Abstract
Introduction and hypothesis
This study was aimed at evaluating the effect of pelvic floor muscle training (PFMT) as a conservative treatment for patients with pelvic organ prolapse (POP).
Methods
A comprehensive search to identify eligible randomized controlled trial (RCT) studies was conducted using electronic databases including PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), and Embase up to 10 June 2021. Results were presented as risk ratio (RR), the weighted mean difference (WMD), with 95% confidence interval (95% CI) using the random effects model. Outcome variables were pooled using Review Manager version 5.3.
Results
Thirteen studies were included. Our results demonstrated that women who received PFMT intervention had a greater improvement than controls in prolapse symptom score (POP-SS; mean difference [MD] −1.66, 95% CI −2.36 to −0.97, p < 0.00001] and POP stages (risk ration [RR] 1.51, 95% CI 1.14–2.01, p = 0.004). The number of participants who felt better after PFMT was higher (RR 1.98, 95% CI 1.21–3.24, p = 0.006). Subgroup analysis showed that the symptoms of prolapse and the degree of prolapse were improved significantly in the short term, but there was no significant difference in the long-term effect. In addition, there was no significant difference in the impact of PFMT on the elderly and the quality of life. More RCTs are needed to evaluate the effect of PFMT on the elderly and whether the quality of life can be improved.
Conclusions
We found that PFMT can improve subjective symptoms and objective POP severity. More research is needed on the long-term effect.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pelvic organ prolapse (POP) is defined as protrusion of pelvic organs into or out of the vagina owing to loss of support from the connective tissue, muscles, or both [1], which includes anterior vaginal wall prolapse (urethrocele, cystocele), posterior vaginal wall prolapse (enterocele, rectocele), and prolapse of the apical segment of the vagina (cervix/cuff, uterine or vault prolapse) [2]. Additionally, women may present with prolapse in one or more of above-mentioned sites, depending on the type and degree of pelvic floor relaxation. However, the severity of prolapse stage is not directly associated with the severity of symptoms, and numerous women with prolapse are asymptomatic [3]. POP is considered a problem only if prolapse results in pressure symptoms with or without a bulge, sexual dysfunction, lower urinary tract dysfunction, or voiding dysfunction [4]. A previous study has reported that the prevalence of POP increases with age [5] and the etiology is believed to be associated with a combination of genetic and environmental risk factors [6]. The prevalence in middle-aged or elderly women is about 30–60%, and women older than 50 years are mostly affected [5]. Approximately 75% of women feel a profound impact on quality of life associated with prolapse symptoms [7]. It is reported that by 2050 the proportion of women suffering from POP will increase by 50%, with the changing demographics of the world’s population [8]. Nowadays, women are more active than they were in the past, and the development of POP adversely affects quality of life and disrupts social and personal activities. It is expected that POP will become a major health concern in the near future.
Currently, therapeutic options used for POP include surgery or nonsurgical management, e.g., vaginal pessary or pelvic floor muscle training (PFMT). It is estimated that the risk of surgery for women with POP in their lifetime is 13% [9]. However, the long-term results of surgery are far from optimal. Prolapse recurrence after any type of POP surgery is possible, and 6–30% recurrence rates have been reported [7]. Owing to the invasive operation, pelvic pain, dyspareunia, persistent vaginal bleeding, or discharge are the common postoperative complications. General treatment often starts with nonsurgical management. Pessary are frequently used as a first-line treatment for POP by many clinicians. However, many women experienced unsuccessful pessary fitting or pessary discontinuation because of difficulties with the insertion and removal of mechanical devices, vaginal discomfort, and the occurrence of complications [10]. And there is no standardized recommendation about the period to replace the device. Compared to surgery and pessary, PFMT has no adverse events virtually. PFMT is usually described as a behavioral therapy, which comprises repeated volitional contraction of the correct pelvic floor muscles and supervised by health professionals [11]. The pelvic floor muscles and ligaments, as well as the fascia, can be depicted as a hammock, where the functional and structural support of the pelvic organs is dependent on the strength of the suspension bridge. Studies suggested that PFM strength was increased after PFMT [12,13,14,15]. The theoretical basis of PFMT originates from two hypothetical mechanisms. First, the intensive training of the PFMs may increase muscle volume and elevate the levator plate to a higher position inside the pelvis by hypertrophying and improving the stiffness of its connective tissues. Second, a conscious, effective contraction before and during an increase in abdominal pressure to prevent leakage [16]. The latest Cochrane Review supports the hypothesis and suggests that PFMT could be included in the first-line options of nonsurgical management for urinary incontinence [11]. In recent years, several meta-analyses have revealed that PFMT is an effective treatment for women with symptomatic mild to moderate POP [17,18,19]. However, it is still unclear about whether PFMT could improve the degree of POP and the long-term effectiveness of the treatment. In addition, the efficacy of PFMT for POP in women aged 55 years or over has been debated, with no consensus to date.
