Abstract
Myocardial PDE2 activity increases in terminal human heart failure and after isoprenaline infusion in rat heart. PDE2 inhibitors do not potentiate the murine sinoatrial tachycardia produced by noradrenaline. We investigated whether isoprenaline infusion induces PDE2 to decrease the chronotropic and inotropic effects of catecholamines in rat heart. Sprague-Dawley rats were infused with isoprenaline (2.4 mg kg−1 day−1) for 3 days. We used spontaneously beating right atria, paced right ventricular strips and left ventricular papillary muscles. The effects of the PDE2 inhibitors EHNA (10 μM) and Bay 60-7550 (0.1–1 μM) were investigated on the cardiostimulation produced by noradrenaline (ICI118551 50 nM present to block β2-adrenoceptors) and adrenaline (CGP20712A 300 nM present to block β1-adrenoceptors). Hydrolysis of cAMP by PDE2 was measured by radioenzyme assay. Bay 60-7550 but not EHNA increased sinoatrial beating. A stable tachycardia elicited by noradrenaline (10 nM) or adrenaline (1 μM) was not increased by the PDE2 inhibitors. Isoprenaline infusion increased the hydrolytic PDE2 activity threefold in left ventricle, reduced the chronotropic and inotropic effects and potency of noradrenaline and abolished the effects of adrenaline. The potency of the catecholamines was not increased by the PDE2 inhibitors. Neither EHNA nor Bay 60-7550 potentiated the effects of the catecholamines. Rat PDE2 decreased basal sinoatrial beating but did not reduce the sinoatrial tachycardia or increases of ventricular force mediated through β1- and β2-adrenoceptors. The β-adrenoceptor desensitization induced by the isoprenaline infusion was not reversed by the PDE2 inhibitors despite the increased hydrolysis of cAMP by PDE2.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The second messenger cAMP is fundamentally involved in cardiac function. The cAMP concentration is particularly high in sinoatrial node cells where it facilitates basal beating (Vinogradova et al. 2006). Cyclic AMP also participates in the increases of sinoatrial beating (Vinogradova et al. 2006, 2008) and ventricular force (Bers 2002) through activation of β-adrenoceptors. Phosphodiesterases PDE1, PDE2, PDE3, PDE4 (Bender and Beavo 2006) and PDE8 (Patrucco et al. 2010) hydrolyse cAMP and are expressed in the heart. PDE2 it is allosterically stimulated by cGMP binding to one of its GAF domains (Martins et al. 1982).
PDE3 and PDE4 do not appear to modify the sinoatrial tachycardia elicited by the agonists through β-adrenoceptors in mouse, rat and rabbit (Galindo-Tovar and Kaumann (2008); Kaumann et al. (2009); Christ et al. (2009); Galindo-Tovar et al. (2016). Inhibition of PDE3 or PDE4 causes sinoatrial tachycardia in a variety of species (Kaumann 2011). The isoenzyme PDE2 is expressed in the murine sinoatrial node and its inhibition with EHNA causes sinoatrial tachycardia (Hua et al. 2012, but see Galindo-Tovar et al. 2016). The PDE2 inhibitor Bay 60-7550 also causes sinoatrial tachycardia (Galindo-Tovar et al. 2016; Vettel et al. 2017).
Several effects of PDE2 have been published for normal heart that generates contractile force. In rat ventricular myocytes, the PDE2 inhibitor EHNA increases the response of L-type Ca2+current (ICa,L) to isoprenaline (Verde et al. 1999). In human atrial myocytes, a high cGMP concentration (5 μM) decreases ICa,L, an effect prevented by EHNA, consistent with activation of PDE2, hydrolysis of cAMP and reduction of PKA-dependent ICa,L (Vandecasteele et al. 2001). In myocytes of new-born mice noradrenaline increases PDE2 activity via β3-adrenoceptor surges of cGMP, thereby conceivably attenuating effects through β1- and β2-adrenoceptors (Mongillo et al. 2006). PDE2 appears to hydrolyse basal cAMP in ventricular myocytes of new-born rats (Di Benedetto et al. 2008). In sinoatrial myocytes of rabbits (Vinogradova et al. 2008) and mice (Hua et al. (2012), a small PDE2 constitutive phosphodiesterase activity restricts the rate of spontaneous basal beating that is inhibited by EHNA. Isoprenaline causes shortening of ventricular myocytes from adult rats that is reduced by cGMP-evoked activation of PDE2 but reversed by Bay 60-7550 (Stangherlin et al. 2011).
Increases of PDE2 activity have been reported in cardiac pathology. Ventricular PDE2 enzymatic activity in rat left ventricle and ventricular weight are increased after 14 days of aortic banding (Yanaka et al. 2003). Ventricular PDE2 expression and hydrolysis of cAMP increased in patients with terminal heart failure associated with hyperfunction of the sympathetic nervous system (Mehel et al. 2013).
In rat cardiomyocytes, Bay 60-7550 did not enhance the increase of ICa,L current by isoprenaline. However, in rats infused with isoprenaline for 4 days PDE2-dependent hydrolysis of cAMP was increased, and Bay 60-7550 increased responses to isoprenaline of ICa,L, Ca2+ transients and sarcomere shortening (Mehel et al. 2013). Thus, adrenergic overstimulation appeared to induce PDE2 activity that in turn reduced these effects.
Two questions prompted our work: 1. Does chronic infusion of isoprenaline induce sinoatrial PDE2 to reduce the sinoatrial tachycardia evoked by catecholamines? 2. Does PDE2 reduce the ventricular inotropic effects of catecholamines? To answer these questions, we investigated whether the sinoatrial tachycardia and ventricular inotropic effects of noradrenaline and adrenaline, mediated through β1- and β2-adrenoceptors respectively, are increased by EHNA and Bay 60-7550.
Methods
All animal care and procedures complied with the guidelines of the European Communities Council Directive of 23 March 1998 (1999/575/CE). Authorizations to perform animal experiments according to this decree were obtained from the Ministerio de Agricultura y Agua, Comunidad Autónoma Región de Murcia, Spain (Nr. B30218 and C30215).
Male Sprague-Dawley rats (220–300 g), maintained at 21 °C on a 12-h light/dark cycle, were allowed free access to standard rodent chow and water. The rats were stunned and exsanguinated. The hearts were dissected and placed in oxygenated, modified Tyrode’s solution at room temperature containing (mM): NaCl 136.9, KCl 5.0, CaCl2 1.8, MgCl2 1.5, NaHCO3 11.9, NaH2PO4 0.4, EDTA 0.04, ascorbic acid 0.2, pyruvate 5 and glucose 5.0. The pH of the solution was maintained at pH 7.4 by bubbling with a mixture of 5% CO2 and 95% O2. Spontaneously beating right atria, right ventricular strips (width ~ 1 mm, length 4–7 mm) and a left ventricular papillary muscle were rapidly dissected, mounted in pairs and attached to Swema 4-45 strain gauge transducers in an apparatus containing above solution at 37 °C. The ventricular tissues were paced at 1 Hz and stretched to Lmax. Atrial beating rate and ventricular force were recorded through PowerLab amplifiers on a Chart for Windows, Version 5.0 recording programme (ADInstruments, Castle Hill, NSW, Australia).
