Abstract
Gluconacetobacter diazotrophicus is a nitrogen-fixing, endophytic bacterium that has the potential to promote plant growth and increase yield. Genetically modified strains might get more benefits to host plants, including through expression of useful proteins, such as Cry toxins from B. thuringiensis, or enzymes involved in phytohormone production, proteins with antagonistic activity for phytopathogens, or that improve nutrient utilization by the plant. For that, expression systems for G. diazotrophicus are needed, which requires active promoters fused to foreign (or innate) genes. This article describes the construction of a G. diazotrophicus PAL5 promoter library using a promoter-less lacZ-bearing vector, and the identification of six active promoters through β-galactosidase activity assays, sequencing and localization in the bacterial genome. The characterized promoters, which are located on distinct regions of the bacterial genome and encoding either sense or antisense transcripts, present variable expression strengths and might be used in the future for expressing useful proteins.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gluconacetobacter diazotrophicus is a nitrogen-fixing, endophytic bacterium that was initially isolated from sugarcane (Cavalcante and Döbereiner 1988; Gillis et al. 1989; Dong et al. 1995), and later from other agronomically important plants, such as wheat, rice, coffee, sweet potato, elephant grass, banana and pineapple (Baldani and Baldani 2005). This bacterium has been considered as one of the possible candidates responsible for the high levels of N2 fixation detected in sugarcane (Boddey et al. 2003). It has been shown that inoculation of G. diazotrophicus strain PAL5 in a consortium with four additional endophytic nitrogen-fixing bacterial strains has the potential to promote sugarcane growth and increase yield (Oliveira et al. 2002, 2006). In addition, there is evidence that, beyond nitrogen fixation, other bacterial benefits might be acting for plant growth promotion (Sevilla et al. 2001), such as phytohormone production (Bastián et al. 1998), antagonistic activity against phytopathogens (Blanco et al. 2005; Saravanan et al. 2007), and improvement of nutrient utilization by the plant (Maheshkumar et al. 1999; Saravanan et al. 2007; Logeshwaran et al. 2009).
If wild-type strains of G. diazotrophicus present potential to be used in inoculants for agronomically important crops, genetically modified G. diazotrophicus strains may get even more beneficial features, exemplified by modifications for expressing Cry proteins from Bacillus thuringiensis with lethality against plague insects (Subashini et al. 2011; Rapulana and Bouwer 2013). In order to generate new recombinant strains for expressing useful proteins, expression systems for G. diazotrophicus are needed, which requires active promoters fused to foreign (or innate) genes.
Strong promoters have been isolated from Leifsonia xyli (basonym Clavibacter xyli) subsp. cynodontis, the causative pathogen of ratoon stunting disease in sugarcane, with the aim to express useful proteins inside plants without having to transform the plant genome (Haapalainen et al. 1996). In Rhizobium etli, which is a nodule-forming, nitrogen-fixing symbiont of bean roots, a collection of active promoters has been isolated from a genomic DNA library constructed in a suited promoter-trap vector, which permitted to characterize the regulon of the main sigma factor σ70 (SigA) in that bacterial species (Ramírez-Romero et al. 2006). Also, in Azospirillum brasilense strain Sp245, a plant growth-promoting rhizobacterium (PGPR) of cereal crops, the promoter-trapping approach of differential fluorescence induction (DFI) has been used to identify promoters that are induced in the presence of wheat seed extracts, revealing genes involved in the interaction with the plant (Pothier et al. 2007). Works like those illustrate the importance of isolating and characterizing promoters from plant-associated bacteria for both knowledge gain and potential agrobiotechnological application.
This article describes the construction of a G. diazotrophicus PAL5 promoter library using a promoter-less lacZ-bearing vector, and the identification of active promoters through β-galactosidase activity assays, sequencing and localization in the bacterial genome. This approach permitted the identification of six G. diazotrophicus active promoters with variable expression strengths, which are located on distinct regions of the bacterial genome, and might be used in the future for expressing useful proteins.
Materials and methods
Determination of minimal inhibitory concentration (MIC) of antibiotic and relative vector stability
In order to determine the tetracycline MIC for G. diazotrophicus PAL5, this strain was grown in a 500-ml erlenmeyer flask containing 150 μl of the culture media DYGS (Rodrigues Neto et al. 1986) (2.0 g l−1 glucose, 1.5 g l−1 bacterial peptone, 2.0 g l−1 yeast extract, 0.5 g l−1 K2HPO4, 0.5 g l−1 MgSO4·7H2O, 1.5 g l−1 glutamic acid, 0.1 mg l−1 biotin, 0.2 mg l−1 pyridoxal-HCl, pH 6.0, 15 g l−1 agar) or LZGD (simplified DYGS medium in which modification was exclusion of both bacterial peptone and yeast extract) in the presence of serial dilutions of tetracycline, in an ELISA plate, for up to 2 day, at 30 °C, 250 rpm. The optical density of the cell culture suspensions was determined using an iEMS microplate reader (Labsystems) at 600 nm.
Vector stability assays comprised in culturing G. diazotrophicus PAL5 harboring pPW452 in DYGS medium under the absence of selective pressure for more than forty generations (estimated following optical density at 600 nm), plating on both the presence and absence of tetracycline, and counting the colony-forming units (CFUs), which revealed the number of resistant (vector-bearing) and total CFUs, respectively.
