Abstract
Summary
Several types of nutrients and foods affect bone mineral density (BMD). However, these nutrients occur together in food groups and dietary patterns, and the overall effects of dietary patterns are not yet well known.
Introduction
We evaluated the associations between dietary patterns and BMD among adults participating in the Health Workers Cohort Study.
Methods
In a cross-sectional analysis, we examined 6915 Mexican adults aged 20–80 years. All participants completed a validated self-administered food frequency questionnaire (FFQ) and had total, hip, and spine BMD measurements assessed by dual-energy X-ray absorptiometry (DXA). The FFQ included 116 foods, which were grouped into 22 categories and entered into a factor analysis to derive dietary patterns.
Results
Three dietary patterns emerged—a Prudent, a Refined foods, and a Dairy and fish pattern. After adjustment for potential confounders, those in the highest quintile of the Prudent pattern had lower odds (OR) of low spine BMD (OR = 0.80; 95 % CI 0.68, 0.94; P for trend = 0.031) compared to those in the lowest quintile. In contrast, participants in the highest quintile of the Refined foods pattern had greater odds of low total BMD (OR = 1.74; 95 % CI 1.10, 2.76; P for trend = 0.016) than those in the lowest quintile. Finally, participants in the highest quintile of the Dairy and fish dietary pattern had significantly lower likelihood of having low BMD.
Conclusion
This study identified specific dietary patterns associated with BMD among a Mexican adult population and highlights the importance of promoting food-based prevention strategies for maintaining bone health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis (OP) is the most common skeletal disease in humans, affecting many individuals, regardless of race or gender, with prevalence increasing with age [1]. OP and related fractures are well recognized as a major public health concern in developed countries and are on the rise in the developing world [2]. Additionally, OP and related fractures contribute to high health care costs. In Mexico, the prevalence of OP among men and women 50 years of age and older is considerable (lumbar spine 17.0 and 9.0 % in women and men, respectively; femoral 16.0 and 6.0 % in women and men, respectively) [3], with an estimated lifetime risk of hip fracture of 8.5 % for women and 3.8 % for men [4]. In 2010, the cost of managing osteopenia and osteoporosis was estimated at 155 million USD, and total costs are expected to increase by 41.7 % by 2020 [5]. The prevention of bone loss is, thus, desirable for both medical and economic reasons.
Bone mineral density (BMD) is affected by genetic, endocrine, mechanical, and lifestyle factors [6]. Of these, diet is one of the most important modifiable risk factors [7]. Until recently, the most common nutritional epidemiologic approach to assessing the relationship between diet and bone health has focused on individual nutrients, particularly calcium, and vitamin D [8–11]. Other nutrients and dietary components, such as phosphorus, potassium, magnesium, vitamin K, several trace minerals [10–12], and fruit and vegetables [11] have also shown beneficial effects. Although this traditional analysis has been valuable for understanding the association between specific nutrients and bone health, there are still gaps in knowledge and several conceptual limitations. As people eat mixtures of foods that contain a complex combination of nutrients, the single nutrient approach may not take into consideration complicated or cumulative intercorrelations and interactions between nutrients, which could lead to inaccurate inferences/conclusions [13, 14]. To address this limitation, a holistic dietary methodology (the measurement of overall diet as a dietary pattern) has been increasingly used to elucidate the relationship between diet and disease [15, 16]. Using this multifactorial approach, interactions among food and nutrients and the associations of some confounding factors are controlled for [17], and the etiologic role of dietary behavior and disease can be better explained [13].
Although several previous studies [14, 18–22] have examined the association of dietary patterns and BMD, with most focusing on adults and the elderly, the findings are contradictory. Data from Tucker [14], Okubo [18], Hardcastle [20], and McNaughton [21] suggest a negative relationship between an “unhealthy or Western dietary pattern” (characterized by red and processed meat, fats, and sweets) and BMD, and a positive association between a “healthy or prudent dietary pattern” (dietary pattern represented by high consumption of fish, olive oil, fruits and vegetables, and low consumption of red meat and candy) and BMD [14, 18, 20, 21]. Others have failed to show such associations [19, 22]. In Mexico, several studies have evaluated the relationship between dietary patterns and other health outcomes [23, 24]; however, no previous study has evaluated the association of dietary patterns to bone health in a Mexican population. The unique characteristics of the Mexican diet and the rapid nutrition transition [25] taking place in Mexico necessitates close investigation of this issue. Therefore, the aim of this research was to examine associations between dietary patterns and BMD among adults participating in the Health Workers Cohort Study.
