Abstract
Purpose
Women who face pregnancy complications may require hospitalization in a high-risk pregnancy unit (HRPU), and this may have a negative impact on the pregnant woman’s mental health. The purpose of this study was to screen for depressive symptoms in pregnant women admitted in a high-risk pregnancy unit and also to investigate associated risk factors.
Methods
This was a cross-sectional study that enrolled pregnant women admitted at ≥24 gestational weeks in a university hospital HRPU, between September 2014 and November 2015. The Edinburgh Postnatal Depression Scale was used to assess depressive symptoms on admission. A cutoff score ≥13 was considered as indicative of depression. Test results were then correlated with the reasons that led to the women’s admission as well as socio-demographic characteristics.
Results
Overall, 157 of the women admitted in the HRPU during the study period were eligible for the study and agreed to complete the questionnaire. The mean age of the pregnant women was 30 ± 6 years, and the mean gestational week at admission was 32 ± 3 weeks. The prevalence of antenatal depression (score ≥13) was 28 % (44/157). In the multivariate analysis, antenatal depression was significantly correlated with smoking (OR = 2.681, 95 % CI 1.089–6.601) and the existence of thoughts for pregnancy termination (OR = 3.624, 95 % CI 1.149–11.427).
Conclusions
Approximately one quarter of pregnant women hospitalized in the HRPU may show signs of depression at the time of admission. Smoking and an unwanted pregnancy were found to be independently associated with antenatal depression, whereas no association was found with any obstetric parameters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence of antenatal depression has been reported to be between 10–15 % in the general pregnant population [1, 2], whereas studies in the UK, the USA, and France have demonstrated a higher prevalence of depressive disorders in high-risk pregnancies ranging from 16 to 25 % [3–6].
Antenatal depression and anxiety have been associated with adverse outcomes for both the mother and the newborn [7]. Women with clinical depression during pregnancy are at increased risk of miscarriage, preterm birth, preeclampsia, intra-uterine growth restriction, and low-birth-weight infants [8, 9]. Antenatal stress and depression may also lead to language problems, impulsivity, attention deficit disorder, behavioral problems, and psychopathology in the offspring during childhood [10, 11]. Furthermore, untreated depression during pregnancy increases the risk for postpartum depression (PPD), which, in turn, has been shown to have negative effects on maternal–infant attachment and child development [12]. Evidence suggests that PPD may, in fact, be part of a continuum of antenatal depression [13–15]. Antenatal depression becomes increasingly prevalent as gestation advances [16]. Results from a systematic review suggested that the rate of depression in the first trimester was similar to that seen in the general female population, whereas rates in the second and third trimesters were twice as high compared with those in the general population [1].
Thus, the American College of Obstetricians and Gynecologists recommends that clinicians screen patients at least once during the perinatal period for depression and anxiety symptoms using standardized, validated tools [17]. Women with high-risk pregnancies may face depression and anxiety symptoms [18]; hospital admission may further increase the stress burden of a high-risk pregnancy [19]. Unfortunately, the psychiatric consultation rate for these women in inpatient obstetric clinics has been reported to be as low as 0.3 % [20].
Given the limited data on antenatal depression in hospitalized high-risk pregnancies and the possible adverse effects of this condition on maternal and fetal health, we conducted a cross-sectional study in an HRPU of a university obstetric clinic to assess the rate of depression and investigate possible risk factors among the demographic and obstetric parameters. To the best of our knowledge, this is the first study ever conducted in Greek women with high-risk pregnancies.
Materials and methods
This cross-sectional study was carried out between September 2014 and November 2015, and recruited pregnant women admitted into the HRPU of the 3rd Academic Department of Obstetrics and Gynaecology, of the Aristotle University of Thessaloniki, Greece. Eligibility criteria for the study were determined based on information obtained from the medical records at the time of admission. Participants were considered eligible for the study if they: (1) were ≥24 and <37 weeks pregnant; (2) had a good understanding of the Greek language; (3) were admitted to the HRPU because of an obstetric complication; and (4) had no mental illness history. During the study period, psychiatric consultation was available when requested by clinical care teams. Two obstetric residents and two obstetric consultants were involved in inviting women to participate in the study.
