold clinical significance. However, further studies are needed before this possibility can be established with greater certainty. It is noteworthy that the development of tolerance in response to chronic use is a characteristic of all the opioid drugs; thus, during ATPM-ET chronic treatment, it is plausible that tolerance develops and complicates its use as pharmacotherapies.
References
Archer S, Glick SD, Bidlack JM (1996) Cyclazocine revisited. Neurochem Res 21:1369–1373
Bahi A (2013) Individual differences in elevated plus-maze exploration predicted higher ethanol consumption and preference in outbred mice. Pharmacol Biochem Behav 105:83–88
Beardsley PM, Howard JL, Shelton KL, Carroll FI (2005) Differential effects of the novel kappa opioid receptor antagonist, JDTic, on reinstatement of cocaine-seeking induced by footshock stressors vs cocaine primes and its antidepressant-like effects in rats. Psychopharmacology (Berl) 183:118–126
Berrocoso E, Ikeda K, Sora I, Uhl GR, Sanchez-Blazquez P, Mico JA (2013) Active behaviours produced by antidepressants and opioids in the mouse tail suspension test. Int J Neuropsychopharmacol 16:151–162
Bodkin JA, Zornberg GL, Lukas SE, Cole JO (1995) Buprenorphine treatment of refractory depression. J Clin Psychopharmacol 15:49–57
Braida D, Capurro V, Zani A, Rubino T, Vigano D, Parolaro D, Sala M (2009) Potential anxiolytic- and antidepressant-like effects of salvinorin A, the main active ingredient of Salvia divinorum, in rodents. Br J Pharmacol 157:844–853
Bruchas MR, Land BB, Lemos JC, Chavkin C (2009) CRF1-R activation of the dynorphin/kappa opioid system in the mouse basolateral amygdala mediates anxiety-like behavior. PLoS One 4, e8528. doi:10.1371/journal.pone.0008528
Bruchas MR, Land BB, Chavkin C (2010) The dynorphin/kappa opioid system as a modulator of stress-induced and pro-addictive behaviors. Brain Res 1314:44–55
Carlezon WA Jr, Beguin C, DiNieri JA, Baumann MH, Richards MR, Todtenkopf MS, Rothman RB, Ma Z, Lee DY, Cohen BM (2006) Depressive-like effects of the kappa-opioid receptor agonist salvinorin A on behavior and neurochemistry in rats. J Pharmacol Exp Ther 316:440–447
Carr GV, Lucki I (2010) Comparison of the kappa-opioid receptor antagonist DIPPA in tests of anxiety-like behavior between Wistar Kyoto and Sprague Dawley rats. Psychopharmacology (Berl) 210:295–302
Dulawa SC, Holick KA, Gundersen B, Hen R (2004) Effects of chronic fluoxetine in animal models of anxiety and depression. Neuropsychopharmacology 29:1321–1330
Falcon E, Maier K, Robinson SA, Hill-Smith TE, Lucki I (2015) Effects of buprenorphine on behavioral tests for antidepressant and anxiolytic drugs in mice. Psychopharmacology (Berl) 232:907–915
Femenía T, Perez-Rial S, Uriguen L, Manzanares J (2011) Prodynorphin gene deletion increased anxiety-like behaviours, impaired the anxiolytic effect of bromazepam and altered GABAA receptor subunits gene expression in the amygdala. J Psychopharmacol 25:87–96
Filliol D, Ghozland S, Chluba J, Martin M, Matthes HW, Simonin F, Befort K, Gaveriaux-Ruff C, Dierich A, LeMeur M, Valverde O, Maldonado R, Kieffer BL (2000) Mice deficient for delta- and mu-opioid receptors exhibit opposing alterations of emotional responses. Nat Genet 25:195–200
Glover EM, Davis M (2008) Anxiolytic-like effects of morphine and buprenorphine in the rat model of fear-potentiated startle: tolerance, cross-tolerance, and blockade by naloxone. Psychopharmacology (Berl) 198:167–180
Gupta D, Radhakrishnan M, Thangaraj D, Kurhe Y (2014) Antidepressant and anti-anxiety like effects of 4i (N-(3-chloro-2-methylphenyl) quinoxalin-2-carboxamide), a novel 5-HT3 receptor antagonist in acute and chronic neurobehavioral rodent models. Eur J Pharmacol 735:59–67
Gupta D, Radhakrishnan M, Thangaraj D, Kurhe Y (2015) Pharmacological evaluation of novel 5-HT3 receptor antagonist, QCM-13 (N-cyclohexyl-3-methoxyquinoxalin-2-carboxamide) as anti-anxiety agent in behavioral test battery. J Pharm Bioallied Sci 7:103–108
Hang A, Wang YJ, He L, Liu JG (2015) The role of the dynorphin/kappa opioid receptor system in anxiety. Acta Pharmacol Sin 36:783–790