Therefore, we aimed to perform a meta-analysis with new randomized trials and previous published data in order to strengthen the evidence base for assessing the efficacy of PFMT on POP, offering a reliable reference for clinicians treating these patients as well as a basis for future studies.
Materials and methods
Search strategy
In order to identify potentially relevant studies, we searched PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), and Embase using the search strategy combining medical subject headings and free-text terms. Limits were used for randomized controlled trails. The last literature search was run on 10 June 2021. The search terms included “Prolapse,” “rectocele,” “Pelvic Floor Disorders,” “prolaps* and (pelvi* or vagin* or genit* or uter* or vault* or apical or urethr* or segment* or wall* or cervi* or urogenital or rect*),” “kegel*,” “PFMT,” and “Physical Therapy Modalities.” An overview of the complete electronic search strategy is presented in Appendix 1.
Study selection
We selected studies based on the following criteria:
-
1.
Population: women aged 18 years or older with no racial restrictions, who were diagnosed with a different stage of POP as determined by the Pelvic Organ Prolapse Quantification System (POP-Q) [2].
-
2.
Intervention: any type of PFMT programs, including various types of PFMT teaching method, type of contractions, and number of contractions.
-
3.
Comparator: other conservative treatments, such as lifestyle advice, as well as muscle contraction without a PFMT program, pessary, or watchful waiting.
-
4.
Outcomes: the publication should include at least one of the outcome measures, such as the number of participants with the improvement in POP syndrome, the change in Pelvic Organ Prolapse Symptom Score (POP-SS) [20], improvement of the prolapse severity stage, the change in total quality of life based on validated Pelvic Floor Distress Inventory-20 (PFDI-20) and Pelvic Floor Impact Questionnaire-7 (PFIQ-7) [21].
-
5.
Studies on randomized controlled trials published in English.
Studies were excluded from this analysis for the following reasons:
-
1.
Participants with stage IV prolapse or postpartum women
-
2.
The role of PFMT intervention as an adjunct to surgery
-
3.
Studies without sufficient information for data analysis
Data extraction
Both authors extracted the data from the studies included independently using a previously tested form designed to capture information specifically. Data extracted included detailed information on the authors, the publication year, patients’ characteristics (age, BMI, number), summary of intervention treatment and control group, and duration of follow-up corresponding to different interest outcome measurements. Any divergence between the two reviewers was resolved by discussion with a third author. In order to obtain missing data that could be included in the analysis, emails were sent to the authors of the eligible studies selected, but no responses were received.
Study quality assessment
Two authors independently assessed all the studies included for risk of bias using Cochrane Handbook for Systematic Reviews of Interventions [22]. The risk of bias included random sequence generation and allocation concealment, blinding, incomplete outcome data, selective reporting, and other biases. The risk of bias was defined as “low,” “high,” or “unclear.” Disagreement was discussed with a third author to reach consensus. The trials included were classified as low-quality, high-quality, or moderate quality according to the following criteria:
-
1.
If one of the following items, such as randomization or allocation concealment or blinding, was assessed to have a high risk of bias, the trial was considered to be of low quality, regardless of the risk of other items.
-
2.