Construction of a lacZ promoter-trap library
Vector pPW452 is a derivative of pMP220 (Spaink et al. 1987), in which the multicloning site (flanked by two HindIII cleavage sites) was inverted (P. Woodley, unpublished). Those are promoter probe vectors containing a multicloning site located upstream to a promoter-less lacZ reporter gene. To construct the promoter library in pPW452, total G. diazotrophicus PAL5 DNA was prepared using the CTAB method (Doyle 1987), and partially digested with Sau3AI. Resulting fragments with 0.5–1.0-kb were excised from an agarose gel, purified using Wizard SV Gel and PCR Clean-Up System (Promega), cloned into the unique BglII site of pPW452, and the resultant recombinant plasmids were transferred into E. coli DH10B cells. Transformants were selected on 5-bromo-4-chloro-3-indolyl-beta-d-galactopyranoside (X-Gal)-containing LB medium supplemented with 10 mg l−1 tetracycline. All colonies of the promoter library were pooled, the plasmid DNA was prepared from the mixed clone cells, and used to transform cells of G. diazotrophicus PAL5 through electroporation, essentially as described (Teixeira et al. 1999). Transformants were selected on X-Gal-containing DYGS medium in the presence of 100 mg l−1 tetracycline. G. diazotrophicus clones were picked using sterilized toothpicks, transferred to 96-well plates, cultured in liquid DYGS medium, and stored in 50 % glycerol at −70 °C.
Clone culture conditions for LacZ activity evaluations
2.5 μl of each clone suspension were transferred from the stock plates at −70 °C onto solid DYGS medium containing 100 mg l−1 tetracycline, and cultured for 5–7 days at 30 °C. The resulting colonies were then picked, using a 96-well format picker, for inoculating into 1 ml of liquid DYGS medium containing 100 mg l−1 tetracycline in 96-deep-well plates. Clones were cultured for 3–4 days at 30 °C, 250 rpm. Then, clones were reinoculated 1:400 into 1 ml of LZGD medium (with glucose or another carbon source when specified) containing 10 mg l−1 tetracycline, in 96-deep-well plates, and then cultured for 3-5 days at 30 °C, 250 rpm. Alternatively, clones were inoculated on one of the following solid media in the presence of X-Gal: DYGS, LGI (Baldani et al. 2005) (pH 6.0), acetic LGI (Sievers and Swings 2005), or BMS (Baldani et al. 2005) (supplemented with 10 % sucrose).
Chromogenic assays of beta-galactosidase activity
Beta-galactosidase activity assays using ortho-nitrophenyl-β-d-galactopyranoside (ONPG) were performed as initially described (Miller 1972), but adapted to the 96-well format (Griffith and Wolf 2002; Schwab et al. 2007). Briefly, 40 μl of the cell suspension were transferred to a well of a 96-deep-well plate containing 360 μl of Z buffer (60 mM Na2HPO4·7H2O, 40 mM Na2H2PO4·H2O, 10 mM KCl, 1 mM MgSO4·7H2O, 0.0027 % SDS, 0.39 % β-mercaptethanol, pH 7.0). For cell permeabilization, 25 μl of chloroform were added in each well. Plates were vortexed, briefly centrifuged using a microplate rotor, and 80 μl of ONPG solution (4.0 g·l−1 in Z buffer) were added into each well. When the reaction solution stained yellow, it was stopped by adding 200 μl of 1 M Na2CO3, and the time required for the reaction was registered. The 96-deep-well plate was centrifuged for 5 min, at 3600×g, and 200 μl of the supernatant were transferred into a well of an ELISA microplate. Absorbance at 414 nm was determined using an iEMS microplate reader (Labsystems). The optical density of the cell culture suspensions (200 μl) was determined using the same microplate reader at 600 nm.
Fluorogenic assays of beta-galactosidase activity
The fluorogenic substrate 5-acetylaminofluorescein di-β-d-galactopyranoside (C2FDG) was used for assaying beta-galactosidase activity of the G. diazotrophicus clones essentially as described for other bacterial species (Rowland et al. 1999). In brief, the cell suspension was centrifuged for 7 min at 3600×g, the supernatant was discarded, and cells were suspended in the same volume of phosphate buffered saline (PBS: 137 mM NaCl, 3 mM KCl, 8 mM Na2HPO4, 1.5 mM KH2PO4). 50 μl of the cell suspension were transferred to a well of an ELISA microplate containing 50 μl of 66 μM C2FDG. The microplate was incubated at 30 °C for 3 h, and then scanned using a Typhoon FLA 9000 manifold (GE Healthcare) with LPB filter. Fluorescence intensity values were obtained through pixel quantification from the image files using ImageQuant TL 7.0 (GE Healthcare). Fluorescence arbitrary units were given as a function of the optical density of the cell culture suspensions, determined as described for chromogenic assays.
Genetic characterization of beta-galactosidase highly active clones
Cell suspensions of the G. diazotrophicus clones were diluted 1:10 in PCR-grade water, boiled for 10 min, and then used as PCR template with primers PPWleft (5′-TTGAGCAACTGACTGAAATGC-3′) and PPWrightV3 (5′-GCTCACCCCAAAAATGGCA-3′), which anneal to the flanks of the multiple cloning site of pPW452. PCR products were precipitated by the addition of 10 % volume of 3 M sodium acetate and 2.5 volumes of ethanol, and dissolved in PCR-grade water.
Alternatively, the cell suspensions in DYGS medium were centrifuged, and the supernatant was discarded. Cells were resuspended in 150μl of GET buffer (50 mM glucose, 10 mM EDTA, 25 mM TRIS–HCl, pH 8.0), and lysed by adding 150 μl of lysis solution (0.4 M NaOH, 2 % SDS). The cell lysate was directly used to transform competent E. coli DH10B cells. Plasmids from the resulting transformant clones were prepared through the alkaline lysis technique (Ish-Horowicz and Burke 1981).
Clones were sequenced with primer PPWleft or PPWrightV3 using a 3500 Genetic Analyzer (Applied Biosystems), or by Macrogen Inc. (South Korea).
Results
Representativeness of the G. diazotrophicus PAL5 promoter-trap library
MIC determination results of tetracycline for G. diazotrophicus strain PAL5 (Fig. 1a) suggested that 100 g·l−1 tetracycline was efficient to select transformants with vector pPW452 and derivatives carrying the promoter library. Also, vector stability assays suggested that under the absence of selective pressure, the vector was rapidly lost (Fig. 1b). Therefore, the use of tetracycline was important to guarantee maintenance of the vector and derivatives in PAL5 cells.
