Abstract
Aims/hypothesis
Experimental studies suggest that the fatty acid palmitoleate may act as an adipocyte-derived lipid hormone (or ‘lipokine’) to regulate systemic metabolism. We investigated the relationship of circulating palmitoleate with insulin sensitivity, beta cell function and glucose tolerance in humans.
Methods
Plasma NEFA concentration and composition were determined in non-diabetic individuals from the Relationship between Insulin Sensitivity and Cardiovascular disease (RISC) study cohort at baseline (n = 1234) and after a 3 year follow-up (n = 924). Glucose tolerance, insulin secretion and beta cell function were assessed during an OGTT. Whole-body insulin sensitivity was measured by a hyperinsulinaemic–euglycaemic clamp (M/I) and OGTT (oral glucose insulin sensitivity index [OGIS]). The liver insulin resistance index was calculated using clinical and biochemical data. Body composition including fat mass was determined by bioelectrical impedance.
Results
Circulating palmitoleate was proportional to fat mass (r = 0.21, p < 0.0001) and total NEFA levels (r = 0.19, p < 0.0001). It correlated with whole-body insulin sensitivity (M/I: standardised regression coefficient [std. β] = 0.16, p < 0.0001), liver insulin resistance (std. β = −0.14, p < 0.0001), beta cell function (potentiation: std. β = 0.08, p = 0.045) and glucose tolerance (2 h glucose: std. β = −0.24, p < 0.0001) after adjustment for age, sex, BMI, adiposity and other NEFA. High palmitoleate concentrations prevented the decrease in insulin sensitivity associated with excess palmitate (p = 0.0001). In a longitudinal analysis, a positive independent relationship was observed between changes in palmitoleate and insulin sensitivity over time (std. β = 0.07, p = 0.04).
Conclusions/interpretation
We demonstrated that plasma palmitoleate is an independent determinant of insulin sensitivity, beta cell function and glucose tolerance in non-diabetic individuals. These results support the role of palmitoleate as a beneficial lipokine released by adipose tissue to prevent the negative effects of adiposity and excess NEFA on systemic glucose metabolism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction

Plasma NEFA concentrations are higher in obese individuals [1] and this may contribute to the pathogenesis of obesity-associated insulin resistance and beta cell dysfunction [2,3,4,5]. However, the distinct and potentially opposite effects of individual NEFA on insulin action and secretion are still poorly understood.
The fatty acid palmitoleate (16:1 n-7), also known as 9-hexadecenoic acid, is the second most abundant monounsaturated fatty acid (MUFA) in human blood and adipose tissue [6]. The two main sources of circulating palmitoleate are endogenous fat synthesis (cis isomer) and dietary whole-fat dairy products (trans isomer). Both palmitoleate isomers have been associated with lower metabolic risk [7,8,9,10,11,12,13]. The enrichment of cis-palmitoleate (hereinafter simply referred to as palmitoleate) in the plasma NEFA pool depends on its endogenous synthesis by subcutaneous adipose tissue, from which palmitoleate can be readily mobilised in response to different metabolic stimuli [9]. Recent studies in rodent models and cell cultures have reported that palmitoleate can directly enhance whole-body glucose disposal [7, 14,15,16,17], attenuate hepatosteatosis induced by a high-fat diet or diabetes [7, 14, 18] and protect beta cells from palmitate-induced apoptosis [19]. These observations support the physiological relevance of palmitoleate as an adipocyte-derived lipid hormone (or ‘lipokine’) by which the adipose tissue can regulate systemic metabolism [7,8,9,10,11,12].
In humans, the potential metabolic benefits of palmitoleate are controversial. A positive relationship between palmitoleate levels and insulin sensitivity has been observed in some studies [8,9,10], but others have reported no or even negative correlations [4, 20,21,22,23]. These discrepancies may be due to small sample sizes, inadequate assessment of insulin sensitivity and beta cell function, and lack of longitudinal data. More importantly, few studies have been able to provide detailed adjustment for potential confounders, such as total NEFA concentration and composition.
The aim of this study was to investigate whether circulating palmitoleate is an independent determinant of insulin sensitivity and beta cell function in humans. To this end, plasma concentration and relative enrichment of the most abundant NEFA were measured by a targeted quantitative metabolomic approach in a large cohort of non-diabetic individuals at baseline and after a 3 year follow-up. Insulin sensitivity and beta cell function variables were determined by means of the hyperinsulinaemic–euglycaemic clamp and by mathematical modelling of plasma glucose and C-peptide concentrations during a 2 h OGTT.
