Abstract
Purpose
Failure to reconstruct the natural footprints of the ruptured anterior cruciate ligament (ACL) may lead to premature graft-failure. Therefore, precise analyses of insertion site anatomy and inter-individual variations of the morphology of the ACL are highly important to facilitate optimal individualized graft placement. Therefore, the purpose of this study was to analyze the inter-individual variation of the morphology of the femoral and tibial ACL footprints.
Methods
Thirty subjects with an intact ACL were included in this study for MR imaging of their knee joint. A three-dimensional (3D) dual-echo steady-state sequence with near 0.8 mm isotropic resolution was acquired on a 3 T system with a 15-channel knee-coil. The ACL was subsequently manually segmented using dedicated medical imaging software (VitreaAdvanced®, Vital Images). The lengths and widths of the footprints were measured after reconstructing an axial oblique (tibial footprint) or coronal oblique (femoral footprint) section at the bone–ligament junction and descriptive analysis was conducted to describe morphology orientation of the footprint.
Results
The femoral footprint measured on average 14 mm ± 2 mm (range 8–19 mm) in length and 5 mm ± 1 mm (range 3–8 mm) in width. The mean value of the tibial footprint measured 10 mm ± 2 mm (range 5–14 mm) in length and 7 mm ± 2 mm (range 5–13 mm) in width. Descriptive analysis showed a stretched, ribbon-like appearance of the femoral footprint, while the tibial footprint revealed larger variability, stretching from anterolateral to posteromedial around the anterior horn of the lateral meniscus.
Conclusion
3D imaging of the ACL footprints reveals a distinct difference in insertion site morphology and fiber bundle orientation between the femoral and tibial footprint. This questions the concept of strict anatomical separation of the ACL into an anteromedial and posterolateral bundle.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several anatomical studies have described the anatomical footprint of the anterior cruciate ligament (ACL) on the femur and tibia [8, 11, 12, 14, 20]. Most studies either described it arthroscopically [11, 14] or during anatomical dissections [8, 12, 15, 20]. The exact description of the actual tendon-to-bone insertion is hindered in arthroscopy due to the two-dimensional nature of the arthroscopic image and the necessity to flex the knee to visualize the femoral insertion site of the ACL. In flexion, the ACL is exhibiting a torsional deformation along its longitudinal axis, further complicating the precise identification of the respective bony footprints. Recently, a group by Smigielski et al. presented thorough anatomical dissections [20] of both the femoral and tibial footprint of the ACL. They found that the ACL had more a band-like rather than a round insertion site at its bony attachments. Especially the tibial footprint was consistently described by them to be a C-shaped structure, extending from posteromedial to anterolateral, which was in contrast to the published literature [4, 6,7,8] that reported the footprints to be round to oval. These previous studies influenced surgical techniques that aimed to restore the footprints of the ACL by placing round tunnels into the footprint of the injured ACL. In addition, the functional separation of the ACL into an anteromedial and posterolateral bundle led to recommendations of respective placement of bone tunnels in the anteromedial and posterolateral area of the femoral and tibial insertion site.
While arthroscopic evaluation of anatomical structures is currently limited to two-dimensional assessment, anatomical dissections are often biased by personal skill and the difficulty to clearly separate different tissue types, such as true ligamentous fibers from synovial membrane or peritendinous tissue, which only play a very minor role in load bearing of the ACL. Magnetic resonance imaging (MRI) has evolved to a technique that can directly visualize three-dimensional anatomy of soft-tissue structures [5, 16, 19]. The increasing spatial resolution of MRI makes this technology suitable for even detailed anatomical descriptions of soft-tissue structures. It allows to clearly differentiate different tissue types and their precise orientation to each other, therefore, eliminating the disadvantages of two-dimensional arthroscopic and purely subjective anatomical assessment. Therefore, it was the aim of this study to accurately describe the location and the shape of the tendon-to-bone insertion of the ACL on the femur and tibia in patients without ACL pathology. This knowledge can provide a basis for surgical reconstruction techniques aiming to restore true ACL anatomy. It was hypothesized that contrary to the accepted notion of the morphology ACL insertion sites, they were not round to oval shaped. Such findings have direct clinical relevance, since current ACL reconstruction techniques do not address such varying footprint anatomy, which in turn affects knee biomechanics.