When randomization and allocation concealment and blinding were assessed as having a low risk of bias, and other items were assessed as having a low or unclear risk of bias in the trial, the trial was considered to be of high quality.
-
3.
If the trial did not meet the high or low risk criteria, the quality of the trial was considered to be moderate.
Statistical analysis
All analyses were performed using Review manager 5.3. For dichotomous data, the numbers of events in control and treatment groups of each of the studies included were used to calculate the risk ratio (RR) and 95% confidence intervals (CI). For continuous variables, means and standard deviations before and after intervention were used to calculate the weighted mean difference (WMD) and 95% CI. A p < 0.05 was considered statistically significant. Statistical heterogeneity was assessed using the Chi-squared test and I2 scores. We applied a random-effects model meta-analysis for all comparisons in order to control effects of unobserved heterogeneity. Because there were fewer than 10 studies on each outcome, we did not use a funnel plot to explore publication bias. Subgroup analyses were performed appropriately to explore the source of heterogeneity and summarize the effect of PFMT on POP by different time points as well as different prolapse compartments. The long-term results were described as ≥ 1 year and the short-term results were described as < 1 year. A sensitivity analysis was performed for primary results by excluding trials of low quality.
Results
Characteristics of the studies included
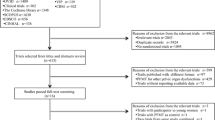
A total of 10,022 records were identified by searching electronic databases, including 1,667 from PubMed, 4,377 from Embase, 3,978 from CENTRAL. After duplicate exclusion, there were 6,324 citations left. Of them, 6,294 were excluded by title and abstract screening. Thirty articles were selected for full-text review, of which 17 were excluded. Table 1 summarized the characteristics of eligible studies. Seven studies were excluded because there were no data available. Eight papers were excluded for not reporting interest outcome measures. Two articles were excluded for language. Finally, we identified 13 studies that met the inclusion criteria and were incorporated into this meta-analysis. The process of selecting articles is presented in Fig. 1.
The main characteristics of the thirteen studies included are shown in Table 1. Of all the trials that assessed the efficacy of PFMT treatment on POP, the duration of follow-up varied between 6 weeks and 2 years [12, 13, 24,24,25,26,27,28,29,30,31,32,33]. There are two citations from the same study, as the main focus of each paper is a different outcome [30, 27]. Two studies that followed up for 2 years evaluated the effectiveness of PFMT treatment in women aged at least 55 years with POP [29, 28]. The study by Panman et al. [29] reported on the 2-year effects of the same population as in the paper by Wiegersma et al. [33]. However, we decided to include both articles, as both of them reported anatomical outcome such as the improvement of prolapse stage, the change of different prolapse compartments, which was one of the primary results of our analysis. The most common frequency of the program is 8 to 12 voluntary contractions in each set, usually with the duration of holding 6 to 10 s per contraction in these trials. All participating physiotherapists were appropriately trained before the beginning of the trials.
Study and data quality
The risk of bias in the studies included is shown in Fig. 2. Of all the studies, one was not clear about sequence generation [24] and one trial was unblinded [25]. One randomized controlled trial was unclear about the blinding of outcome assessment [12]. Attrition bias, selection bias, and other sources of bias were not found in any of the studies.
Outcomes
Pelvic organ prolapse symptom score
Three studies [31,27,23] with a total of 662 patients reported the change in prolapse symptoms after PFMT treatment as measured by the POP-SS [20], which consists of seven items relating to prolapse symptoms. Each item is scored from 0 (never) to 4 (all of the time), with a total ranging from 0 to 28. The pooled results showed that the POP-SS was significantly lower in women in the PFMT group than in controls (mean difference [MD] −1.66, 95% CI −2.36 to −0.97; Fig. 3).
Self-reported change in prolapse symptoms
Six studies [27, 26,28,29,30,28] with a total of 901 patients self-reported a change in symptoms. PFMT was considered successful if women reported their symptoms to be “better” and was considered unsuccessful if women reported their symptoms to be “the same” or “worse” [34]. The pooled results showed a significant change in the self-reported symptoms to better after PFMT treatment compared with the control group (RR 1.98, 95% CI 1.21–3.24; Fig. 4a).