Materials and methods
Study population
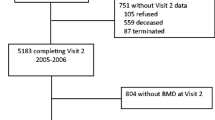
A cross-sectional analysis was conducted using data from participants in the baseline assessment of the Health Workers Cohort Study (HWCS). Details of the study design and methodology, as well as the baseline characteristics of the study population, have been published previously [23]. In brief, the HWCS focuses on lifestyle and chronic diseases. It was initiated in 2004 with the enrollment of 10,769 employees and their relatives from three health and academic institutions in Morelos and other Mexico states. For the present analysis, we excluded participants aged <20 or >80 years (n = 1557), those with >10 % blank items on their food intake information (n = 642), or with daily energy intake values outside of 600–7000 kcal/day (n = 114), determined with the standard deviation method suggested by Rosner [26]. Additionally, we excluded participants with incomplete bone mineral density (n = 1306) or with missing information (n = 235) on other important covariates (tobacco, body mass index (BMI), use of supplements and, for women, estrogen use). A total of 6915 (1948 men and 4967 women) were included in our final analysis.
This study was managed according to the Declaration of Helsinki guidelines. The ethics committees of all participating institutions [Comité de Ética e Investigación, Instituto Mexicano del Seguro Social (No. 12CEI0900614); Comité de Ética e Investigación, Instituto Nacional de Salud Pública (No. 13CEI1700736); Comité de Ética, Centro de Investigación en Ciencias Médicas (No. 1233008X0236)] reviewed and approved the study protocol and informed consent forms.
Dietary intake
Usual dietary intake was assessed using a semi-quantitative, 116-item food frequency questionnaire (FFQ) [27]. The FFQ was previously validated by being administered twice, at a 1-year interval, to 134 women residing in Mexico City; the results were then compared with those from a set of four dietary recalls given at 3-month intervals. The FFQ describes the frequency of consumption of foods during the previous year. The intake frequency reported for each food was converted to portions per day. To calculate energy intake (kcal/day), and micronutrient intakes, the daily frequency of consumption (portions/day) of each food was multiplied by the food’s energy or micronutrient content [28]. Finally, use of specific types and brands of multivitamins was determined by asking about the weekly number of multivitamins taken.
Dietary pattern derivation
Energy consumed from each food was converted into a proportion of the total consumption of energy per day, and subsequently standardized using Z-scores [29, 30]. The foods and beverages on the questionnaire were categorized into 22 food groups, which were used as the basis for the dietary pattern derivation. In short, the criteria for assigning a food to a particular food group were similarity of nutrient content (e.g., fat, protein, carbohydrate) and dietary fiber. Other groups were classified based on their profile of fatty acids (e.g., vegetable oils). Finally, some individual foods were considered to be groups in and of themselves because of their unique nutritional composition and/or frequency of consumption (e.g., corn tortillas and Mexican foods, potatoes, eggs, tomato juice) (Online Resource 1).
Dietary patterns were derived with principal components analysis of the food groups [30]. The resulting factors were orthogonally rotated (varimax rotation) to improve their interpretation. The number of factors retained was determined by eigenvalues >1.5, screen plots, and interpretability. In addition, each factor was defined by a subset of at least five food groups with an absolute factor loading ≥0.2 (considering that ≥0.2 factor loadings contributed significantly to the dietary pattern) as suggested in previous analyses [23, 24, 29]. The factor scores for each dietary pattern were estimated by adding the consumption of the food groups weighted by their factor loading, and each participant received a score for each of the identified patterns.
