Abstract
Tributyltin (TBT) is a widespread environmental contaminant that is present in human blood and other tissues. It has been shown to disrupt the immune function of human natural killer (NK) cells and to alter the secretion of a number of pro-inflammatory cytokines from immune cells. Secretion of both interleukin 1β (IL-1β) and interleukin 6 (IL-6) from human lymphocytes can be increased dependent upon the level of TBT exposure. This study shows that the TBT-induced increases in secretion of both cytokines are due to TBT-induced increases in the synthesis of these proteins and not simply because of the release of pre-existing cytokine. Furthermore, the data indicate that these TBT-induced increases in IL-1β and IL-6 synthesis require MAP kinase signaling pathways. Additionally, elevated synthesis of IL-1β and IL-6 seen at the highest exposures to TBT (200, 200, 50 nM) were accompanied by increases in the mRNA for these cytokines. TBT-induced increases in IL-1β and IL-6 mRNAs were also shown to be dependent on MAP kinase signaling. The study suggests that TBT has the capacity to increase immune cell production of these 2 important pro-inflammatory cytokines and that this increase is in part explained by increased mRNA for the cytokines.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Due to its multitude of uses as a biocide, tributyltin (TBT) widely contaminates the environment and has been found in human tissues including blood (as high as 261 nM in blood) (Antizar-Ladislao 2008; Kannan et al. 1995, 1999; Whalen et al. 1999). It was used as an antifouling agent in paints applied to the hulls of ships (Loganathan et al. 2001; Loganathan 2016). Its use was banned by the International Convention on the Control of Harmful Antifouling Systems on Ships in 2008 (Gipperth 2009), but it is still used by countries that are not part of the convention. It is also found in some household goods such as siliconized-paper baking parchments and shower curtains (Yamada et al. 1993). TBT exposure can cause increased incidences of tumors and thymic atrophy in mammals (Wester et al. 1990; Snoeij et al. 1988) and decrease the ability of human natural killer (NK) lymphocytes to destroy tumor cells (Dudimah et al. 2007). Additionally, TBT has been shown to alter the secretion of several important pro-inflammatory cytokines from human lymphocytes. These include interferon gamma (IFNγ), tumor necrosis factor alpha (TNFα), IL-1β, and IL-6 (Hurt et al. 2013; Lawrence et al. 2015; Brown and Whalen 2015; Brown et al. 2018).
IL-1β regulates the inflammatory response and promotes cellular growth, and tissue repair (Apte and Voronov 2002; Arend 2002; Dinarello 1996, 2005, 2009). It is produced by monocytes, macrophages, T cells, NK cells, neutrophils, keratinocytes, and fibroblasts (Guma et al. 2009; Burger and Dayer 2002; Dinarello 2005; Apte and Voronov 2002; Voronov et al. 2002). IL-1β can act as a stimulator of the production of other pro-inflammatory cytokines, including IFNγ and IL-6 (Tosato and Jones 1990; Cooper et al. 2001; Luo et al. 2003). When IL-1β levels are elevated inappropriately it contributes to chronic inflammation (Dinarello 2011). Elevated IL-1β is associated with a number of diseases including multiple sclerosis, rheumatoid arthritis, and cancer (Rossi et al. 2012; Choy and Panayi 2001; Arlt et al. 2002; Elaraj et al. 2006; Jin et al. 1997; Lewis and Varghese 2006; Muerkoster et al. 2006). A drug that decreases IL-1β levels is an approved treatment for rheumatoid arthritis (Dinarello 2011).
Like IL-1β, IL-6 is a pro-inflammatory cytokine (22–27 kDA) (Kishimoto 1989) produced by a variety of cells types including lymphocytes, monocytes/macrophages, and granulocytes (Akira et al. 1993; Li and He 2006; Hall et al. 2010; Riedemann et al. 2004; Zimmermann et al. 2015, 2016). As with IL-1β, inappropriately elevated levels of IL-6 can lead to chronic inflammation (Tanaka et al. 2014) and increased IL-6 activity contributes to chronic inflammatory diseases such as colon cancer, colitis, rheumatoid arthritis, systemic lupus erythematosus, psoriasis, and Crohn’s disease (Gabay 2006; Atreya et al. 2000; Hurst et al. 2001). An antibody to the IL-6 receptor, tocilizumab, has been approved to treat rheumatoid arthritis (Tanaka et al. 2014).
TBT has been shown to alter the secretion of both IL-1β and IL-6 from human lymphocytes (Brown and Whalen 2015; Brown et al. 2018). The current study examines whether these alterations in secretion are due to changes in the lymphocytes’ synthesis of these cytokines or if it is simply due to changes in the secretion of pre-existing cytokine. To address this, both secreted and intracellular levels of each cytokine were measured from the same cells in response to exposures to TBT. TBT concentrations that caused increases in cytokine synthesis were then examined to determine if this reflected an increase in the mRNA for the cytokine. Additionally, signaling pathways known to be utilized in cellular production of each of the cytokines were examined for potential roles in any TBT-induced increases in either cytokine. These included, mitogen activated protein kinases (MAPKs) and nuclear factor kappa B (NFκB) (Gaestel et al. 2009).