Pelvic organ prolapse
Six trials [24, 26,28,29,30,28] enrolling 911 patients reported prolapse severity improvement defined by the Pelvic Organ Prolapse Quantification System (POP-Q). The pooled results showed that there was a statistically significant difference in POP-Q stage improvement after PFMT treatment compared with the control group (RR 1.51, 95% CI 1.14–2.01; Fig. 5a).
Subgroup analyses
Subgroup analysis of the subjective improvement outcome according to different follow-up duration found a significant difference in the self-reported improved symptom change within 6 months between the two groups (RR 3.0, 95% CI 1.96–4.59), but no significant variation for participants followed up for 1 year or 2 years after PFMT intervention (RR 1.43, 95% CI 0.75–2.73; Fig. 4b).
Subgroup analysis of the objective improvement outcome reported significant improvement in POP-Q stage after PFMT intervention compared with controls within a 6-month follow-up period (RR 1.86, 95% CI 1.03–3.36), but no statistically significant improvement in participants followed up for 2 years (RR 1.40, 95% CI 0.86–2.28; Fig. 5b).
Subgroup analysis according to different prolapse compartments [12, 13, 32, 33] showed a remarkable difference in the improvement of anterior prolapse stage between the PFMT group and the controls (RR 2.06, 95% CI 1.44–2.94; Fig. 5c), but no remarkable difference in the posterior prolapse (RR 1.23, 95% CI 0.66–2.27; Fig 5d).
The effect of PFMT on the elderly
Two trials [29, 28] focus on the effect of PFMT intervention in older women aged 55 years or over. The pooled results showed no difference in self-reported improved symptom change (RR 1.52, 95% CI 0.41–5.65; Fig. 4c). In addition, there was no significant improvement in prolapse severity between the two groups (RR 1.40, 95% CI 0.86–2.28; Fig. 5b2).
Quality of life
Three studies [30, 29, 28] reported on prolapse-specific quality of life using the PFDI-20 and the PFIQ-7; however, the data of one trial [28] were not available to the meta-analysis because participants had not completed the intervention followed up by the questionnaire. The PFDI-20 scale contains 20 items about anorectal, prolapse and urinary symptoms, whose total scores range from 0 to 300. With the PFIQ-7 scale with 21 items, each item can be scored from 0 to 3. For two scales, higher scores indicate a greater symptom burden or a more impaired quality of life. The other two trials reported a negative change in total score of the PFDI-20 questionnaire and PFIQ-7 questionnaire from the baseline, indicating improved symptoms or quality of life, but the pooled results showed no remarkable difference between the two groups in the change of PFDI-20 scale after PFMT intervention (MD −3.71, 95% CI −26.11 to 18.69; Fig. 6a) and no significant difference in the change of PFIQ-7 scale between the two groups (MD −6.34, 95% CI −12.88 to 0.21; Fig. 6b). Limited by the small number of studies, the quality of life cannot be further analyzed.
Sensitivity analysis
We conducted a sensitivity analysis by repeating the main outcome of interest of only high-quality studies. The pooled results did not change remarkably compared with the original analysis when the study [25] was removed (Fig. 7).
Discussion
This analysis summarized and reported the existing evidence for the effect of PFMT on POP. For participants, we found that PFMT treatment had a positive effect on prolapse symptoms and prolapse severity. Subgroup analysis showed that women benefited from PFMT treatment in the short term, but the long-term effects were not significant.
Also, the findings of our study revealed that there was no significant effect on elderly POP women and no remarkable difference in the quality of life after PFMT treatment. However, the main reason may be the small sample size in the meta-analysis (only two studies).
