Abstract
Rationale
Attention-deficit hyperactivity disorder (ADHD) is the most frequently diagnosed neuropsychiatric disorder in childhood. Currently available ADHD drugs include the psychostimulants methylphenidate (MPH) and d-amphetamine (D-AMP), acting on norepinephrine and dopamine transporters/release, and atomoxetine (ATX), a selective norepinephrine uptake inhibitor. Recent evidence suggests an involvement of glutamate neurotransmission in the pathology and treatment of ADHD, via mechanisms to be clarified.
Objective
We have investigated how ADHD drugs could modulate, through interaction with catecholamine receptors, basal and glutamate-induced excitability of pyramidal neurons in the prefrontal cortex (PFC), a region which plays a major role in control of attention and impulsivity.
Methods
We have used the technique of extracellular single-unit recording in anaesthetised rats coupled with microiontophoresis.
Results
Both MPH (1–3 mg/kg) and d-AMP (1–9 mg/kg) increased the firing activity of PFC neurons in a dopamine D1 receptor-dependent manner. ATX administration (1–6 mg/kg) also increased the firing of neurons, but this effect is not significantly reversed by D1 (SCH 23390) or alpha1 (prazosin) receptor antagonists but potentiated by alpha2 antagonist (yohimbine). All drugs induced a clear potentiation of the excitatory response of PFC neurons to the microiontophoretic application of the glutamate agonist N-methyl-d-aspartate (NMDA), but not to the glutamate agonist α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA). The potentiating effect of d-AMP on NMDA-induced activation of PFC neurons was partially reversed or prevented by dopamine D1 receptor blockade.
Conclusion
Our data shows that increase in excitability of PFC neurons in basal conditions and via NMDA receptor activation may be involved in the therapeutic response to ADHD drugs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Attention-deficit hyperactivity disorder (ADHD) is a common psychological disturbance which affects an average of 5 % of school-aged children worldwide (with very variable rate of prevalence ranging from 1 to 20 %) (Polanczyk et al. 2007). It is characterised by inattention, impulsivity and hyperactivity that can persist into adulthood. Currently available ADHD treatments include psychostimulants, such as d-amphetamine (D-AMP) and methylphenidate (MPH), which when administered at the adequate dose, have a powerful and immediate therapeutic effect. However, as indirect dopamine agonists, they also have the potential to affect the motor system and disrupt dopamine-dependent behaviours (Swanson and Volkow 2003). It may therefore not be without risk to administer such compounds to a developing brain. An alternative treatment to psychostimulants is the selective norepinephrine reuptake inhibitor atomoxetine (ATX). This drug may be devoid of action on the motor system and drug abuse liability, but its onset of action is particularly long, usually 4–6 weeks (Bushe and Savill 2014).
The mechanism by which ADHD drugs exert their therapeutic effects, particularly on attention and cognition processes, is not well understood. ATX selectively blocks the norepinephrine transporter (NET, Ki = 3.7–21 nM) but has some affinity (Ki = 79 nM) for the serotonin transporter (Easton et al. 2007; Shen et al. 2013). Both D-AMP and MPH are strong inhibitors of the synaptic reuptake of dopamine and norepinephrine (MPH: Ki = 46–340 and 238–427 nM; D-AMP: Ki = 190–206 and 55–257 nM, for DAT and NET, respectively) (Easton et al. 2007; Upadhyaya et al. 2013). Other effects of D-AMP include the blockade of the vesicular transport of catecholamine (Heal et al. 2009).
One of the main brain regions involved in the behavioural-calming and cognition-enhancing effects of ADHD drugs is the prefrontal cortex (PFC) (Gamo et al. 2010). It plays a critical role in the control of higher cognitive function such as vigilance, attention, impulsivity and behavioural inhibition (Kieling et al. 2008), as well as on the activity of the basal ganglia which control movement and emotional behaviours.
According to microdialysis studies, all ADHD drugs increased the release of dopamine in the PFC, at therapeutic doses (Koda et al. 2010). Prefrontal dopamine, at the adequate concentration range, is thought to play a major positive role in cognition, attention and working memory, mainly through stimulation of dopamine D1 receptors (Floresco 2013).
Recent evidence supports a role for the glutamatergic system in the pathology and treatment of ADHD (Chang et al. 2014), particularly in the PFC, which main cellular constituents are glutamate pyramidal neurons. In the PFC, there is evidence for both functional and anatomical interaction between catecholamine and glutamate systems. Most prefrontal catecholamine innervations, originating from the brainstem, terminate onto pyramidal glutamate projecting neurons and modulate various cortico-cortical circuits, as well as cortical excitatory descending pathways. This includes the massive prefrontal projections to the basal ganglia, known to regulate, as part of a series of complex feedback loops, executive, motor and affective functions, impaired in ADHD (Carli and Invernizzi 2014). As a result, change in PFC catecholamine levels could affect these excitatory drives and in turn impact on catecholamine neurotransmission in many subcortical structures (Sullivan and Brake 2003). Furthermore, there is evidence in the PFC for colocalization of glutamate and catecholamine receptors, which can interact with each other on common intracellular signalling pathways and modulate synaptic transmission (Tseng and O’Donnell 2004; Urban et al. 2013).
Imaging studies in drug-naive ADHD patients indicate dysregulation of glutamatergic frontostriatal circuits, which are strongly modulated by dopamine and norepinephrine, as well as reduction in thickness and hypofunction of some specific subregions of the PFC (Cubillo et al. 2012). Attention has been driven to the glutamate/N-Methyl-d-aspartate (NMDA) receptor. It has been well established that NMDA receptors have critical roles in excitatory synaptic transmission and plasticity underlying cognitive processes, short-term and long-term memory, as well as motor functioning. There is evidence for genetic polymorphism of both the NR2A and the NR2B subunits of the NMDA receptor with ADHD (Turic et al. 2004; Dorval et al. 2007). Although not a pharmacological model for ADHD, glutamate NMDA receptor antagonists induce hyperlocomotion, disrupt attention and impulsivity control (Amitai and Markou 2010), and mitigate some of the neurochemical and behavioural effects of psychostimulants in animal models (Bristow et al. 1994).
Using the single-unit in vivo electrophysiology method, with the aim to identify a possible common mechanism by which ADHD drugs exert their therapeutic effects, we have compared the electrophysiological effects of the main ADHD drugs (ATX, D-AMP, MPH) on the excitability of PFC neurons, in basal conditions and during glutamate receptor activation, and attempt to pharmacologically characterise the different effects produced by these drugs.
Material and methods
Subjects
Adult male Sprague–Dawley rats (Charles River, UK), 250–350 g, housed in groups of 2 to 4 in standard conditions of temperature humidity, and light/dark cycle, with water and food ad libitum, were used in all experiments. Experiments were conducted with permission from the UK Home Office and had approval from the De Montfort University ethics committee.