Near 8100 colonies of E. coli DH10B cells were obtained presenting variable degrees of blue color intensity on X-Gal-containing medium after transformation with the PAL5 promoter library. A HindIII restriction analysis involving a random sample of 44 clones revealed the presence of inserts with the expected size range (0.5–1.0 kb) in 33 clones, suggesting the presence of desired inserts in approximately 75 % of the promoter library. Extrapolating that percentage to the whole library, the success-rate was estimated to be approximately 8100 × 75 % = 6100 valid clones. Therefore, the genomic relative coverage of the promoter library was calculated as previously described (Clarke and Carbon 1976), but considering both DNA strands for transcriptional orientation reasons relating to lacZ, which would double the genome size (factor “2”):
in which f is the fraction of the total genome represented by a fragment with a given average size, and N is the number of positive clones in the library.
The promoter library was then extracted from the pooled E. coli clones, and transferred into G. diazotrophicus cells by electroporation, a procedure that resulted in roughly 7400 tetracycline-resistant colonies of clones. The probability P 2 of a given cloned plasmid in E. coli has been included in the ~7400 colonies of the PAL5 transformed cells, considering that all had the same chance, follows the same statistics as above:
Considering the relative genomic coverage of the promoter library calculated in E. coli (44 %), in G. diazotrophicus the relative genomic coverage of the promoter library would be reduced to 44 % × 60 % ≅ 26 %.
To validate those estimations, a random sample of 71 clones in G. diazotrophicus was analyzed through DNA sequencing of their inserts. Of those, 30 clones presented empty vector sequence, while 41 showed G. diazotrophicus PAL5 genome sequences (Table 1). Thus, the percentage of positive clones was 41/71 = 58 %, in contrast to 75 % in E. coli, as shown above. Reduction of positive clones after transferring the promoter library to another bacterial strain might result from the greater probability of smaller plasmids (empty or small insert-containing vector) to enter host cells (Hanahan 1983). In accordance with this, among the positive G. diazotrophicus clones, most had fragment sizes near the minimal limit of the selected length to construct the promoter library (~500 bp).
The 41 insert-containing clones showed some redundancy, actually representing 21 distinct constructions involving the G. diazotrophicus PAL5 genome (Table 1). Although this redundancy was statistically expected, one might suppose that the five distinct constructions that appeared more than once among clones would be “hot spots” for cloning. Nevertheless, one of those constructions, appearing ten times, is a chimera of three regions of the G. diazotrophicus PAL5 genome, and that combination with a determined configuration should be a rare event for a random cloning process. Thus, probably that redundancy is associated to the transfer process of the promoter library between host bacteria, and maybe in that case its high frequency should be a coincidence within the sample of clones analyzed.
Another observation was the presence of chimeras, which appeared six times among 21 distinct constructions (~29 %). That was acceptable considering that it could not damp our goal of isolating active promoters, although eventually it might difficult to precisely identify and localize them on the right place of the PAL5 genome.
Optimal conditions for β-galactosidase activity assays
Initial evaluations of β-galactosidase expression involving clones on DYGS medium supplemented with 100 mg·l−1 tetracycline and X-Gal resulted in faintly blue colonies, suggesting that other growth conditions, including other media and/or lower concentration of antibiotic, might favour reporter enzyme expression. Hence, the following tests aimed to choose appropriate culture conditions for the promoter expression/β-galactosidase activity assays. Two random clones previously presenting β-galactosidase activity on X-Gal-containing DYGS medium, arbitrarily named p1 and p2, plus the clone of the empty vector pPW452, and also non-transformed PAL5, were all plated on other media for G. diazotrophicus in the presence of two different concentrations of tetracycline (Fig. 2). On DYGS medium and under the lower concentration (10 mg·l−1) of tetracycline, β-galactosidase expression (based on blue stain intensity) seemed to be improved (Fig. 2a), when compared to the higher concentration of antibiotic (100 mg·l−1). Non-transformed PAL5 was unable to grow neither on DYGS nor BMS at 10 mg·l−1 tetracycline, in contrast to LGI, and acetic LGI, in which growth was observed under that antibiotic concentration. On the latter media, little or no blue staining at all was observed, and the same was true on BMS, probably due to the high levels of gum produced by the bacteria in this medium.
Evaluation of the adequate growth conditions to assay for β-galactosidase activity. Within each Petri plate, clockwise, non-transformed PAL5 (upper right), PAL5 (pPW452) (empty vector), PAL5 (p1), and PAL5 (p2) were plated on: a, b DYGS medium amended with 10 or 100 mg·l−1 tetracycline (Tc10 or Tc100), respectively; c, d BMS Tc10 or Tc100; e–j Acetic LGI with 20 mmol·l−1 NH4Cl, or 20 mmol·l−1 KNO3, or 20 mg·l−1 yeast extract as N source amended with Tc10 or Tc100, alternately; and k–p LGI with 20 mmol·l−1 NH4Cl, or 20 mmol·l−1 KNO3, or 20 mg·l−1 yeast extract as N source amended with Tc10 or Tc100, alternately
It is well known that MIC is not constant for a given antibiotic, as it can be affected by the growth medium composition, among other factors. Considering that DYGS containing 10 mg·l−1 tetracycline seemed attractive for β-galactosidase activity assays, the finding that non-transformed PAL5 was sensitive to that antibiotic concentration would guarantee the exclusive presence of transformed PAL5 cells in the bacterial culture under those conditions. Nevertheless, DYGS medium limits comparative studies of gene expression levels, as it is a complex medium. Thus, complex ingredients were removed from DYGS composition, and a simpler medium, LZGD (for assaying LacZ activity in G. d iazotrophicus), was developed and tested. First, MIC of tetracycline for G. diazotrophicus strain PAL5 was evaluated in liquid LZGD medium, and results suggested that 100 or even 10 g·l−1 tetracycline was more than enough to select transformants with vector pPW452 and derivatives carrying the promoter library (Fig. 3a). Then, the same four strains of Fig. 3a were plated on LZGD in the presence of two different concentrations of tetracycline and three different N sources (Fig. 3b–g). Similarly to DYGS medium, in LZGD under the lower concentration (10 mg·l−1) of tetracycline, β-galactosidase expression seemed to be improved, when compared to the higher concentration of the antibiotic (100 mg·l−1), especially with sodium glutamate as N source (which is also present in DYGS composition). However, in contrast to DYGS, in LZGD non-transformed PAL5 was capable to grow at 10 mg·l−1 tetracycline on solid medium, suggesting that a higher antibiotic concentration, such as 100 mg·l−1, was necessary to guarantee the exclusive presence of plasmid-bearing bacteria, a condition that avoids underestimation of β-galactosidase activity of the library clones. The contrasting results of tetracycline sensitivity between solid and liquid LZGD media (Fig. 3a, f) led to further investigation, which confirmed that non-transformed PAL5 is sensitive to Tc10 in liquid LZGD, when compared to the transformed strain (Fig. 3h). This was important for the subsequent β-galactosidase activity assays.
