Abstract
This study assessed the effectiveness of limestone-montmorillonite-rapeseed residue-Si fertilizer compound amendment on the bioavailability and crop uptake of cadmium (Cd) and enzyme activities in acidic paddy soils. Applying the compound amendment at ratios of 1%–3% increased soil pH by 0.1–1.9 units, decreased leaching ratios of soil Cd 4.0%–22%, and decreased exchangeable and carbonated Cd 42%–55% and 27%–49%, respectively. Organic matter-bound Cd increased 47%–62% (p < 0.05). Cadmium concentrations decreased in the roots, culms, leaves, and grains of rice grown in the Cd-contaminated soils by 37%–81%, 18%–73%, 29%–64% and 27%–63%, respectively, (p < 0.05). Catalase and urease activities increased 2.5%–63% and 3.9%–36%, (p < 0.05), respectively. Applying this compound amendment may significantly mitigate soil acidification and decrease the bioavailability and crop uptake of Cd in acidic Cd-contaminated paddy soils.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Soil cadmium (Cd) pollution has recently received increasing attention because of its impacts on soil quality, food security and human health (Li et al. 2017a; Rizwan et al. 2017; Shakir et al. 2017). According to the survey of the Ministry of Agriculture of the People’s Republic of China, over 1.3 × 104 ha of the farmlands from 11 provinces have been contaminated by Cd in China (Zhou et al. 2012; Sun et al. 2013). Many remediation techniques on Cd-contaminated soils have been developed and reported, although most of them are characterized as being highly difficult or expensive (Dermont et al. 2008; Hu et al. 2016; Liu et al. 2018). Therefore, the development of cost-effective, environmentally friendly remediation technologies for Cd-contaminated soils in agricultural settings is urgently needed.
Considered one of the most effective remediation technologies for Cd-contaminated soils, immobilization of soil Cd in situ is commonly used to reduce the mobility and bioavailability of soil Cd with various amendments (Guo et al. 2006; Kumpiene et al. 2008). Alkaline materials, such as lime and calcium carbonate, can effectively mitigate soil acidification and reduce Cd bioavailability in acidic soils (Zhu et al. 2016; He et al. 2017). Soil minerals, such as montmorillonite, apatite, bentonite, and sepiolite, may significantly reduce the mobility and bioavailability of soil Cd (Hamidpour et al. 2010; Tica et al. 2011; Du et al. 2016). Organic materials (e.g. crop residues) can decrease the availability of soil Cd but may also increase soil enzymatic activities (Zeng et al. 2011; Lee et al. 2013; Abad-Valle et al. 2017). Additionally, Si, Zn, and Se fertilization have been reported to reduce Cd bioavailability and restrict Cd transportation in crops (Kirchmann et al. 2009; Shao et al. 2017; Zare et al. 2017). Several studies focused on the development of an immobilization agent composed of a single effective component, as well as combined amendments; however, cost-effective, environmentally friendly compound amendments with the synergistic effects are less known.
Change in the soil metal speciation is typically used to assess amendment efficacy on the remediation of metal-contaminated soils. Soluble and exchangeable (or leachable) metal fractions are available to crops roots, whereas the bound, residual fractions have less bioavailability (Chavez et al. 2016). Soil enzyme and microbial activities have been used as indicators to investigate and assess effects of amendments on soil quality. Soil urease and catalase, involving nitrogen and hydrogen peroxide transformation, are important indices reflecting soil quality (Stepniewska et al. 2009; Gao et al. 2010).
Microcosm experiments with double factors were conducted to evaluate the efficacies of an original compound amendment [limestone-montmorillonite-rapeseed residue-Si fertilizer (LMRrSf)] on Cd-contaminated acidic paddy soils. The present study aimed to evaluate impacts on Cd immobilization, enzyme activities and crop uptake.