The Edinburgh Postnatal Depression Scale (EPDS), a self-administered 10-item screening questionnaire, was used for the study [21]. The EPDS was originally developed to assist in identifying potential symptoms of depression in the postnatal period [21], and it is the only rating scale for depression that has been further validated for both the antenatal period and the postnatal period [22]. The EPDS has been translated and validated for the Greek population by two independent research groups [23, 24] and showed a very high overall internal consistency (Cronbach’s alpha value for the total scale: 0.804, CI 0.108–1.642, p < 0.0001) [24].
The ten symptoms of depression assessed are: inability to laugh and look forward to things with enjoyment, blaming oneself unnecessarily, being anxious or worried, being scared or panicky, suffering from inability to cope, having difficulty in sleeping, feeling sad, or miserable, crying, and having thoughts of harming oneself [21]. Each question has four possible answers, the grading of which depends on the severity or duration of each symptom [21]. Every answer is scored on a 4-point scale (0–3), and the result is a total score ranging between 0–30 [21]. Respondents are asked to rate the intensity of depressive symptoms during the preceding 7 days; higher scores indicate more severe depressive symptoms. Elevated EPDS scores may not be specific for depression, but rather detect a whole range of psychiatric disorders, including anxiety disorders [25]. Cox et al. [21] reported a sensitivity of 86 % and a specificity of 78 % using a cutoff ≥10 for “possible” depression and ≥13 for “probable” depression. In a systematic review of 37 validation studies of the EPDS for postpartum depression (PPD), Gibson et al. [26] noted that a cutoff score of less than 9/10 provides “strong evidence” for ruling out minor and/or major depression and that a cutoff of more than 12/13 provides “convincing diagnostic evidence” for minor/major depression, because the accuracy of diagnosing PPD increases. In a Greek validation study, it was found that the optimal cutoff for postpartum minor depression is 8/9 (77 % sensitivity and 68 % specificity) and 12/13 for major depression (88 % sensitivity and 86 % specificity) [24]. The cutoff point of EDPS among pregnant women is usually higher than among postpartum women [26]. Thus, in this study, we selected 12/13 as the optimal cutoff point to detect antenatal depression.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki (World Medical Association, 2008). The four investigators responsible for the recruitment of the sample (TD, IT, AM, and FC) informed all the participants about the aims and objectives of the study. A comprehensive information leaflet was also given to the participants with the questionnaire pack. Informed consent was necessary for participation in the study.
Measurements
Medical and obstetric data were collected from the patients’ records. We further interviewed women to ascertain socio-demographic characteristics, including origin, educational level, marital status, employment status, monthly income, planned pregnancy, abortion thoughts, and the quality of the relationship with the partner, via a fixed questionnaire developed by the study staff.
All questionnaires were fully completed (no unanswered items). All participants that remained in the hospital for more than a week were asked to repeat the EPDS questionnaire on a weekly basis until their discharge or delivery, to detect possible changes during their hospital stay. Women who were readmitted during their pregnancy were invited to continue participating in the study and complete the questionnaire weekly.
Participants with an EPDS score ≥13 were categorized as having a high probability of suffering from depression and were offered psychiatric consultation, so as to discuss the results of the screening and to be offered mental health services. Psychiatric consultation to assess the patient’s safety was also offered if a participant scored positive on the suicide-screening question (#10) of the EPDS.
The reasons for admission were divided into six categories: threatened preterm labor, intrauterine growth restriction (IUGR), preterm premature rupture of membranes, gestational diabetes mellitus, hypertensive disorder, or other. The categorization of the other obstetric and socio-demographic parameters is shown in Table 1.
Statistical analysis
Descriptive statistics are displayed as mean ± SD for continuous variables, while absolute (n) and relative frequencies (%) are presented for qualitative variables. The primary outcome variable of interest was the EPDS score on the date of admission in the HRPU. The EPDS score was used as a categorical variable using the cutoff limit of 13. Univariate analysis between categorical dependent and independent variables was performed using Pearson’s Chi-square test for categorical variables. Using significant variables (p < 0.05) from the univariate analysis, a multivariate logistic regression model was fitted to identify the independent predictors of antenatal depression. Estimated associations are described in terms of relative risks (RRs) and odds ratios (ORs) with 95 % confidence intervals (CIs). The level of statistical significance was set at 0.05. Statistical analysis was performed using SPSS v.22.0.