Our study confirmed that PFMT produced a significant improvement in subjective prolapse symptoms and objective anatomical changes in women with stages I–III symptomatic POP, which was consistent with a previous meta-analysis [18]. However, different than previous several meta-analyses, we performed a subgroup analysis of self-reported improvement in symptoms and POP severity by different time points. Our pooled results showed that PFMT intervention had a positive effect in the short term, but no significant difference in long-term follow-up. In the meta-analysis, subgroup analysis showed either no significance or high heterogeneity. The long-term effects have to be interpreted with caution owing to the existence of heterogeneity. Three previous meta-analyses demonstrated that PFMT as an adjuvant treatment to surgery shows no additional improvement for patients and the conclusion was the same in the latest review [35]. Therefore, this part of the article was not included in the study; more high-quality RCTs are needed to evaluate the impact of PFMT on surgery. Previous meta-analyses focused less on the elderly and the impact on quality of life. We analyzed the results of POP severity improvement and self-reported symptom improvement in the elderly with no significant difference. The interpretation of the subjective measure of prolapse in the elderly became challenging because of the heterogeneity between the two articles included. There were many studies using various tools to assess changes in quality of life. This analysis summarized the impact of quality of life using the PFDI-20 and PFIQ-7 questionnaires. It was concluded that there was no positive effect on the improvement of quality of life by PFMT intervention compared with controls. However, the small sample size (just two trials) may have introduced bias into the results. In addition, the evaluation of the heterogeneity of the two papers may not be significant, also because of the small sample size.
In the present study, our findings showed that there was a remarkable improvement in prolapse symptoms and severity over the short term after PFMT treatment, but no significant difference in the lasting effects. This decrease in efficacy should not necessarily be attributed to the waning effectiveness of the training itself. Long-term trials may lose some participants, leading to a risk of bias. The common reasons for poor training persistence including unrealistic expectations, forgetting to exercise, and lack of time [36]. In the future, more RCTs should study the benefits of long-term pelvic floor training under the supervision of a physical therapist. Motivation and adherence are the most important principles for treatment effectiveness, because of the slow process of muscle fiber enlargement as with other physical exercises. The guidance suggests supervised PFMT for at least 16 weeks as a first option for women with symptomatic POP-Q stage I or stage II prolapse [37]. But there is no standardized treatment intensity and number of PFM contractions in a set or length of each contraction for patients to adhere to. However, the latest review mentioned that it is not recommended to use a strictly standard PFMT dose, because the intensity of the PFM may be different for each woman experiencing different POP stages [35]. Therefore, the dose of PFMT should be further studied for specific subgroups of different PFM strength.
Our findings showed that PFMT intervention produced no obvious efficacy for women aged ≥55 years compared with controls. These findings must be interpreted with caution owing to the heterogeneity. Additionally, the relatively small sample size in the study may introduce bias and reduce the reliability of the results. Wiegersma et al. found that the odds of treatment success decreased with age, owing to two hypotheses: that the older women were less able to train PFM strength, and that they were more accepting of POP symptoms and less likely to insist on home exercise [38]. The purpose of PFMT intervention is to increase the intensity of PFMT and improve support to the pelvic organs, yet older women have less muscle mass than younger women. A hypoestrogenic environment in the pelvic organs leads to alterations in the composition and strength of collagen [39]. However, previous study has shown that the PFMT program could be an effective way in postmenopausal women of increasing PFM contractility as well as improving POP [13]. Also, Tosun et al. reported that strength increase could be achieved at all stages of the menopause with PFMT [40]. It is hypothesized that beneficial effects of the training program on pelvic floor symptoms were not limited to improving pelvic floor support by increasing muscle volume [41]. Therefore, more trials are needed to get robust evidence for a pathophysiological explanation of the benefits of pelvic floor muscle exercises.
The quality of life is one of the most important clinical outcomes for assessing the effect of training exercise on POP. Each participant of the two trials included completed two condition-specific quality-of-life questionnaires (PFDI-20, PFIQ-7), which included three subscales on the bladder, bowel, and vaginal bulging syndromes. However, no differences in the improvement of quality of life were found. Owing to the existence of heterogeneity, the quality-of-life outcomes have to be interpreted with caution once again. Whether the quality of life improves is an important reason for further treatment. Given our small sample size, the reliability of our conclusion may be reduced. Further research is needed to assess the quality of life after the training program more conclusively.