All tissues were exposed to phenoxybenzamine (5 μM) for 90 min followed by washout, to irreversibly block α-adrenoceptors and tissue uptake of the catecholamines (Gille et al. 1985). Experiments with (-)-noradrenaline were carried out in the presence of ICI118551 (50 nM) to block β2-adrenoceptors. Salbutamol is an effective partial agonist at β2-adrenoceptors of human atrial tissues (Hall et al. 1990). We therefore investigated the effects of a high concentration (10 μM) of salbutamol on ventricular tissues. Experiments with (-)-adrenaline and salbutamol were carried out in the presence of CGP20712A (300 nM) to selectively block β1-adrenoceptors and conceivably uncover CGP20712A-resistant effects, mediated through β2-adrenoceptors (Kaumann 1986; Hall et al. 1990).
To assess the time-course of the chronotropic response, right atria from untreated rats were exposed to 10 nM noradrenaline or 1 μM adrenaline during 30 min. The PDE2 inhibitors EHNA (10 μM) or Bay 60-7550 (100 nM) were incubated 30 min before the administration of the agonists and also for 20 min in the presence of an agonist, pre-incubated for 10 min. Cumulative concentration-effect curves for noradrenaline and adrenaline were also determined in the absence and presence of the PDE2 inhibitors on right atria from normal rats or rats infused with isoprenaline. Experiments were terminated by the administration of a concentration of (-)-isoprenaline (200 μM) that saturates β-adrenoceptors.
For inotropic studies, cumulative concentration-effect curves for the catecholamines were determined on ventricular tissues as described above for beating atria. Following the administration of isoprenaline (200 μM), the experiments were terminated by elevating the CaCl2 concentration to 9 mM (Galindo-Tovar and Kaumann 2008).
Isoprenaline infusion
Rats were infused for 3 days with isoprenaline (2.4 mg kg−1 day−1) through osmotic mini-pumps (ALZET model 2001, USA), implanted subcutaneously under ether anesthesia. Right and left ventricular tissues were frozen in liquid nitrogen and stored at − 80 °C until use.
PDE2 assay
Frozen ventricular strips were homogenized in ice cold Tris-HCl (20 mM, pH = 7.5), Mg acetate 2.0 mM, EGTA 5.0 mM, dithiotreitol 1.0 mM, 10 μg/ml of soya trypsin inhibitor, 2000 U/ml of aprotinine and pefabloc 0.33 mM. PDE activity was measured by a two-step radioenzymatic assay (Thompson and Appelman 1971) as described by Keravis et al. (2005). Briefly, cAMP hydrolysis was measured 2.5 min (during the linear range of enzyme reaction) at 30 °C in a 250-μl reaction medium at a substrate concentration of 1 μM cAMP in the presence of 150,000 dpm 3H-cAMP as tracer, in the absence and presence of the PDE2 inhibitor EHNA (10 μM). The incubation was stopped by the immersion of the tubes into ice immediately followed by the addition of an excess of cAMP (5 mM) and cGMP (5 mM) in the presence of EDTA (50 mM) and the non-selective PDE inhibitor IBMX (100 mM). The second step of the assay involved the conversion of the 5’AMP formed into adenosine by incubating for 20 min Crotalus atrox toxin at 30 °C. The reaction was stopped by the addition of an excess of adenosine (0.1 mM) and guanosine (0.1 mM) in the presence of EDTA (15 mM), followed by separation by ion exchange chromatography on columns with QAE-Sephadex A-25. Since adenosine is not fixed to the column, the radioactivity obtained corresponds to the final product 3H-adenosine. Total PDE activity was expressed as nmoles of cAMP hydrolysed per min per mg protein. Results are mean ± SEM, expressed as the percentage of the total cAMP-PDE activity remaining following treatment with EHNA.
Statistics
-LogEC50(M) values of the catecholamines were estimated from fitting a Hill function with variable slopes to concentration-effect curves of catecholamines from individual experiments. Data were expressed as mean ± SEM of n = number of rats. Significance between means of two groups was assessed with paired or non-paired Student’s t test (GraphPad 5.02, GraphPad Software Inc., San Diego, CA), considering P < 0.05 statistically significant. When three groups were compared, we used one-way ANOVA, preceded by a test of homogeneity of variance. A P < 0.05 of ANOVA indicated differences between groups, which were identified by multiple comparisons of means post hoc test (IBM SPSS statistics Version 21.0).
Drugs
(-)-Noradrenaline, (-)-adrenaline, (-)-isoprenaline, phenoxybenzamine, EHNA (erythro-9-(2-hydroxy-3-nonyl) adenine) and IBMX (3-isobutyl-1-methylxanthine) were purchased from Sigma (Poole, Dorset, UK); CGP20712A (2-hydroxy-5-[2-[[2-hydroxy-3-[4-[1-methyl-4-(trifluorometyl)-1H-imidazol-2-yl]phenoxy]propyl]amino]ethoxy]-benzamide) was a gift from Novartis (Basel, Switzerland). ICI118551 (1-[2,3-dihydro-7-methyl-1H-inden-4-yl)oxy-3-[(1-methylethyl)amino]-2-butanol) was purchased from Tocris (Bristol, UK); Bay 60-7550 (2-[(3,4-dimethoxyphenyl)methyl]-7-[(2R,3R)-2-hydroxy-6-phenylhexan-3-yl]-5-methyl-1H-imidazo[5,1-f][1,2,4]triazin-4-one) was a gift of Bayer (Wuppertal, Germany).
Results
EHNA and Bay 60-7550 do not increase the noradrenaline-induced sinoatrial tachycardia, mediated through β1-adrenoceptors
The experiments were carried out on spontaneously beating right atria to investigate whether phosphodiesterase PDE2 reduces the sinoatrial tachycardia caused by noradrenaline. Atrial beating was measured as described in Methods. Noradrenaline (10 nM) produced stable tachycardia of 45 ± 6 beats min−1, which did not fade (Fig. 1a). EHNA did neither change basal sinoatrial beating nor increase the responses to noradrenaline (Fig. 1b). Noradrenaline produced a smaller tachycardia of 29 ± 6 beats min−1 (P < 0.05, unpaired t test) in the presence of EHNA (Fig. 1b) than absence of EHNA (Fig. 1a). In other experiments, EHNA was administrated on top of a stable response to noradrenaline (Fig. 1c). A noradrenaline-induced tachycardia of 30 ± 7 beats min−1 was not decreased by EHNA (P = 0.24 paired t test).