Materials and methods
Preparation of MD-PBMCs
PBMCs were isolated from Leukocyte filters (PALL- RCPL or RC2D) obtained from the Red Cross Blood Bank Facility (Nashville, TN, USA) as described in Meyer et al. 2005. Leukocytes were retrieved from the filters by back-flushing them with sterile PBS containing 5 mM disodium EDTA and 2.5% [w/v] sucrose and collecting the eluent. The eluent was layered onto Ficoll-Hypaque (1.077 g/mL) and centrifuged at 1200g for 30–50 min. Mononuclear cells were collected and washed with PBS (500 g, 10 min). Following washing, the cells were layered on bovine calf serum for platelet removal. Monocyte-depleted PBMCs were prepared by incubating the cells in glass Petri dishes (150 × 15 mm) at 37 °C and air/CO2, 19:1 for 1.5 h. This cell preparation is referred to as MD-PBMCS cells.
Chemical preparation
Tributyltin chloride (TBT) was purchased from Sigma-Aldrich (96%) (St. Louis, MO). Desired concentrations of TBT were prepared by dilution of the stock into cell culture media. TBT was a neat standard, dissolved initially in deionized water to give a 1 mM solution.
Inhibitor preparation
Enzyme inhibitors were purchased from Fisher Scientific (Pittsburgh, PA). The stock solution for each inhibitor was a 50 mM solution in dimethylsulfoxide (DMSO). Capase-1 inhibitor II (50 µM), MEK 1/2 pathway inhibitor (PD98059) (50 µM), p38 inhibitor (SB202190) (25 µM), JNK pathway inhibitor (JNK X BI78D3) (0.05 µM), and NFκB inhibitor (BAY11-7085) (0.3125–1.25 µM) were prepared by dilution of the stock solution into cell culture media.
Cell treatments
MD-PBMCs were treated with TBT and appropriate control(s). Following the incubations, the cells were pelleted and the supernatants were collected and frozen at − 70 °C until assay. Cell pellets were lysed and stored at − 70 °C for Western blot analysis.
IL-1 β and IL-6 secretion assays
IL-1 β and IL-6 levels were measured using the BD OptEIA™ Human enzyme-linked immunosorbent assay (ELISA) kits (BD-Pharmingen, San Diego, CA, USA).
Cell lysis
MD-PBMCs (at a concentration of 4–6 million cells/mL) were treated with 2.5–200 nM TBT or control for 10, 30 min, 6, or 24 h. Following the treatments, the cells were centrifuged, supernatants collected, and the cell pellets were lysed using 133 µL of lysis buffer (Active motif, Carlsbad, CA). For inhibitor studies, MD-PBMCs (at a concentration of 3 million cells/mL) were either treated with 5, 10, 25 nM TBT (IL-1β studies) or 50, 100, 200 nM TBT (IL-6 studies). The cell lysates were stored frozen at − 80 °C.
Immunoblotting analysis
Cell lysates were run on 10% SDS-PAGE (sodium dodecylsulfate polyacrylamide gel electrophoresis) and transferred to a PVDF (polyvinylidene difluoride) membrane. The PVDF was immunoblotted with specific primary antibodies: anti-IL-1β, anti-IL-6, and anti-β actin (Cell Signaling Technologies, Beverly, MA). Antibodies were visualized using an ECL chemiluminescent detection system (Amersham, Piscataway, NJ, USA) and UVP Imager. β-actin levels were determined for each condition to verify that equal amounts of protein were loaded. In addition, the density of each protein band was normalized to β-actin to correct for slight differences in protein loading among the lanes.
RNA isolation and RT-qPCR
RNA from MD-PBMCs (at a concentration of 4–6 million cells/mL) was extracted with RNeasy Mini Kit (Qiagen). PCR primers were designed using Primer Express 2.0 (Applied Biosystems). All RT-qPCR assays were conducted using QuantiTect SYBR Green RT-PCR kit (Qiagen). Reaction was done in 20 µL containing 50 ng of total RNA and 0.4 µM of each primer.
Gene | Forward sequence (5′) | Reverse sequence (3′) |
|---|---|---|
IL-1β | GCACGATGCACCTGTACGAT | AGACATCACCAAGCTTTTTTGCT |
IL-6 | CCGGGAACGAAAGAGAAGCT | GCGCTTGTGGAGAAGGAGTT |
Statistical analysis
Statistical analysis of the data was performed using ANOVA and Student’s t test. Data were initially compared within a given experimental setup by ANOVA. A significant ANOVA was followed by pairwise analysis of control versus exposed data using Student’s t test, and a p value of less than 0.05 was considered significant.
Results
Effects of TBT exposures on the synthesis (secretion + intracellular levels) of IL-1β
10 min exposures to TBT
The effects of 10 min exposures to TBT on intracellular and secreted levels of IL-1β were examined in MD-PBMCs from 3 separate donors. There were no increases in the synthesis of IL-1β (secretion + intracellular levels) after 10 min of exposure to TBT (data not shown).