Viability of using LZGD medium for screening clones of the G. diazotrophicus PAL5 promoter library. a Minimal inhibitory concentration (MIC) of tetracycline (with 20 mmol·l−1 sodium glutamate as nitrogenous source). b–g Adequate growth conditions to assay for β-galactosidase activity with 20 mmol·l−1 NH4Cl, or 20 mmol·l−1 KNO3, or 20 mmol·l−1 sodium glutamate as N source amended with Tc10 or Tc100, alternately [within each Petri plate, clockwise: non-transformed PAL5 (upper right), PAL5 (pPW452) (empty vector), PAL5 (p1), and PAL5 (p2)]. h Growth profiles of PAL5 (pPW452) and PAL5 in LZGD medium with glucose or sugarcane juice as C source supplemented with 20 mmol·l−1 sodium glutamate as N source in the presence of 10 mg·l−1 tetracycline
To verify if the general tendencies observed for β-galactosidase activity among different solid media would be also detected among their liquid forms, 96 clones were grown in four liquid media for three days, and assayed for β-galactosidase activity using either the chromogenic or the fluorogenic substrates. Table 2 shows the results for an active clone only, suggesting quite the same tendencies observed on solid media. Therefore, for practical purposes, LZGD, a simplified form of DYGS medium, amended with 10 mg·l−1 tetracycline, has been considered a good option for β-galactosidase activity assays with the cost-effective ONPG substrate.
β-Galactosidase active clones
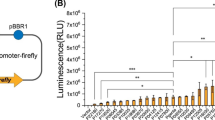
Following the approach of a previous work involving a mutant library of Herbaspirillum seropedicae strain Z78 (Schwab et al. 2007), the initial strategy to screen the G. diazotrophicus promoter library for β-galactosidase active clones was plating them on solid LZGD medium in the presence of X-Gal and observing development of blue colour. Then, clones qualitatively selected would be quantitatively assayed for β-galactosidase activity using ONPG or C2FDG, in order to statistically validate the colour-based observations. Our goal in the current work was to isolate promoters constitutively expressed and/or activated under a specific condition. A set of 480 clones (five 96-well plates from the promoter library) were plated on LZGD medium in the presence of X-Gal and supplemented with one of diverse carbon sources, all appropriate to support G. diazotrophicus growth (Li and MacRae 1991; Dong et al. 1995; Tejera et al. 2004; Barbosa et al. 2006). Alternatively, sterilized juice from two sugarcane varieties (either commercial or wild) was used as carbon source instead. Also, tetracycline was added at 10 mg·l−1, an antibiotic concentration that has resulted in better development of blue colour (Fig. 3f–g). There were only small differences in blue colour development among colonies grown under different carbon sources; apparently, this was due to gum production or non-uniform growth (Fig. 4). Nevertheless, 15 among 480 screened clones were selected for further characterization through β-galactosidase activity assays, because they seemed to present high enzymatic activity in the presence of at least one of the carbon sources tested. Results of those assays showed that two out of fifteen pre-selected clones had significantly increased activity when compared to the empty vector pPW452 (Fig. 5a). Clone pPW59-B09 (position B09 of the 96-well plate number 59) presented increased levels of β-galactosidase activity in the presence of organic acids and polyalcohols, and lower levels of enzymatic activity with glucose and the sugarcane juices. In turn, clone pPW13-D09 presented a similar pattern of enzymatic activity to pPW59-B09, except that, in the presence of glucose, its activity was the second highest among the carbon sources tested. Clone p2, which has been selected for its LacZ activity on solid media (Figs. 2, 3) and was included in the assays, presented intermediate levels of enzymatic activity when compared to both pPW59-B09 and pPW13-D09 clones in liquid medium supplemented with the carbon sources tested.
β-Galactosidase activity of 96 clones (plate PPW59) from the G. diazotrophicus promoter library on diverse carbon sources: a ethanol, b glucose, c mannitol, d sorbitol, e potassium tartrate, f potassium propionate, g sugarcane juice, commercial variety SP701143, and h sugarcane juice, wild variety Chunee; i and j, a zoom in two clones on sorbitol and potassium tartrate, respectively, illustrating different aspects assumed by the colonies on solid media with X-Gal
β-Galactosidase activity of clones grown in liquid media with the carbon sources indicated. a Clones pre-selected from the qualitative tests on X-Gal-containing solid media; clone p2 is the same as in Fig. 3 (see details in text). b A random sample of 96 clones (or a 96-well plate) from the promoter library. Error bars are standard deviation of four replicates of the enzymatic assays
Although the results of β-galactosidase activity assays permitted the identification of two highly active clones, they did not correspond well to the qualitative results on solid media: under this condition, most pre-selected clones seemed (through X-Gal-derived blue color) significantly active, but results with ONPG suggested that LacZ qualitative tests were quite elusive. Thus, screening of the promoter library for new highly active and/or regulated clones was performed through the more laborious β-galactosidase activity assays. To simplify the screening procedure, less growth conditions were tested. Once useful promoters would be those active when in contact with sugarcane compounds, as they might be used afterwards in planta for expression of target proteins, sterilized juice from a commercial variety was used in the growth medium to screen a 96-well plate of the promoter library, in comparison with glucose-containing medium. Using directly the quantitative procedure, two new clones showed high β-galactosidase activity under both conditions tested (Fig. 5b): pPW73-F07 and pPW73-B04.