Although we tried to avoid bias and mistakes in identifying research, there were some limitations to our research. One important limitation was that the components of the PFMT program varied, such as contraction type, training frequency, duration, which might be the key reason for evaluating the heterogeneity of effects between PFMT intervention and POP. Another limitation might be that the articles included were restricted to English. This may limit the generality of our outcomes. Finally, the same limitation as was most common in other reviews was the quality of the original data.
In conclusion, we found that the PFMT program decreased the prolapse stage and improved prolapse symptoms significantly, especially in the short term. Nevertheless, more long-term studies are needed to evaluate the effect of long-term treatment, as well as more research in the elderly, and more RCTs are needed to explore the intensity and frequency of training for specific subgroups of different pelvic floor muscle strengths to confirm the results of our meta-analysis.
References
ACOG technical bulletin. Pelvic organ prolapse. American College of Obstetricians and Gynecologists. Int J Gynaecol Obstet. 1996;52(2):197–205.
Bump RC, Mattiasson A, Bø K, et al. The standardization of terminology of female pelvic organ prolapse and pelvic floor dysfunction. Am J Obstet Gynecol. 1996;175(1):10–7. https://doi.org/10.1016/s0002-9378(96)70243-0.
Ghetti C, Gregory WT, Edwards SR, Otto LN, Clark AL. Pelvic organ descent and symptoms of pelvic floor disorders. Am J Obstet Gynecol. 2005;193(1):53–7. https://doi.org/10.1016/j.ajog.2004.12.004.
Pelvic Organ Prolapse. ACOG Practice Bulletin, Number 214. Obstet Gynecol. 2019;134(5):e126–42. https://doi.org/10.1097/aog.0000000000003519.
Nygaard I, Barber MD, Burgio KL, et al. Prevalence of symptomatic pelvic floor disorders in US women. JAMA. 2008;300(11):1311–6. https://doi.org/10.1001/jama.300.11.1311.
Altman D, Forsman M, Falconer C, Lichtenstein P. Genetic influence on stress urinary incontinence and pelvic organ prolapse. Eur Urol. 2008;54(4):918–22. https://doi.org/10.1016/j.eururo.2007.12.004.
Mouritsen L, Larsen JP. Symptoms, bother and POPQ in women referred with pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct. 2003;14(2):122–7. https://doi.org/10.1007/s00192-002-1024-1.
Wu JM, Vaughan CP, Goode PS, et al. Prevalence and trends of symptomatic pelvic floor disorders in U.S. women. Obstet Gynecol. 2014;123(1):141–8. https://doi.org/10.1097/aog.0000000000000057.
Wu JM, Matthews CA, Conover MM, Pate V, Jonsson Funk M. Lifetime risk of stress urinary incontinence or pelvic organ prolapse surgery. Obstet Gynecol. 2014;123(6):1201–6. https://doi.org/10.1097/aog.0000000000000286.
Yimphong T, Temtanakitpaisan T, Buppasiri P, Chongsomchai C, Kanchaiyaphum S. Discontinuation rate and adverse events after 1 year of vaginal pessary use in women with pelvic organ prolapse. Int Urogynecol J. 2018;29(8):1123–8. https://doi.org/10.1007/s00192-017-3445-x.
Dumoulin C, Cacciari LP, Hay-Smith EJC. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2018;10(10):CD005654. https://doi.org/10.1002/14651858.CD005654.pub4.
Stüpp L, Resende AP, Oliveira E, Castro RA, Girão MJ, Sartori MG. Pelvic floor muscle training for treatment of pelvic organ prolapse: an assessor-blinded randomized controlled trial. Int Urogynecol J. 2011;22(10):1233–9. https://doi.org/10.1007/s00192-011-1428-x.
Alves FK, Riccetto C, Adami DB, et al. A pelvic floor muscle training program in postmenopausal women: A randomized controlled trial. Maturitas. 2015;81(2):300–5. https://doi.org/10.1016/j.maturitas.2015.03.006.