Results
Socio-demographic and obstetric characteristics
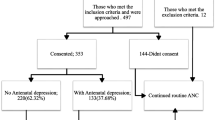
Overall, 254 women were admitted in the HRPU during the 14 month study period; 161 of them fulfilled the criteria, and of those 157 (97.5 %) agreed to participate in the study. The mean age of the women was 30 years (SD:6, range 14–47 years); 71.3 % (n = 112) were below the age of 35; the mean gestational age was 33 weeks (range 24–36, SD:3); and the mean body mass index (BMI) before pregnancy was 26.4 kg/m2 (SD = 9.9) with 80.9 % (n = 127) less than 30 kg/m2. The most common reason for hospital admission was threatened preterm labor (41.4 % n = 65). One hundred thirty-four (85.4 %) of the women were married, 55 (35.0 %) had a high educational level, 61 (38.9 %) were employed, and 60 (38.2 %) had a monthly family income of ≥€1000. The majority of the respondents (68.2 %, n = 107) reported that the relationship with their partner was good, and in 102 (65 %) cases, the pregnancy was planned. Fourteen (8.9 %) of the women had thoughts of abortion, and 25 (15.9 %) were smoking during pregnancy (Table 1).
Prevalence of antenatal depression
The mean EPDS score was 9.85 (SD = 5.5; range 0–24; 95 % CI 8.98–10.72; SE:0.44). The prevalence of antenatal depression (EPDS score ≥13) was 28 % (n = 44). Six (4 %) participants scored positive on the suicide-screening question (#10) of the EPDS. The median length of hospitalization was 5 days (range 1–72 days). The majority (72.6 %, n = 114) of the women stayed in the HRPU for less than 1 week and, therefore, completed the EPDS only once. For the rest of the women who completed the questionnaire more than once, no significant differences were observed (data available upon request).
Factors associated with antenatal depression
The following characteristics were not significantly associated with antenatal depression in the univariate analysis: reason for admission, maternal age, gestational age, BMI before pregnancy, marital status, maternal education, employment status, family monthly income, relationship with the partner, and planned pregnancy. On the contrary, abortion thoughts and smoking were statistically associated with depression in the univariate analysis (Table 2). In the multivariate analysis, these two factors were independently associated with antenatal depression (Table 3). Antenatal depression was significantly higher among women who had abortion thoughts than those who wanted the pregnancy (OR 3.624, 95 % CI 1.149–11.427); smoking during pregnancy was also associated with a higher risk for antenatal depression (OR 2.681, 95 % CI 1.089–6.601) (Table 3).
Discussion
Cross-sectional data reported here were derived from a larger prospective study, the first one ever conducted in Greece, on maternal antenatal depression among pregnant women hospitalized in a high-risk pregnancy unit. Our results showed that there was a high prevalence of antenatal depression among pregnant women who were hospitalized in the HRPU. In fact, 28 % of the participants had antenatal depression based on their EPDS score. This rate is higher than previously reported in hospitalized pregnant women [3, 27]. Socio-demographic and economic differences, especially at this time of severe economic crisis, may explain the different prevalence of antenatal depression between this study and previous studies from other countries.
It is well known that pregnancy does not protect women from depression [28, 29] and that a prior history of mental disease is a risk factor for antenatal depression [30]. In this study, all women with a previous history of mental illness were excluded. Our findings possibly show the stressful influence of pregnancy complications on the woman’s mental health.
In our study, antenatal depression was significantly higher among women who had abortion thoughts. These women were four times more likely to have antenatal depression than those without abortion thoughts. Our results are in compliance with the results of previous studies among pregnant women in various countries [31–33] and with a recently published systematic review and meta-analysis that showed that the prevalence of perinatal depression is increased twofold in women with an unintended pregnancy [34]. Moreover, a previous study has shown that women who had thoughts about having an abortion were also more prone to develop postpartum depression [35].
The association between smoking and depression is well-recognized in the general population. In cross-sectional studies, smoking is associated with a nearly twofold increase in the risk for depression compared with both never-smokers and former-smokers [36]. Smoking in pregnancy has also been documented as a significant factor linked to antenatal depression. Numerous cross-sectional studies that compared mental health level between pregnant smokers and non-smokers reported a higher prevalence of depressive symptoms among smokers [7, 32, 37–40]. In our study, smoking during pregnancy was also significantly associated with a higher prevalence of antenatal depression.