In vivo electrophysiological recordings
Animals were initially deeply anaesthetised with urethane (1.2–1.7 g/kg, ip, with additional doses if necessary), secured to a stereotaxic frame and maintained at 36–37 °C. A hole was drilled through the bone at the coordinates of the PFC according to the atlas of Paxinos and Watson (1997). Electrodes, manufactured in house from borosilicate capillaries (1.5 mm, Harvard Apparatus, UK) or from five-barrel glass micropipettes (ASI, USA), were pulled on a PP-830 electrode puller (Narishige, Japan). Recording channels were filled by hand with an electrolyte solution (NaCl 147 mM), and sidebarrels (if present) were filled with NMDA (30 mM, pH 7–8) and/or α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA, 5 mM, pH 8) and NaCl 2 M for current balancing. Single and multibarrel electrodes had their tip broken down to an external diameter of 1–1.5 and 5–7 μm, respectively (typical resistance of the recording channel 4–8 MΩ). NMDA pulses of −10 nA or AMPA pulses of −5 nA were applied during 40–70 s onto the PFC neuron at regular intervals (80–100 s). Output from the electrode were sent to a Neurolog AC pre-amplifier and amplifier (Digitimer, UK). Signals were filtered and sent to an audio amplifier, a digital storage oscilloscope and a 1401 interface connected to a computer running Spike 2 (CED, Cambridge, UK) for data capture and analysis. Descent of the electrode was accomplished using a hydraulic micromanipulator (Narishige). Coordinates for the PFC were as follows: anteroposterior 2.5–3.7 mm, lateral 0.3–2 mm, 1.5–4 mm below cortical surface. Neurons were identified according to previous electrophysiological criteria established from studies carried on formally identified pyramidal neurons (Hajos et al. 2003; Puig et al. 2005; Tseng et al. 2006; Kargieman et al. 2007; Wang et al. 2011): a broad action potential (>1 ms), with a biphasic or triphasic, large waveform, starting with a positive inflection, a slow firing rate typically between 1 and 50 spikes/10 s and irregular firing pattern, often with burst activity.
Chemicals
All chemicals were purchased from Sigma-Aldrich (UK) except ATX (Sequoia Research Products, UK). Drugs, dissolved in saline, were administered via the lateral tail vein.
Data analysis
All data are expressed as the mean ± standard error of the mean (SEM). Statistical analyses were performed using paired or unpaired Student’s t tests or one-way analysis of variance (ANOVA), followed, if appropriate, by Neuman–Keuls tests. Probabilities smaller than 0.05 were considered to be significant; n values refer to the number of cells tested.
The mean basal firing activity was evaluated after the neuron had attained a stable firing rate, generally after at least 10 min of recording. Predrug values of firing rate were obtained by averaging the firing rate over a period of at least 4 min immediately prior to the iv administration, postdrug values were obtained by averaging the firing over a period of 5 min following the administration. Individual change in basal firing activity was considered significant following drug (or saline) administration when the postdrug value was significantly different from the predrug value (determined as indicated above, unpaired Student’s t test analysis). When appropriate, portion of recording involving iontophoretic application of excitatory substances were excluded from these calculations. Proportions of a specific type of response in two different groups of animals were also compared using the Fisher’s exact test (comparing proportions of responses and no response/opposite responses in two groups).
The response of neurons to iontophoretic applications of NMDA or AMPA were assessed by subtracting the mean baseline activity measured immediately before the application (typically during the last 50 s before the application) of the drug from the mean activity obtained during the application of the drug (typically during the first 50 s of the application). Responses were expressed as the number of supplementary spikes (over baseline activity) per 10 nA of NMDA or per 5 nA of AMPA (as these currents were systematically administered to neurons). Effects of drugs/saline were examined by comparing the averaged response of at least three subsequent applications of NMDA/AMPA occurring immediately before or after the administration. For an individual neuron, a change in NMDA or AMPA averaged response (defined as the % difference between predrug and postdrug values) of more than 20 % was considered significant.
Results
A total of 125 neurons were included. The basal firing activity of the neurons recorded was variable (0.5–60 spikes/10 s) but typically below 20 spikes/10 s. All neurons tested exhibited the electrophysiological characteristics of pyramidal neurons corresponding to the criteria presented in “Material and methods”. More specifically, all the neurons included in the present study exhibited large biphasic or triphasic action potentials (at least 2 mV amplitude) starting with a positive inflection, and with long duration (>1.1 ms). According to different in vivo electrophysiological studies combining electrophysiology and labelling techniques of GABA and/or glutamate neurons, PFC neurons with action potential duration shorter than 0.75 ms are likely GABAergic neurons (Tseng et al. 2006; Tierney et al. 2008; Gui et al. 2011), while neurons with relatively low firing activity and action potentials larger than 0.9 ms can be considered pyramidal neurons (Tseng et al. 2006), a finding confirmed by electrophysiological studies on PFC neurons identified by antidromic stimulation (Hajos et al. 2003; Puig et al. 2005; Kargieman et al. 2007). It is therefore anticipated that the types of neurons included in the present study are pyramidal neurons, which are by far the more represented type of neurons in the PFC (Povysheva et al. 2008). However, other slightly different criteria for classifications of PFC pyramidal/interneurons exist in literature (Bartho et al. 2004; Gobbi and Janiri 2006) and one could not rule out that a small minority of interneurons has been misclassified as pyramidal neurons in our study, as the recorded neurons were not labelled with glutamatergic or GABAergic markers.
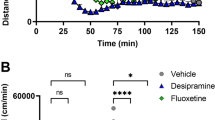
Only one neuron per rat was tested with each of the ADHD drugs. All neurons included in the present data were recorded in the dorsal part of the medial PFC in subregions, which include the cingulated and the prelimbic cortex and the medial part of frontal cortex. Some recording sites were marked by ejection of pontamine blue (n = 13, Fig. 1) and were confirmed to be within the PFC at similar location from what was determined from stereotaxic measurement. The effects of ADHD drugs was investigated on neurons that were spontaneously active (n = 77) and on neurons with virtually no firing activity (n = 41, firing rate <4 spikes/10 s). Silent neurons were detected by recording neuronal activity using an iontophoretic electrode and applying every 100 s (during 40–70 s) a small current of NMDA or AMPA which induced reversible neuronal activation. Spontaneously active neurons were usually recorded with single-barrel electrodes and silent neurons with multibarrel ones. However, a few spontaneously active neurons (n = 21) were also recorded with multibarrel electrodes. Silent neurons and spontaneously active neurons did not differ in their action potential characteristics. Figure 2 shows that the three ADHD drugs tested (ATX, MPH, and D-AMP) can significantly increase the basal activity of PFC neurons.