Measurement of bone mineral density
A trained technician used a Lunar DPX NT densitometer to perform dual-energy X-ray absorptiometry (DXA) to measure the bone mineral density (BMD) of the hip (femoral neck and total hip), the lumbar spine (L1-L4), and whole body. International Society of Clinical Densitometry (ISCD) procedures were strictly followed. Standard calibration of instruments was performed daily using the phantom provided by the manufacturer; technicians ensured that the daily coefficient of variation (CV) was within normal operational standards and that the in vivo CV was lower than 1.5 %. Results obtained by DXA are expressed as grams of hydroxyapatite per square centimeter (WHO Technical Report Series: Prevention and Management of Osteoporosis). Therefore, calculation of a BMD T-score was adopted, which represents the comparison between the individual’s BMD and the mean BMD of the healthy young adult population of the same age and sex. For the present analysis, low BMD (osteopenia/osteoporosis) was defined as ≤−1.0 T-score [31].
Measurement of confounders
Information on sociodemographic characteristics, medical history, supplement use (yes or no), current use of hormone replacement therapy (yes or no), menopausal status, parity (times, continuous), age at menarche, as well as lifestyle variables, including consumption of alcohol (g/d), tobacco (never, past or current), and physical activity level, were collected with self-administered questionnaires. Physical activity was assessed using a validated physical activity questionnaire, adapted for use in a Mexican population [32]. Participants reported the time they spent each week on activities such as running and walking, during a typical week in the previous year. Each activity was given a value in metabolic equivalent tasks (METs) and total METs/week were computed.
Body weight was measured with a calibrated electronic scale (model BC-533; Tanita, Tokyo, Japan), with minimal clothing and no shoes. Height was measured using a conventional stadiometer (SECA brand), on barefoot participants standing with their shoulders in a normal position. Measurements were taken with the tape in a horizontal plane perpendicular to the vertical scale, touching the top of the head at the moment of inspiration. BMI was computed as a ratio of weight in kilograms divided by height in meters squared. The definition for normal weight was BMI <25.0, participants with BMI ≥25.0–<30.0 were classified as overweight, and those with BMI ≥30.0 were classified as obese.
Statistical analysis
We performed descriptive analysis of the principal characteristics of the study population across each of the dietary patterns, grouped into quintiles. Continuous variables are presented as means and standard deviations, and categorical variables as percentages and 95 % confidence intervals. To investigate differences in participant characteristics, we compared continuous variables using ANOVA, and categorical variables using chi-square tests.
Odds ratios (OR) and their 95 % confidence interval (95 % CIs) were estimated using multivariate logistic regression analysis to assess the magnitude of the association between specific dietary patterns and the likelihood of having low bone mineral density (BMD), adjusting for age (continuous), sex, physical activity (MET/day), use of multivitamin supplements (yes/no), cigarette smoking (never, past, or current), estrogen use (yes/no), menopausal status (yes/no), parity (times, continuous), age at menarche, BMI (kg/m2), and energy intake (kcal/day). In all multivariate models, the first quintile of the dietary pattern was considered the reference category. Sensitivity analyses were conducted to repeat models by age (20–50 and >50 years) and sex groups. The Mantel-Haenszel extension chi-square test was used to assess the overall trend across increasing quintiles of dietary pattern scores. All P values shown are two-tailed; P < 0.02 was considered statistically significant; in this particular case, the P value was adjusted to reduce the chance of incorrectly declaring a statistical significance. All statistical analyses were carried out using the statistical software Stata, version 14.0 (StataCorp, College Station, TX, USA).
Results
We entered food intake data for 22 predefined food groups into the factor analysis procedure, using principal components. A three-factor solution was obtained, which explained 26.1 % of the total variance (Table 1). The patterns were labeled according to the food groups with high loadings. The first factor, which accounted for 9.8 % of the variance, loaded positively on fresh vegetables (0.68), fruit (0.53) tomato (0.52), oils (0.40), legumes (0.28), fish (0.25), and whole grains (0.23), and negatively on refined grains (−0.55), soft drinks (−0.25), and other sweetened beverages (−0.25), and was labeled as a “Prudent” dietary pattern. Factor two, which we labeled as “Refined foods”, showed positive loadings for red meat, fats, sugar and sweets, soft drinks, eggs, refined grains, alcoholic beverages, and negative loadings for milk and fresh fruit, accounting for 8.3 % of the variance. Lastly, the third factor, defined as “Dairy and fish,” accounted for 8.0 % of the total variance and was characterized by positive loadings for fish and other seafood (0.45), dairy foods (0.45), whole grains (0.38), and milk (0.30) and negative loadings for corn tortillas and Mexican food (−0.65), legumes (−0.46), and refined grains (−0.25).