Materials and methods
Thirty patients were enrolled and underwent MRI examinations of the knee for a suspected pathology other than an injury to the ACL. These pathologies were meniscus tears, cartilage injury, patella dislocations, and unclear medial side knee pain. Patients with a previous history of ACL injury or abnormality of the ACL on MR imaging as well as patients with progressed degenerative changes of the knee joint were excluded from this study. Median age of the patients was 31 ± 17 years. 13 patients were female, and 17 patients were male. Written consent was given by all patients that imaging data could be used for scientific studies and the institutional ethics review board approved this study.
All examinations were performed with a 3-T MR system (Magnetom Skyra, Siemens Health Care, Erlangen, Germany) with a dedicated 15-channel transmit–receive knee-coil with the knee positioned in full extension. A high spatial resolution T2-weighted three-dimensional dual-echo steady-state (3D DESS) sequence was used for the analysis of the ACL. This sequence was acquired in sagittal orientation with the following parameters: repetition time (TR): 14.1 ms; time-to-echo (TE): 5 ms; 1 average; flip-angle 25°; and bandwidth 250 Hz/pixel. The spatial resolution was near-isotropic measuring 0.78 × 0.63 × 0.8 mm and the acquisition time was 6 min and 8 s.
The 3D DESS sequence was postprocessed using the VitreaAdvanced™ application of the Vitrea Enterprise Suite (Version 6.7.2.) software (Vital Images, Inc., Minnetonka, MN, USA).
The following postprocessing steps were performed to accurately describe ligamentous fiber alignment and their true anatomic insertion to bone and to differentiate this from locations, where they were adjacent, but without physical connection to the bone:
-
1.
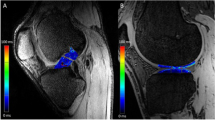
The entire ACL of each patient was manually segmented on sagittal 3D DESS images (Fig. 1a) by an experienced orthopaedic surgeon, who was trained in manual segmentation using this specific type of software.
-
2.
The manual segmentation was reviewed by an experienced musculoskeletal radiologist for precision, including axial and coronal reconstructions of the ACL to guarantee the most exact segmentation result. The manually segmented volume was color-coded (blue, Fig. 1b) by the software algorithm and was used as the basis for further analysis. In addition, the software calculated the volume of each segmented ACL.
-
3.
An obliquely reformatted plane was constructed orthogonal to the course of the ACL. The tendon-to-bone insertion was then determined as the last orthogonal section, showing ACL structure with the next subsequent section showing bone. This orthogonal section of the respective footprint was stored and used for subsequent measurements and anatomical descriptions.
-
4.
The maximum two-dimensional size of the footprint was measured in mm and the surface area of the footprint was measured in square mm using the planimetric tool of the software.
-
5.
The exact anatomical location of the tibial footprint was assessed with respect to the anterior root of the lateral meniscus and with respect to the midline of the tibial plateau. Location of the midline was determined as an anterior–posterior line crossing orthogonally the midpoint of a tangent line to the posterior border of the tibial plateau.
The mean values of ACL volumes (mm3), the two-dimensional size (mm), and the surface areas (mm2) of the footprints were calculated.
The ACL was manually segmented using a dedicated workstation (Vitrea Enterprise Suite, product version 6.7.2, Vital Images Inc. Minnetonka, Minnesota), which automatically calculated the volume of the segmented structure (here, ACL). Based on the segmentation, the footprints were reconstructed in a double-oblique plane, which was used to measure the area of the footprint by manual planimetry using the same software. The maximum (and vertical) diameter of the footprint was also measured manually.
Two orthopaedic surgeons subspecialized in ACL surgery reviewed the femoral and tibial footprint reconstructions in a consensus reading session. First, they scored the shape of the femoral footprint as ribbon-like, ovoid, round, or undetermined (Fig. 2a). Second, they scored the shape of the tibial footprint as c-shaped, double-C-shaped, ovoid, or undetermined (Fig. 2b–d). Third, they scored the exact anatomical insertion location of the tibial footprint with respect to the midline (in the midline, left of midline, right of midline, and undetermined) and with respect to the anterior root of the lateral meniscus (anterior to the root, at the level of the root, posterior to the level of the root, undetermined).
This study received IRB approval by the Charité, University Medicine Berlin with the ID number EA4/084/13.