Characterization of active promoters
Plasmids from clones presenting promoter activity, including both p1 and p2, were submitted to insert sequencing. All the six clones sequenced revealed distinct genomic regions (Fig. 6). Clone pPW73-B04 bears a fragment that spans from most of the 3′ region of ORF GDI_0566, which encodes a nitrate reductase, and part of its 3′ intergenic region, which would be the upstream region of the neighboring ORF GDI_0565, coding for a glyoxalase. A putative RpoD-dependent promoter was identified in that region, which probably is the glyoxalase gene promoter, and might be responsible for the LacZ activity detected in clone pPW73-B04.
G. diazotrophicus PAL5 genomic regions presenting promoter activity identified in this work. Transcriptional orientation of the ORFs (black arrows) in each representation of the genomic regions (above) is maintained in the respective representation of the transcriptional fusion with lacZ (white arrow) in the constructed plasmids (below). Restriction sites involved in cloning procedures are indicated. Annotated gene products (Bertalan et al. 2009) of the locus tags are: GDI_0565 = glyoxalase; GDI_0566 = nitrate reductase; GDI_1267 = putative coenzyme PQQ synthesis protein E; GDI_1623 = phytoene synthase; GDI_1624 = glycosyl transferase; GDI_2195 = hypothetical protein; GDI_0612 = AroD/3-dehydroquinate dehydratase; GDI_3657 = putative exported protein; GDI_3812 = putative signalling protein, GGDEF family; GDI_2606 = IstB-like ATP-binding protein; GDI_2607 = hypothetical protein. Sequences of putative RpoD-dependent promoters are shown in boxes, with characteristic −35/−10 regions underlined. Physical maps are not to scale
Clone pPW59-B09 contains the 3′-terminal of ORF GDI_1624, coding for a glycosyl transferase, and the 5′-terminal of GDI_1623, which encodes a phytoene synthase, in the opposite transcription orientation of lacZ. In spite of this apparently incongruent finding, significant levels of promoter activity were detected through β-galactosidase activity assays (Fig. 5a). Also, two putative promoter sequences were identified in the 3′-terminal of GDI_1624, and specially one is very similar to the consensus sequence of RpoD-type promoters (Lisser and Margalit 1993). Thus, it is possible that the lacZ gene in clone pPW59-B09 is transcribed from an antisense RNA (asRNA) promoter located in the complementary strand, 3′-terminus, of a glycosyl transferase gene.
Sequencing of clone pPW73-F07 revealed a chimera of two distinct regions of the G. diazotrophicus PAL5 genome, in which the mid part of the ORF GDI_2195, encoding a hypothetical protein, was fused to the mid part also of GDI_0612 or aroD, which codes for a 3-dehydroquinate dehydratase. The former has the same transcription orientation as lacZ, while the latter is in the opposing direction in the middle of the construction. A search for putative promoter sequences revealed two probable RpoD-type promoters within the GDI_0612 sequence, in the complementary strand, which resembles the situation of clone pPW59-B09, mentioned above. Hence, the lacZ gene in clone pPW73-F07 might be transcribed from an antisense RNA (asRNA) promoter within the aroD gene.
Clone pPW13-D09 comprehends ORF GDI_3657, which encodes a putative exported protein, and its upstream intergenic region, in which a putative RpoD-like promoter was found. Thus, the high levels of β-galactosidase activity detected in that clone (Fig. 5a) could be ascribed to this probable promoter.
Clone p1, which was randomly selected for previously presenting β-galactosidase activity on X-Gal-containing DYGS medium, contains the upstream region plus the 5′-terminal of ORF GDI_3812, coding for a diguanylate cyclase. Two putative, overlapping RpoD-type promoters, and also a typical RpoE-like promoter (not shown), were found upstream to that ORF, so probably lacZ gene in p1 clone would be transcribed from at least one of those promoters.
Finally, clone p2 bears most part of the 5′-terminus of ORF GDI_2607, which encodes a hypothetical protein, and a 3′ fragment of its neighbor ORF GDI_2606, coding for an IstB-like ATP-binding protein, including an intergenic region between both ORFs. Those ORFs are in the opposite transcription orientation to lacZ. Putative RpoD-type promoters were found in the complementary 3′-terminus of GDI_2606 and the intergenic region, both in the same transcription orientation as lacZ. That finding suggests another situation similar to clones pPW73-F07 and pPW59-B09, so the lacZ gene in clone p2 would be transcribed from an antisense RNA (asRNA) promoter located in the intergenic region and/or the 3′-terminus of ORF GDI_2606.
Discussion
Transcriptional promoters have been isolated and characterized from diverse bacteria for scientific and/or technological purposes. In this work we isolated and characterized six G. diazotrophicus PAL5 promoters with variable expression strengths that might be used for expressing useful proteins: while the promoter of clone pPW73-F07 showed the highest levels of LacZ expression through the enzymatic assays, the other five showed to be moderate to weak promoters. We used a promoter-less lacZ-bearing vector to construct a promoter library in PAL5 cells, and identified those active promoters through β-galactosidase activity assays, sequencing and localization in the bacterial genome. Interestingly, three out of the six clones presented promoters probably involved in transcription of antisense RNA, including that presenting the highest expression strength.