30 min exposures to TBT
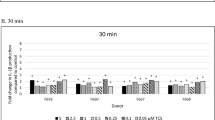
Figure 1a summarizes the effects of 30 min exposure to TBT on intracellular and secreted IL-1β in MD-PBMCs from 4 separate donors. Cells from each of the 4 donors exhibited slight increases in synthesis of IL-1β (the combination of fold increase in intracellular + fold increase in secreted IL-1β > 1) at one or more TBT concentrations. For example, cells from donor F219 showed increases of 1.5, 1.2, 1.4, and 1.8 fold in IL-1β synthesis with exposures to 200, 10, 5, and 2.5 nM TBT.
Effects of exposures to TBT on IL-1 β synthesis in monocyte-depleted PBMCs. a 30 min exposure to 0–200 nM TBT. The blots with accompanying secretion data are shown for the cells from 4 different donors. An increase in secretion or in intracellular level is a number greater than 1; a decrease in secretion compared to the control is a number less than 1. The control is arbitrarily set at 1. A combined fold increase (secretion + intracellular) greater than 1 indicates an increase in synthesis. b 6 h exposure to 0–200 nM TBT. The blots with accompanying secretion data are shown for the cells from 4 different donors. c 24 h exposure to 0–200 nM TBT. The blots with accompanying secretion data are shown for the cells from 5 different donors
6 h exposures to TBT
The effects of 6 h exposures to TBT on IL-1β synthesis are shown in Fig. 1b. As was seen at 30 min, cells from all donors exhibited increased production (a combined intracellular fold increase + secreted fold increase of greater than 1). However, the extent of the increases was somewhat greater than that seen after 30 min and it occurred at 3 or more exposure concentrations.
24 h exposures to TBT
The changes in intracellular and secreted IL-1β after 24 h exposures to TBT are shown in Fig. 1c. When the effects on secretion + intracellular levels of IL-1β were examined, there was a notable increase in its production in the cells from each of the 4 donors. Every TBT exposure caused an increase in production many of which were quite large (ranging from 1.1 to 26.4 fold) indicating that after 24 h of exposure TBT stimulates very consistent and quite substantial increases in IL-1β production in MD-PBMCs.
Effects of exposures to TBT on the synthesis of IL-1β by MD-PBMCs treated with signaling pathway inhibitors
Mitogen activated protein kinase kinase (MAP2K), MEK, inhibitor (PD98059)
The effects of exposures to 5, 10, and 25 nM TBT synthesis (intracellular + secreted levels) of IL-1β from MD-PBMCs where p44/42 MAPK function had been inhibited with PD98059 are shown in Fig. 2a. Cells exposed to TBT when the inhibitor was present showed diminished TBT-stimulated synthesis of IL-1β. For instance, when cells from donor F300 were exposed to 5, 10, and 25 nM TBT in the absence of PD98059 inhibitor there were 4.2, 4.1, and 8.4 fold increases in IL-1β production. When the inhibitor was present, those same TBT exposures caused no increase in synthesis at the 5 and 10 nM exposures, and only a twofold increase in IL-1β production at the 25 nM exposure. This same effect was seen in the cells from 3 other donors tested. Overall, the results indicate that the p44/42 MAPK pathway is being utilized to some extent by TBT to lead to increases in IL-1β synthesis in the cells from the majority of donors.
Effects of the inhibition of selected signaling pathways on the synthesis (secretion + intracellular levels) of IL-1β from human MD-PBMCs after 24 h exposure to TBT. a MEK inhibitor (PD98059); b p38 inhibitor (SB202190). An increase in secretion or in intracellular level is a number greater than 1 a decrease in secretion compared to the control is a number less than 1. The control is arbitrarily set at 1. Cells treated with inhibitor are compared to control cells treated with inhibitor. A combined fold increase (secretion + intracellular) greater than 1 indicates an increase in synthesis
p38 inhibitor (SB202190)
The effect of p38 MAPK pathway inhibition on TBT-induced increases in IL-1β synthesis from MD-PBMCs is shown in Fig. 2b. All donors consistently showed lowered TBT-induced IL-1β synthesis in the presence of SB202190. For example, donor F325 showed 2.3, 3.0, and 4.9 fold increases when MD-PBMCs were exposed to 5, 10, and 25 nM TBT in the absence of the p38 inhibitor. When the inhibitor was present, those same TBT exposures caused 1.5, 2.1, and 1.9 fold change. This indicates that the p38 pathway is being utilized by TBT to stimulate IL-1β production.
Nuclear factor kappa B (NFκB) inhibitor (BAY 11-7085) and JNK inhibitor (JNK X BI78D3)
In contrast to the results seen with MEK and p38, inhibition of either the NFκB pathway or the JNK pathway caused no consistent loss of TBT-induced increases in IL-1β synthesis (data not shown). This indicated that neither of these pathways are consistently needed for TBT to increase IL-1β synthesis.
Effects of exposures to TBT on IL-1β mRNA levels in MD-PBMCs
Table 1 shows IL-1β mRNA expression tested in 4 separate donors from MD-PBMCs with TBT concentrations ranging from 2.5 to 200 nM. No significant increases were seen after 6 h for 3 of the 4 donors.
The effects of 24 h exposures to TBT on IL-1β mRNA levels in MD-PBMCs are also shown in Table 1. Higher concentrations of TBT (in the range of 25–200 nM) caused increased mRNA expression compared to the control for all donors at two or more TBT concentrations with fold increases ranging from 1.3 to 5.3. These results suggest that the TBT-induced increases in IL-1β synthesis are due to increases in IL-1β mRNA at levels of TBT of 25 nM or more.