The PDE2 inhibitors EHNA (10 μM) or Bay 60-7550 (BAY, 100 nM), administered 30 min before the addition of noradrenaline (NA, 10 nM), do not increase the chronotropic response to NA, mediated through β1-adrenoceptors (ICI118551 50 nM present). Data from spontaneously beating right atria from which beating rate was measured. The increases in beating rate to NA in the absence and presence of EHNA are shown in a and b, respectively. In c, EHNA was added 10 min after the NA administration. Panels d and e show the responses to NA in the presence of BAY, administered 30 min before or 10 min after the response to NA respectively. Left-hand panel and middle panels show a representative experiment and mean ± SEM data, respectively. Right-hand columns show further statistical results and number of rats with P values compared to basals over the middle columns and P values over the right columns compared to the middle columns. The experiments were terminated with isoprenaline 200 μM (ISO)
We recently reported that Bay 60-7550 did not increase the sinoatrial tachycardia elicited by noradrenaline in murine right atria (Galindo-Tovar et al. 2016). Here we investigated whether Bay 60-7550 affected the noradrenaline-induced tachycardia in the rat. Bay 60-7550 (100 nM) caused a small tachycardia of 11 ± 3 beats min−1 (P < 0.05 paired t test) in the absence (Fig. 1d), but not presence of noradrenaline (Fig. 1e), respectively. Bay 60-7550 (100 nM), administrated before or on top of the response to noradrenaline did not increase the catecholamine-induced tachycardia (Fig. 1d, e). Noradrenaline in the presence of Bay 60-7550 caused a tachycardia of 38 ± 6 beats min−1, not different from the noradrenaline-induced tachycardia in the absence of Bay 60-7550 (45 ± 6 beats min−1) (P = 0.47 unpaired t test) (Fig. 1d). Noradrenaline, administered before the addition of Bay 60-7550 on the response plateau caused a tachycardia of 36 ± 5 beats min−1 which did not differ from the control response of 45 ± 6 beats min−1 (P = 0.3 unpaired t test) (Fig. 1e).
As reported previously in mice (Galindo-Tovar et al. 2016), increasing tenfold the Bay 60-7750 concentration to 1 μM also caused moderate sinoatrial tachycardia in the presence of ICI118551 of 24 ± 6 beats min−1 (P = 0.009 paired t test) (Fig. 2b) in the rat but did not potentiate the noradrenaline-induced tachycardia (Fig. 2b). BAY added 10 min later on top of the NA response produced an increase of additional 37 ± 5 beats min−1 (Fig. 2c) which was not different from the BAY-induced tachycardia before administration of NA, P = 0.132, unpaired t test). The noradrenaline-induced tachycardia at the plateau of the BAY-evoked tachycardia was not increased compared to the noradrenaline-induced tachycardia in the absence of BAY (Fig. 2a, b). The noradrenaline-induced tachycardia was 45 ± 6 and 45 ± 4 beats min−1 in the absence or presence of BAY, respectively (Fig. 2a, b).
Bay 60-7550 (BAY, 1 μM) causes tachycardia but does not increase the chronotropic response to noradrenaline (NA, 10 nM), mediated through β1-adrenoceptors. Data from spontaneously beating right atria. The responses to NA in the absence and presence of BAY are shown in a and b, respectively (the experiments in a are those of Fig. 1a). In c, BAY was added 10 min after the NA administration. For further details, see legend to Fig. 1
The results of EHNA and Bay 60-7550 are inconsistent with a reduction by PDE2 of the noradrenaline-induced tachycardia mediated through β1-adrenoceptors.
EHNA and Bay 60-7550 do not increase the sinoatrial adrenaline-induced tachycardia
To investigate whether or not PDE2 reduces the tachycardia mediated by adrenaline through β2-and β1-adrenoceptors, we also used the PDE2 inhibitors. Adrenaline (1 μM) produced a stable tachycardia of 29 ± 5 beats min−1 which did not fade (Fig. 3a). EHNA did not change basal sinoatrial beating or increase the adrenaline-induced tachycardia (Fig. 3b, c). Adrenaline produced a tachycardia of 26 ± 3 beats min−1 in the presence of EHNA not different from the tachycardia by adrenaline alone (P = 0.65 unpaired t test) (Fig. 3b). In other experiments, EHNA was administrated on top of a stable response to adrenaline. An adrenaline-induced tachycardia 32 ± 8 beats min−1 was not changed by EHNA (Fig. 3c).
The PDE2 inhibitors EHNA (10 μM) or Bay 60-7550 (BAY, 100 nM) do not increase the chronotropic response to adrenaline (ADR), mediated through β2-adrenoceptors (CGP20712A 300 nM present). Data from spontaneously beating right atria. The sinoatrial adrenaline (1 μM)-induced tachycardia in the absence and presence of EHNA, pre-incubated for 30 min, is shown in a and b, respectively. In c, EHNA was added 10 min after the ADR administration. Panels d and e show the responses to ADR in the presence of BAY, administered 30 min before or 10 min after the response to ADR respectively. For further details, see legend to Fig. 1
Bay 60-7550 (100 nM) caused a marginal tachycardia of 17 ± 8 beats min−1 (P > 0.05 paired t test) (Fig. 3d). Bay 60-7550 (100 nM), administrated before or on top of the response to adrenaline did not change the adrenaline-induced tachycardia (Fig. 1d, e). The adrenaline-induced tachycardia in the absence and presence of Bay 60-7550 was 26 ± 3 and 16 ± 6 beats min−1, respectively (P = 0.11 unpaired t test) (Fig. 3a, d). In another experiment adrenaline produced a tachycardia of 41 ± 7 beats min−1 which was not altered by Bay 60-7550 administered 10 min later on top the plateau of the adrenaline response (Fig. 3e).
Bay 60-7550 (1 μM) caused a tachycardia of 20 ± 3 beats/min−1 in the presence of the β1-adrenoceptor-selective antagonist CGP20712A (300 nM) (Fig. 4b), not different from the tachycardia in the presence of ICI118551 (24 ± 6 beats min−1). Bay 60-7550 did not change the adrenaline-induced tachycardia (Fig. 4b). The adrenaline-induced tachycardia was 29 ± 5 and 24 ± 1 beats min−1 in the absence or presence of Bay 60-7550, respectively (P = 0.46, unpaired t test). The responses to ADR in the absence or presence of BAY did not differ (P > 0.05 unpaired t test). ADR caused a tachycardia of 21 ± 2 beats min−1 and BAY, administered on the plateau of the ADR response, elicited an additional tachycardia of 11 ± 2 beats min−1 (Fig. 4c). The adrenaline-induced tachycardia in the presence and absence of BAY was not different (P > 0.05 unpaired t test). The BAY-induced tachycardia was not different in the absence or presence of adrenaline (P > 0.05 unpaired t test).