Characterization in this work of such a proportion of antisense promoters in PAL5 leads to infer that antisense RNA is omnipresent in G. diazotrophicus. In diverse bacteria, promoters located in the complementary strand of genes drive transcription of antisense RNAs with regulatory functions (Georg and Hess 2011; Sesto et al. 2012). Those molecules have important roles in nutrient metabolism, stress responses, toxin synthesis, and bacterial pathogenesis. In the plant pathogen Agrobacterium tumefaciens, a deep sequencing survey showed the presence of 76 cis-antisense sRNAs, suggesting considerable antisense transcription in that alpha-proteobacterium (Wilms et al. 2012). In another alpha-proteobacterium, the symbiotic nitrogen-fixing Sinorhizobium meliloti, a genome-wide survey revealed 117 cis-encoded antisense sRNAs (Schlüter et al. 2010). To our knowledge, there are no reports of the presence of those molecules in G. diazotrophicus or any other member of the Acetobacteraceae, an alpha-proteobacterial family. Also, recent reports have mapped transcription start sites (TSSs) to unexpected locations in bacterial genomes, including the non-coding strand, a phenomenon that has been termed as pervasive transcription (Wade and Grainger 2014). In E. coli, it has been demonstrated that there are more than 1000 intragenic TSSs that are positioned in the antisense orientation (Dornenburg et al. 2010). The genome of G. diazotrophicus strain PAL5 encodes diverse sigma subunits of RNA polymerase that might be involved in pervasive transcription (Table 3); also, proteins probably suppressive of transcription were annotated. A further investigation should be conducted in G. diazotrophicus in order to confirm the presence and function of antisense RNA, and to study possible regulatory mechanisms governing transcription from their promoters. For that purpose, transcriptional profiling through RNA-Seq could be a good choice, taking advantage of having the bacterial genome sequence available for mapping of sequencing reads.
Genome sequencing of G. diazotrophicus strain PAL5 by two independent groups (Bertalan et al. 2009; Giongo et al. 2010) has opened room to further genetic and functional genomics analysis of bacterial traits related to plant association and growth promotion. For instance, genome surveys has revealed the presence, and permitted genetic characterization, of: (1) a gum gene cluster, for which has been shown that its product, exopolysaccharide, is required for biofilm formation and plant colonization (Meneses et al. 2011); (2) homologs to genes from the alternative asparagine biosynthesis pathway, which affects nitrogenase activity and thus might be essential to bacterial survival during interaction with plant (Alquéres et al. 2012); (3) luxR and luxI homologs composing a quorum sensing system, which has a role on the expression of a number of proteins including few probably involved in microbial interactions, host colonization and stress survival (Bertini et al. 2014); (4) homologs for reactive oxygen species (ROS)-detoxifying enzymes, including superoxide dismutase and glutathione reductase, which play an important role in the endophytic colonization of rice plants (Alquéres et al. 2013); and (5) several genes probably involved in iron metabolism, including tonB, which has a role in nitrogenase activity and biofilm formation capability, beyond siderophore accumulation (de Paula Soares et al. 2015). Those studies have been assisted by molecular tools, previously developed, such as the use of suite vectors for labelling PAL5 strain in plant association studies (Rouws et al. 2010), and/or a transposon-mediated mutagenesis system in genetic characterization studies (Rouws et al. 2008). However, there was a necessity of expression systems for availing on genetic characterization studies and/or developing useful genetically modified strains. By using one of the six promoters characterized here with a determined expression strength, a protein (gene) of interest might be cloned just downstream to one of those promoters already cloned in the pPW452 plasmid; alternatively, a certain promoter might be easily isolated through digestion with HindIII enzyme, which has two cleavage sites flanking the multicloning site of that vector, and cloned into another vector bearing the protein (gene) of interest.
Gene expression systems have been developed in more or less extent for acetic acid bacteria (Acetobacteraceae), the family in which G. diazotrophicus is included (Saichana et al. 2014). For the acetobacterium Gluconobacter oxydans, development of those systems is well advanced, thanks to active promoters that had been characterized (Kallnik et al. 2010; Shi et al. 2014). As concluded by those authors, the G. oxydans promoters could be used as a tool for overexpression of enzymes involved in industrial production processes, increasing even more the bacterial potential in industrial bioconversion. Similarly, the approach used in the current work permitted to characterize active promoters of G. diazotrophicus strain PAL5, with potential to be used in expression systems for useful proteins in agriculture. Among these, beyond proteins already mentioned above and previously characterized in PAL5 strain, we anticipate Cry toxins from B. thuringiensis, as already described with Cry1Ac (Subashini et al. 2011; Rapulana and Bouwer 2013). Also, induced homologous expression of enzymes involved in phytohormone production (Bastián et al. 1998), antagonistic activity for phytopathogens (Blanco et al. 2005; Saravanan et al. 2007), and improvement of nutrient utilization by the plant (Maheshkumar et al. 1999; Saravanan et al. 2007; Logeshwaran et al. 2009) might also be expressed using engineered genetic constructions.