Summarizing, clinicians should be aware of the high prevalence of depression in women admitted in high-risk pregnancy units, as early identification of depressive symptoms may lead to appropriate treatment and help protect maternal and fetal health. As mentioned before, the American College of Obstetricians and Gynecologists has recommended that all women should be screened at least once for depression symptoms. This strategy needs to be tested for its efficacy and further investigated for the appropriate time of conducting the screening.
This study has certain limitations. The relatively small sample size does not allow for adequate subdivision of different reasons for admission and the association with pregnancy outcome. The small sample size could also explain the wide CIs noticed in the multivariate regression analysis. Thus, caution should be exercised when extrapolating the findings beyond the study population. However, this is an ongoing study, and in time, these comparisons may reveal the relative burden of each pregnancy complication on maternal mental health and pregnancy outcome. Furthermore, as EDPS is not a diagnostic tool but rather a screening method, making a definite diagnosis of antenatal depression based on the EDPS scale without psychiatric examination is not appropriate. It is essential that pregnant women who score high receive further assessment by a psychiatrist to confirm or refute the suspected diagnosis. Last, the exclusion of women with a previous history of depression may underestimate the total prevalence of antenatal depression, as a previous history of depression is an important risk factor for the development of antenatal depression [41, 42]. However, considering that in Greece, there is a lack of any registries concerning major depression or other psychiatric disorders, we decided to exclude pregnant women without a previously confirmed diagnosis or prescribed medication by a mental health specialist. The number of women with a confirmed medical history of depression was very small in our sample (3 %), and they were excluded from our study.
Conclusions
Our results further support previous studies that found a higher prevalence rate of depression symptoms in women with high-risk pregnancies than that reported in low-risk pregnancy populations. Furthermore, we identified smoking and having thoughts of abortion as risk factors for depression. Given the high rates of antenatal depression, especially in complicated pregnancies, and given its association with adverse maternal and fetal outcome, there is a need for further studies that will examine the effect of screening and appropriate psychiatric consultation on pregnancy outcome. Further research is also needed to determine the factors, which place hospitalized pregnant women at risk for depression and the optimal approach for detecting and treating major depression among such patients. A new model of cooperation between the obstetrician and the psychiatrist in the HRPU may be necessary to promote the mental health of pregnant women.
References
Bennett HA, Einarson A, Taddio A, Koren G, Einarson TR (2004) Prevalence of depression during pregnancy: systematic review. Obstet Gynecol 103:698–709. doi:10.1097/01.AOG.0000116689.75396.5f
Evans J, Heron J, Francomb H, Oke S, Golding J (2001) Cohort study of depressed mood during pregnancy and after childbirth. BMJ 323:257–260
Adouard F, Glangeaud-Freudenthal NM, Golse B (2005) Validation of the Edinburgh postnatal depression scale (EPDS) in a sample of women with high-risk pregnancies in France. Arch Womens Ment Health 8:89–95. doi:10.1007/s00737-005-0077-9
Brandon AR, Trivedi MH, Hynan LS, Miltenberger PD, Labat DB, Rifkin JB, Stringer CA (2008) Prenatal depression in women hospitalized for obstetric risk. J Clin Psychiatry 69:635–643
Powers PS, Johnson T, Knuppel R, Cupoli JM, Achenbach KE (1986) Psychiatric disorders in high-risk pregnancy. Compr Psychiatry 27:159–164