Ouchi M, Kato K, Gotoh M, Suzuki S. Physical activity and pelvic floor muscle training in patients with pelvic organ prolapse: a pilot study. Int Urogynecol J. 2017;28(12):1807–15. https://doi.org/10.1007/s00192-017-3356-x.
Liu YJ, Ting SW, Hsiao SM, Huang CM, Wu WY. Efficacy of bio-assisted pelvic floor muscle training in women with pelvic floor dysfunction. Eur J Obstet Gynecol Reprod Biol. 2020;251:206–11. https://doi.org/10.1016/j.ejogrb.2020.04.050.
Bø K. Pelvic floor muscle training is effective in treatment of female stress urinary incontinence, but how does it work? Int Urogynecol J Pelvic Floor Dysfunct. 2004;15(2):76–84. https://doi.org/10.1007/s00192-004-1125-0.
Hagen S, Stark D. Conservative prevention and management of pelvic organ prolapse in women. Cochrane Database Syst Rev. 2011;12:CD003882. https://doi.org/10.1002/14651858.CD003882.pub4.
Li C, Gong Y, Wang B. The efficacy of pelvic floor muscle training for pelvic organ prolapse: a systematic review and meta-analysis. Int Urogynecol J. 2016;27(7):981–92. https://doi.org/10.1007/s00192-015-2846-y.
Ge J, Wei XJ, Zhang HZ, Fang GY. Pelvic floor muscle training in the treatment of pelvic organ prolapse: a meta-analysis of randomized controlled trials. Actas Urol Esp (Engl Ed). 2021;45(1):73–82. https://doi.org/10.1016/j.acuro.2020.01.012.
Hagen S, Glazener C, Sinclair L, Stark D, Bugge C. Psychometric properties of the pelvic organ prolapse symptom score. BJOG. 2009;116(1):25–31. https://doi.org/10.1111/j.1471-0528.2008.01903.x.
Barber MD, Walters MD, Bump RC. Short forms of two condition-specific quality-of-life questionnaires for women with pelvic floor disorders (PFDI-20 and PFIQ-7). Am J Obstet Gynecol. 2005;193(1):103–13. https://doi.org/10.1016/j.ajog.2004.12.025.
Higgins JP, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. https://doi.org/10.1136/bmj.d5928.
Hagen S, Stark D, Glazener C, Sinclair L, Ramsay I. A randomized controlled trial of pelvic floor muscle training for stages I and II pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20(1):45–51. https://doi.org/10.1007/s00192-008-0726-4.
Braekken IH, Majida M, Engh ME, Bø K. Can pelvic floor muscle training reverse pelvic organ prolapse and reduce prolapse symptoms? An assessor-blinded, randomized, controlled trial. Am J Obstet Gynecol. 2010;203(2):170.e1–7. https://doi.org/10.1016/j.ajog.2010.02.037.
Kashyap R, Jain V, Singh A. Comparative effect of 2 packages of pelvic floor muscle training on the clinical course of stage I–III pelvic organ prolapse. Int J Gynaecol Obstet. 2013;121(1):69–73. https://doi.org/10.1016/j.ijgo.2012.11.012.
Hagen S, Stark D, Glazener C, et al. Individualised pelvic floor muscle training in women with pelvic organ prolapse (POPPY): a multicentre randomized controlled trial. Lancet. 2014;383(9919):796–806. https://doi.org/10.1016/S0140-6736%2813%2961977-7.
Due U, Brostrøm S, Lose G. Lifestyle advice with or without pelvic floor muscle training for pelvic organ prolapse: a randomized controlled trial. Int Urogynecol J. 2016;27(4):555–63. https://doi.org/10.1007/s00192-015-2852-0.
Panman CM, Wiegersma M, Kollen BJ, et al. Effectiveness and cost-effectiveness of pessary treatment compared with pelvic floor muscle training in older women with pelvic organ prolapse: 2-year follow-up of a randomized controlled trial in primary care. Menopause. 2016;23(12):1307–18. https://doi.org/10.1097/gme.0000000000000706.
Panman C, Wiegersma M, Kollen BJ, et al. Two-year effects and cost-effectiveness of pelvic floor muscle training in mild pelvic organ prolapse: a randomised controlled trial in primary care. BJOG. 2017;124(3):511–20. https://doi.org/10.1111/1471-0528.13992.