References
Alquéres SM, Cardoso AM, Brito-Moreira J et al (2012) Transfer RNA-dependent asparagine biosynthesis in Gluconacetobacter diazotrophicus and its influence on biological nitrogen fixation. Plant Soil 356:209–216
Alquéres S, Meneses CHSG, Rouws L et al (2013) The bacterial superoxide dismutase and gluthatione reductase are crucial for endophytic colonization of rice roots by Gluconacetobacter diazotrophicus strain PAL5. Mol Plant Microbe Interact 26:937–945
Baldani JI, Baldani VLD (2005) History on the biological nitrogen fixation research in graminaceous plants: special emphasis on the Brazilian experience. An Acad Bras Ciênc 77:549–579
Baldani J, Krieg NR, Baldani VLD et al (2005) Genus II. Azospirillum. In: Brenner DJ, Krieg NR, Staley JT (eds) Bergey’s manual® of systematic bacteriology, 2nd edn. Springer, New York, pp 22–24
Barbosa EA, Perin L, Reis VM (2006) Uso de diferentes fontes de carbono por estirpes de Gluconacetobacter diazotrophicus isoladas de cana-de-açúcar. Pesqui Agropecu Bras 41:827–833
Bastián F, Cohen A, Piccoli P et al (1998) Production of indole-3-acetic acid and gibberellins A1 and A3 by Acetobacter diazotrophicus and Herbaspirillum seropedicae in chemically-defined culture media. Plant Growth Regul 24:7–11
Bertalan M, Albano R, de Pádua V et al (2009) Complete genome sequence of the sugarcane nitrogen-fixing endophyte Gluconacetobacter diazotrophicus Pal5. BMC Genom 10:450
Bertini EV, Peñalver CGN, Leguina AC et al (2014) Gluconacetobacter diazotrophicus PAL5 possesses an active quorum sensing regulatory system. Antonie Van Leeuwenhoek 106:497–506
Blanco Y, Blanch M, Piñón D et al (2005) Antagonism of Gluconacetobacter diazotrophicus (a sugarcane endosymbiont) against Xanthomonas albilineans (pathogen) studied in alginate-immobilized sugarcane stalk tissues. J Biosci Bioeng 99:366–371
Boddey RM, Urquiaga S, Alves BJR, Reis V (2003) Endophytic nitrogen fixation in sugarcane: present knowledge and future applications. Plant Soil 252:139–149
Cavalcante VA, Döbereiner J (1988) A new acid-tolerant nitrogen-fixing bacterium associated with sugarcane. Plant Soil 108:23–31
Clarke L, Carbon J (1976) A colony bank containing synthetic CoI EI hybrid plasmids representative of the entire E. coli genome. Cell 9:91–99
de Paula Soares C, Rodrigues EP, de Paula Ferreira J et al (2015) Tn5 insertion in the tonB gene promoter affects iron-related phenotypes and increases extracellular siderophore levels in Gluconacetobacter diazotrophicus. Arch Microbiol 197:223–233
Dong Z, Heydrich M, Bernard K, McCully ME (1995) Further evidence that the N2-fixing endophytic bacterium from the intercellular spaces of sugarcane stems is Acetobacter diazotrophicus. Appl Environ Microbiol 61:1843–1846
Dornenburg JE, DeVita AM, Palumbo MJ, Wade JT (2010) Widespread antisense transcription in Escherichia coli. MBio 1:e00024-10
Doyle JJ (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Georg J, Hess WR (2011) cis-antisense RNA, another level of gene regulation in bacteria. Microbiol Mol Biol Rev 75:286–300
Gillis M, Kersters K, Hoste B et al (1989) Acetobacter diazotrophicus sp. nov., a nitrogen-fixing acetic acid bacterium associated with sugarcane. Int J Syst Bacteriol 39:361–364
Giongo A, Tyler HL, Zipperer UN, Triplett EW (2010) Two genome sequences of the same bacterial strain, Gluconacetobacter diazotrophicus PAl 5, suggest a new standard in genome sequence submission. Stand Genomic Sci 2:309–317. doi:10.4056/sigs.972221
Griffith KL, Wolf RE (2002) Measuring β-galactosidase activity in bacteria: cell growth, permeabilization, and enzyme assays in 96-well arrays. Biochem Biophys Res Commun 290:397–402
Haapalainen M, Karp M, Metzler MC (1996) Isolation of strong promoters from Clavibacter xyli subsp. cynodontis using a promoter probe plasmid. Biochim Biophys Acta 1305:130–134
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J Mol Biol 166:557–580
Ish-Horowicz D, Burke JF (1981) Rapid and efficient cosmid cloning. Nucleic Acids Res 9:2898–2989
Kallnik V, Meyer M, Deppenmeier U, Schweiger P (2010) Construction of expression vectors for protein production in Gluconobacter oxydans. J Biotechnol 150:460–465
Li RP, MacRae IC (1991) Specific association of diazotrophic acetobacters with sugarcane. Soil Biol Biochem 23:999–1002
Lisser S, Margalit H (1993) Compilation of E. coli mRNA promoter sequences. Nucleic Acids Res 21:1507–1516
Logeshwaran P, Thangaraju M, Rajasundari K (2009) Hydroxamate siderophores of endophytic bacteria Gluconacetobacter diazotrophicus isolated from sugarcane roots. Aust J Basic Appl Sci 3:3564–3567
Maheshkumar KS, Krishnaraj PU, Alagawadi AR (1999) Mineral phosphate solubilizing activity of Acetobacter diazotrophicus: a bacterium associated with sugarcane. Curr Sci 76:874–875
Meneses CHSG, Rouws LFM, Simões-Araújo JL et al (2011) Exopolysaccharide production is required for biofilm formation and plant colonization by the nitrogen-fixing endophyte Gluconacetobacter diazotrophicus. Mol Plant Microbe Interact 24:1448–1458
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Oliveira ALM, Urquiaga S, Döbereiner J, Baldani JI (2002) The effect of inoculating endophytic N2-fixing bacteria on micropropagated sugarcane plants. Plant Soil 242:205–215. doi:10.1023/A:1016249704336
Oliveira ALM, Canuto EL, Urquiaga S et al (2006) Yield of micropropagated sugarcane varieties in different soil types following inoculation with diazotrophic bacteria. Plant Soil 284:23–32. doi:10.1007/s11104-006-0025-0