Effects of TBT exposures on IL-1β mRNA levels in MD-PBMCs treated with p44/42 (PD98059) and p38 (SB 202,190) pathway inhibitors
Based on its role in TBT-induced synthesis of IL-1β, we examined if the p44/42 MAPK pathway was needed for TBT-induced increases in IL-1β mRNA. Results indicated that p44/42 pathway is not consistently utilized by TBT to lead to increased levels of IL-1β mRNA (data not shown). However, when the p38 MAPK pathway was inhibited by SB202190, TBT-stimulated increases in IL-1β mRNA levels were blocked (Fig. 3). The ability of TBT to increase IL-1β mRNA was diminished in cells from all donors. There were 1.2, 1.9, and 1.7 fold increases in IL-1β mRNA when cells were exposed to 50, 100, and 200 nM TBT in the absence of inhibitor. There were no TBT-induced increases in IL-1β mRNA expression when the inhibitor was present at 50 and 200 nM TBT exposures and the increase at 100 nM went from 1.9 fold to 1.1 fold (Fig. 3). These results were replicated in cells from 3 additional donors. This indicates that p38 pathway is needed to cause TBT-induced increases of IL-1β mRNA levels.
Effects of 24 h TBT exposure to 50, 100, and 200 nM on IL-1β mRNA from monocyte-depleted PBMCs treated with MAPK pathway inhibitors. a MEK Inhibitor (PD98059) Donor F465; b MEK Inhibitor (PD98059) donor F479; c p38 inhibitor (SB202190) Donor F479. Fold changes in mRNA levels compared to the appropriate control are shown above the bars. (+) indicates a significant increase (p < 0.05) above the appropriate control and (*) indicates a significant decrease (p < 0.05) compared to the appropriate control
Effects of TBT exposures on the synthesis (secretion + intracellular levels) of IL-6
10 min exposures to TBT
As was seen with TBT’s effect on IL-1β production, there were no consistent increases in IL-6 synthesis seen after a 10 min exposure to TBT (data not shown).
30 min exposures to TBT
After a 30 min exposure to TBT, cells from all donors showed increases in IL-6 production at a minimum of 3 concentrations. The changes in intracellular levels paired with the changes in fold secretion are shown in Fig. 4a. These results indicate that TBT induces generally small but consistent increases in IL-6 synthesis after 30 min of exposure to TBT.
Effects of exposures to TBT on IL-6 synthesis in monocyte-depleted PBMCs. a 30 min exposure to 0–200 nM TBT. The blots with accompanying secretion data are shown for the cells from 4 different donors. An increase in secretion or in intracellular level is a number greater than 1 a decrease in secretion compared to the control is a number less than 1. The control is arbitrarily set at 1. A combined fold increase (secretion + intracellular) greater than 1 indicates an increase in synthesis. b 6 h exposure to 0–200 nM TBT. The blots with accompanying secretion data are shown for the cells from 4 different donors. c 24 h exposure to 0–200 nM TBT. The blots with accompanying secretion data are shown for the cells from 5 different donors
6 h exposures to TBT
When MD-PBMCs from 4 distinct donors were exposed to TBT for 6 h there was also typically a small but consistent increase in IL-6 synthesis at 1 or more exposure (Fig. 4b). This was evidenced by the combined fold increases in intracellular and secreted levels being greater than 1. Increases ranged from 1.3 fold for cells from F234 at the 2.5 nM exposure to 9.7 fold for cells from F246 at the 200 nM concentration.
24 h exposures to TBT
Figure 4c shows the effects of 24 h exposure to TBT on synthesis of IL-6. Dramatic increases were seen in IL-6 synthesis at the higher TBT concentrations of 50, 100, and 200 nM. Fold increases ranged from 28 to 852 (200 nM TBT). There was not a commensurate increase in the secretion of IL-6 from these cells. This indicates that while TBT is able to increase the production of IL-6, it must also be to some extent blocking its secretion.
Effects of exposures to TBT on the synthesis of IL-6 by MD-PBMCs treated with signaling pathway inhibitors
MEK, inhibitor (PD98059)
The effects of inhibiting the p44/42 mitogen activated protein kinase (MAPK) pathway with PD98059 (50 µM) on TBT-induced increases in IL-6 synthesis are shown in Fig. 5a (shows fold increases in intracellular and secreted levels). When this pathway was inhibited, the ability of TBT to stimulate IL-6 synthesis was decreased at one or more TBT concentration in cells from all 4 donors. For example, cells from donor F325 exposed to 50, 100, and 200 nM TBT in the absence of the inhibitor showed 1.6, 2.0, and 4.0 fold increases in IL-6 production. In the presence of inhibitor these same cells showed reduced TBT-induced production of IL-6 of 1.1, 1.2, and 2.3 fold at these same concentrations. This indicates that the p44/42 MAPK pathway is being utilized to some extent by TBT to increase IL-6 production.