Results
Quantitative analysis
All results below are reported as mean values ± standard deviation. The mean volume of the ACL measured 1476 mm3 ± 484 mm3 (range 608–2290 mm3).
The femoral footprint measured 14 mm ± 2 mm (range 10–19 mm) in length and 5 mm ± 1 mm (range 3–8 mm) in width. The surface area of the femoral footprint measured 54 mm2 ± 21 mm2 (range 27–118 mm2).
The tibial footprint measured 10 mm ± 2 mm (range 5–14 mm) in length and 7 mm ± 2 mm (range 5–13 mm) in width. The surface area of the tibial footprint measured 59 mm2 ± 20 mm2 (range 33–118 mm2).
Qualitative analysis
The shape of the femoral footprint was very consistent and rated as ribbon-like in all 30 patients (Fig. 2a).
The shape of the tibial footprint displayed more variations and was rated as C-shaped (Fig. 2c) in 19 patients, ovoid in 9 patients (Fig. 2b) and double-C-shaped in 2 patients (Fig. 2d).
The location of the tibial footprint with respect to the tibial midline was rated “in the midline” in 22 patients and “medial of midline” in 8 patients.
The location of the anatomical tibial footprint of the ACL was found to be anterior in respect to the anterior root of the lateral meniscus in all 30 patients.
Discussion
The most important finding of this study was that the morphology of especially the tibial, but also femoral footprint of the intact ACL varied from descriptions of most previous studies and cannot be fully reconstructed with round bone tunnels as it is typically done in today´s ACL reconstruction techniques. Therefore, our hypothesis was confirmed that the femoral footprint of the ACL was not round to oval, but rather rectangular. The same observation was made for the tibial footprint, which did not display a round morphology, but with some variations a C-shaped or ribbon-like appearance. It was also found that the strict anatomical separation of the ACL in an anteromedial and posterolateral bundle, reflected by its respective insertions site areas could not be confirmed by the findings of this 3D MRI analysis.
Current ACL reconstruction techniques place bone tunnels in the center of the footprint of the femoral and tibial insertions sites. Specially designed drill guides have been developed to facilitate anatomical placement of these tunnels. Round drill bits are used for placement of round to oval shaped bone tunnels with a diameter between 6 and 10 mm, matching the graft´s diameter. In the 2000s, clinical and basic science studies found that the non-isometric behaviour of the ACL could be explained by different functional bundles with different insertion areas of the respective footprints of the ACL [1, 17]. Most studies agreed on the existence of two main bundles [3, 17], the anteromedial and posterolateral, while others found a third or even more bundles [1, 13]. These observations were put in question by the group of Smigieski et al. [20] and others [15], who carried out anatomical dissections and histological analyses to determine the true area of the ligament-to-bone attachment of the ACL. They found that the morphology of the femoral footprint was not round, but more rectangular or as they termed it “ribbon-like” [18, 20]. Similar findings were reported for the tibial footprint, which was also found not be round, but to fan out from anterior to posteromedial, resembling a C-shaped like structure [18, 20]. The authors were not able to replicate the strict morphological separation of anteromedial and posterolateral structures with their respective tibial insertion sites as it had been described in previously published studies [4, 6,7,8], questioning the generalized concept of two distinct functional bundles. However, criticism was raised about Smigelski´s group study design, especially, that their anatomical dissections might have altered the ACL´s structure, possibly creating artificial, rather than true anatomic footprints [9]. Therefore, we designed a new study, which did not alter the morphology of the ACL during measurements, using non-invasive 3D MR imaging analysis. Other studies have shown that 3D MR imaging has almost identical accuracy compared to direct measurements of the ACL´s footprints, such as laser scanning [2] or direct anatomic identification [10].
In this study, the morphology of the femoral insertion site of the ACL was found not to be round, but rather rectangular or ovally stretched. Its length was between 10 and 19 mm (average 14 mm) and its width was between 3 and 8 mm (average 6 mm). Similar measurements were shown during anatomical dissections by Colombet at al [4]. They found the length of the femoral footprint to be 13.9 mm and the width to be 9 mm on average, separating the footprint into an anteromedial and posterolateral area. It was found that the femoral footprint occupied a region on the lateral notch wall from anteromedial to posterolateral. Edwards et al. [7] reported from anatomical dissections that wide variations were found in size and shape for the femoral footprint of the ACL [7]. They stated that a clear separation of anteromedial and posterolateral areas was detected for the ACL attachment on the femur. They measured an average length of 14 mm, an average width of 7 mm for the femoral attachment, and values very similar to our study. However, the morphology of the femoral footprints did not show large variations in shape. None of them were round, all of them were more rectangular with a slight curvature. These findings were in line with the descriptive findings of our 3D MRI study, which showed a very consistent morphology and anatomical orientation at the lateral notch wall.