Bay 60-7550 (BAY, 1 μM) causes tachycardia but does not increase the chronotropic response to adrenaline (ADR, 1 μM), mediated through rat β2-adrenoceptors (Kaumann 1986, CGP20712A 300 nM present). Data from spontaneously beating right atria. The responses to ADR 1 μM in the absence and presence of BAY are shown a and b, respectively (the experiments in a are those of Fig. 3a). In c, BAY was added 10 min after ADR was administered. For further details, see legend to Fig. 1
Since part of the response to 1 μM adrenaline is mediated through β2-adrenoceptors (Kaumann 1986), the results with EHNA and Bay 60-7550 appear inconsistent with a control by PDE2.
Isoprenaline infusion increases PDE2 hydrolytic activity in left ventricle
To investigate whether the enzymatic activity of PDE2 is enhanced by chronic stimulation with a catecholamine, we infused rats with isoprenaline. The 3-day infusion of isoprenaline caused a threefold increase of the hydrolytic activity of PDE2 that can be inhibited by EHNA in left ventricle (Fig. 5). P < 0.0048 (unpaired t test) compared to rats not infused with isoprenaline. The PDE2 activity of right ventricle was not increased (P = 0.27).
Isoprenaline infusion (ISO) induces the hydrolysis of cAMP by PDE2 in left ventricle (LV) (P = 0.0048, unpaired t test), presumably mainly mediated through β1-adrenoceptors, but not right ventricle (RV). The percentage of PDE2 activity was calculated from the activity obtained in the absence and presence of EHNA (10 μM). Results are mean ± SEM of PDE2 activity. Number of rats in columns
Sinoatrial β-adrenoceptor desensitization by infusions of isoprenaline is not reversed by PDE2 inhibitors
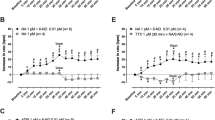
The 3-day infusion of isoprenaline did not change the –log EC50 of noradrenaline-induced increases of sinoatrial beating (7.12 ± 0.33 n = 5 compared to 7.16 ± 0.06 n = 6 in untreated rats). The maximum response (Emax) to noradrenaline was reduced to 296 ± 15 beats min−1 compared to 415 ± 4 beats min−1 in normal rats (P < 0.05, unpaired t test) (Fig. 6a, c). The Emax to adrenaline was 285 ± 8 n = 6 beats min−1 after isoprenaline infusion compared to 320 ± 20 n = 6 beats min−1 (P = 0.1046, unpaired t test) in normal rats (Fig. 6b, d). EHNA and Bay 60-7550 did not change the chronotropic responses to the catecholamines in normal rats or rats infused with isoprenaline (Fig. 6a–d).
Long-lasting stimulation of sinoatrial β1- or β2-adrenoceptors causes desensitization unaffected by PDE2 inhibitors. Data from spontaneously beating right atria. Concentration-effect curves for the increase of sinoatrial beating rate by noradrenaline (mediated through β1-adrenoceptors in the presence of ICI118551, panels a and c) and adrenaline (mediated through β2-adrenoceptors in the presence of CGP20712A, panels b and d) are shown at left and right panels, respectively. Data from untreated rats and rats infused with isoprenaline are shown on top and bottom panels, respectively. Curves in the absence and presence of EHNA (1 μM) or Bay 60-7550 (BAY, 100 nM) are shown in black, red, and blue, respectively. The B at the bottom of the ordinate is basal beating rate. I/T stands for PDE2 inhibitor (I) over time matched control (T)
Left ventricular force responses to noradrenaline, reduced by isoprenaline-infusions, are not improved by PDE2 inhibitors
The –logEC50 was reduced to 5.9 ± 0.1 (n = 8) in left ventricular papillary muscles of rats infused with isoprenaline compared to 7.2 ± 0.06 (n = 9) (P < 0.05 unpaired t test) in normal rats (Fig. 7a, c). The force responses to noradrenaline in papillary muscles were reduced by infusion of isoprenaline through β1-adrenoceptor desensitization (Fig. 7e, g). The inotropic responses to Ca2+ (9 mM) were larger than rats infused with isoprenaline than non-isoprenaline rats (Fig. 7e, f and g, h). For unknown reasons, the responses to Ca2+ were larger than the response to isoprenaline in the presence PDE inhibitors. Neither EHNA nor Bay 60-7550 increased the response to noradrenaline in normal rats or rats infused with isoprenaline (Fig. 7a, c). However, for unknown reasons, Bay 60-7550 caused a small right-ward shift of the concentration-effect curve of noradrenaline (P < 0.05 ANOVA) (Fig. 7a).
Long-lasting stimulation of β1- or β2-adrenoceptors of left ventricular papillary muscle causes desensitization unaffected by the PDE2 inhibitors EHNA (1 μM) or Bay 60-7550 (BAY, 100 nM). Concentration-effect curves for the contractile effects of noradrenaline (mediated through β1-adrenoceptors in the presence of ICI118551, panels a and c) and adrenaline (mediated through β2-adrenoceptors in the presence of CGP20712A, panels b and d) are shown at left and right panels, respectively. Data (Mean ± SEM) from untreated rats and rats infused with isoprenaline (ISO) are shown on top and second from top panels, respectively. Curves in the absence and presence of EHNA or BAY are shown in black, red, and blue, respectively. Force responses in mN to ISO, 200 μM, open columns) and Ca2+ 9 mM (black columns) after the highest concentrations of noradrenaline (panels e and g) or adrenaline (panels f and h) are shown from untreated rats (e, f) and rats chronically infused with ISO (g, h). Responses to Ca2+ tended to be greater than the corresponding responses to ISO. Actual P values are shown between some black and white columns. I/T stands for PDE2 inhibitor (I) over time matched control (T)
Small inotropic responses to adrenaline in papillary muscles from normal rats were virtually abolished in rats that underwent isoprenaline infusions (Fig. 7b, d). As observed with noradrenaline, the inotropic responses to Ca2+ (9 mM) tended to be larger than the responses to isoprenaline in papillary muscles from both normal rats and rats infused with isoprenaline (Fig. 7f, h). Neither EHNA nor Bay 60-7550 increased the response to adrenaline in untreated rats or isoprenaline-infused rats (Fig. 7b, d).