Recording coordinates of 13 neurons located in the prefrontal cortex. Ejection of pontamine blue (−400 nA, 20 min) was performed after the recordings. Each black dot represents the location of one recording, n = 13. This diagram is a stack of recordings ranging from +2 to +3.5 mm anteroposterior from bregma. Scales represent distances (in mm) from the midline and the surface of the brain. Reproduced from Paxinos and Watson (1997). M1/M2 medial prefrontal cortex, Cg1 cingulate cortex area, PrL prelimbic cortex
Prefrontal cortex neuron electrical activity following cumulative administration of ADHD drugs. a MPH (3 mg/kg) significantly increased neuronal firing rate, confirming previous data from our laboratory (Gronier 2011). Cumulative dose of D-AMP (b) and ATX (c) also significantly increased PFC firing rate. d Saline injections did not change firing rate. Each histogram represents mean firing rate value, and n indicates the number of neurons tested. The same neurons were studied in the complete sequence. *P < 0.05, **P < 0.01 versus respective baseline. Newman–Keuls after significant ANOVA
Effect of MPH on PFC neurons (Fig. 2a)
The excitatory effect of MPH on the basal firing activity of PFC neurons has previously been demonstrated in a large population (n = 28) of PFC neurons (Gronier 2011) and was found to be reversed in 55 % of neurons following the administration of the D1 antagonist SCH 23390 (0.6 mg/kg), a drug that exerted no effects on its own on the basal firing activity of PFC neurons (Gronier 2011). In the present study, we have confirmed these previous data in a smaller population on neurons (n = 13), showing that MPH (3 mg/kg) significantly increased the basal firing activity of PFC neurons (p < 0.01, Student’s paired t test, n = 13; Fig. 2a). We have also observed significant reduction (of more than 10 spikes/10s) of this excitatory effect following the administration of SCH 23390, in three out of five neurons tested this way (60 %, not shown).
Dose-dependent activating effects of D-AMP (Fig. 2b)
In a group of 23 neurons, D-AMP was cumulatively administered up to a dose of 9 mg/kg, by injecting subsequently intravenous doses of 1, 2, 3, and 3 mg/kg. D-AMP induced a significant increase of 59 and 118 % in mean firing activity of PFC neurons, versus initial basal firing, at the cumulative dose of 3 and 6 mg/kg, respectively (p < 0.05, p < 0.01 compared to baseline levels for the 3 and 6 mg/kg doses, respectively, Newman–Keuls after significant repeated measures ANOVA, n = 32). Activity remained unchanged at the highest dose (9 mg/kg) (p < 0.01 compared to baseline levels, Newman–Keuls after significant repeated measures ANOVA, n = 23). Nine neurons (30 %) remained insensitive, and only one neuron exhibited a progressive reduction in firing activity during the course of the cumulative administration of the drug. When tested at lower doses (0.5–1 mg/kg) in a larger population of neurons (n = 36), D-AMP did not have an overall significant effect on basal activity, though 12/36 neurons (33 %) had their firing increasing significantly compared to their respective baseline levels (p < 0.05, compared saline response, Fisher’s exact test).
Neurons treated with saline (n = 28) did not display any significant change in firing (Fig. 2d), even within a long recording period (>2000 s), as already observed (Gronier 2011).
The activating effect of D-AMP is partially dopamine D1 receptor-dependent (Fig. 3)
d-Amphetamine increased the firing rate of the PFC neurons through a D1 receptor mechanism. a Administration of the D1 receptor antagonist SCH 23390 (0.6 mg/kg) significantly decreased the activation of firing rate elicited by D-AMP. b, c Typical firing rate histograms of prefrontal cortex neurons during D-AMP and SCH 23390 administration. Boxed is shown a typical action potential shape of a pyramidal neuron. The same neurons were studied in the complete sequence. *P < 0.05, ***P < 0.001 versus baseline; ++P < 0.01 versus corresponding values obtained immediately before administration of SCH23390. Newman–Keuls after significant repeated measures ANOVA
Figure 3a shows that the increase in firing activity elicited by cumulative doses of D-AMP (6–9 mg/kg) is significantly attenuated by the administration of 0.6 mg/kg of the D1 receptor antagonist SCH 23390 (p < 0.01 compared to levels obtained after 6–9 mg/kg of D-AMP, Newman–Keuls after significant repeated measures ANOVA). Complete reversal occurred in 5 out of 14 neurons tested this way (36 %), while 4 neurons had their firing that remains insensitive, or keeps increasing, following SCH 23390, despite D-AMP was found to more than double their basal firing activity (Table 1).
Dose-dependent activating effects of ATX (Fig. 2c)
ATX was cumulatively administered up to a dose of 9 mg/kg, by injecting subsequently doses of 1, 2, 3, and 3 mg/kg. ATX did not increase significantly the basal activity of PFC neurons at the dose of 1 and 3 mg/kg, though in a larger population of 30 neurons, a dose of 3 mg/kg increased significantly the mean basal activity by 38 % (p < 0.01, Student’s paired t test, n = 30, Table 1). Cumulative doses of 6 mg/kg provide a significant increase in firing by 98 % over baseline activity (P < 0.001 compared to baseline levels for the 6 and 9 mg/kg doses; Newman–Keuls test after significant repeated measures ANOVA, n = 24). The dose of 9 mg/kg did not enhance further the firing activity, compared to the 6 mg/kg dose (Fig. 2c).
Effect of selective catecholamine antagonists on ATX-induced activation of firing (Fig. 4)
Effects of selective monoamine receptor antagonists on atomoxetine-induced activation of PFC neurons. a, c, e Neither D1 (SCH 23390), α1 (prazosin) or α2 receptor antagonists did significantly reverse ATX-dependent activation of PFC neurons. The α2 receptor antagonist yohimbine further increased the activation of firing rate induced by a cumulative dose of 3 mg/kg of ATX, and this activation is partially dependent on dopamine D1 receptor (e). b, d, f Firing rate histograms of individual PFC neuron during the administration of ATX, followed by the administration of antagonists. Neuron shown in d had its activity dramatically reduced following the administration of prazosin, unlike other neurons tested this way (b). *P < 0.05, **P < 0.01 versus respective baseline; ++P < 0.01 vs. values obtained immediately before administration of yohimbine. Newman–Keuls test after significant repeated measures ANOVA. The same neurons were studied in the complete sequence, except in Fig. 4e where the D1 antagonist has not been administered following yohimbine administration in three neurons
We have found previously that the different selective monoamine receptor antagonists—SCH 23390, prazosin, yohimbine—exerted no effects on their own on the basal firing activity of PFC neurons (Gronier 2011). We have confirmed these previous data on small groups of neurons from naive rats (yohimbine 1 mg/kg, n = 5; SCH 23390 0.6 mg/kg, n = 5; prazosin 1 mg/kg, n = 4; not significantly different (NS) compared to respective baseline levels, paired Student t test, data not shown).
Administration of the D1 dopamine receptor antagonist SCH 23390 did not significantly reduce the mean excitatory effect produced by cumulative doses of ATX (NS, Newman–Keuls after significant repeated measures ANOVA, Fig. 4a, Table 1). However, in three out of seven neurons tested, a significant reduction of activation by more than 30 % was observed.
Similarly, administration of the α1 adrenoceptor antagonist prazosin, following cumulative doses of ATX, had no significant effect on the neuronal activation induced by ATX. Yet, in three neurons out of seven tested, ATX-induced activation was reduced by more than 50 % following prazosin administration (Fig. 4c–d). Figure 4d represents the firing histogram of such a neuron whose firing activation induced by ATX was reduced by prazosin.