Following analysis of the tibial insertion site of the ACL, we found that the area of attachment was in midline or medial of the midline of the tibial plateau in all patients. The morphology of the tibial footprint showed larger variations than on the femoral site. Its shape varied between predominantly curved [C-shape/double-C-shape (21/30)] and ovoid (9/30). In all cases, the anterior root of the lateral meniscus was in close posterolateral proximity to the tibial ACL insertion. An anteromedial and posterolateral insertion area of the ACL on the tibial plateau could not be confirmed as it was previously published [4, 6, 8]. Our findings are in agreement with the observations made by Smigielski et al. and Siebold et al. [18, 20], describing a mere curved morphology, forming an anterolateral and posteromedial area of the tibial insertion site. Such an observation is of critical importance, since the previous concepts of double-bundle ACL reconstructions were based on establishing anteromedial and posterolateral bone tunnels on the tibial plateau and at the lateral notch wall of the femur. In consequence of the results of this study, such tunnel placement would result in non-anatomic reconstruction of the ACL.
The ultimate goal of a knee surgeon is to restore the ACL´s insertion sites as accurately as possible. The findings of this study suggest that a more rectangular single bone tunnel or two small bone tunnels should be placed on the femur in anteromedial and posterolateral orientation. On the tibia, different concepts would be possible. A round tunnel could be drilled at the anatomical footprint of the tibial insertion site, placing a fixation device in the posterolateral area of the bone tunnel close to the joint aperture, thereby pressing the graft in semilunar fashion against the antero/posteromedial aspect of the footprint. Alternatively, two smaller tunnels could be drilled, placing one anterior and a second one posteromedially. Future studies are warranted to biomechanically analyze this new approach in tibial tunnel placement, compare it to current double-bundle or single-bundle ACL reconstruction techniques and determine its impact on knee kinematics.
The findings of this study have clinical relevance as they suggest that current ACL reconstruction techniques do not restore the morphology of the anatomic ACL insertion sites, This might lead to new concepts of optimized “anatomic” ACL reconstruction techniques.
Conclusion
In conclusion, this study shows that anatomical quantitative analysis of intraarticular structures of the ACL is possible using 3D MRI technology. This technique allows for non-invasive measurements without the disadvantages of altering structures during tissue preparation or the difficulty to separate different types of tissues during direct inspection. Furthermore, it puts in question currently accepted notion that ACL consists of strict anteromedial / posterolateral bundles with round to oval insertion sites. As a consequence, surgeons need to reconsider current approach for anatomic ACL reconstruction.
References
Amis AA, Dawkins GP (1991) Functional anatomy of the anterior cruciate ligament. Fibre bundle actions related to ligament replacements and injuries. J Bone Joint Surg Br 73:260–267
Araki D, Thorhauer E, Tashman S (2017) Three-dimensional isotropic magnetic resonance imaging can provide a reliable estimate of the native anterior cruciate ligament insertion site anatomy. Knee Surg Sports Traumatol Arthrosc 37:1904
Boisgard S, Levai JP, Geiger B, Saidane K, Landjerit B (1999) Study of the variations in length of the anterior cruciate ligament during flexion of the knee: use of a 3D model reconstructed from MRI sections. Surg Radiol Anat 21:313–317
Colombet P, Robinson J, Christel P, Franceschi J-P, Djian P, Bellier G, Sbihi A (2006) Morphology of anterior cruciate ligament attachments for anatomic reconstruction: a cadaveric dissection and radiographic study. Arthroscopy 22:984–992