Effects of the inhibition of selected signaling pathways on the production (secretion + intracellular levels) of IL-6 from human MD-PBMCs after 24 h exposure to TBT. a MEK inhibitor (PD98059); b p38 inhibitor (SB202190). An increase in secretion or in intracellular level is a number greater than 1, a decrease in secretion compared to the control is a number less than 1. The control is arbitrarily set at 1. Cells treated with inhibitor are compared to control cells treated with inhibitor. A combined fold increase (secretion + intracellular) greater than 1 indicates an increase in synthesis
p38 inhibitor (SB202190)
Figure 5b shows the effects of exposures to 50, 100, and 200 nM TBT on intracellular/secreted levels of IL-6 from MD-PBMCs where p38 MAPK function had been inhibited with SB202190. When the inhibitor was present, TBT-stimulated synthesis of IL-6 was completely blocked in MD-PBMCs from all donors tested. For instance, when cells from donor F405 were exposed to 50, 100, and 200 nM TBT in the absence of the inhibitor, there were 0.9, 1.4, and 3.9 fold increases in IL-6 production and no TBT-induced increases in IL-6 production when the inhibitor was present. These results indicate that the p38 MAPK pathway is being utilized by TBT to lead to increases in IL-6 production.
NFκB inhibitor (BAY 11-7085) and JNK inhibitor (JNK X BI78D3)
When either the NFκB pathway or JNK pathway was inhibited in MD-PBMCs with their respective inhibitors, the ability of TBT to increase IL-6 synthesis was not consistently decreased. Thus, it appears that as was seen with IL-1β, neither the NFκB pathway nor the JNK pathway was consistently used by TBT to induce increases in IL-6 synthesis.
Effects of exposures to TBT on IL-6 mRNA levels in MD-PBMCs
The effects of 6 h exposures to TBT on IL-6 mRNA expression in MD-PBMCs are shown in Table 2. Tested in cells from 4 separate donors, no statistically significant increases were seen at any of the TBT exposures after 6 h. Table 2 also shows the effects of 24 h exposures to TBT on IL-6 mRNA levels. Higher concentrations of TBT (100 and 200 nM) showed increased mRNA expression compared to the control for 3 of the 4 donors. Fold increases at the highest TBT concentration were 4.0 fold (F277), 7.5 fold (F266), and 54.7 fold (F322). These data indicate that TBT may be increasing IL-6 synthesis, at least in part, by increasing the mRNA for IL-6.
Effects of TBT exposures on IL-6 mRNA levels in MD-PBMCs treated with p44/42 (PD98059) and p38 (SB 202190) pathway inhibitors
Figure 6 shows the effects of p38 MAPK pathway inhibition on TBT-stimulated increases in IL-6 mRNA levels. When this pathway was inhibited, the ability of TBT to increase IL-6 mRNA was blocked. Cells showed 1.3, 1.3, and 2.7 fold increases in IL-6 mRNA when exposed to 50, 100, and 200 nM TBT in the absence of inhibitor. There were no TBT-induced increases in IL-6 mRNA expression when the inhibitor was present (Fig. 6). This result was replicated in cells from 3 additional donors. Thus, TBT-induced increases in IL-6 mRNA appear to be dependent on the p38 pathway. As was seen with TBT-induced increases in IL-1β mRNA, inhibition of the p44/42 pathway with the MEK inhibitor (PD98059) did not effect TBT’s ability to increase IL-6 mRNA levels in MD-PBMCs (data not shown).
Effects of 24 h TBT exposure to 50, 100, and 200 nM on IL-6 mRNA from monocyte-depleted PBMCs treated with MAPK pathway inhibitors. p38 inhibitor (SB202190) donor F480. Fold changes in mRNA levels compared to the appropriate control are shown above the bars. (+) indicates a significant increase (p < 0.05) above the appropriate control and (*) indicates a significant decrease (p < 0.05) compared to the appropriate control
Discussion
TBT has been shown to increase the secretion of IL-1β and IL-6 within 24 h of exposure (Brown and Whalen 2015; Brown et al. 2018). However, it was not clear if TBT-induced increases in secretion of IL-1β or IL-6 were due to increased synthesis of the cytokines or simply release of pre-existing IL-1β or IL-6. Due to its ability to increase secretion of these 2 important pro-inflammatory proteins, TBT has the capacity to produce “sterile inflammation” (inflammation in the absence of microorganisms, Chen and Nunez 2010) which could then contribute to chronic inflammation. Chronic inflammation is a known promoter of a number of disease states including rheumatoid arthritis, Crohn’s disease, multiple sclerosis, and cancer (Choy and Panayi 2001; Dinarello 2011; Rossi et al. 2012; Arlt et al. 2002; Elaraj et al. 2006; Jin et al. 1997; Lewis and Varghese 2006; Muerkoster et al. 2006; Voronov et al. 2002; Chirivi et al. 1996; Vidal-Vanaclocha et al. 1996; Gabay 2006; Atreya et al. 2000; Hurst et al. 2001; Culig et al. 2005; Nilsson et al. 2005). If TBT is increasing the secretion of existent cytokine in the absence of increased cellular synthesis, then increased levels of these cytokines would be somewhat self-limiting. However, if TBT is also stimulating immune cells to produce these potent inflammatory proteins, then very significant “sterile inflammation” could result in response to TBT exposures.