Taken together, these experiments are inconsistent with an effect of PDE2 on the positive inotropic effect of catecholamines, not even when left ventricular PDE2 activity is enhanced after adrenergic overstimulation.
Force responses to salbutamol of left ventricular papillary muscles are not increased by EHNA and vanish after isoprenaline infusion
Due to the small effects of low adrenaline concentrations, we could not estimate the contribution of β2-adrenoceptors from the responses to adrenaline of Fig. 7b. Instead, we investigated the effects of the β2-adrenoceptor-selective partial agonist salbutamol. Salbutamol tended to cause small increases of force in left ventricular papillary muscles from normal rats in the presence of CGP20712A (Fig. 8b (P = 0.21 paired t test), Fig. 8d (P = 0.0021, paired t test)). The response to salbutamol was not observed in tissues obtained from rats infused with isoprenaline (Fig. 8a, c). EHNA did not increase the salbutamol responses in papillary muscles from normal rats (Fig. 8b) and rats infused with isoprenaline (Fig. 8a).
Salbutamol tends to increase force of left ventricular papillary muscles through β2-adrenoceptors from untreated rats (b, d) but not from isoprenaline(ISO)-infused rats (a, c). No effect of EHNA (a, b). Left-hand tracings are representative experiments. Right-hand columns show data as mean ± SEM. Papillary muscles of a (ISO-infused) and b (untreated) set up as pairs in one organ bath, muscles c (ISO-infused) and d (untreated) in another organ bath. Experiments carried out in the presence of CGP20712A (300 nM). Numbers of rats are shown in the right-hand columns. (TMC) Time match control
Right ventricular force responses to noradrenaline, reduced by isoprenaline-infusions, are not improved by PDE2 inhibitors
The inotropic potency of noradrenaline in right ventricular strips was reduced by the isoprenaline infusion. The –logEC50 decreased to 6.1 ± 0.2 (n = 5) in rats infused with isoprenaline compared to 6.7 ± 0.1 (n = 8) (P < 0.05 unpaired test) in normal rats (Fig. 9a, c). The inotropic responses to isoprenaline were smaller than the responses to Ca2+ in ventricular strips from both normal rats and rats infused with isoprenaline (Fig. 9e, g).
Long-lasting stimulation of β1- or β2-adrenoceptors of right ventricular strips causes desensitization unaffected by the PDE2 inhibitors EHNA (1 μM) or Bay 60-7550 (BAY, 100 nM). Concentration-effect curves for the contractile effects of noradrenaline, mediated through β1-adrenoceptors (In the presence of ICI118551, panels a and c) and adrenaline, mediated in part through β2-adrenoceptors (In the presence of CGP20712A, panels b and d) are shown at left and right panels, respectively. Data (Mean ± SEM) from untreated rats and rats chronically infused with isoprenaline (ISO) are shown on top and second from top panels, respectively. Curves in the absence and presence of EHNA or BAY are shown in black, red, and blue, respectively. Force responses in mN to ISO (200 μM, open columns) and Ca2+ 9 mM (black columns) after the highest concentrations to noradrenaline (panels e and g) or adrenaline (panels f and h) are shown from untreated rats (e, f) and rats chronically infused with ISO (g, h). Actual P values are shown between some black and white columns. I/T stands for PDE2 inhibitor (I) over time matched control (T)
Neither EHNA nor Bay 60-7550 increased the responses to noradrenaline in untreated rats and rats infused with isoprenaline (Fig. 9a, c).
Marginal inotropic responses to adrenaline in right ventricular strips from normal rats were abolished in rats with isoprenaline infusions (Fig. 9b, d). The inotropic responses to isoprenaline were smaller than the responses to Ca2+ in ventricular strips from both normal rats and rats infused with isoprenaline (Fig. 9f, h). Neither EHNA nor Bay 60-7550 increased the response to adrenaline in untreated rats or rats infused with isoprenaline (Fig. 9b, d). However, for unknown reasons Bay 60-7550 produced a small right-ward shift of the concentration-effect curves for noradrenaline (P < 0.05 ANOVA) (Fig. 9a). As in left ventricle, these experiments are inconsistent with a control of PDE2 of right ventricular β-adrenoceptors-mediated force responses, not even after adrenergic overstimulation.
Discussion
Our experiments are inconsistent with a decrease by phosphodiesterase PDE2 of the chronotropic and inotropic effects mediated through β-adrenoceptors. A 3-day exposure to isoprenaline increases the hydrolysis of cAMP by PDE2 in rat left ventricle. Surprisingly however, the inotropic effects of noradrenaline and adrenaline were not enhanced by the PDE2 inhibitors. These findings raise the question about the relevance of the enhanced ventricular PDE2 activity after adrenergic overstimulation.
Is the sinoatrial tachycardia, mediated through Gs-coupled receptors, modulated by phosphodiesterases?
Consistent with recent work on mice (Galindo-Tovar et al. 2016), the PDE2 inhibitors did not enhance the noradrenaline-induced sinoatrial tachycardia in normal rats. After the chronic infusion of isoprenaline, the maximum effect of the noradrenaline-induced sinoatrial tachycardia mediated through β1-adrenoceptors, was markedly reduced (Fig. 6) but not affected by the PDE2 inhibitors. Therefore, an influence of PDE2 is doubtful, even after adrenergic hyper-stimulation. PDE2 is expressed in murine sinoatrial cells (Hua et al. 2012) but unfortunately it is still unknown whether the enzyme activity is enhanced by adrenergic hyper-stimulation as found in left ventricle.
Inhibitors of PDE3 and PDE4 do not increase catecholamine-induced sinoatrial tachycardia catecholamines in the mouse, rat and rabbit (Kaumann 2011). The lack of a reduction by PDE2, PDE3 and PDE4 of the tachycardia mediated through β-adrenoceptors (which involves the participation of cAMP, Fig. 10), suggests that these three phosphodiesterases do not have access to a relevant physiological cAMP pool (Fig. 10). The sinoatrial tachycardia in the rat probably precludes an effect of any cAMP-hydrolysing phosphodiesterase at least during our 30 min observation period. It probably would be unlikely that a PDE enzyme or even several PDE enzymes could maintain fast associations and dissociations with and from cAMP to allow a perfect equilibrium chronotropic response for 30 min, as observed on the cAMP-dependent response i.e. tachycardia for 30 min. It is plausible that efflux of cAMP of the cell is mediated by cyclic nucleotide transporters. These are multidrug resistance proteins (MRP) that belong to the ATP-binding cassette (ABC) transporter superfamily C. MRP4 aka ABCC4, MRP5 aka ABCC5 and MRP8 aka ABCC11 actively extrude cAMP and cGMP from the cell (Kruh and Belinsky 2003 Oncogene 22: 7537–7552). MRP4 mediates cAMP efflux from cardiomyocytes. In cardiomyocytes from MRP4-deficient mice, cAMP formation and contractility is increased and cardiac hypertrophy produced (Sassi et al. 2012). Due to this evidence, stable tachycardia could conceivably be maintained without PDE activity.