King NM, Chambers J, O’Donnell K, Jayaweera SR, Williamson C, Glover VA (2010) Anxiety, depression and saliva cortisol in women with a medical disorder during pregnancy. Arch Womens Ment Health 13:339–345. doi:10.1007/s00737-009-0139-5
Zuckerman B, Amaro H, Bauchner H, Cabral H (1989) Depressive symptoms during pregnancy: relationship to poor health behaviors. Am J Obstet Gynecol 160:1107–1111
Alder J, Fink N, Bitzer J, Hosli I, Holzgreve W (2007) Depression and anxiety during pregnancy: a risk factor for obstetric, fetal and neonatal outcome? A critical review of the literature. J Matern Fetal Neonatal Med 20:189–209. doi:10.1080/14767050701209560
Grote NK, Bridge JA, Gavin AR, Melville JL, Iyengar S, Katon WJ (2010) A meta-analysis of depression during pregnancy and the risk of preterm birth, low birth weight, and intrauterine growth restriction. Arch Gen Psychiatry 67:1012–1024. doi:10.1001/archgenpsychiatry.2010.111
Van den Bergh BR, Mulder EJ, Mennes M, Glover V (2005) Antenatal maternal anxiety and stress and the neurobehavioural development of the fetus and child: links and possible mechanisms. A review. Neurosci Biobehav Rev 29:237–258. doi:10.1016/j.neubiorev.2004.10.007
Pearson RM, Bornstein MH, Cordero M, Scerif G, Mahedy L, Evans J, Abioye A, Stein A (2015) Maternal perinatal mental health and offspring academic achievement at age 16: the mediating role of childhood executive function. J Child Psychol Psychiatry. doi:10.1111/jcpp.12483
Logsdon MC, Wisner KL, Pinto-Foltz MD (2006) The impact of postpartum depression on mothering. J Obstet Gynecol Neonatal Nurs 35:652–658. doi:10.1111/j.1552-6909.2006.00087.x
Austin MP (2004) Antenatal screening and early intervention for “perinatal” distress, depression and anxiety: where to from here? Arch Womens Ment Health 7:1–6. doi:10.1007/s00737-003-0034-4
Gotlib IH, Whiffen VE, Mount JH, Milne K, Cordy NI (1989) Prevalence rates and demographic characteristics associated with depression in pregnancy and the postpartum. J Consult Clin Psychol 57:269–274
Koutra K, Vassilaki M, Georgiou V, Koutis A, Bitsios P, Chatzi L, Kogevinas M (2014) Antenatal maternal mental health as determinant of postpartum depression in a population based mother-child cohort (Rhea Study) in Crete, Greece. Soc Psychiatry Psychiatr Epidemiol 49:711–721. doi:10.1007/s00127-013-0758-z
Field T, Diego M, Hernandez-Reif M, Figueiredo B, Deeds O, Ascencio A, Schanberg S, Kuhn C (2010) Comorbid depression and anxiety effects on pregnancy and neonatal outcome. Infant Behav Dev 33:23–29. doi:10.1016/j.infbeh.2009.10.004
The American College of Obstetricians and Gynecologists (2015) Screening for Perinatal Depression. Committee Opinion Number 630
Littleton HL, Breitkopf CR, Berenson AB (2007) Correlates of anxiety symptoms during pregnancy and association with perinatal outcomes: a meta-analysis. Am J Obstet Gynecol 196:424–432. doi:10.1016/j.ajog.2007.03.042
Heaman M (1992) Stressful life events, social support, and mood disturbance in hospitalized and non-hospitalized women with pregnancy-induced hypertension. Can J Nurs Res 24:23–37
Tsai SJ, Lee YC, Yang CH, Sim CB (1996) Psychiatric consultations in obstetric inpatients. J Obstet Gynaecol Res 22:603–607
Cox JL, Holden JM, Sagovsky R (1987) Detection of postnatal depression. Development of the 10-item Edinburgh postnatal depression scale. Br J Psychiatry 150:782–786
Murray L, Carothers AD (1990) The validation of the Edinburgh post-natal depression scale on a community sample. Br J Psychiatry 157:288–290
Leonardou AAZY, Papageorgiou CC, Marks MN, Tsartsara EC, Antsaklis A, Christodoulou GN, Soldatos CR (2009) Validation of the Edinburgh postnatal depression scale and prevalence of postnatal depression at 2 months postpartum in a sample of Greek mothers. J Reprod Infant Psyc 27(1):28–39
Vivilaki VG, Dafermos V, Kogevinas M, Bitsios P, Lionis C (2009) The Edinburgh postnatal depression scale: translation and validation for a Greek sample. BMC Public Health 9:329. doi:10.1186/1471-2458-9-329