Jutkiewicz EM, Rice KC, Traynor JR, Woods JH (2005) Separation of the convulsions and antidepressant-like effects produced by the delta-opioid agonist SNC80 in rats. Psychopharmacology (Berl) 182:588–596
Jutkiewicz EM, Torregrossa MM, Sobczyk-Kojiro K, Mosberg HI, Folk JE, Rice KC, Watson SJ, Woods JH (2006) Behavioral and neurobiological effects of the enkephalinase inhibitor RB101 relative to its antidepressant effects. Eur J Pharmacol 531:151–159
Karp JF, Butters MA, Begley AE, Miller MD, Lenze EJ, Blumberger DM, Mulsant BH, Reynolds CF 3rd (2014) Safety, tolerability, and clinical effect of low-dose buprenorphine for treatment-resistant depression in midlife and older adults. J Clin Psychiatry 75:785–793
Knoll AT, Carlezon WA Jr (2010) Dynorphin, stress, and depression. Brain Res 1314:56–73
Knoll AT, Meloni EG, Thomas JB, Carroll FI, Carlezon WA Jr (2007) Anxiolytic-like effects of kappa-opioid receptor antagonists in models of unlearned and learned fear in rats. J Pharmacol Exp Ther 323:838–845
Knoll AT, Muschamp JW, Sillivan SE, Ferguson D, Dietz DM, Meloni EG, Carroll FI, Nestler EJ, Konradi C, Carlezon WA Jr (2011) Kappa opioid receptor signaling in the basolateral amygdala regulates conditioned fear and anxiety in rats. Biol Psychiatry 70:425–433
Kordjazy N, Haj-Mirzaian A, Amiri S, Ostadhadi S, Kordjazy M, Sharifzadeh M, Dehpour AR (2015) Elevated level of nitric oxide mediates the anti-depressant effect of rubidium chloride in mice. Eur J Pharmacol 762:411–418
Kudryavtseva NN, Gerrits MA, Avgustinovich DF, Tenditnik MV, Van Ree JM (2004) Modulation of anxiety-related behaviors by mu- and kappa-opioid receptor agonists depends on the social status of mice. Peptides 25:1355–1363
Kudryavtseva NN, Gerrits MA, Alekseenko OV, Van Ree JM (2005) Chronic cocaine injections attenuate behavioral response of kappa-opioid receptors to U-50,488H agonist. Bull Exp Biol Med 140:320–322
Kurhe YV, Radhakrishnan M, Thangaraj D, Gupta D (2014) Anti-anxiety effect of a novel 5-HT(3) receptor antagonist N-(benzo[d]thiazol-2-yl)-3-ethoxyquinoxalin-2-carboxamide (6k) using battery tests for anxiety in mice. Indian J Pharmacol 46:100–104
Kuzmin A, Madjid N, Terenius L, Ogren SO, Bakalkin G (2006) Big dynorphin, a prodynorphin-derived peptide produces NMDA receptor-mediated effects on memory, anxiolytic-like and locomotor behavior in mice. Neuropsychopharmacology 31:1928–1937
Lutz PE, Kieffer BL (2013) Opioid receptors: distinct roles in mood disorders. Trends Neurosci 36:195–206
Mague SD, Pliakas AM, Todtenkopf MS, Tomasiewicz HC, Zhang Y, Stevens WC Jr, Jones RM, Portoghese PS, Carlezon WA Jr (2003) Antidepressant-like effects of kappa-opioid receptor antagonists in the forced swim test in rats. J Pharmacol Exp Ther 305:323–330
Mesfin M, Asres K, Shibeshi W (2014) Evaluation of anxiolytic activity of the essential oil of the aerial part of Foeniculum vulgare Miller in mice. BMC Complement Altern Med 14:310. doi:10.1186/1472-6882-14-310
Naidu PS, Lichtman AH, Archer CC, May EL, Harris LS, Aceto MD (2007) NIH 11082 produces anti-depressant-like activity in the mouse tail-suspension test through a delta-opioid receptor mechanism of action. Eur J Pharmacol 566:132–136
Nyhuis PW, Gastpar M, Scherbaum N (2008) Opiate treatment in depression refractory to antidepressants and electroconvulsive therapy. J Clin Psychopharmacol 28:593–595. doi:10.1097/JCP.0b013e31818638a4
Peters MF, Zacco A, Gordon J, Maciag CM, Litwin LC, Thompson C, Schroeder P, Sygowski LA, Piser TM, Brugel TA (2011) Identification of short-acting kappa-opioid receptor antagonists with anxiolytic-like activity. Eur J Pharmacol 661:27–34
Pfeiffer A, Brantl V, Herz A, Emrich HM (1986) Psychotomimesis mediated by kappa opiate receptors. Science 233:774–776
Rogala B, Li Y, Li S, Chen X, Kirouac GJ (2012) Effects of a post-shock injection of the kappa opioid receptor antagonist norbinaltorphimine (norBNI) on fear and anxiety in rats. PLoS One 7, e49669. doi:10.1371/journal.pone.0049669
Rojas-Corrales MO, Berrocoso E, Gibert-Rahola J, Mico JA (2002) Antidepressant-like effects of tramadol and other central analgesics with activity on monoamines reuptake, in helpless rats. Life Sci 72:143–152