Materials and Methods
Bulk surface soils (0–20 cm, dry weight 120 kg) were collected from paddy fields in Ningxiang (28°12′N, 112°27′E), Hunan province, China. The region has a subtropical monsoon climate, with an annual average temperature of 17°C and rainfall of 1500 mm. Ultisoils (USDA Soil Taxonomy) had a particle size composition of 45%, 8.8% and 46% (sand, silt, clay, respectively), a pH of 5, contained 0.9% organic matter and a total Cd concentration of 1.0 mg kg−1. Cadmium chloride [AR grade, crystal, Sangon Biotech (Shanghai) Co., Ltd] was gradually supplemented into 60 kg of soil to obtain a soil Cd level of 5.0 mg kg−1. Results from 2-year spiking experiments indicated that Cd bioavailability and geochemical speciation were not markedly affected by aging (Zhong and Wang 2006). Therefore, the spiking procedure for Cd-added soil generally requires a month for equilibration (Sun et al. 2013; Xia et al. 2016; Li et al. 2017b). Thus, the Cd-spiked paddy soil was maintained around 70% (w/w) of water holding capacity at 25°C for 5 weeks (Sun et al. 2013).
Double-factor microcosm experiments were carried out with four application amounts of LMRrSf compound amendment and two soil Cd levels (1.0 and 5.0 mg kg−1). For each soil, four levels of a compound amendment [0%, 1%, 2%, and 3% (w/w)] were applied and completely mixed (Huang et al. 2017). Each treatment was established in triplicate and placed randomly in four blocks. The rice cultivar “II You 93” was transplanted to each microcosm at a planting density of four seedlings per microcosm. During the growth period, 70% water holding capacity was maintained with deionized water.
The compound amendment was composed of limestone, montmorillonite, rapeseed residues, and Si fertilizer with ratios of 4:3:2:1. The montmorillonite contained mainly (Al,Mg)2[Si4O10](OH)2·nH2O, had a surface area of 25 m2 g−1, average pore size of 1.5 nm, and cation exchange capacity (CEC) of 90 meq 100 g−1. Limestone was 80% CaCO3. Rapeseed residues had 42% total C, 1.2% total N, and a C:N ratio of 35:1. The Si fertilizer was a commercial fertilizer and had water soluble Si of 55 mg kg−1 and a pH of 9.
When the rice crop of each treatment was harvested, roots, culms, leaves and grains (polished) were divided, oven-dried (75°C) to a constant weight, and then ground to < 0.25 mm. After harvest, soil for each treatment was collected, air-dried (25°C) and ground to < 2 and 0.15 mm to determine soil pH and extractable Cd, and total Cd, respectively.
Soil pH was directly determined using a pH meter (IQ150, Spectrum) at planting, tillering, filling, and harvesting stages. Soil texture was determined by the pipette method, and organic matter was measured by the Walkley–Black method (Tica et al. 2011). Soil and crop Cd were digested using mixtures of aqua regia–HClO4 and HNO3–HClO4 (open system), respectively. Concentrations of Cd in digested solutions were determined by Atomic Absorption Spectrophotometry (Solaar M6, Thermo Fisher Scientific, USA). Toxicity characteristic leaching procedure (TCLP) was conducted to assess the mobility of soil Cd, as described by Sun et al. (2013). A sequential extraction procedure was applied to determine Cd fractions in the soil (Lestan et al. 2003). Soil urease activity was determined with urea as substrate, incubating at pH 7 and 37°C for 24 h, and expressed as NH4–N g−1 h−1 (Gu et al. 2009). Soil catalase activity was determined using the titration method (Stepniewska et al. 2009).
Quality assurance and quality control measures involved reagent blanks, triplicate samples and Certified Reference Materials (CRMs) for soil (GBW 07401) and rice (GBW 10045). All reagents were analytical grade. All glass and polyethylene bottles were previously soaked overnight in HNO3 (10%) and rinsed thoroughly with deionized water before use. Elemental recoveries for CRMs were within 95% ± 5% for soil and 100% ± 5% for rice, respectively. Cd limits of detection and quantification for AAS were 0.02 and 0.08 mg kg−1, respectively.