Participants were divided into quintiles by the factor score of each dietary pattern. Frequencies and means were calculated across quintiles. Sample characteristics of the Health Workers Cohort Study participants, across quintiles (Q1, Q3, and Q5) of each dietary pattern, are shown in Table 2. Those in the highest quintile of the “Prudent” dietary pattern tended to be older, female, to never have smoked, to exercise, and to use multivitamins, relative to those in the lowest quintile. Participants in the highest quintile of the “Refined foods” dietary pattern tended to be younger, male, to smoke, and to be less likely to use multivitamins. They had higher intakes of carbohydrates and lower intakes of vitamin D, calcium, vitamin K, vitamin C, magnesium, and potassium. Participants in the highest quintile of the “Dairy and fish” dietary pattern were younger, more likely to be women, and to less likely to be overweight/obese than those in the lowest quintile. These participants also had relatively higher intakes of protein, vitamin D, calcium, vitamin C, vitamin K, magnesium, and potassium, and lower intakes of carbohydrate than those in the lowest quintile.
After adjusting for age, sex, physical activity, use of multivitamin supplements, cigarette smoking, estrogen use, menopausal status, parity, age at menarche, BMI, and energy intake, those in the highest category of the “Dairy and fish” dietary pattern had significantly lower odds of having low total BMD (OR = 0.51; 95 % CI 0.40–0.66; P for trend = <0.001), or low spine BMD (OR = 0.69; 95 % CI 0.56–0.85; P for trend = <0.001), compared to those in the lowest category (Table 3). Participants in the highest quintile of the “Refined foods” dietary pattern had significantly higher likelihood of having low whole body (OR = 1.74; 95 % CI 1.10–2.76; P for trend = 0.016), hip (OR = 1.91; 95 % CI 1.19–3.04; P for trend <0.01), and lumbar spine BMD (OR = 1.61; 95 % CI 1.06–2.45; P for trend = 0.027), compared with those in the lowest quintile. Finally, those in the highest category of the “Prudent” dietary pattern had lower odds of having low BMD at the hip (OR = 0.71; 95 %CI 0.44–0.97) and lumbar spine (OR = 0.80; 95 %CI 0.68–0.94), relative to those in the lowest quintile (Table 3). Sensitivity analyses by age and sex showed similar patterns of results to those for the full sample (data not shown).
Discussion
Using factor analysis, an approach that considers overall eating patterns, we identified three distinct dietary patterns in this adult Mexican population, and found that these dietary patterns were associated with BMD. Our findings suggest that individuals with a diet high in dairy foods, fish and other seafood, and whole grains, and low in refined grains and soft drinks (Dairy and fish dietary pattern) had lower probability of having low BMD. In contrast, those consuming diets high in refined grains, sugar and sweets, soft drinks, red meat, fats, and alcoholic beverages had significantly higher odds of low BMD. Finally, the “Prudent” pattern, characterized by high intake of fruit, vegetables, and whole grains, showed a positive significantly protective association with BMD.
Our findings of significant associations between dietary patterns and BMD are in line with those observed in previous studies. We identified a “Refined foods” dietary pattern represented by high consumption of refined grains, sugar and sweets, soft drinks, red meat, fats, and alcoholic beverages, which is similar to dietary patterns identified in other studies [14, 21, 22, 33]. For example, McNaughton and coworkers [21], identified a “high-energy nutrient-poor” pattern characterized by high intakes of refined cereals, soft drinks, fried potatoes, processed meat, and beer, and low consumption of vegetables, fruit and whole grains cereals, which was significantly inversely associated with total body BMD (β = −15.4; 95 %CI −27.4, −3.3). Similarly, the “Candy” pattern, observed by Tucker et al. [14], in the Framingham Cohort Study, found that a diet rich in refined foods and lacking in nutrient-dense foods may be detrimental to bone health in men. Furthermore, a recent study conducted in Brazil found that a “sweet foods, coffee, and tea” pattern was inversely correlated with BMD [34]. Additionally, our “Refined foods” pattern was also represented by high intakes of meat and fats, comparable to the “Western” dietary pattern identified by Okubo [18], which showed a borderline inverse association with BMD among Japanese women.