Saitoh A, Kimura Y, Suzuki T, Kawai K, Nagase H, Kamei J (2004) Potential anxiolytic and antidepressant-like activities of SNC80, a selective delta-opioid agonist, in behavioral models in rodents. J Pharmacol Sci 95:374–380
Shippenberg TS, Herz A (1986) Differential effects of mu and kappa opioid systems on motivational processes. NIDA Res Monogr 75:563–566
Shirayama Y, Ishida H, Iwata M, Hazama GI, Kawahara R, Duman RS (2004) Stress increases dynorphin immunoreactivity in limbic brain regions and dynorphin antagonism produces antidepressant-like effects. J Neurochem 90:1258–1268
Smith JS, Schindler AG, Martinelli E, Gustin RM, Bruchas MR, Chavkin C (2012) Stress-induced activation of the dynorphin/kappa-opioid receptor system in the amygdala potentiates nicotine conditioned place preference. J Neurosci 32:1488–1495
Steru L, Chermat R, Thierry B, Simon P (1985) The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology (Berl) 85:367–370
Sun JF, Wang YH, Li FY, Lu G, Tao YM, Cheng Y, Chen J, Xu XJ, Chi ZQ, Neumeyer JL, Zhang A, Liu JG (2010) Effects of ATPM-ET, a novel kappa agonist with partial mu activity, on physical dependence and behavior sensitization in mice. Acta Pharmacol Sin 31:1547–1552
Sun JF, Wang YH, Chai JR, Li FY, Hang A, Lu G, Tao YM, Cheng Y, Chi ZQ, Neumeyer JL, Zhang A, Liu JG, Wang YJ (2014) Pharmacological characterization and therapeutic potential for the treatment of opioid abuse with ATPM-ET, an N-ethyl substituted aminothiazolomorphinan with kappa agonist and mu agonist/antagonist activity. Eur J Pharmacol 740:455–463
Tejedor-Real P, Mico JA, Maldonado R, Roques BP, Gibert-Rahola J (1995) Implication of endogenous opioid system in the learned helplessness model of depression. Pharmacol Biochem Behav 52:145–152
Valdez GR, Harshberger E (2012) kappa opioid regulation of anxiety-like behavior during acute ethanol withdrawal. Pharmacol Biochem Behav 102:44–47
Van’t Veer A, Carlezon WA Jr (2013) Role of kappa-opioid receptors in stress and anxiety-related behavior. Psychopharmacology (Berl) 229:435–452
Vergura R, Balboni G, Spagnolo B, Gavioli E, Lambert DG, McDonald J, Trapella C, Lazarus LH, Regoli D, Guerrini R, Salvadori S, Calo G (2008) Anxiolytic- and antidepressant-like activities of H-Dmt-Tic-NH-CH(CH2-COOH)-Bid (UFP-512), a novel selective delta opioid receptor agonist. Peptides 29:93–103
Wilson MA, Burghardt PR, Ford KA, Wilkinson MB, Primeaux SD (2004) Anxiolytic effects of diazepam and ethanol in two behavioral models: comparison of males and females. Pharmacol Biochem Behav 78:445–458
Wittmann W, Schunk E, Rosskothen I, Gaburro S, Singewald N, Herzog H, Schwarzer C (2009) Prodynorphin-derived peptides are critical modulators of anxiety and regulate neurochemistry and corticosterone. Neuropsychopharmacology 34:775–785
Yoo JH, Lee SY, Loh HH, Ho IK, Jang CG (2004) Altered emotional behaviors and the expression of 5-HT1A and M1 muscarinic receptors in micro-opioid receptor knockout mice. Synapse 54:72–82
Zan GY, Wang Q, Wang YJ, Liu Y, Hang A, Shu XH, Liu JG (2015) Antagonism of kappa opioid receptor in the nucleus accumbens prevents the depressive-like behaviors following prolonged morphine abstinence. Behav Brain Res 291:334–341
Acknowledgments
We thank professor Ao Zhang, Shanghai Institute of Materia Medica, Shanghai, China 201203, for kindly providing compound ATPM-ET. This research was supported by grants 2013CB835100 and 2015CB553500 (to J.-G. L.) from the Ministry of Science and Technology of China, by grants 81130087, 91232716 (to J.-G. L.), and 81401107 (to Y.-J. W.) from the National Natural Science Foundation of China, and by grant 13JC140680 (to J.-G. L.) from the Committee of Science and Technology of Shanghai.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Qian Wang and Yu Long contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Q., Long, Y., Hang, A. et al. The anxiolytic- and antidepressant-like effects of ATPM-ET, a novel κ agonist and μ partial agonist, in mice. Psychopharmacology 233, 2411–2418 (2016). https://doi.org/10.1007/s00213-016-4292-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4292-z