Pothier JF, Wisniewski-Dyé F, Weiss-Gayet M et al (2007) Promoter-trap identification of wheat seed extract-induced genes in the plant-growth-promoting rhizobacterium Azospirillum brasilense Sp245. Microbiology 153:3608–3622
Ramírez-Romero MA, Masulis I, Cevallos MA et al (2006) The Rhizobium etli σ70 (SigA) factor recognizes a lax consensus promoter. Nucleic Acids Res 34:1470–1480
Rapulana T, Bouwer G (2013) Toxicity to Eldana saccharina of a recombinant Gluconacetobacter diazotrophicus strain carrying a truncated Bacillus thuringiensis cry1Ac gene. Afr J Microbiol Res 7:1207–1214
Rodrigues Neto J, Malavolta VA Jr, Victor O (1986) Meio simples para o isolamento e cultivo de Xanthomonas campestris pv. citri tipo B. Summa Phytopathol 12:32
Rouws LFM, Simões-Araújo JL, Hemerly AS, Baldani JI (2008) Validation of a Tn5 transposon mutagenesis system for Gluconacetobacter diazotrophicus through characterization of a flagellar mutant. Arch Microbiol 189:397–405
Rouws LFM, Meneses CHSG, Guedes HV et al (2010) Monitoring the colonization of sugarcane and rice plants by the endophytic diazotrophic bacterium Gluconacetobacter diazotrophicus marked with gfp and gusA reporter genes. Lett Appl Microbiol 51:325–330. doi:10.1111/j.1472-765X.2010.02899.x
Rowland B, Purkayastha A, Monserrat C et al (1999) Fluorescence-based detection of lacZ reporter gene expression in intact and viable bacteria including Mycobacterium species. FEMS Microbiol Lett 179:317–325
Saichana N, Matsushita K, Adachi O et al (2014) Acetic acid bacteria: a group of bacteria with versatile biotechnological applications. Biotechnol Adv. doi:10.1016/j.biotechadv.2014.12.001
Saravanan VS, Kalaiarasan P, Madhaiyan M, Thangaraju M (2007) Solubilization of insoluble zinc compounds by Gluconacetobacter diazotrophicus and the detrimental action of zinc ion (Zn2+) and zinc chelates on root knot nematode Meloidogyne incognita. Lett Appl Microbiol 44:235–241
Schlüter J-P, Reinkensmeier J, Daschkey S et al (2010) A genome-wide survey of sRNAs in the symbiotic nitrogen-fixing alpha-proteobacterium Sinorhizobium meliloti. BMC Genom 11:245
Schwab S, Ramos HJ, Souza EM et al (2007) Identification of NH4 +-regulated genes of Herbaspirillum seropedicae by random insertional mutagenesis. Arch Microbiol 187:379–386
Sesto N, Wurtzel O, Archambaud C et al (2012) The excludon: a new concept in bacterial antisense RNA-mediated gene regulation. Nat Rev Microbiol 11:75–82
Sevilla M, Burris RH, Gunapala N, Kennedy C (2001) Comparison of benefit to sugarcane plant growth and 15N2 incorporation following inoculation of sterile plants with Acetobacter diazotrophicus wild-type and nif mutant strains. Mol Plant Microbe Interact 14:358–366
Shi L, Li K, Zhang H et al (2014) Identification of a novel promoter gHp0169 for gene expression in Gluconobacter oxydans. J Biotechnol 175:69–74
Sievers M, Swings J (2005) Genus VIII. Gluconacetobacter. In: Brenner DJ, Krieg NR, Staley JT (eds) Bergey’s manual® of systematic bacteriology, 2nd edn. Springer, New York, pp 72–77
Spaink HP, Okker RJH, Wijffelman CA et al (1987) Promoters in the nodulation region of the Rhizobium leguminosarum Sym plasmid pRL1JI. Plant Mol Biol 9:27–39
Subashini M, Moushumi Priya A, Sundarakrishnan B, Jayachandran S (2011) Recombinant Gluconacetobacter diazotrophicus containing cry1Ac gene codes for 130-kDa toxin protein. J Mol Microbiol Biotechnol 20:236–242
Teixeira KRS, Wülling M, Morgan T et al (1999) Molecular analysis of the chromosomal region encoding the nifA and nifB genes of Acetobacter diazotrophicus. FEMS Microbiol Lett 176:301–309
Tejera NA, Ortega E, Rodés R, Lluch C (2004) Influence of carbon and nitrogen sources on growth, nitrogenase activity, and carbon metabolism of Gluconacetobacter diazotrophicus. Can J Microbiol 50:745–750
Wade JT, Grainger DC (2014) Pervasive transcription: illuminating the dark matter of bacterial transcriptomes. Nat Rev Microbiol 12:647–653
Wilms I, Overlöper A, Nowrousian M et al (2012) Deep sequencing uncovers numerous small RNAs on all four replicons of the plant pathogen Agrobacterium tumefaciens. RNA Biol 9:446–457
Acknowledgments
The authors acknowledge Luc Rouws for critical revision of this manuscript, Janaína Rouws and Daniel Ferreira for statistical considerations in constructing the promoter library, and Emanuel M. de Souza for kindly providing vector pPW452. This work has been funded by The National Council for Scientific and Technological Development (CNPq—“National Institute of Science and Technology of Biological Nitrogen Fixation”, grant nº 573.828/2008-3), Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (Faperj—A.A.L.B. “Iniciação Científica” fellowship, nº E-26/200.930/2015, and grant “Desenvolvimento Científico e Tecnológico Regional”, nº E-26/110.235/2011), and Brazilian Agricultural Research Corporation (Embrapa—Infoseg nº 03.07.09.026.00.00).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jorge Membrillo-Hernández.
Kátia Regina dos Santos Teixeira: In memoriam.
Rights and permissions
About this article
Cite this article
Schwab, S., Pessoa, C.A., de Lima Bergami, A.A. et al. Isolation and characterization of active promoters from Gluconacetobacter diazotrophicus strain PAL5 using a promoter-trapping plasmid. Arch Microbiol 198, 445–458 (2016). https://doi.org/10.1007/s00203-016-1203-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-016-1203-y