Scheme for two classes of cAMP compartments in a rat sinoatrial cell containing both β1 and β2-adrenoceptors (Adapted from Kaumann (2011) and Galindo-Tovar et al. (2016)). Cyclic AMP increases sinoatrial beating rate by augmenting the hyperpolarization-activated current If both directly and through PKA-catalyzed phosphorylation (Liao et al. 2010). PKA also phosphorylates Ik channels, L-type Ca2+ channels (ICa,L), phospholamban (PLB) and possibly ryanodine channels (RyR2). Phosphodiesterases PDE3 and PDE4 are phosphorylated by PKA (Smith et al. 1991; MacKenzie et al. 2002). Ca2+ movements in the cytoplasm and sarcoplasmic reticulum (SR) are depicted in red and reviewed in detail elsewhere (Kaumann 2011). Two hypothetical classes of cAMP compartments exist in the sinoatrial cell. In one compartment, cAMP is formed by Ca2+-stimulatable adenylyl cyclases AC1 or AC8 and hydrolysed to 5’AMP (yellow arrow) by PDE2, PDE3 and/or PDE4. The cAMP available from its formation and degradation tonically maintains basal sinoatrial beating. In other compartments, the cyclic AMP generated by Ca2+-inhibitable AC5/6 through stimulation of the Gs protein-coupled β1-adrenoceptor and β2-adrenoceptor appears unaffected by PDE2, PDE3 and PDE4 and causes the tachycardia elicited by noradrenaline (NA) and adrenaline (ADR)
Tonic decrease of basal sinoatrial heart rate by PDE2
A high cGMP concentration (5 μM) has been reported to decrease ICa,L current in human atrial myocytes, an effect completely reversed by EHNA (Vandecasteele et al. 2001). Since the hydrolysis of cAMP by PDE2 is markedly enhanced by cGMP (Bender and Beavo 2006), Vandecasteele et al. (2001) suggested a tonic modulation of the ICa,L current by PDE2. The PDE2 inhibitor EHNA produces a moderate tachycardia in murine sinoatrial myocytes at room temperature (Hua et al. 2012), but we did not find that EHNA causes tachycardia at 37 °C in beating atria from mice (Galindo-Tovar et al. 2016) and rats (this work). However, even at room temperature (21–22 °C), in rabbit spontaneously beating sinoatrial node cells Vinogradova et al. (2008) found a very small increase of spontaneous beating rate with EHNA. The chronotropic effect of PDE2 inhibition with high concentrations of Bay 60-7550 on basal beating is remarkably similar in 3 species, 24 beats min−1 both in mice (Galindo-Tovar et al. 2016) and rats (this paper) and 22 beats min−1 in beagle dogs (Vettel et al. 2017). The work of Vinogradova et al. (2008) and Hua et al. (2012), together with that of (Galindo-Tovar et al. 2016), this paper and (Vettel et al. 2017) showing that the PDE2 inhibitor Bay 60-7550 caused sinoatrial tachycardia, are consistent with a partial tonic control by PDE2 of basal sinoatrial beating (Fig. 10).
PDE2 does not reduce increases of ventricular force mediated through β1- and β2-adrenoceptors
The inotropic effects of noradrenaline, mediated through β1-adrenoceptors, were not increased by the PDE2 inhibitors in left ventricular papillary muscles and right ventricular strips. The chronic infusion of isoprenaline caused desensitization, as shown by rightward shifts of the inotropic concentration-effect curves of noradrenaline. Neither EHNA nor Bay 60-7550 enhanced the reduced effects of noradrenaline in the left and right ventricular preparations from isoprenaline-infused rats, inconsistent with a control by PDE2. The results are in contrast to the results of Mehel et al. (2013) with rats after a 4-day infusion of isoprenaline. These authors reported that the ICa,L current, Ca-transients and sarcomere shortening responses to isoprenaline were increased by Bay 60-7550 in ventricular cardiomyocytes from rats chronically infused with isoprenaline. They attributed their findings to the increase in PDE2 expression and hydrolytic activity caused by the adrenergic hyper-function. Furthermore, more recently Vettel et al. (2017) reported that after chronic isoprenaline infusion in mice Bay 60-7550 reversed β-adrenoceptor-mediated inotropy and chronotropy. Although we found the increase of the hydrolytic activity in left ventricle after the 3-day isoprenaline infusion, our results are inconsistent with a reduction by PDE2 of the inotropic and chonotropic effects of noradrenaline, mediated through β1-adrenoceptors.
We did not detect possible β2-adrenoceptor-mediated effects of adrenaline in left ventricular papillary muscles and right ventricular strips in the absence or presence of the PDE2 inhibitors, not even after the infusion of isoprenaline. In agreement with findings of Hall et al. (1990) on human atrium, we found small or marginal responses to salbutamol on rat papillary muscles that were resistant to blockade by β1-selectivive CGP20712A and probably mediated through β2-adrenoceptors. The slow kinetic of the responses to ISO in Fig. 8a, c are probably due to the dissociation of CGP20712A from the desensitized β-adrenoceptors. The fast kinetics of ISO in Fig. 8b, d are due to the contribution of both β1- and β2-adrenoceptors in the absence of desensitization.
A report of Soler et al. (2015) claimed that salbutamol increased the force of left ventricular papillary muscles of the rat in the presence but not absence of EHNA. For unknown reasons we were, however, unable to reproduce the results of Soler et al. (2015). Our results showed that salbutamol (10 μM) caused small force increases in the absence of EHNA in left ventricular papillary muscles from normal rats but not from rats infused with isoprenaline (Fig. 6). Furthermore, the effects of salbutamol were not enhanced by EHNA. The response to salbutamol was abolished in papillary muscles from rats infused with isoprenaline but not restored by EHNA.
Taken together, the above results rule out a reduction by PDE2 of the ventricular inotropic effects mediated through rat β1- and β2-adrenoceptors.