Statistical significance was determined using ANOVA, followed by the least significant difference (LSD) test at p < 0.05. Pearson correlation was applied for all the correlations. Statistical analyses were performed using SPSS 16.0 (SPSS Inc. Chicago, USA).
Results and Discussion
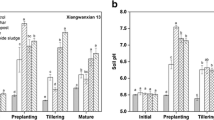
Soil acidity is an important factor controlling the speciation, solubility, mobility and bioavailability of metals in soils (Lee et al. 2013). Paddy soils had a pH of around 5.0 (Table 1). Applying the compound amendment increased soil pH across the whole rice growth stage by 0.1–1.9 units (p < 0.05, Table 1). Furthermore, soil pH increased with the ratios of the LMRrSf compound amendment 19%–22% (Table 1). These results indicate application of the compound amendment may significantly increase soil pH and mitigate soil acidification in acidic Cd-contaminated paddy soils. The compound amendment can directly neutralize the H+ in soils (Zhang et al. 2008; Wu et al. 2016). Montmorillonite and rapeseed residues have huge surface areas, abundant pores and may adsorb H+ and indirectly increase soil pH (Lee et al. 2013; Yang et al. 2016). These effects result in the increase of soil pH. Previous studies have shown an increasing soil pH leads to an increase of negative surface charges, thereby inducing the precipitation of metals (Madejón et al. 2006; Querol et al. 2006; Lee et al. 2009).
TCLP was used to assess the mobility and phytoavailability of soil Cd (Zhou et al. 2016). For paddy soils without any amendment, leaching ratios of soil Cd increased 62% and 48% at the two levels (1.0 and 5.0 mg kg−1) of soil Cd, respectively (Table 1). This indicates high Cd phytoavailability and potential leaching risk in the acidic paddy soils. However, application of the compound amendment reduced leaching ratio of soil Cd by 4.0%–22% (p < 0.05, Table 1), respectively. Moreover, the TCLP-extracted Cd decreased with an increase in the application ratio (Table 1). Alkaline composed materials provide OH− and directly precipitate the active Cd fractions (Zhu et al. 2016; He et al. 2017). Application of the amendment may significantly reduce the mobility and phytoavailability of Cd in the acidic paddy soils.
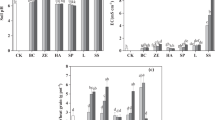
For paddy soils without amendment, exchangeable Cd was 0.26 and 1.21 mg kg−1, accounting for almost 25% of the total Cd (Fig. 1). This indicates the high phytoavailability of Cd in the paddy soils. For soils with the compound amendment, exchangeable Cd decreased 42%–55%, while carbonated Cd decreased 27%–49%. Organic matter-bound Cd increased 47%–62% (p < 0.05, Fig. 1). Application of the amendment may transform the exchangeable and carbonated Cd to the organic matter-bound Cd and consequently reduce the phytoavailability of Cd in paddy soils. Organic materials (i.e. rapeseed residues) have abundant pores and functional groups and may directly adsorb Cd and increase organic matter-bound Cd (Lee et al. 2013; Abad-Valle et al. 2017; Tang et al. 2017). Inputs of montmorillonites and rapeseed residues may increase organo-mineral complexes and enhance metal adsorption properties (Wu et al. 2011; Du et al. 2016). The alkaline condition of the compound amendment may enhance the adsorption and precipitation as mentioned previously (Cheng and Hseu 2002; Madejón et al. 2006; Querol et al. 2006; Lee et al. 2009; Wu et al. 2016).