In a group of seven neurons initially insensitive to ATX (cumulative dose of 3 mg/kg produced no significant change in firing activity), the α2 adrenergic antagonist yohimbine (1 mg/kg) promoted a significant activation of firing (p < 0.01, compared to value obtained in baseline condition or after ATX, Newman–Keuls test after significant repeated measures ANOVA, Fig. 4e). Interestingly, a further administration of the dopamine D1 antagonist SCH 23390 reversed this stimulatory effect in two out of the four neurons that could have been tested this way (Fig. 4e–f).
ADHD drugs preferentially stimulate the firing activity of spontaneously active PFC neurons
Only 36 % (9/25) of the neurons with very low or no baseline activity became spontaneously active or had their firing activity significantly increased, following the administration of the ADHD drugs, regardless of the type of drug tested. On the other hand, most spontaneously active neurons (77 %) were responsive to any of the ADHD drug tested.
ADHD drugs potentiate NMDA-induced activation (Fig. 5)
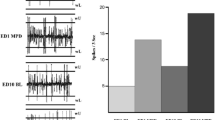
Effect of ADHD drugs on NMDA-induced activation of PFC neurons. a Mean NMDA-induced increase in firing rates of PFC neurons before and following ADHD drugs and saline administration. All three ADHD drugs induced a significant increase in NMDA-induced firing activities. b Mean AMPA-induced firing rates of PFC neurons is not significantly changed following D-AMP administration. *P < 0.04, **P < 0.001 versus respective controls (white bars). Paired Student’s t test
When applied at currents of −10 nA, NMDA produced generally a large increase in firing activity which typically increased baseline levels by 40–120 additional spikes/10 s during the ejection period. Only neurons which application of NMDA produced consistent neuronal activations over time were selected. After at least four to seven consistent responses to NMDA, saline or ADHD drugs were administered at cumulative doses.
Figure 5 shows that the activation of firing induced by NMDA was significantly increased following the iv administration of MPH (3 mg/kg, P < 0.04, n = 9, paired Student’s t test), ATX (3 mg/kg, P < 0.03, paired Student’s t test, n = 11) and D-AMP (3 mg/kg, P < 0.001, paired Student’s t test, n = 15) but remained unchanged following the administration of saline (Fig. 5a). On the other hand, the response of neurons to AMPA application (−5 nA) was also tested on a group of neurons treated with D-AMP (n = 6). No significant effect was observed, though two neurons exhibited an increase in both their NMDA and AMPA responses after D-AMP administration (1–3 mg/kg) by more than 30 % (Fig. 5b).
Most of the neurons ((14/18 in total) that were silent in basal conditions had their responses to NMDA significantly augmented in response to the administration of the ADHD drugs but remained silent or with a very low baseline activity during the course of the experiment (Fig. 6a, c). On the other hand, spontaneously active neurons generally showed an increase in both baseline and NMDA-induced firing (Fig. 6b).
Individual responses of PFC neurons to NMDA following ADHD drugs. a, b, c Typical firing rate histograms of representative PFC neurons showing their response to NMDA during the administration of ADHD drugs. Each top horizontal bar represents the pulsed current applied onto the neuron, therefore inducing transient neuronal activation (NMDA −10 nA, AMPA −5 nA)
Lower doses of D-AMP (0.5–1 mg/kg), closer to therapeutic ranges, did not change significantly the mean NMDA response. However, it was found to increase by more than 20 % the NMDA response of 7 neurons out of 17 (41 %, Fig. 7c, Table 1), while a higher proportion of neurons responded to the 3 mg/kg dose (11/15, 73 %, Table 1). Figure 7a, c, d shows that the potentiating effect of D-AMP on the NMDA response was significantly reduced following the administration of the D1 receptor antagonist SCH 23390 in a group of six neurons (P < 0.05 when comparing values obtained immediately before and after administration of SCH 23390; Newman–Keuls test after significant repeated measures ANOVA). On the other hand, in one other neuron tested (not included in the graph), the NMDA response that was further increased following SCH 23390. When neurons were pretreated with SCH 23390 (0.6 mg/kg), the administration of D-AMP did not change significantly the neuronal response to NMDA, though two neurons out of the five tested still had their response increased by 20 % following D-AMP (Fig. 7b).
Effect of dopamine D1 receptor blockade on NMDA-induced activation of PFC neurons induced by D-AMP. a Mean NMDA-induced firing rates of PFC neurons before and after D-AMP and following administration of SCH 23390, in a group of six neurons. D-AMP induced a significant increase in NMDA-induced firing activation that was significantly reduced following administration of SCH 23390. b Mean NMDA-induced firing rates of PFC neurons following D-AMP administration in neurons pretreated with the dopamine D1 receptor antagonist SCH 23390. Administration of SCH 23390 prevents the effect of D-AMP on NMDA-induced firing (though two neurons of this group had their NMDA response still augmented after D-AMP administration). c, d Firing rate histogram in basal and NMDA-stimulated condition of a PFC neuron before and after the administration of D-AMP at low (c) or medium (d) doses, and following SCH 23390. The neuron showed an increase in NMDA response caused by D-AMP; this was partially reversed following the D1 receptor antagonist. The same neurons were recorded during the complete sequence. **P < 0.01 versus baseline; *P < 0.05 versus corresponding values obtained immediately before administration of SCH 23390. Newman–Keuls test after significant repeated measures ANOVA
Table 1 summarises all data obtained in the present study, showing the proportion of neurons tested responding to the administration of the different drugs.
Discussion
The present study demonstrates that the ADHD drugs MPH, D-AMP and ATX modulate the firing activity and glutamate excitability of PFC pyramidal neurons.
As already demonstrated (Gronier 2011), we confirmed that MPH excitatory effects on PFC neurons are largely dependent on dopamine D1 receptors. Nonetheless, the present study is the first to demonstrate that D-AMP also stimulated PFC firing activity in a dose-dependent manner which is also partially dependent on the stimulation of D1 receptors. This is compatible with the facts that both D-AMP and MPH, by interacting with the synaptic catecholamine transporters and increasing dopamine efflux, act as indirect dopamine D1 agonists and that D1 receptor activation can generate increase in neuronal excitability (Tseng and O’Donnell 2007; Wang et al. 2011; Gronier et al. 2013). As a matter of fact, we found in a previous investigation that the systemic administration of D1 selective agonists, and to a lesser extent their local application, can stimulate the firing activity of PFC neurons in vivo (Gronier et al. 2013).
The magnitude of the increase in firing activity elicited by both psychostimulants, MPH and D-AMP, does not seem directly proportional to the amount of dopamine release that these drugs can induce. According to microdialysis studies, D-AMP is by far more potent than MPH in inducing dopamine release but does not produce a more dramatic increase in firing activity, indicating that D1-dependent activation of pyramidal neurons is likely at its maximum levels, when the two drugs are already administered at intermediate doses (3–6 mg/kg).