Most dietary pattern studies have revealed positive and significant associations between a diet rich in vegetables and/or fruit and BMD [14, 18, 20–22]. Tucker et al. [11] found that intakes of fruit and vegetables were positively correlated with BMD in both elderly men and women. In accordance with previous findings, a similar association was observed between our prudent dietary pattern and BMD. Despite many observational studies [14, 18, 20–22] have found a positive relationship between vegetabes and/or fruit intakes with BMD, Macdonald et al. [35] suggested that health benefits of vegetables and fruits may be limited to people who eat little or no vegetables and fruits, but apparently no added benefit for those who are already eating a few portions a day.
In this study, a third pattern, characterized by high intakes of milk, other dairy products, whole grains, and fish emerged. This dietary pattern was related with 49 % lower odds of low total BMD and 31 % lower odds of low spine BMD. This finding is consistent with a Korean study conducted in adolescents [36], which detected a significant relationship between a “milk and cereal” pattern and BMD. Additionally, a study of postmenopausal women found that those in the highest quintile of a “dairy and fruit” pattern had 53 % lower odds of osteoporosis of the lumbar spine (53 %), compared with those in the lowest quintile (OR = 0.47; 95 % CI 0.35, 0.65; P for trend = 0.001).
Our study has a number of strengths; to our knowledge, this is the first study to evaluate the association of dietary patterns and BMD in a Mexican adult population. Furthermore, the similarity of some of the dietary patterns in the present study with those identified in previous research simplified comparisons of results between studies. In addition, the large number of participants, aged from 20 to 80 years, the use of standardized procedures, and the inclusion of both men and women are other important strengths.
There are a few potential limitations; first, cross-sectional studies of diet and disease associations cannot fully explore lifetime dietary intake or dietary change and cannot completely exclude confounding. Consequently, our results cannot determine a causal relationship between dietary patterns and BMD. Second, measurement error in dietary intake is inevitable and limitations in assessing dietary intake by FFQ are noted. However, this is unlikely to have significantly influenced our findings because random errors in dietary assessment measures tend to account for a lack of associations, but not the reverse [37]. Additionally, the most commonly used method of dietary assessment in dietary pattern analysis has been the FFQ, and Denova et al. [38] reported that the FFQ used was found to be valid in terms of the dietary pattern derived from a Mexican adult population. Third, the use of principal components analysis to estimate dietary patterns has a few downsides. It requires decisions regarding the number and type of foods that make up the groups and the cutoff points in the number of patterns, and these decisions may influence the results and their interpretation [39]. However, as mentioned above, the patterns in our study were similar to the patterns observed across different populations [14, 18, 20–22, 33, 34]. Additionally, while we adjusted for potential confounding factors, the possibility of residual or unmeasured confounding cannot be ruled out. Also, the participants in the present study are adults from a particular segment of the Mexican population: working class, apparently healthy individuals. The participants included in this analysis vary in age (from 20 to 80 years); therefore, a percentage of the population may be at low risk of osteoporosis. However, stratifying by age groups (20–50 and >50 years) and gender revealed no differences in the pattern of results with dietary pattern. While they may not be considered representative of the Mexican adult population as a whole, they may be considered representative of middle- to low-income men and women between 20 to 80 years residing in the urban areas of central Mexico.
In summary, among a Mexican adult population, dietary patterns were significantly associated with BMD. A dietary pattern high in fresh fruit and vegetables (prudent pattern), as well as a dietary pattern high in milk, other dairy foods, fish, and whole grains, and low in refined grains and soft drinks (dairy and fish pattern), may contribute to better BMD. In contrast, a dietary pattern represented by high consumption of refined grains, sugar and sweets, alcoholic beverages, red meat, fats, and soft drinks was significantly associated with higher likelihood of low BMD. Overall, this study provides evidence that will contribute to potential food-based prevention strategies for maintaining bone health. Further, longitudinal studies in the Mexican adult population are required to confirm these results.