Brouwers EP, van Baar AL, Pop VJ (2001) Does the Edinburgh postnatal depression scale measure anxiety? J Psychosom Res 51:659–663
Gibson J, McKenzie-McHarg K, Shakespeare J, Price J, Gray R (2009) A systematic review of studies validating the Edinburgh postnatal depression scale in antepartum and postpartum women. Acta Psychiatr Scand 119:350–364. doi:10.1111/j.1600-0447.2009.01363.x
Thiagayson P, Krishnaswamy G, Lim ML, Sung SC, Haley CL, Fung DS, Allen JC Jr, Chen H (2013) Depression and anxiety in Singaporean high-risk pregnancies–prevalence and screening. Gen Hosp Psychiatry 35:112–116. doi:10.1016/j.genhosppsych.2012.11.006
Fishell A (2010) Depression and anxiety in pregnancy. J Popul Ther Clin Pharmacol 17:e363–e369
Pearlstein T (2008) Perinatal depression: treatment options and dilemmas. J Psychiatry Neurosci 33:302–318
Redshaw M, Henderson J (2013) From antenatal to postnatal depression: associated factors and mitigating influences. J Womens Health (Larchmt) 22:518–525. doi:10.1089/jwh.2012.4152
Hickey CA, Cliver SP, Goldenberg RL, McNeal SF, Hoffman HJ (1995) Relationship of psychosocial status to low prenatal weight gain among nonobese black and white women delivering at term. Obstet Gynecol 86:177–183
Silva RA, Jansen K, Souza LD, Moraes IG, Tomasi E, Silva Gdel G, Dias Mde S, Pinheiro RT (2010) Depression during pregnancy in the Brazilian public health care system. Rev Bras Psiquiatr 32:139–144
Kitamura T, Yoshida K, Okano T, Kinoshita K, Hayashi M, Toyoda N, Ito M, Kudo N, Tada K, Kanazawa K, Sakumoto K, Satoh S, Furukawa T, Nakano H (2006) Multicentre prospective study of perinatal depression in Japan: incidence and correlates of antenatal and postnatal depression. Arch Womens Ment Health 9:121–130. doi:10.1007/s00737-006-0122-3
Abajobir AA, Maravilla JC, Alati R, Najman JM (2016) A systematic review and meta-analysis of the association between unintended pregnancy and perinatal depression. J Affect Disord 192:56–63. doi:10.1016/j.jad.2015.12.008
Moraes IG, Pinheiro RT, Silva RA, Horta BL, Sousa PL, Faria AD (2006) Prevalence of postpartum depression and associated factors. Rev Saude Publica 40:65–70
Luger TM, Suls J, Vander Weg MW (2014) How robust is the association between smoking and depression in adults? A meta-analysis using linear mixed-effects models. Addict Behav 39:1418–1429. doi:10.1016/j.addbeh.2014.05.01
Smedberg J, Lupattelli A, Mardby AC, Overland S, Nordeng H (2015) The relationship between maternal depression and smoking cessation during pregnancy–a cross-sectional study of pregnant women from 15 European countries. Arch Womens Ment Health 18:73–84. doi:10.1007/s00737-014-0470-3
Goodwin RD, Keyes K, Simuro N (2007) Mental disorders and nicotine dependence among pregnant women in the United States. Obstet Gynecol 109:875–883
Orr ST, Blazer DG, Orr CA (2012) Maternal prenatal depressive symptoms, nicotine addiction, and smoking-related knowledge, attitudes, beliefs, and behaviors. Matern Child Health J 16:973–978. doi:10.1007/s10995-011-0822-9
Linares Scott TJ, Heil SH, Higgins ST, Badger GJ, Bernstein IM (2009) Depressive symptoms predict smoking status among pregnant women. Addict Behav 34:705–708. doi:10.1016/j.addbeh.2009.04.003
Dayan J, Creveuil C, Dreyfus M, Herlicoviez M, Baleyte JM, O’Keane V (2010) Developmental model of depression applied to prenatal depression: role of present and past life events, past emotional disorders and pregnancy stress. PLoS One 5:e12942. doi:10.1371/journal.pone.0012942
Johanson R, Chapman G, Murray D, Johnson I, Cox J (2000) The North Staffordshire Maternity Hospital prospective study of pregnancy-associated depression. J Psychosom Obstet Gynaecol 21:93–97
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Dagklis, T., Papazisis, G., Tsakiridis, I. et al. Prevalence of antenatal depression and associated factors among pregnant women hospitalized in a high-risk pregnancy unit in Greece. Soc Psychiatry Psychiatr Epidemiol 51, 1025–1031 (2016). https://doi.org/10.1007/s00127-016-1230-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00127-016-1230-7