Thus, intracellular levels and accompanying secretion of IL-1β and IL-6 were examined at 10, 30 min, 6, and 24 h. The measurement of both secreted and intracellular levels of the cytokine from the same cells made it possible to determine if TBT affected synthesis of these 2 cytokines in immune cells. The results indicated that TBT at several exposure levels caused small but consistent increases in IL-1β production within 6 h of exposure and these changes were very robust after 24 h. Results after 24 h showed that TBT increased IL-1β synthesis at every concentration.
The effects of TBT exposures on IL-6 synthesis were similar to those seen with IL-1β in that increased IL-6 production was noticeable after 6 h and most clearly seen after 24 h with substantial increases seen at every TBT concentration. In contrast to the effect of TBT on IL-1β production, the increased production of IL-6 stimulated by TBT was not accompanied by increased secretion of the cytokine. This indicates that TBT while greatly activating the synthesis of IL-6 (at higher concentration) is simultaneously having an inhibitory effect on its secretion.
Synthesis of both IL-6 and IL-1β is regulated by MAP kinase pathways (Gaestel et al. 2009). The current studies indicate that TBT is utilizing both the p44/42 and p38 pathways to lead to increased IL-1β production, with a greater dependence on the p38 pathway. The pathway dependence of TBT-induced increases in IL-6 at 50, 100, and 200 nM was also examined. As was seen with IL-1β, both the p44/42 and p38 are utilized by TBT to lead to increased IL-6 production. The p38 pathway was necessary for TBT-induced increases in IL-6 and the p44/42 pathway contributed to TBT-induced increases but was not as crucial as p38, similar to what was seen with IL-1β.
The increases that were seen in IL-1β and IL-6 synthesis in response to TBT exposures could be due to increased transcription, stabilization of mRNA, increased translation, or a combination of these factors. Results indicated that increased mRNA levels of both cytokines were generally seen after 24 h at TBT concentrations of 50, 100, and 200 nM. However, increased protein levels were seen at all TBT concentrations after 24 h exposures. This suggests that an increase in mRNA is at least in part responsible for the TBT-induced increases in these 2 cytokines seen at higher TBT concentrations. However, TBT-induced increases in the mRNA of either IL-1β or IL-6 do not appear to explain the TBT-induced increases in IL-1β and IL-6 synthesis seen at the lower concentrations of TBT. It is possible that these increases are due to TBT effects on the translational process. TBT utilizes the MAPK pathways for increased production and it is known that MAPKs can lead to increased translation due to activation of the eukaryotic initiation factor 4E (eIF4E) (Shveygert et al. 2010).
As both p44/42 and p38 appear to have a role in the TBT-induced increases in IL-1β and IL-6 production, we investigated whether these signaling pathways were required for TBT-induced increases in IL-1β and IL-6 mRNA expression. The results suggest that p38 pathway is needed for TBT-induced increases of IL-1β and IL-6 mRNA levels.
In summary, the current study indicates that TBT induces increases in the cellular synthesis of both IL-1β and IL-6. Furthermore, it shows that elevated synthesis is detectable within 6 h of exposure. TBT causes synthesis of IL-1β, which then leads to increased secretion. In contrast, TBT-stimulated synthesis of IL-6 but did not increase secretion, indicating that TBT causes at least a partial inhibition of IL-6 secretion, which was not seen with IL-1β. TBT-induced synthesis of each of these cytokines is primarily dependent on the p38 MAPK pathway and to a lesser extent the p44/42 pathway. The highest levels of TBT caused increased mRNA for both IL-1β and IL-6, which could account for the TBT-elevated synthesis. However, lower concentrations of TBT did not increase the mRNAs for these cytokines even though they stimulated the synthesis of the proteins.
References
Akira S, Taga T, Kishimoto T (1993) Interleukin-6 in biology and medicine. Adv Immunol 54:1–78
Antizar-Ladislao B (2008) Environmental levels, toxicity and human exposure to tributyltin (TBT)-contaminated marine environment. Environ Int 34:292–308
Apte RN, Voronov E (2002) Interleukin-1—a major pleiotropic cytokine in tumor–host interactions. Semin Cancer Biol 12:277–290
Arend WP (2002) The balance between IL-1 and IL-1Ra in disease. Cytokine Growth Factor Rev 13:323–340
Arlt A, Vorndamm J, Müerköster S, Yu H, Schmidt WE, Fölsch UR, Schäfer H (2002) Autocrine production of interleukin 1β confers constitutive nuclear factor κB activity and chemoresistance in pancreatic carcinoma cell lines. Can Res 62:910–916