Soil cadmium fractions with amendments in the contaminated paddy soils from Ningxiang, China. Data are means ± standard deviations. The letter following each value indicates a significant difference between values in each line (p < 0.05). WS water soluble Cd, SE exchangeable Cd, WSA carbonate-bound Cd, OX Fe/Mn-bound Cd, OM organic matter-bound Cd, RES residual Cd
Rice grains from the two paddy soils contained 0.5–1.0 mg kg−1 Cd, which were 2.5–5.0 times the Chinese allowable limit (0.2 mg kg−1, GB2762—2017). This reflects the high risk in food security and human health in the study region. When the compound amendment was applied, Cd in rice grains decreased 27%–63%, while Cd in the roots, culms and leaves also decreased 18%–81% (Fig. 2a, b). These trends increased along with the application ratios of the amendment. This indicates application of the amendment may efficiently inhibit Cd uptake and translocation in rice plants and grains.
Mean cadmium concentrations (a, b) and bioaccumulation factors (c, d) for the roots, culms, leaves, and grains in contaminated paddy soils from Ningxiang, China. Bars indicate standard deviations. The letter following each value indicates a significant difference between values in each line (p < 0.05)
The bioaccumulation factor (BCF) is used to evaluate the accumulation of metals in plants (Sun et al. 2015). For paddy soils without amendment, the BCF for grains was 0.2–0.5, while the total BCF was 3.0–9.5. This reflects significant bioaccumulation and a high risk in food security. However, applying the compound amendment reduced the BCF for roots, culms, leaves, grains, and rice plants by 37%–81%, 17%–73%, 29%–64%, 27%–63%, and 30%–74%, respectively (p < 0.05, Fig. 2c, d). These trends increased along with the application ratios of the amendment. The input of water soluble Si (5.5 mg kg−1 in the compound amendment) in soils may inhibit Cd uptake in roots and its transportation to aerial and edible parts (Dresler et al. 2015; Farooq et al. 2016).
Soil enzyme activities have been used to assess soil quality, especially in metal contaminated soils (Bandick and Dick 1999; Shen et al. 2005; Gu et al. 2009). Compared to the values in soils without amendment, urease and catalase activities in soils applied with the compound amendment (1.0 and 5.0 mg kg−1) increased 3.9%–36% and 2.5%–63% (p < 0.05), respectively (Fig. 3a, b). These results are consistent with those reported by Garau et al. (2007), in which urease activity increased in the zeolite, red mud (p < 0.05) and lime-amended (p < 0.05) soils, but the unamended soil showed low mean values. These results indicate the soil amendment may not only remediate Cd-contaminated acidic paddy soils, but they may also enhance soil quality.
In this study, soil pH was significantly correlated with soil urease activity and rice grain Cd (p < 0.05, Fig. 4a, b). Rice grain Cd was significantly correlated with soil pH, exchangeable Cd and TCLP-Cd (p < 0.05, Fig. 4a, c, d). These results further reflect that soil pH and active Cd fractions control Cd phytoavailability in acidic paddy soils and Cd accumulation in rice grains. The compound amendment significantly increased soil pH, decreased soil Cd phytoavailability, and consequently reduced Cd uptake and accumulation in rice plants and grains.
Pearson correlations among the tested parameters. a Correlation between soil pH and Cd concentration in seeds. b Correlation between soil pH and soil urease activity. c h1: correlation between TCLP-Cd and Cd concentration in seeds; h2: correlation between TCLP-Cd and Cd concentration of exchangeable fraction. d Correlation between Cd concentration in seeds and Cd concentration of exchangeable fraction
It is possible to mass produce the compound amendment, because its components are inexpensive and accessible. The ratio of 2% (equivalent to 4.5 t ha−1) is recommended for potential application of the LMRrSf compound amendment to remediate Cd-contaminated acidic paddy soils in rice fields. Applying LMRrSf compound amendment may significantly mitigate soil acidification, reduce the leaching ratio of soil Cd, decrease the mobility and phytoavailability of Cd, increase urease and catalase activities, and inhibit Cd uptake and translocation in rice plants and Cd accumulation in rice grains in acidic Cd-contaminated paddy soils. It should be also noted that as these results are from microcosm experiments, further field experiments should be conducted in the future, due to the variability of rice fields in subtropical China.