Methods
Study design and participants
The Relationship between Insulin Sensitivity and Cardiovascular disease (RISC) study is a multicentre, prospective, observational study involving 1566 participants at baseline and 1059 participants at follow up between 30 and 60 years of age, recruited in 19 centres in 14 European countries [24]. Major exclusion criteria were diabetes, dyslipidaemia, hypertension, class III obesity, chronic lung, hepatic or kidney diseases and neoplastic and inflammatory diseases. Information regarding medical history, drug use and family history of diabetes (i.e. any first-degree family member with type 2 diabetes) was collected using standardised self-reported questionnaires. The examination protocol included anthropometry, blood pressure measurements, a fasting blood test, a euglycaemic−hyperinsulinaemic clamp at baseline and an OGTT at baseline and after a 3 year follow-up. Anthropometric, clinical and biochemical measurements as well as metabolic tests were performed in all centres according to standardised methods [24].
For the purpose of this study, we excluded individuals whose fasting blood samples were not available for NEFA measurement (n = 330) and those individuals with total NEFA inappropriately elevated (higher than 2000 μmol/l, n = 2). This resulted in a population of 1234 participants for the cross-sectional study and 924 participants for the prospective analysis.
The study was conducted according to the principles expressed in the Declaration of Helsinki and approved by the local ethics committee of each centre. Written informed consent was obtained before recruitment from all participants.
Metabolic tests
OGTTs were performed at baseline and at follow-up. Blood samples were collected at fasting and at 30, 60, 90 and 120 min after the ingestion of 75 g glucose for measurement of glucose, insulin and C-peptide concentrations. Normal glucose tolerance (NGT), impaired fasting glucose (IFG), impaired glucose tolerance (IGT) and type 2 diabetes were defined according to the current ADA criteria [25].
A euglycaemic−hyperinsulinaemic clamp was performed on a separate day within 1 month of the baseline OGTT. Insulin was administered as a primed-continuous infusion at a rate of 240 pmol min−1 m−2. Plasma glucose was clamped at 4.5–5.5 mmol/l with a variable 20% dextrose infusion adjusted every 5 min. Blood was sampled every 20 min for determination of insulin concentrations. OGTT and clamp procedures were standardised across centres.
Insulin sensitivity and beta cell function variables
Insulin sensitivity was expressed as the ratio of the average glucose infusion rate (M value) during the final 40 min of the clamp normalised to the fat-free mass (FFM) and to the mean plasma insulin concentration measured during the same time interval (M/I, in units of μmol kgFFM−1 min−1 [nmol/l]−1). Insulin sensitivity was also measured by the oral glucose insulin sensitivity index (OGIS) calculated during the baseline and follow-up OGTTs [26]. The liver insulin resistance index was calculated in all participants with an algorithm based on OGTT insulin levels, percentage fat mass (FM%), BMI and HDL-cholesterol [27]. This index strongly correlates with the tracer-derived endogenous glucose production relative to insulin at baseline (r = 0.65, p < 0.001) and during clamp (r = 0.59, p < 0.001) in these individuals without diabetes [27].
Basal and glucose-stimulated insulin secretion rate was estimated by C-peptide deconvolution [28]. Total insulin output throughout the OGTT was calculated by integrating insulin secretion rate over the duration of the test. Variables of beta cell function were calculated by mathematical modelling of insulin secretion and glucose concentrations, as previously reported [29,30,31]. This model describes the relationship between beta cell insulin secretion and glucose concentration as the sum of two components. The first component represents the dependence of insulin secretion on absolute glucose concentration through a dose–response function relating the two variables. The slope of this dose–response function is calculated and called beta cell glucose sensitivity. The dose–response is modulated by a potentiation factor, which accounts for the physiological processes that can acutely modify insulin secretion (e.g. protracted hyperglycaemia, non-glucose substrates, gastrointestinal hormones, neural modulation). The second component of beta cell function represents the dependence of insulin secretion on the rate of change of glucose concentration and is determined by a single variable (beta cell rate sensitivity), which is related to early insulin release.
Body composition
Body weight and FFM were assessed by electrical bioimpedance using a Body Composition Analyzer (model TB-300; Tanita, Tokyo, Japan). Fat mass was obtained as the difference between body weight and FFM; FM% was calculated as the ratio of fat mass to body weight. Waist circumference was measured at the narrowest circumference between the lower rib margin and anterior superior iliac crest. Hip circumference was measured around the widest portion of the buttocks, and the waist/hip ratio was calculated.
Biochemical measurements
Blood glucose was measured at the bedside by the glucose oxidase technique. Plasma samples were divided into aliquots and stored at −80°C until analysis. Centralised biochemical analyses were performed in predefined core laboratories to minimise assay errors and variability [24]. Plasma insulin and C-peptide were measured by fluoroimmunoassay (AutoDELFIA Insulin kit; Wallac Oy, Turku, Finland). Total plasma NEFA were measured by a fluorometric method (Wako, Neuss, Germany). Plasma concentrations of the six most prevalent NEFA, including saturated fatty acids (SFA: palmitate [16:0] and stearate [18:0]), monounsaturated fatty acids (MUFA: palmitoleate [16:1 n-7] and oleate [18:1 n-9]), and polyunsaturated fatty acids (PUFA: linoleate [18:2] and linolenate [18:3]), were measured by a targeted quantitative metabolomic approach using isotope dilution ultrahigh-performance liquid chromatography coupled to tandem MS (MS/MS), as previously reported [32].