Atreya R, Mudter J, Finotto S, Müllberg J, Tostock T, Wirtz S, Schütz M, Bartsch B, Holtmann M, Becker C, Strand D, Czaja J, Schlaak JF, Lehr HA, Autschbach F, Schürmann G, Nishimoto N, Yoshizaki K, Ito H, Kishimoto T, Galle PR, Rose-John S, Neurath MF (2000) Blockade of interleukin 6 trans signaling suppresses T-cell resistance against apoptosis in chronic intestinal inflammation: evidence in Crohn disease and experimental colitis in vivo. Nat Med 6:583–588
Brown S, Whalen M (2015) Tributyltin alters secretion of interleukin 1 beta from human immune cells. J Appl Toxicol 35:895–908
Brown S, Wilburn WJ, Martin T, Whalen MM (2018) Butyltin compounds alter secretion of interleukin 6 from human immune cells. J Appl Toxicol 38:201–218
Burger D, Dayer J (2002) Cytokines, acute-phase proteins, and hormones. Ann N Y Acad Sci 473:464–473
Chen GY, Nunez G (2010) Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol 10:826–837
Chirivi RG, Chiodoni C, Musiani P, Garofalo A, Bernasconi S, Colombo MP, Giavazzi R (1996) IL-1α gene-transfected human melanoma cells increase tumor-cell adhesion to endothelial cells and their retention in the lung of nude mice. Int J Cancer 67:856–863
Choy EHS, Panayi GS (2001) Cytokine pathways and joint inflammation in rheumatoid arthritis. N Eng J Med 344:907–916
Cooper MA, Fehniger TA, Ponnappan A, Mehta V, Wewers MD, Caligiun MA (2001) Interleukin-1β costimulates interferon-γ production by human natural killer cells. Eur J Immunol 31:792–801
Culig Z, Steiner H, Bartsch G, Hobisch A (2005) Interleukin-6 regulation of prostate cancer cell growth. J Cell Biochem 95:497–505
Dinarello CA (1996) Biologic basis for IL-1 in disease. J Am Soc Hematol 87:2095–2147
Dinarello CA (2005) Blocking IL-1 in systemic inflammation. J Exp Med 201:1355–1359
Dinarello CA (2009) Immunological and inflammatory functions of the interleukin-1 Family. Annu Rev Immunol 27:519–550
Dinarello CA. 2011, Interleukin-1 in the pathogenesis and treatment of inflammatory diseases blood 117: 3720–3732
Dudimah FD, Odman-Ghazi SO, Hatcher F, Whalen MM (2007) Effect of tributyltin (TBT) on ATP levels in human natural killer (NK) cells: Relationship to TBT-induced decreases in NK function. J Appl Toxicol 27:86–94
Elaraj DM, Weinreich DM, Varghese S, Puhlmann M, Hewitt SM, Carroll NM, Feldman ED, Turner EM, Alexander HR (2006) The role of interleukin 1 in growth and metastasis of human cancer xenografts. Clin Cancer Res 12:1088–1096
Gabay C (2006) Interleukin-6 and chronic inflammation. Arthritis Res Ther 8:S3
Gaestel M, Kotlyarov A, Kracht M (2009) Targeting innate immunity protein kinase signaling in inflammation. Nat Rev Drug Discov 8:480–481
Gipperth L (2009) The legal design of the international and European Union ban on tributyltin antifouling paint: Direct and indirect effects. J Environ Manag 90:S86–S95
Guma M, Ronacher L, Liu-Bryan R, Takai S, Karin M (2009) Caspase 1-independent activation of interleukin-1 beta in neutrophil-predominant inflammation. Arthritis Rheum 60:3642–3650
Hall LJ, Clare S, Dougan G (2010) NK cells influence both innate and adaptive immune responses after mucosal immunization with antigen and mucosal adjuvant. J Immunol 84:4327–4337
Hurst SM, Wilkinson ST, McLouglin RM, Jones S, Horiuchi S, Yamamoto N, Rose-John S, Fuller GM, Topley N, Jones SA (2001) IL-6 and its soluble receptor orchestrate a temporal switch in the pattern of leukocyte recruitment seen during acute inflammation. Immunity 14:705–714
Hurt K, Hurd-Brown T, Whalen M (2013) Tributyltin and dibutyltin alter secretion of tumor necrosis factor alpha from human natural killer cells and a mixture of T cells and natural killer cells. J Appl Toxicol 33:503–510
Jin L, Yuan RQ, Fuchs A, Yao Y, Joseph A, Schwall R, Schnitt SJ, Guidae A, Hastings HM, Andres J, Turkel G, Polverini PJ, Goldberg ID, Rosen EM (1997) Expression of interleukin-1β in human breast carcinoma. Cancer 80:421–434
Kannan K, Tanabe S, Tatsukawa R (1995) Occurrence of butyltin residues in certain foodstuffs. Bull Environ Contam Toxicol 55:510–516
Kannan K, Senthilkumar K, Giesy JP (1999) Occurrence of butyltin compounds in human blood. Environ Sci Technol 33:1776–1779
Kishimoto T (1989) The biology of interleukin-6. Blood J 74:1–10
Lawrence S, Reid J, Whalen MM (2015) Secretion of interferon gamma from human immune cells is altered by exposure to tributyltin and dibutyltin. Environ Toxicol 30:559–571
Lewis A, Varghese S (2006) Interleukin-1 and cancer progression: the emerging role of interleukin-1 receptor antagonist as a novel therapeutic agent in cancer treatment. J Transl Med 12:1–12
Li T, He S (2006) Induction of IL-6 release from human T cells by PAR-1 and PAR-2 agonists. Immunol Cell Biol 84:461–466