Dong Y, Mou Z, Huang Z, Hu G, Dong Y, Xu Q (2013) Three-dimensional reconstruction of subject-specific knee joint using computed tomography and magnetic resonance imaging image data fusions. Proc Inst Mech Eng H 227(10):1083–1093
Edwards A, Bull AMJ, Amis AA (2007) The attachments of the anteromedial and posterolateral fibre bundles of the anterior cruciate ligament: Part 1: tibial attachment. Knee Surg Sports Traumatol Arthrosc 15:1414–1421
Edwards A, Bull AMJ, Amis AA (2008) The attachments of the anteromedial and posterolateral fibre bundles of the anterior cruciate ligament. Part 2: femoral attachment. Knee Surg Sports Traumatol Arthrosc 16:29–36
Ferretti M, Doca D, Ingham SM, Cohen M, Fu FH (2011) Bony and soft tissue landmarks of the ACL tibial insertion site: an anatomical study. Knee Surg Sports Traumatol Arthrosc 20:62–68
Hohmann E (2017) Editorial Commentary: The ribbon theory. Another quantum leap? The anterior cruciate ligament is twisted and in fact a flat structure. Or not? Arthroscopy 33:1710–1711
Hui C, Pi Y, Swami V, Mabee M, Jaremko JL (2016) A validation study of a novel 3-dimensional MRI modeling technique to identify the anatomic insertions of the anterior cruciate ligament. Orthop J Sports Med 4:2325967116673797
Irarrázaval S, Albers M, Chao T, Fu FH (2017) Gross, arthroscopic, and radiographic anatomies of the anterior cruciate ligament: foundations for anterior cruciate ligament surgery. Clin Sports Med 36:9–23
Iriuchishima T, Ryu K, Aizawa S, Fu FH (2015) Proportional evaluation of anterior cruciate ligament footprint size and knee bony morphology. Knee Surg Sports Traumatol Arthrosc 23:3157–3162
Iwahashi T, Shino K, Nakata K, Nakamura N, Yamada Y, Yoshikawa H, Sugamoto K (2008) Assessment of the “functional length” of the three bundles of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc 16:167–174
Middleton KK, Muller B, Araujo PH, Fujimaki Y, Rabuck SJ, Irrgang JJ, Tashman S, Fu FH (2015) Is the native ACL insertion site “completely restored” using an individualized approach to single-bundle ACL-R? Knee Surg Sports Traumatol Arthrosc 23:2145–2150
Noailles T, Boisrenoult P, Sanchez M, Beaufils P, Pujol N (2017) Torsional appearance of the anterior cruciate ligament explaining “Ribbon” and double-bundle concepts: a cadaver-based study. Arthroscopy 33:1703–1709
Pass B, Robinson P, Hodgson R, Grainger AJ (2015) Can a single isotropic 3D fast spin echo sequence replace three-plane standard proton density fat-saturated knee MRI at 1.5 T? Br J Radiol 88:20150189
Sakane M, Fox RJ, Woo SL, Livesay GA, Li G, Fu FH (1997) In situ forces in the anterior cruciate ligament and its bundles in response to anterior tibial loads. J Orthop Res 15:285–293
Siebold R, Schuhmacher P, Fernandez F, Śmigielski R, Fink C, Brehmer A, Kirsch J (2015) Flat midsubstance of the anterior cruciate ligament with tibial “C-”shaped insertion site. Knee Surg Sports Traumatol Arthrosc 23:3136–3142
Swami VG, Cheng-Baron J, Hui C, Thompson RB, Jaremko JL (2015) Reliability of 3D localisation of ACL attachments on MRI: comparison using multi-planar 2D versus high-resolution 3D base sequences. Knee Surg Sports Traumatol Arthrosc 23:1206–1214
Śmigielski R, Zdanowicz U, Drwięga M, Ciszek B, Ciszkowska-Łysoń B, Siebold R (2015) Ribbon like appearance of the midsubstance fibres of the anterior cruciate ligament close to its femoral insertion site: a cadaveric study including 111 knees. Knee Surg Sports Traumatol Arthrosc 23:3143–3150
Funding
No external source of funding was used.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest related to this study.
Ethical approval
This study received IRB approval by the Charité, University Medicine Berlin with the ID number EA4/084/13.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Scheffler, S.U., Maschewski, K., Becker, R. et al. In-vivo three-dimensional MR imaging of the intact anterior cruciate ligament shows a variable insertion pattern of the femoral and tibial footprints. Knee Surg Sports Traumatol Arthrosc 26, 3667–3672 (2018). https://doi.org/10.1007/s00167-018-4939-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-018-4939-x