Uncertainties about the function of PDE2
Mehel et al. (2013) postulated that the upregulation of PDE2 after clinical (heart failure) and experimental catecholamine stress reduces β-adrenoceptor responsiveness, arrhythmias and hypertrophy. They observed that PDE2 overexpression decreases the isoprenaline-induced stimulation of ICa,L in cardiomyocytes, an effect reversed by Bay 60-7550. According to these authors, one would expect that the inhibition of PDE2 would partially restore the reduced β-responsiveness after the adrenergic hyper-function which, however, our experiments did not reveal in the rat model. Therefore, the function of the upregulated PDE2 appears uncertain. Furthermore, there is also controversy as to the function of PDE2 with experimental catecholamine-induced hypertrophy or hypertrophy due to transverse aortic constriction (Zoccarato et al. 2015). While Mehel et al. (2013) proposed that the PDE2 upregulation protects against hypertrophy, Zoccarato et al. (2015) demonstrated that this leads to hypertrophy. Conversely, inhibition of PDE2 mediates anti-hypertrophic effects (Zoccarato et al. 2015) through the formation of a cAMP pool and activation of PKAII that phosphorylates the pro-hypertrophic transcription factor nuclear factor of activated T cells, blocking its nuclear translocation. These discrepancies are under intensive debate (Wagner et al. 2016 vs Zoccarato et al. 2016) but remain unresolved.
Conclusions
Catecholamines cause stable sinoatrial tachycardia, unaffected by PDE2, not even after adrenergic hyper-function caused by the isoprenaline infusion. Since PDE2, PDE3 and PDE4 do not reduce the tachycardia evoked by catecholamines, the tachycardia probably rules out access of any phosphodiesterase to the cAMP at the receptor compartment. However, PDE2 contributes together with PDE3 and PDE4, to reduce basal sinoatrial beating in another cAMP compartment (Fig. 10). The ventricular inotropic effects, mediated through β1- and β2-adrenoceptors, are not reduced by PDE2 not even after the isoprenaline infusion which increased threefold the left ventricular hydrolytic activity of PDE2. The relevance of these results is currently under investigation in human myocardium.
References
Bender AT, Beavo JA (2006) Cyclic nucleotide phosphodiesterases: molecular regulation to clinical use. Pharmacol Rev 58:488–520
Bers DM (2002) Cardiac excitation-contraction coupling. Nature 415:198–205. https://doi.org/10.1124/pr.58.3.5
Christ T, Galindo-Tovar A, Thoms M, Ravens U, Kauman AJ (2009) Inotropy and L-type Ca2+ current, activated by beta1- and beta2-adrenoceptors, are differently controlled by phosphodiesterases 3 and 4 in rat heart. Br J Pharmacol 156:62–83. https://doi.org/10.1111/j.1476-5381.2008.00015.x
Di Benedetto G, Zoccarato A, Lissandron V, Terrin A, Li X, Houslay MD et al (2008) Protein kinase A type I and type II define distinct intracellular signalling compartments. Circ Res 103:836–844. https://doi.org/10.1161/CIRCRESAHA.108.174813
Galindo-Tovar A, Kaumann AJ (2008) Phosphodiesterase-4 blunts inotropism and arrhythmias but not sinoatrial tachycardia of adrenaline mediated through mouse cardiac β1-adrenoceptors. Br J Pharmacol 153:710–720. https://doi.org/10.1038/sj.bjp.0707631
Galindo-Tovar A, Vargas ML, Kaumann AJ (2016) Inhibitors of phosphodiesterases PDE2, PDE3, and PDE4 do not increase sinoatrial tachycardia of noradrenaline and prostaglandin PGE1 in mice. Naunyn Schmiedeberg's Arch Pharmacol 389:177–186. https://doi.org/10.1007/s00210-015-1178-2
Gille E, Lemoine H, Ehle B, Kaumann AJ (1985) The affinity of (-)-propranolol for β1- and β2-adrenoceptors of human heart. Differential antagonism of the positive inotropic effects of adenylate cyclase stimulation by (-)-nodradrenaline and (-)-adrenaline. Naunyn Schmiedeberg's Arch Pharmacol 331:60–70
Hall JA, Kaumann AJ, Brown MJ (1990) Selective beta 1-adrenoceptor blockade enhances positive inotropic responses to endogenous catecholamines mediated through beta 2-adrenoceptors in human atrial myocardium. Circ Res 66:1610–1623
Hua R, Adamcyk A, Robbins C, Ray G, Rose RA (2012) Distinct patterns of constitutive phosphodiesterase activity in mouse sinoatrial node and atrial myocardium. PLoS 7:e47652. https://doi.org/10.1371/journal.pone.0047652
Kaumann AJ (1986) The β1-adrenoceptor antagonist CGP 20712A unmasks β2-adrenoceptors activated by (-)-adrenaline in rat sinoatrial node. Naunyn Schmiedeberg's Arch Pharmacol 332:406–409
Kaumann AJ (2011) Phosphodiesterases reduce spontaneous sinoatrial beating but do not the “fight or flight” tachycardia elicited by agonists through Gs protein-coupled receptors. Trends Pharmacol Sci 32:277–283. https://doi.org/10.1016/j.tips.2011.03.003
Kaumann AJ, Galindo-Tovar A, Escudero E, Vargas ML (2009) Phosphodiesterases do not limit β1-adrenoceptor-mediated sinoatrial tachycardia: evidence with PDE3 and PDE4 in rabbits and PDE1-5 in rats. Naunyn Schmiedeberg's Arch Pharmacol 380:421–430. https://doi.org/10.1007/s00210-009-0445-5
Keravis T, Thaseldar-Roumié R, Lugnier C (2005). Assessment of phosphodiesterase isoenzyme contribution in cell and tissue extracts in: phosphodiesterase methods and protocols. Ed C Lugnier. Humana Press Totowa, New Jersey, pgs 63–74. https://doi.org/10.1385/1-59259-839-0:063
Kruh GD, Belinsky MG (2003) The MRP family of drug pumps. Oncogene 22:7537–7552. https://doi.org/10.1038/sj.onc.1206953
Liao Z, Lockhead D, Larson ED, Proenza C (2010) Phosphorylation and modulation of hyperpolarization-activated HCN4 channels by protein kinase A in the mouse sinoatrial node. J Gen Physiol 136:247–258. https://doi.org/10.1085/jgp.201010488
MacKenzie SJ, Baillie GS, MacPhee I, MacKenzie C, Seamons R, McSorley T et al (2002) Long PDE4 cAMP specific phosphodiesterases are activated by protein kinase A-mediated phosphorylation of a single serine in upstream conserved region 1 (UCR1). Br J Pharmacol 136:421–433. https://doi.org/10.1038/sj.bjp.0704743