Loganathan BG (2016) Persistent organic chemicals in the Pacific Basin countries: an overview. In. Persistent organic chemicals in the environment: status and trends in the pacific Basin Countries I. Loganathan BG, Khim JS, Kodavanti PR, Masunaga S (eds) ACS Symposium Series Vol. 1243. American Chemical Society and Oxford University Press, pp 1–15
Loganathan BG, Kannan K, Sajwan KS, Owen DA (2001) Butyltin compounds in freshwater ecosystems. In: Lipnick RL, Hermens J, Jones K, Muir D (eds) Persistent, bioaccumulative and toxic chemicals i: fate and exposure, ACS Monograph Series, vol 772. American Chemical Society, Washington, DC, 134–149
Luo G, Hershko DD, Robb BW, Wray CJ, Hasselgren PO (2003) IL-1β stimulates IL-6 production in cultured skeletal muscle cells through activation of MAP kinase signaling pathway and NF-κB. Am J Physiol Regul Integr Comp 284:1249–1254
Meyer TP, Zehnter I, Hofmann B, Zaisserer J, Burkhart J, Rapp S, Weinauer F, Schmitz J, Illert WE (2005) Filter Buffy Coats (FBC): a source of peripheral blood leukocytes recovered from leukocyte depletion filters. J Immunol Methods 307:150–166
Muerkoster SS, Lust J, Arlt A, Hasler R, Witt M, Sebens T, Schreiber S, Folsch UR, Schafer H (2006) Acquired chemoresistance in pancreatic carcinoma cells: induced secretion of IL-1beta and NO lead to inactivation of caspases. Oncogene 25:3973–3981
Nilsson MB, Langley RR, Fidler IJ (2005) Interleukin-6, secreted by human ovarian carcinoma cells, is a potent proangiogenic cytokine. Can Res 65:10794–10800
Riedemann NC, Guo R-F, Hollmann TJ, Gao H, Neff TA, Reuben JS, Speyer CL, Sarma JV, Wetsel RA, Zetoune FS, Ward PA (2004) Regulatory role of C5a in LPS-induced IL-6 production by neutrophils during sepsis. FASEB J 18:370–372
Rossi S, Furlan R, De Chiara V, Motta C, Studer V, Mori F, Musella A, Bergami A, Muzio L, Bernardi G, Battistini L, Martino G, Centonze D (2012) Interleukin-1β causes synaptic hyperexcitability in multiple sclerosis. Ann Neurol 71(1):76–83. https://doi.org/10.1002/ana.22512
Shveygert M, Kaiser C, Bradrick SS, Gromeier M (2010) Regulation of eukaryotic initiation factor 4E (eIF4E) by mitogen-activated protein kinase occurs through moducationof Mnk- 1- eIFG interaction. Mol Cell Biol 30:5160–5167
Snoeij NJ, Penninks AH, Seinen W (1988) Dibutyltin and tributyltin compounds induce thymus atrophy in rats due to selective action on thymic lymphoblasts. Int J Immunopharmacol 10:891–899
Tanaka T, Narazaki M, Kishimoto T (2014) IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol 6:1–16
Tosato G, Jones KD (1990) Interleukin-1 induces interleukin-6 production in peripheral blood monocytes. Blood J 75:1305–1310
Vidal-Vanaclocha F, Alvarez A, Asumendi A, Urcelay B, Tonino P, Dinarello CA (1996) Interleukin 1 (IL-1)-dependent melanoma hepatic metastasis in vivo; increased endothelial adherence by IL-1-induced mannose receptors and growth factor production in vitro. J Natl Cancer Inst 88:198–205
Voronov E, Shouval DS, Krelin Y, Cagnano E, Benharroch D, Iwakura Y, Dinarello CA, Apte RN (2002) IL-1 is required for tumor invasiveness and angiogenesis. Proc Natl Acad Sci USA 100:2645–2650
Wester PW, Krajnc EI, van Leeuwen FXR, Loeber JG, van der Heijden CA, Vaessen HA, Helleman PW (1990) Chronic toxicity and carcinogenicity of bis(tri-n-butyltin)oxide (TBTO) in the rat. Food Chem Toxicol 28:179–196
Whalen MM, Loganathan BG, Kannan K (1999) Immunotoxicity of environmentally relevant concentrations of butyltins on human natural killer cells in vitro. Environ Res 81:108–116
Yamada S, Fuji Y, Mikami E, Kawamura N, Hayakawa J (1993) Small-scale survey of organotin compounds in household commodities. J AOAC Int 76:436–441
Zimmermann M, Bianchetto-Aguilera F, Castellucci M, Rossato M, Costa S, Lunardi C, Ostuni R, Girolomoni G, Natoli G, Bazzoni F, Tamassia N, Cassatella MA (2015) Chromatin remodelling and autocrine TNFa are required for optimal interleukin-6 expression in activated human neutrophils. Nat Commun 6:6061. https://doi.org/10.1038/ncomms7061
Zimmermann M, Arruda-Silva F, Bianchetto-Aguilera F, Finotti G, Calzetti F, Scapini P, Lunardi C, Cassatella MA, Tamassia N (2016) IFNα enhances the production of IL-6 by human neutrophils activated via TLR8. Sci Rep 6:19674. https://doi.org/10.1038/srep19674
Acknowledgements
Grants U54CA163066 and 2T34GM007663 from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Brown, S., Boules, M., Hamza, N. et al. Synthesis of interleukin 1 beta and interleukin 6 in human lymphocytes is stimulated by tributyltin. Arch Toxicol 92, 2573–2586 (2018). https://doi.org/10.1007/s00204-018-2248-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2248-2