However, the present study also shows that these large activations occur at doses that slightly exceed therapeutic levels. Lower doses of MPH and D-AMP closer to the therapeutic range (0.5–1 mg/kg for both drugs) produced milder electrical activation only in a subset of PFC neurons (Table 1; see also previous studies for MPH (Gronier et al. 2009; Gronier 2011). Although it is difficult to compare doses in human and rodents, which have powerful metabolism capacity, we can expect that, in human, therapeutic doses of D-AMP and MPH are likely to produce moderate dopamine D1-dependent increase in excitability of PFC neurons. Larger activation of PFC neurons occurring at higher doses is likely to be associated with the well-known motor and cognitive impairments induced by these drugs (Swanson and Volkow 2003). Indeed, the D1 receptor is known to exert biphasic effect on PFC function, with moderate activation producing cognition improvement and sharpening synaptic transmission, while excessive stimulation impairs cognitive function and neuronal network activity (Goldman-Rakic et al. 2000; Seamans and Yang 2004).
However, D1 receptor stimulation is likely not the only catecholamine receptor involved in psychostimulant-induced activation of PFC neurons. As a matter of fact, only 60–65 % of the neurons activated by MPH or D-AMP had their activation significantly reversed by D1 receptor antagonist, and in some of these D1-sensitive neurons, the effect of the antagonist was only partial. Therefore, contribution of other catecholamine receptors is also likely. To note, we found in a previous study that β1 adrenergic and dopamine D2 receptors could contribute to MPH-induced activation of PFC neurons in a subset of neurons. Nevertheless, in the present study blockade of D1 receptor was the only pharmacological manipulation that significantly reduced the excitatory effects of psychostimulants.
On the other hand, ATX-induced neuronal activation of PFC neurons did not seem to be primarily dependent on D1 receptors. Whether this can be associated with the lack of immediate effect of ATX, over psychostimulants, in ADHD patients is an interesting question to address. However, ATX is known to promote, with similar potency as MPH, large amount of dopamine release in the PFC in in vivo condition (Bymaster et al. 2002), likely via its action on the NET, which plays a key role in the clearance of dopamine in the PFC (Devoto and Flore 2006). ATX-induced effect may result from a more complex combination of activation of different catecholamine receptors. As a matter of fact, contribution of the α1 adrenoceptor has been demonstrated in our study in a fraction of the neurons tested (Fig. 4c–d). Furthermore, initial blockade of α2 receptors by yohimbine (Fig. 4e–f) was found to potentiate the effect of ATX on firing activity, at least in a subset of neurons. ATX, by potently blocking NET, may initially indirectly activate inhibitory α2 receptors on the pyramidal cell and therefore attenuate possible excitatory effects mediated by other catecholamine receptors, including the D1 receptors, as demonstrated in some neurons tested in the present study (Fig. 4e). Therefore, by this way, ATX may not be able to promote the same dopamine D1 receptor-dependent excessive activation of PFC neurons which may be associated to the well-described behavioural disturbances elicited by psychostimulants when administered at high doses (Spiller et al. 2013).
Nevertheless, the fact that a significant proportion of neurons (compared to saline-treated) are activated by therapeutic doses of ADHD drugs may well contribute to improve ADHD symptoms. Moderate enhancement of PFC neuronal excitability may help to compensate for the well-documented cortical hypofunction observed in ADHD, as evidenced by reduced cortical thickness and metabolic activity in some subregions of the PFC in drug-free patients (McLaughlin et al. 2013; Fernandez-Jaen et al. 2014).
In parallel with some of our latest observation with D1 receptor agonist (Gronier et al. 2013), we found that ADHD drugs preferentially increased the basal activity of spontaneously active PFC neurons but had more limited effects on silent neurons (neurons with no spontaneous activity). This is compatible with the fact that dopamine D1 receptor may not directly stimulate the activity of PFC pyramidal neurons but rather may function by strengthening a tonic excitatory regulation of firing activity of PFC neurons. Such tonic excitatory input is likely to be mainly maintained by glutamatergic neurotransmission, as the spontaneous activity of PFC neurons has been shown to be mediated by mutual excitation between glutamatergic pyramidal neurons (Kritzer and Goldman-Rakic 1995). As a matter of fact, we found that ADHD drugs powerfully stimulate the excitability of PFC neurons mediated by glutamate receptor activation.
Indeed, the present study, for the first time, demonstrates that ADHD drugs promote in vivo a potentiation of the excitatory response of PFC neurons to the glutamate agonist NMDA, an effect not associated with change in basal firing activity. Ionotropic glutamate receptors in PFC are essential for attention processes, impulse control and for proper memory function (Nakanishi 1992). Antagonism of NMDA receptor has been shown to prevent some of the acute and chronic neurochemical and behavioural effects of psychostimulants (Hemrick-Luecke et al. 1992; Bristow et al. 1994; Gaytan et al. 2000; Yang et al. 2000), indicating that NMDA receptors may play a crucial role in stimulant-induced effects. Moreover, recent behavioural evidence shows that administration of NMDA antagonists directly in the PFC induced a profound deficit in attention and increased impulsivity in rodents (Carli and Invernizzi 2014).
As indicated previously, imaging studies have shown that children with ADHD exhibit hypoactivity in prefrontal region, as well as anatomical and functional dysfunction in frontostriatal circuits, associated with white matter loss (Liston et al. 2011; Cubillo et al. 2012). The elevated NMDA receptor function induced by ADHD drugs may lead to an increase in excitability of PFC neurons that may be essential in regulating glutamatergic corticostriatal transmission, and strengthen synaptic transmission at corticostriatal synapses. This may impact on cortical as well as on striatal synaptic plasticity and possibly, via this way, exert beneficial effects on various aspects of cognitive and executive functions which are impaired in ADHD. As a matter of fact, glutamate neurotransmission in ventral and dorsal stritatum, originating mainly from glutamate frontostriatal projections, plays a major role in controlling cognitive flexibility ((Ding et al. 2014).
The question arises as to whether pharmacological manipulation that can enhance NMDA receptor function can be of therapeutic benefit for ADHD children. For instance, one can speculate that cognition enhancers such as direct or indirect glycineB agonists of the NMDA receptor (e.g., GlyT1 glycine uptake inhibitors) could be of any clinical use (Cheng et al. 2014). Such agents have been shown to improve cognitive flexibility and working memory (Bado et al. 2011; Kuriyama et al. 2011; Nikiforuk et al. 2011), as well as attention deficits caused by NMDA receptor blockade (Chang et al. 2014). Despite that these drugs have not proven clear therapeutical benefit on negative deficits in schizophrenia (Chue 2013; Schoemaker et al. 2014), it cannot be ruled out that they can contribute to alleviate some of the symptoms of ADHD. However, increase in NMDA receptor function may be only one component of the complex mechanisms that can lead to therapeutic responses, and it is now necessary to establish whether this effect can persist after the chronic administration of ADHD drugs.