References
Abad-Valle P, Iglesias-Jiménez E, Álvarez-Ayuso E (2017) A comparative study on the influence of different organic amendments on trace element mobility and microbial functionality of a polluted mine soil. J Environ Manag 188:287–296
Bandick AK, Dick RP (1999) Field management effects on soil enzyme activities. Soil Biol Biochem 31:1471–1479
Chavez E, He ZL, Stoffella PJ, Mylavarapu RS, Li YC, Baligar VC (2016) Chemical speciation of cadmium: an approach to evaluate plant-available cadmium in Ecuadorian soils under cacao production. Chemosphere 150:57–62
Cheng SF, Hseu ZY (2002) In-situ immobilization of cadmium and lead by different amendments in two contaminated soils. Water Air Soil Pollut 140:73–84
Dermont G, Bergeron M, Mercier G, Richer-Laflèche M (2008) Soil washing for metal removal: a review of physical/chemical technologies and field applications. J Hazard Mater 152:1–31
Dresler S, Wojcik M, Bednarek W, Hanaka A, Tukiendorf A (2015) The effect of silicon on maize growth under cadmium stress. Russ J Plant Physiol 62:86–92
Du H, Chen W, Cai P, Rong X, Dai K, Peacock CL, Huang Q (2016) Cd(II) Sorption on montmorillonite-humic acid-bacteria composites. Sci Rep 6:19499
Farooq MA, Detterbeck A, Clemens S, Dietz KJ (2016) Silicon-induced reversibility of cadmium toxicity in rice. J Exp Bot 67:3573–3585
Gao Y, Zhou P, Mao L, Zhi Y, Zhang C, Shi W (2010) Effects of plant species coexistence on soil enzyme activities and soil microbial community structure under Cd and Pb combined pollution. J Environ Sci 22:1040–1048
Garau G, Castaldi P, Santona L, Deiana P, Melis P (2007) Influence of red mud, zeolite and lime on heavy metal immobilization, culturable heterotrophic microbial populations and enzyme activities in a contaminated soil. Geoderma 14:47–57
Gu Y, Wang P, Kong CH (2009) Urease, invertase, dehydrogenase and polyphenoloxidase activities in paddy soil influenced by allelopathic rice variety. Eur J Soil Biol 45:436–441
Guo G, Zhou Q, Ma LQ (2006) Availability and assessment of fixing additives for the in situ remediation of heavy metal contaminated soils: a review. Environ Monit Assess 116:513–528
Hamidpour M, Kalbasi M, Afyuni M, Shariatmadari H, Holm PE, Hansen HC (2010) Sorption hysteresis of Cd(II) and Pb(II) on natural zeolite and bentonite. J Hazard Mater 181:686–691
He YB, Huang DY, Zhu QH, Wang S, Liu SL, He HB, Zhu HH, Xu C (2017) A three-season field study on the in-situ remediation of Cd-contaminated paddy soil using lime, two industrial by-products, and a low-Cd-accumulation rice cultivar. Ecotoxicol Environ Saf 136:135–141
Hu Y, Cheng H, Tao S (2016) The challenges and solutions for cadmium-contaminated rice in china: a critical review. Environ Int 92–93:515–532
Huang SH, Yang Y, Li Q, Su Z, Yuang CY, Yang KO (2017) Evaluation of the effects of lime–bassanite–charcoal amendment on the immobilization of cadmium in contaminated soil. Bull Environ Contam Toxicol 98:433–438
Kirchmann H, Mattsson L, Eriksson J (2009) Trace element concentration in wheat grain: results from the Swedish long-term soil fertility experiments and national monitoring program. Environ Geochem Health 31:561–571