References
Lupsa BC, Insogna K (2015) Bone health and osteoporosis. Endocrinol Metab Clin N Am 44(3):517–530
Sambrook P, Cooper C (2006) Osteoporosis. Lancet 367:2010–2018
Clark P, Cons-Molina F, Deleze M et al (2009) The prevalence of radiographic vertebral fractures in Latin American countries: the Latin American Vertebral Osteoporosis Study (LAVOS). Osteoporos Int 20(2):275–282
Clark P, Calos F, Vázquez-Martínez JL (2010) Epidemiology, costs and burden of osteoporosis in Mexico. Arch Osteoporos 5:9–17. doi:10.1007/s11657-010-0042-8
Carlos F, Clark P, Galindo-Suárez RM, Chico-Barba LG (2013) Health care costs of osteopenia, osteoporosis, and fragility fractures in Mexico. Arch Osteoporos 8:125. doi:10.1007/s11657-013-0125-4
McGuigan FE, Murray L, Gallagher A et al (2002) Genetic and environmental determinants of peak bone mass 9 in young men and women. J Bone Miner Res 17:1273–1279
Kitchin B, Morgan S (2003) Nutritional considerations in osteoporosis. Curr Opin Rheumatol 15:476–480
Bischoff-Ferrari HA, Dawson-Hughes B, Baron JA et al (2007) Calcium intake and hip fracture risk in men and women: a meta-analysis of prospective cohort studies and randomized controlled trials. Am J Clin Nutr 86(6):1780–1790
Bischoff-Ferrari HA, Willett WC, Orav EJ et al (2012) A pooled analysis of vitamin D dose requirements for fracture prevention. N Engl J Med 367(1):40–49
Cashman KD (2007) Diet, nutrition and bone health. J Nutr 137(11 Suppl):2507S–2512S
Tucker KL, Hannan MT, Chen H, Cupples LA, Wilson PW, Kiel DP (1999) Potassium, magnesium and fruit and vegetable intakes are associated with greater bone mineral density in elderly men and women. Am J Clin Nutr 69(4):727–736
Dermience M, Lognay G, Mathieu F, Goyens P (2015) Effects of thirty elements on bone metabolism. J Trace Elem Med Biol 32:86–106
Hu FB (2002) Dietary pattern analysis: a new direction in nutritional epidemiology. Curr Opin Lipidol 13:3–9
Tucker KL, Chen H, Hannan MT et al (2002) Bone mineral density and dietary patterns in older adults: the Framingham osteoporosis study. Am J Clin Nutr 76(1):245–252
Newby PK, Tucker KL (2004) Empirically derived eating patterns using factor or cluster analysis: a review. Nutr Rev 62:177–203
Kant AK (2004) Dietary patterns and health outcomes. Am J Diet Assoc 104:615–635
Jacobs DR Jr, Steffen LM (2003) Nutrients, foods and dietary patterns as exposure in research: a framework for food synergy. Am J Clin Nutr 78(suppl 3):508S–513S
Okubo H, Sasaki S, Horiguchi H et al (2006) Dietary patterns associated with bone mineral density in premenopausal Japanese farmwomen. Am J Clin Nutr 83(5):1185–1192
Kontogianni MD, Melistas L, Yannakoulia M, Malagaris I, Panagiotakos DB, Yiannakouris N (2009) Association between dietary patterns and indices of bone mass in a sample of Mediterranean women. Nutrition 25(2):165–171
Hardcastle AC, Aucott L, Fraser WD, Reid DM, Macdonald HM (2011) Dietary patterns, bone resorption and bone mineral density in early post-menopausal Scottish women. Eur J Clin Nutr 65(3):378–385
McNaughton SA, Wattanapenpaiboon N, Wark JD, Nowson CA (2011) An energy-dense, nutrient-poor dietary pattern is inversely associated with bone health in women. J Nutr 141:1516–1523
Langsetmo L, Poliquin S, Hanley DA et al (2010) Dietary patterns in Canadian men and women ages 25 and older: relationship to demographics, body mass index, and bone mineral density. BMC Musculoskelet Disord 11:20. doi:10.1186/1471-2474-11-20
Denova-Gutierrez E, Castañon S, Talavera JO et al (2010) Dietary patterns are associated with metabolic syndrome in an urban Mexican population. J Nutr 140:1855–1863