Interestingly, we found that, for D-AMP, this increase in NMDA neurotransmission may occur, at least partially, via an interaction with dopamine D1 receptors as we found in a significant number of neurons tested (but not all) that the selective D1 antagonist SCH 23390 reversed or prevented the potentiation of NMDA response elicited by D-AMP. Dopamine is known to facilitate glutamatergic transmission onto PFC neurons via interaction with both AMPA and NMDA receptors. Intracellular electrophysiological studies have shown that NMDA receptor currents are potentiated by dopamine D1 receptor activation (Sarantis et al. 2009), while D1 receptor stimulation may facilitate AMPA receptor synaptic insertion in the PFC (Sun et al. 2005). Postsynaptic dopamine D1 and some NMDA receptors colocalised in the PFC and have been shown to interact synergically (Goldman-Rakic et al. 2000; Wang and O’Donnell 2001; Kruse et al. 2009). It is generally believed that dopamine promotes NMDA receptor trafficking through phosphorylation induced by D1 receptor-like signalling cascade (Yang and Chen 2005; Hu et al. 2010; Trepanier et al. 2012; Urban et al. 2013). Nevertheless, the role of dopamine on NMDA receptor function can be very complex in the PFC, with differential modulation dependent on receptor subtypes (Urban and Gao 2013). In addition, not all neurons tested were responsive to the dopamine antagonist after potentiation of their NMDA response by D-AMP. Therefore, other mechanisms may also contribute to explain the increase in glutamate/NMDA excitability generated by ADHD drugs (Zhang et al. 2012).
Our results, showing that ATX also enhances NMDA-mediated response on PFC neurons, stand in contrast with a recent in vitro electrophysiological study, demonstrating that ATX, at relatively low concentration (5 μM), inhibits NMDA-induced current when studied on PFC slices, via an open-channel blocking mechanism (Ludolph et al. 2010). Similar results were observed with D-AMP which could act as a low affinity inhibitor of the NMDA receptor complex (Yeh et al. 2002). However, ATX, like D-AMP, enhances dopamine efflux in PFC according to microdialysis studies. Even if D1 receptor stimulation moderately contributes to enhance the basal activity of PFC neurons in ATX-treated animals, ATX may generate enough D1 receptor activation to interact with NMDA receptors and compensate for the weak inhibitory effects of this drug on NMDA receptor.
Finally, our data differs from two other electrophysiological investigations carried out in anaesthetised rats, using different recording techniques, showing that D-AMP (2 mg/kg) and MPH (0.25–15 mg/kg) do not elicit activation of firing of PFC neurons but rather, in the case of D-AMP, tend to have suppressant effects (Devilbiss and Berridge 2008; Wood et al. 2012). Such divergence might be associated with differences in the techniques of recording, in particular regarding the use of probes of different sensitivity for recording the neuronal electrical signals. Nevertheless, the study of Devilbiss and Berridge (2008) shows that low dose of MPH (0.5 mg/kg) increased the evoked excitatory responses of PFC neurons following hippocampus stimulation. Such stimulatory effect, likely to involve glutamate-dependent processes, is not in contradiction with our study.
Our data agrees, in part, with a recent in vitro electrophysiological study showing that an acute administration of low dose of MPH can increase NMDA-dependent excitatory postsynaptic potentials on PFC slices (Cheng et al. 2014). However, the authors did not demonstrate any contribution of the D1 receptor in this modulation and, rather, suggested an involvement of alpha1/2 adrenoceptors.
In conclusion, our electrophysiological studies indicate that all three ADHD drugs equally stimulate the excitability of PFC pyramidal neurons, in basal and NMDA-evoked conditions. While the electrophysiological effects elicited by psychostimulants may be primarily dependent on dopamine D1 receptor activation, those induced by ATX may also rely on other mechanisms. Finally, our data support the assumption that NMDA receptor activation may be required for the therapeutic effect of ADHD drugs and psychostimulants.
Abbreviations
- ATX:
-
Atomoxetine
- D-AMP:
-
d-Amphetamine
- DAT:
-
Dopamine transporter
- MPH:
-
Methylphenidate
- NET:
-
Norepinephrine transporter
- NMDA:
-
N-Methyl-d-aspartate
- PFC:
-
Prefrontal cortex
References
Amitai N, Markou A (2010) Disruption of performance in the five-choice serial reaction time task induced by administration of N-methyl-D-aspartate receptor antagonists: relevance to cognitive dysfunction in schizophrenia. Biol Psychiatry 68:5–16
Bado P, Madeira C, Vargas-Lopes C, Moulin TC, Wasilewska-Sampaio AP, Maretti L, de Oliveira RV, Amaral OB, Panizzutti R (2011) Effects of low-dose D-serine on recognition and working memory in mice. Psychopharmacology (Berl) 218:461–470
Bartho P, Hirase H, Monconduit L, Zugaro M, Harris KD, Buzsaki G (2004) Characterization of neocortical principal cells and interneurons by network interactions and extracellular features. J Neurophysiol 92:600–608
Bristow LJ, Thorn L, Tricklebank MD, Hutson PH (1994) Competitive NMDA receptor antagonists attenuate the behavioural and neurochemical effects of amphetamine in mice. Eur J Pharmacol 264:353–359
Bushe CJ, Savill NC (2014) Systematic review of atomoxetine data in childhood and adolescent attention-deficit hyperactivity disorder 2009–2011: focus on clinical efficacy and safety. J Psychopharmacol 28:204–211
Bymaster FP, Katner JS, Nelson DL, Hemrick-Luecke SK, Threlkeld PG, Heiligenstein JH, Morin SM, Gehlert DR, Perry KW (2002) Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology 27:699–711
Carli M, Invernizzi RW (2014) Serotoninergic and dopaminergic modulation of cortico-striatal circuit in executive and attention deficits induced by NMDA receptor hypofunction in the 5-choice serial reaction time task. Front Neural Circ 8:58
Chang JP, Lane HY, Tsai GE (2014) Attention deficit hyperactivity disorder and N-methyl-D-aspartate (NMDA) dysregulation. Curr Pharm Des 20(32):5180–5185
Cheng J, Xiong Z, Duffney LJ, Wei J, Liu A, Liu S, Chen GJ, Yan Z (2014) Methylphenidate exerts dose-dependent effects on glutamate receptors and behaviors. Biol Psychiatry 76(12):953–962. doi:10.1016/j.biopsych.2014.04.003