Kumpiene J, Lagerkvist A, Maurice C (2008) Stabilization of As, Cr, Cu, Pb and Zn in soil using amendments—a review. Waste Manag 28:215–225
Lee SH, Lee JS, Choi YJ, Kim JG (2009) In situ stabilization of cadmium-, lead-, and zinc-contaminated soil using various amendments. Chemosphere 77:1069–1075
Lee SS, Lim JE, El-Azeem SA, Choi B, Oh SE, Moon DH, Ok YS (2013) Heavy metal immobilization in soil near abandoned mines using eggshell waste and rapeseed residue. Environ Sci Pollut Res Int 20:1719–1726
Lestan D, Grcman H, Zupan M, Bacac N (2003) Relationship of soil properties to fractionation of Pb and Zn in soil and their uptake into Plantago lanceolata. Soil Sediment Contam 12:507–522
Li H, Luo N, Li YW, Cai QY, Li HY, Mo CH, Wong MH (2017a) Cadmium in rice: transport mechanisms, influencing factors, and minimizing measures. Environ Pollut 224:622–630
Li Y, Pang HD, He LY, Wang Q, Sheng XF (2017b) Cd immobilization and reduced tissue Cd accumulation of rice (Oryza sativa wuyun-23) in the presence of heavy metal-resistant bacteria. Ecotoxicol Environ Saf 138:56–63
Liu L, Li W, Song W, Guo M (2018) Remediation techniques for heavy metal-contaminated soils: principles and applicability. Sci Total Environ 633:206–219
Madejón E, de Mora AP, Felipe E, Burgos P, Cabrera F (2006) Soil amendments reduce trace element solubility in a contaminated soil and allow regrowth of natural vegetation. Environ Pollut 139:40–52
Querol X, Alastury A, Moreno N, Alvarez-ayuso E, Garía-Sánchez A, Cama J, Ayora C, Simón M (2006) Immobilization of heavy metals in polluted soils by the addition of zeolitic material synthesized from coal fly ash. Chemosphere 62:171–180
Rizwan M, Ali S, Adrees M, Ibrahim M, Tsang DCW, Zia-Ur-Rehman M, Zahir ZA, Rinklebe J, Tack FMG, Ok YS (2017) A critical review on effects, tolerance mechanisms and management of cadmium in vegetables. Chemosphere 182:90–105
Shakir SK, Azizullah A, Murad W, Daud MK, Nabeela F, Rahman H, Ur Rehman S, Häder DP (2017) Toxic metal pollution in Pakistan and its possible risks to public health. Rev Environ Contam Toxicol 242:1–60
Shao JF, Che J, Yamaji N, Shen RF, Ma JF (2017) Silicon reduces cadmium accumulation by suppressing expression of transporter genes involved in cadmium uptake and translocation in rice. J Exp Bot 68:5641–5651
Shen GQ, Cao LK, Lu YT, Hong JB (2005) Influence of phenanthrene on cadmium toxicity to soil enzymes and microbial growth. Environ Sci Pollut Res 12:259–263
Stepniewska Z, Wolińska A, Ziomek J (2009) Response of soil catalase activity to chromium contamination. J Environ Sci 21:1142–1147
Sun Y, Sun G, Xu Y, Wang L, Liang X, Lin D, Hu F (2013) Assessment of natural sepiolite on cadmium stabilization, microbial communities, and enzyme activities in acidic soil. Environ Sci Pollut Res Int 20:3290–3299
Sun YB, Li Y, Xu YM, Liang XF, Wang L (2015) In situ stabilization remediation of cadmium (Cd) and lead (Pb) co-contaminated paddy soil using bentonite. Appl Clay Sci 105–106:200–206
Tang W, Zhong h, Xiao L, Tan Q, Zeng Q, Wei Z (2017) Inhibitory effects of rice residues amendment on Cd phytoavailability: a matter of Cd-organic matter interactions? Chemosphere 186:227–234
Tica D, Udovic M, Lestan D (2011) Immobilization of potentially toxic metals using different soil amendments. Chemosphere 85(4):577–583