Martins TJ, Mumby MC, Beavo JA (1982) Purification and characterization of acyclic GMP-stimulated cyclic nucleotide phosphodiesterase from bovine tissues. J Biol Chem 257:1973–1979
Mehel H, Emons J, Vettel C, Wittkörper K, Seppelt D, Dewenter M et al (2013) Phosphodiesterase-2 is up-regulated in human failing hearts and blunts β-adrenergic responses in cardiomyocytes. JACC 62:1596–1606. https://doi.org/10.1016/j.jacc.2013.05.057
Mongillo M, Tochetti CG, Terrin A, Lissandron V, Cheung YF, Dostmann WR et al (2006) Compartamentalized phosphodiesterase-2 activity blunts beta adrenergic cardiac inotropy via an NO/cGMP-dependent pathway. Circ Res 98:226–234. https://doi.org/10.1161/01.RES.0000200178.34179.93
Patrucco E, Albergine MS, Santana LF, Beavo JA (2010) Phosphodiesterase 8A (PDE8A) regulates excitation-contraction coupling in ventricular myocytes. J Mol Cell Cardiol 49:330–333. https://doi.org/10.1016/j.yjmcc.2010.03.016
Sassi Y, Abi-Gerges A, Fauconnier J, Mougenot N, Reiken S, Haghighi K, Kranias EG, Marks AR, Lacampagne A, Engelhardt S, Hatem SN, Lompre AM, Hulot JS (2012) Regulation of cAMP homeostasis by the efflux protein MRP4 in cardiac myocytes. FASEB J 26:1009–1017. https://doi.org/10.1096/fj.11-194027
Smith CJ, Vasta V, Degerman E, Belfrage P, Manganiello VC (1991) Hormone sensitive cyclic GMP-inhibited cyclic AMP phosphodiesterase PDE3 in rat adipocytes. J Biol Chem 266:13385–13390
Soler F, Fernández-Belda F, Pérez-Schindler J, Handschin C, Fuente T, Hernandez-Cascales J (2015) PDE2 activity differs in right and left rat ventricular myocardium and differentially regulates β2 adrenoceptor-mediated effects. Exp Biol Med (Maywood) 240(9):1205–1213. https://doi.org/10.1177/1535370214560969
Stangherlin A, Gesellchen F, Zoccarato A, Terrin A, Fields LA, Berrera M et al (2011) cGMP signals modulate cAMP levels in a compartment-specific manner to regulate catecholamine-dependent signaling in cardiac myocytes. Circ Res 108:929–939. https://doi.org/10.1161/CIRCRESAHA.110.230698
Thompson WJ, Appleman MM (1971) Characterization of cyclic nucleotide phosphodiesterases of rat tissues. J Biol Chem 246:3145–3150
Vandecasteele G, Verde I, Rücker-Martin C, Donzeau-Gouge P, Fischmeister R (2001) Cyclic GMP regulation of the L-type Ca2+ channel current in human atrial myocytes. J Physiol 533:329–340
Verde I, Vandecasteele G, Lezoualc’h F, Fischmeister R (1999) Characterization of the cyclic nucleotide phosphodiesterase subtypes involved in the regulation of the Ca2+ current in rat ventricular myocytes. Br J Pharmacol 127:65–74. https://doi.org/10.1038/sj.bjp.0702506
Vettel C, Lindner M, Dewenter M, Lorenz K, Schanbacher C, Riedel M et al (2017) Phosphodiesterase 2 protects against catecholamine-induced arrhythmia and preserves contractile function after myocardial infarction. Circ Res 120:120–132. https://doi.org/10.1161/CIRCRESAHA.116.310069
Vinogradova TM, Lyashkov AE, Zhu W, Ruknudin AM, Sirenko S, Yang D, Deo S, Barlow M, Johnson S, Caffrey JL, Zhou YY, Xiao RP, Cheng H, Stern MD, Maltsev VA, Lakatta EG (2006) High basal protein kinase A-dependent phosphorylation drives rhythmic internal Ca2+ store oscillations and spontaneous beating of cardiac pacemaker cells. Circ Res 98:505–514. https://doi.org/10.1161/01.RES.0000204575.94040.d1
Vinogradova TM, Sirenko S, Lyashkov AE, Younes A, Li Y, Zhu W, Yang D, Ruknudin AM, Spurgeon H, Lakatta EG (2008) Constitutive phosphodiesterase activity restricts spontaneous beating rate of cardiac pacemaker by suppressing local Ca2+ releases. Circ Res 102:761–769. https://doi.org/10.1161/CIRCRESAHA.107.161679
Wagner M, Mehel H, Fischmeister R, El-Armouche A (2016) Phosphodiesterase 2: anti-adrenergic friend or hypertrophic foe in heart disease? Naunyn Schmiedeberg's Arch Pharmacol 389:1139–1141. https://doi.org/10.1007/s00210-016-1289-4
Yanaka N, Kurosawa Y, Minami K, Kawai E, Omori K (2003) cGMP-phosphodiesterase activity is upregulated in response to pressure overload of rat ventricles. Biosci Biotechnol Biochem 67:973–979. https://doi.org/10.1271/bbb.67.973
Zoccarato A, Surdo NC, Aronsen JM, Fields LA, Mancuso L, Dodoni G, Stangherlin A, Livie C, Jiang H, Sin YY, Gesellchen F, Terrin A, Baillie GS, Nicklin SA, Graham D, Szabo-Fresnais N, Krall J, Vandeput F, Movsesian M, Furlan L, Corsetti V, Hamilton G, Lefkimmiatis K, Sjaastad I, Zaccolo M (2015) Cardiac hypertrophy is inhibited by a local pool of cAMP regulated by phophodiesterase 2. Circ Res 117:707–719. https://doi.org/10.1161/CIRCRESAHA.114.305892
Zoccarato A, Fields LH, Zaccolo M (2016). Response to Wagner et al.: phopsdiesterase-2-anti-adrenergic friend or foe of heart disease? Naunyn-Schmiedeberg’s Arch Pharmacol 389:1144–1145. https://doi.org/10.1007/s00210-016-1301-z
Acknowledgments
We thank Juana María Hidalgo Cespedes for technical assistance and Antonio Piñuela for constructing Fig. 10. This work was supported by a grant of the Ministerio de Ciencia e Innovación (SAF/FEDER 2013-47076) and grant Plan Propio de Investigación PMAFI/07/14 of the Universidad Católica San Antonio de Murcia.
Author information
Authors and Affiliations
Contributions
A.G., ML.V. and A.K. performed the research. A.G., ML.V. and A.K. designed the research study. A.G., ML.V. and A.K. analyzed the data. A.K. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
None.
Rights and permissions
About this article
Cite this article
Galindo-Tovar, A., Vargas, M.L. & Kaumann, A. Phosphodiesterase PDE2 activity, increased by isoprenaline, does not reduce β-adrenoceptor-mediated chronotropic and inotropic effects in rat heart. Naunyn-Schmiedeberg's Arch Pharmacol 391, 571–585 (2018). https://doi.org/10.1007/s00210-018-1480-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-1480-x