Chue P (2013) Glycine reuptake inhibition as a new therapeutic approach in schizophrenia: focus on the glycine transporter 1 (GlyT1). Curr Pharm Des 19:1311–1320
Cubillo A, Halari R, Smith A, Taylor E, Rubia K (2012) A review of fronto-striatal and fronto-cortical brain abnormalities in children and adults with Attention Deficit Hyperactivity Disorder (ADHD) and new evidence for dysfunction in adults with ADHD during motivation and attention. Cortex 48:194–215
Devilbiss DM, Berridge CW (2008) Cognition-enhancing doses of methylphenidate preferentially increase prefrontal cortex neuronal responsiveness. Biol Psychiatry 64:626–635
Devoto P, Flore G (2006) On the origin of cortical dopamine: is it a co-transmitter in noradrenergic neurons? Curr Neuropharmacol 4:115–125
Ding X, Qiao Y, Piao C, Zheng X, Liu Z, Liang J (2014) N-methyl-D-aspartate receptor-mediated glutamate transmission in nucleus accumbens plays a more important role than that in dorsal striatum in cognitive flexibility. Front Behav Neurosci 8:304
Dorval KM, Wigg KG, Crosbie J, Tannock R, Kennedy JL, Ickowicz A, Pathare T, Malone M, Schachar R, Barr CL (2007) Association of the glutamate receptor subunit gene GRIN2B with attention-deficit/hyperactivity disorder. Genes Brain Behav 6:444–452
Easton N, Steward C, Marshall F, Fone K, Marsden C (2007) Effects of amphetamine isomers, methylphenidate and atomoxetine on synaptosomal and synaptic vesicle accumulation and release of dopamine and noradrenaline in vitro in the rat brain. Neuropharmacology 52:405–414
Fernandez-Jaen A, Lopez-Martin S, Albert J, Fernandez-Mayoralas DM, Fernandez-Perrone AL, Tapia DQ, Calleja-Perez B (2014) Cortical thinning of temporal pole and orbitofrontal cortex in medication-naive children and adolescents with ADHD. Psychiatry Res 224(1):8–13. doi:10.1016/j.pscychresns.2014.07.004
Floresco SB (2013) Prefrontal dopamine and behavioral flexibility: shifting from an “inverted-U” toward a family of functions. Front Neurosci 7:62
Gamo NJ, Wang M, Arnsten AF (2010) Methylphenidate and atomoxetine enhance prefrontal function through alpha2-adrenergic and dopamine D1 receptors. J Am Acad Child Adolesc Psychiatry 49:1011–1023
Gaytan O, Nason R, Alagugurusamy R, Swann A, Dafny N (2000) MK-801 blocks the development of sensitization to the locomotor effects of methylphenidate. Brain Res Bull 51:485–492
Gobbi G, Janiri L (2006) Sodium- and magnesium-valproate in vivo modulate glutamatergic and GABAergic synapses in the medial prefrontal cortex. Psychopharmacology (Berl) 185:255–262
Goldman-Rakic PS, Muly EC 3rd, Williams GV (2000) D (1) receptors in prefrontal cells and circuits. Brain Res Brain Res Rev 31:295–301
Gronier B (2011) In vivo electrophysiological effects of methylphenidate in the prefrontal cortex: involvement of dopamine D1 and alpha 2 adrenergic receptors. Eur Neuropsychopharmacol 21:192–204
Gronier B, Aston J, Liauzun C, Zetterstrom T (2009) Age-dependent effects of methylphenidate in the prefrontal cortex: evidence from electrophysiological and Arc gene expression measurements. J Psychopharmacol 24:1819–1827
Gronier B, Waters S, Ponten H (2013) The dopaminergic stabilizer pridopidine increases neuronal activity of pyramidal neurons in the prefrontal cortex. J Neural Transm 120:1281–1294
Gui ZH, Zhang QJ, Liu J, Zhang L, Ali U, Hou C, Fan LL, Sun YN, Wu ZH, Hui YP (2011) Unilateral lesion of the nigrostriatal pathway decreases the response of fast-spiking interneurons in the medial prefrontal cortex to 5-HT1A receptor agonist and expression of the receptor in parvalbumin-positive neurons in the rat. Neurochem Int 59:618–627
Hajos M, Gartside SE, Varga V, Sharp T (2003) In vivo inhibition of neuronal activity in the rat ventromedial prefrontal cortex by midbrain-raphe nuclei: role of 5-HT1A receptors. Neuropharmacology 45:72–81
Heal DJ, Cheetham SC, Smith SL (2009) The neuropharmacology of ADHD drugs in vivo: insights on efficacy and safety. Neuropharmacology 57:608–618
Hemrick-Luecke SK, Henderson MG, Fuller RW (1992) MK801 antagonism of the prolonged depletion of striatal dopamine by amphetamine in iprindole-treated rats. Life Sci 50:PL31–PL33
Hu JL, Liu G, Li YC, Gao WJ, Huang YQ (2010) Dopamine D1 receptor-mediated NMDA receptor insertion depends on Fyn but not Src kinase pathway in prefrontal cortical neurons. Mol Brain 3:20
Kargieman L, Santana N, Mengod G, Celada P, Artigas F (2007) Antipsychotic drugs reverse the disruption in prefrontal cortex function produced by NMDA receptor blockade with phencyclidine. Proc Natl Acad Sci U S A 104:14843–14848
Kieling C, Goncalves RR, Tannock R, Castellanos FX (2008) Neurobiology of attention deficit hyperactivity disorder. Child and adolescent psychiatric clinics of North America 17:285–307
Koda K, Ago Y, Cong Y, Kita Y, Takuma K, Matsuda T (2010) Effects of acute and chronic administration of atomoxetine and methylphenidate on extracellular levels of noradrenaline, dopamine and serotonin in the prefrontal cortex and striatum of mice. J Neurochem 114:259–270
Kritzer MF, Goldman-Rakic PS (1995) Intrinsic circuit organization of the major layers and sublayers of the dorsolateral prefrontal cortex in the rhesus monkey. J Comp Neurol 359:131–143
Kruse MS, Premont J, Krebs MO, Jay TM (2009) Interaction of dopamine D1 with NMDA NR1 receptors in rat prefrontal cortex. Eur Neuropsychopharmacol 19:296–304
Kuriyama K, Honma M, Shimazaki M, Horie M, Yoshiike T, Koyama S, Kim Y (2011) An N-methyl-D-aspartate receptor agonist facilitates sleep-independent synaptic plasticity associated with working memory capacity enhancement. Sci Rep 1:127
Liston C, Malter Cohen M, Teslovich T, Levenson D, Casey BJ (2011) Atypical prefrontal connectivity in attention-deficit/hyperactivity disorder: pathway to disease or pathological end point? Biol Psychiatry 69:1168–1177
Ludolph AG, Udvardi PT, Schaz U, Henes C, Adolph O, Weigt HU, Fegert JM, Boeckers TM, Fohr KJ (2010) Atomoxetine acts as an NMDA receptor blocker in clinically relevant concentrations. Br J Pharmacol 160:283–291
McLaughlin KA, Sheridan MA, Winter W, Fox NA, Zeanah CH, Nelson CA (2013) Widespread reductions in cortical thickness following severe early-life deprivation: a neurodevelopmental pathway to attention-deficit/hyperactivity disorder. Biol Psychiatry 76(8):629–638. doi:10.1016/j.biopsych.2013.08.016