Wu PX, Zhang Q, Dai YP, Zhu NW, Dang Z, Li P, Wu JH, Wang XD (2011) Adsorption of Cu(II), Cd(II) and Cr(III) ions from aqueous solutions on humic acid modified Ca-montmorillonite. Geoderma 164:215–219
Wu YJ, Zhou H, Zou ZJ, Zhu W, Yang WT, Peng PQ, Zeng M, Liao BH (2016) A three-year in-situ study on the persistence of a combined amendment (limestone + sepiolite) for remedying paddy soil polluted with heavy metals. Ecotoxicol Environ Saf 130:163–170
Xia Q, Peng C, Lamb D, Mallavarapu M, Naidu R, Ng JC (2016) Bioaccessibility of arsenic and cadmium assessed for in vitro bioaccessibility in spiked soils and their interaction during the Unified BARGE Method (UBM) extraction. Chemosphere 147:444–450
Yang WT, Gu JF, Zou JL, Zhou H, Zeng QR, Liao BH (2016) Impacts of rapeseed dregs on Cd availability in contaminated acid soil and Cd translocation and accumulation in rice plants. Environ Sci Pollut Res Int 23:20853–20861
Zare AA, Khoshgoftarmanesh AH, Malakouti MJ, Bahrami HA, Chaney RL (2017) Root uptake and shoot accumulation of cadmium by lettuce at various Cd:Zn ratios in nutrient solution. Ecotoxicol Environ Saf 148:441–446
Zeng F, Ali S, Zhang H, Ouyang Y, Qiu B, Wu F, Zhang G (2011) The influence of pH and organic matter content in paddy soil on heavy metal availability and their uptake by rice plants. Environ Pollut 159:84–91
Zhang C, Wang L, Nie Q, Zhang W, Zhang F (2008) Long-term effects of exogenous silicon on cadmium translocation and toxicity in rice (Oryza sativa L.). Environ Exp Bot 62:300–307
Zhong H, Wang WX (2006) Influences of aging on the bioavailability of sediment-bound Cd and Zn to deposit-feeding sipunculans and soldier crabs. Environ Toxicol Chem 25:2775–2780
Zhou YF, Haynes RJ, Naidu R (2012) Use of inorganic and organic wastes for in situ immobilisation of Pb and Zn in a contaminated alkaline soil. Environ Sci Pollut Res Int 19:1260–1270
Zhou G, Xia X, Wang H, Li LQ, Wang GJ, Zheng SX, Liao SJ (2016) Immobilization of lead by Alishewanella sp. WH16-1 in pot experiments of Pb-contaminated paddy soil. Water Air Soil Pollut 227:339
Zhu H, Chen C, Xu C, Zhu Q, Huang D (2016) Effects of soil acidification and liming on the phytoavailability of cadmium in paddy soils of central subtropical China. Environ Pollut 219:99–106
Acknowledgements
This project was financially supported by the Science Research Project of the Science and Technology Department in Hunan Province (2017NK2144 and 2016NK2036), Changsha Municipal Science and Technology Major Project (kq1703010), Opening Foundation of the Hunan Engineering Research Center of Safe and Efficient Utilization of Heavy Metal Contaminated Arable Land, Changsha, 410083, China (TGOP-001) and Hunan Agricultural Science and Technology Innovation Alliance Project (2017LM0305).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shan, S., Guo, Z., Lei, P. et al. Impacts of a Compound Amendment on Cd Immobilization, Enzyme Activities and Crop Uptake in Acidic Cd-Contaminated Paddy Soils. Bull Environ Contam Toxicol 101, 243–249 (2018). https://doi.org/10.1007/s00128-018-2379-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-018-2379-4