Due U, Brostrøm S, Lose G. The 12-month effects of structured lifestyle advice and pelvic floor muscle training for pelvic organ prolapse. Acta Obstet Gynecol Scand. 2016;95(7):811–9. https://doi.org/10.1111/aogs.12884.
Hagen S, Glazener C, McClurg D, et al. Pelvic floor muscle training for secondary prevention of pelvic organ prolapse (PREVPROL): a multicentre randomised controlled trial. Lancet. 2017;389(10067):393–402. https://doi.org/10.1016/s0140-6736(16)32109-2.
Resende APM, Bernardes BT, Stüpp L, et al. Pelvic floor muscle training is better than hypopressive exercises in pelvic organ prolapse treatment: An assessor-blinded randomized controlled trial. Neurourol Urodyn. 2019;38(1):171–9. https://doi.org/10.1002/nau.23819.
Wiegersma M, Panman CM, Kollen BJ, Berger MY, Lisman-Van Leeuwen Y, Dekker JH. Effect of pelvic floor muscle training compared with watchful waiting in older women with symptomatic mild pelvic organ prolapse: randomised controlled trial in primary care. BMJ. 2014;349:g7378. https://doi.org/10.1136/bmj.g7378.
Burgio KL, Goode PS, Richter HE, Locher JL, Roth DL. Global ratings of patient satisfaction and perceptions of improvement with treatment for urinary incontinence: validation of three global patient ratings. Neurourol Urodyn. 2006;25(5):411–7. https://doi.org/10.1002/nau.20243.
Basnet R. Impact of pelvic floor muscle training in pelvic organ prolapse. Int Urogynecol J. 2021;32(6):1351–60. https://doi.org/10.1007/s00192-020-04613-w.
Fine P, Burgio K, Borello-France D, et al. Teaching and practicing of pelvic floor muscle exercises in primiparous women during pregnancy and the postpartum period. Am J Obstet Gynecol. 2007;197(1):107.e101–5. https://doi.org/10.1016/j.ajog.2007.02.052.
NICE Guidance—Urinary incontinence and pelvic organ prolapse in women: management: © NICE. Urinary incontinence and pelvic organ prolapse in women: management. BJU Int. 2019;123(5):777–803. https://doi.org/10.1111/bju.14763.
Wiegersma M, Panman C, Hesselink LC, Malmberg AGA, Berger MY, Kollen BJ, et al. Predictors of success for pelvic floor muscle training in pelvic organ prolapse. Phys Ther. 2019;99(1):109–17. https://doi.org/10.1093/ptj/pzy114.
Jackson SR, Avery NC, Tarlton JF, Eckford SD, Abrams P, Bailey AJ. Changes in metabolism of collagen in genitourinary prolapse. Lancet. 1996;347(9016):1658–61. https://doi.org/10.1016/s0140-6736(96)91489-0.
Tosun Ö, Mutlu EK, Tosun G, et al. Do stages of menopause affect the outcomes of pelvic floor muscle training? Menopause. 2015;22(2):175–84. https://doi.org/10.1097/gme.0000000000000278.
Roovers JP. Female urology: the benefits of pelvic floor muscle training for prolapse. Nat Rev Urol. 2014;11(4):192–3. https://doi.org/10.1038/nrurol.2014.35.
Acknowledgements
We thank Mr Z.B. Wang in Beijing University of Chinese Medicine for his kind advice on the project and writing assistance.
Author information
Authors and Affiliations
Contributions
Ting Wang: project development, data collection, manuscript writing; Zhengfang Wen: manuscript revision; Meng Li: data collection.
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOC 34 kb)
Rights and permissions
About this article
Cite this article
Wang, T., Wen, Z. & Li, M. The effect of pelvic floor muscle training for women with pelvic organ prolapse: a meta-analysis. Int Urogynecol J 33, 1789–1801 (2022). https://doi.org/10.1007/s00192-022-05139-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-022-05139-z