Nakanishi S (1992) Molecular diversity of glutamate receptors and implications for brain function. Science 258:597–603
Nikiforuk A, Kos T, Rafa D, Behl B, Bespalov A, Popik P (2011) Blockade of glycine transporter 1 by SSR-504734 promotes cognitive flexibility in glycine/NMDA receptor-dependent manner. Neuropharmacology 61:262–267
Paxinos G, Watson C (1997) The rat brain in stereotaxic coordinates. Academic Press, San Diego
Polanczyk G, de Lima MS, Horta BL, Biederman J, Rohde LA (2007) The worldwide prevalence of ADHD: a systematic review and metaregression analysis. Am J Psychiatry 164:942–948
Povysheva NV, Zaitsev AV, Rotaru DC, Gonzalez-Burgos G, Lewis DA, Krimer LS (2008) Parvalbumin-positive basket interneurons in monkey and rat prefrontal cortex. J Neurophysiol 100:2348–2360
Puig MV, Artigas F, Celada P (2005) Modulation of the activity of pyramidal neurons in rat prefrontal cortex by raphe stimulation in vivo: involvement of serotonin and GABA. Cereb Cortex 15:1–14
Sarantis K, Matsokis N, Angelatou F (2009) Synergistic interactions of dopamine D1 and glutamate NMDA receptors in rat hippocampus and prefrontal cortex: involvement of ERK1/2 signaling. Neuroscience 163:1135–1145
Schoemaker JH, Jansen WT, Schipper J, Szegedi A (2014) The selective glycine uptake inhibitor org 25935 as an adjunctive treatment to atypical antipsychotics in predominant persistent negative symptoms of schizophrenia: results from the GIANT trial. J Clin Psychopharmacol 34:190–198
Seamans JK, Yang CR (2004) The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 74:1–58
Shen F, Tsuruda PR, Smith JA, Obedencio GP, Martin WJ (2013) Relative contributions of norepinephrine and serotonin transporters to antinociceptive synergy between monoamine reuptake inhibitors and morphine in the rat formalin model. PLoS One 8:e74891
Spiller HA, Hays HL, Aleguas A Jr (2013) Overdose of drugs for attention-deficit hyperactivity disorder: clinical presentation, mechanisms of toxicity, and management. CNS Drugs 27:531–543
Sullivan RM, Brake WG (2003) What the rodent prefrontal cortex can teach us about attention-deficit/hyperactivity disorder: the critical role of early developmental events on prefrontal function. Behav Brain Res 146:43–55
Sun X, Zhao Y, Wolf ME (2005) Dopamine receptor stimulation modulates AMPA receptor synaptic insertion in prefrontal cortex neurons. J Neurosci 25:7342–7351
Swanson JM, Volkow ND (2003) Serum and brain concentrations of methylphenidate: implications for use and abuse. Neurosci Biobehav Rev 27:615–621
Tierney PL, Thierry AM, Glowinski J, Deniau JM, Gioanni Y (2008) Dopamine modulates temporal dynamics of feedforward inhibition in rat prefrontal cortex in vivo. Cereb Cortex 18:2251–2262
Trepanier CH, Jackson MF, MacDonald JF (2012) Regulation of NMDA receptors by the tyrosine kinase Fyn. FEBS J 279:12–19
Tseng KY, O’Donnell P (2004) Dopamine-glutamate interactions controlling prefrontal cortical pyramidal cell excitability involve multiple signaling mechanisms. J Neurosci 24:5131–5139
Tseng KY, O’Donnell P (2007) Dopamine modulation of prefrontal cortical interneurons changes during adolescence. Cereb Cortex 17:1235–1240
Tseng KY, Mallet N, Toreson KL, Le Moine C, Gonon F, O’Donnell P (2006) Excitatory response of prefrontal cortical fast-spiking interneurons to ventral tegmental area stimulation in vivo. Synapse 59:412–417
Turic D, Langley K, Mills S, Stephens M, Lawson D, Govan C, Williams N, Van Den Bree M, Craddock N, Kent L, Owen M, O’Donovan M, Thapar A (2004) Follow-up of genetic linkage findings on chromosome 16p13: evidence of association of N-methyl-D aspartate glutamate receptor 2A gene polymorphism with ADHD. Mol Psychiatry 9:169–173
Upadhyaya HP, Desaiah D, Schuh KJ, Bymaster FP, Kallman MJ, Clarke DO, Durell TM, Trzepacz PT, Calligaro DO, Nisenbaum ES, Emmerson PJ, Schuh LM, Bickel WK, Allen AJ (2013) A review of the abuse potential assessment of atomoxetine: a nonstimulant medication for attention-deficit/hyperactivity disorder. Psychopharmacology (Berl) 226:189–200
Urban KR, Gao WJ (2013) Methylphenidate and the juvenile brain: enhancement of attention at the expense of cortical plasticity? Med Hypotheses 81:988–994
Urban KR, Li YC, Gao WJ (2013) Treatment with a clinically-relevant dose of methylphenidate alters NMDA receptor composition and synaptic plasticity in the juvenile rat prefrontal cortex. Neurobiol Learn Mem 101:65–74
Wang J, O’Donnell P (2001) D (1) dopamine receptors potentiate nmda-mediated excitability increase in layer V prefrontal cortical pyramidal neurons. Cereb Cortex 11:452–462
Wang Y, Liu J, Gui ZH, Ali U, Fan LL, Hou C, Wang T, Chen L, Li Q (2011) alpha2-Adrenoceptor regulates the spontaneous and the GABA/glutamate modulated firing activity of the rat medial prefrontal cortex pyramidal neurons. Neuroscience 182:193–202
Wood J, Kim Y, Moghaddam B (2012) Disruption of prefrontal cortex large scale neuronal activity by different classes of psychotomimetic drugs. J Neurosci 32:3022–3031
Yang CR, Chen L (2005) Targeting prefrontal cortical dopamine D1 and N-methyl-D-aspartate receptor interactions in schizophrenia treatment. Neuroscientist 11:452–470
Yang P, Swann A, Dafny N (2000) NMDA receptor antagonist disrupts acute and chronic effects of methylphenidate. Physiol Behav 71:133–145
Yeh GC, Chen JC, Tsai HC, Wu HH, Lin CY, Hsu PC, Peng YC (2002) Amphetamine inhibits the N-methyl-D-aspartate receptor-mediated responses by directly interacting with the receptor/channel complex. J Pharmacol Exp Ther 300:1008–1016
Zhang CL, Feng ZJ, Liu Y, Ji XH, Peng JY, Zhang XH, Zhen XC, Li BM (2012) Methylphenidate enhances NMDA-receptor response in medial prefrontal cortex via sigma-1 receptor: a novel mechanism for methylphenidate action. PLoS One 7:e51910
Acknowledgments
This study was supported by internal DMU funds. The authors thank Mrs Anita O’Donoghue for excellent technical support and Dr. Florence Serres (Oxford University) for carefully reading the manuscript.
Conflict of interest
Authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Miceli, M., Gronier, B. Psychostimulants and atomoxetine alter the electrophysiological activity of prefrontal cortex neurons, interaction with catecholamine and glutamate NMDA receptors. Psychopharmacology 232, 2191–2205 (2015). https://doi.org/10.1007/s00213-014-3849-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3849-y