Denova-Gutiérrez E, Hernández-Ramírez R, López-Carrillo L (2014) Dietary patterns and gastric cancer risk in Mexico. Nutr Cancer 66:369–376
Rivera JA, Barquera S, González-Cossío T, Olaiz G, Sepúlveda J (2004) Nutrition transition in Mexico and in other Latin American countries. Nutr Rev 62:S149–S157
Rosner B (1983) Percentage points for a generalized ESD many-outlier procedure. Technometrics 25:165–172
Hernández-Avila M, Romieu I, Parra S, Hernández-Avila J, Madrigal H, Willett W (1998) Validity and reproducibility of a food frequency questionnaire to assess dietary intake of women living in Mexico City. Salud Publica Mex 40:133–140
Hernández-Avila M, Resoles M, Parra S (2014) Sistema de Evaluación de Hábitos Nutricionales y Consumo de Nutrimentos (SNUT). INSP, Cuernavaca
Newby PK, Weismayer C, Akesson A, Tucker KL, Wolk A (2006) Long-term stability of food patterns identified by use of factor analysis among Swedish women. J Nutr 136:626–633
Kim JO, Muller C (1984) Factor analysis. Sage Publications, Inc., Newbury Park
Kanis JA (2002) Diagnosis of osteoporosis and assessment of fracture risk. Lancet 359:1929–1936
Méndez-Hernández P, Flores Y, Siani C et al (2009) Physical activity and risk of metabolic syndrome in an urban Mexican cohort. BMC Public Health 9:276
Whittle CR, Woodside JV, Cardwell CR et al (2012) Dietary patterns and bone mineral status in young adults: the Northern Ireland Young Hearts Project. Br J Nutr 108(8):1494–1504
de França NA, Camargo MB, Lazaretti-Castro M, Peters BS, Martini LA (2016) Dietary patterns and bone mineral density in Brazilian postmenopausal women with osteoporosis: a cross-sectional study. Eur J Clin Nutr 70(1):85–90
Macdonald HM, Black AJ, Aucott L et al (2008) Effect of potassium citrate supplementation or increased fruit and vegetable intake on bone metabolism in healthy postmenopausal women: a randomized controlled trial. Am J Clin Nutr 88(2):465–474
Shin S, Hong K, Kang SW, Joung H (2013) A milk and cereal dietary pattern is associated with a reduced likelihood of having a low bone mineral density of the lumbar spine in Korean adolescents. Nutr Res 33:59–66
Hu FB, Stampfer MJ, Rimm E et al (1999) Dietary fat and coronary heart disease: a comparison of approaches for adjusting for total energy intake and modeling repeated dietary measurements. Am J Epidemiol 149:531–540
Denova-Gutiérrez E, Tucker KL, Salmerón J, Flores M, Barquera S. Relative validity of a semi-quantitative food frequency questionnaire to identify dietary patterns in an adult Mexican population. Forthcoming 2016
Denova-Gutiérrez E, Tucker KL, Flores M, Barquera S, Salmerón J (2016) Dietary patterns are associated with predicted cardiovascular disease risk in an urban Mexican adult population. J Nutr 146(1):90–97
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was managed according to the Declaration of Helsinki guidelines. The ethics committees of all participating institutions [Comité de Ética e Investigación, Instituto Mexicano del Seguro Social (No. 12CEI0900614); Comité de Ética e Investigación, Instituto Nacional de Salud Pública (No. 13CEI1700736); Comité de Ética, Centro de Investigación en Ciencias Médicas (No. 1233008X0236)] reviewed and approved the study protocol and informed consent forms.
Conflicts of interest
None.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
(DOCX 58 kb)
Rights and permissions
About this article
Cite this article
Denova-Gutiérrez, E., Clark, P., Tucker, K.L. et al. Dietary patterns are associated with bone mineral density in an urban Mexican adult population. Osteoporos Int 27, 3033–3040 (2016). https://doi.org/10.1007/s00198-016-3633-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-016-3633-4