Statistical analysis
Continuous variables are presented as mean ± SD and nominal variables are expressed as percentages. Variables with a skewed distribution are presented as median (interquartile range) and were log-transformed in multivariable analyses. Differences between groups (i.e. tertile of plasma palmitoleate at baseline or tertile of palmitoleate change at follow-up) were tested using χ2 for nominal variables and using ANOVA or Kruskal–Wallis test for normally or non-normally distributed continuous variables, respectively. Post hoc pairwise comparisons were tested by Tukey’s honest significant difference (HSD) test or by Steel–Dwass test, as appropriate. Differences between participants’ characteristics at baseline and at follow-up were assessed by paired Student’s t test or Wilcoxon signed-rank test. Correlations were tested using Pearson or Spearman correlation coefficients. Multivariable linear regression analysis tested the effect of palmitoleate on insulin sensitivity, beta cell function and glucose tolerance while controlling for potential confounders.
Cross-sectional analyses were adjusted for age, sex, BMI, FM% and total NEFA (Model 1). They were further corrected by adding the percentage enrichments of all measured NEFA as covariates (Model 2). The effect modification by sex was examined by adding a product term to all regression models (palmitoleate × sex), which was eventually removed as it showed no significant effect. The effect of palmitoleate on the expected [33] negative correlation between insulin sensitivity and palmitate was tested by including plasma palmitate, palmitoleate tertile and a product term between the two variables as factors. Longitudinal analyses were adjusted for baseline OGIS and percentage changes in BMI, FM% and total NEFA. They were further corrected by sex, age, follow-up duration and a product term between baseline OGIS and changes in palmitoleate to estimate the effect modification by baseline insulin sensitivity. Longitudinal analyses were also repeated using baseline M/I instead of baseline OGIS as covariate. To quantify the relative contribution of palmitoleate with respect to other factors, standardised regression coefficients (std. β), which indicate how many SDs the dependent variable changes per SD change in the predictor variable, were obtained from models on standardised variables. Statistical analyses were performed using JMP Pro 13.2.1 software (SAS Institute, Cary, NC, USA). A two-sided p < 0.05 was considered statistically significant.
Results
Cross-sectional analyses
Baseline clinical and metabolic characteristics of study participants (n = 1234) stratified by tertile of percentage palmitoleate enrichment in the total NEFA pool (PO%) are shown in Table 1.
The mean (± SD) value of total NEFA was 531 ± 210 μmol/l (range 70–1585 μmol/l). Total NEFA were higher in women than men (585 ± 214 and 465 ± 184 μmol/l, respectively, p < 0.0001) and associated with age (r = 0.09, p < 0.0001). They were also associated with FM% (r = 0.28, p < 0.0001), clamp-derived whole-body insulin sensitivity (M/I: r = −0.16, p < 0.0001) and liver insulin resistance (r = 0.11, p < 0.0001), but not with BMI (p = 0.40) or beta cell function variables (p > 0.14 for all).
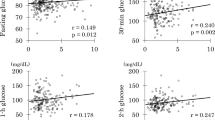
Plasma palmitoleate concentration was 13 ± 8 μmol/l (range 1–61 μmol/l), being higher in women than men (15 ± 8 μmol/l vs 10 ± 6 μmol/l, respectively, p < 0.0001) and increased with age (r = 0.10, p = 0.0003). PO% was 2.3 ± 0.8% (range 0.3–5.9%) and correlated with absolute palmitoleate concentration (r = 0.71, p < 0.0001). Consistently, PO% was higher in women (2.5 ± 0.9 % vs 2.1 ± 0.7 %, p < 0.0001) and in older people (age: r = 0.10, p = 0.0003). PO% was positively correlated with total NEFA concentration (r = 0.19, p < 0.0001) and adiposity (FM%: r = 0.21, p < 0.0001), while other major NEFA decreased or remained stable as total NEFA and FM% increased (Fig. 1; subgroup analyses by sex are shown in electronic supplementary material [ESM] Fig. 1). Furthermore, PO% was associated with plasma adiponectin concentration (r = 0.21, p < 0.0001) and negatively correlated with waist/hip circumference ratio (r = −0.19, p < 0.0001) and alanine aminotransferase levels (r = −0.15, p < 0.0001).
The M/I, which provides an accurate estimate of whole-body insulin sensitivity, was significantly higher in those with higher PO% after stratification for total NEFA (Fig. 2a) or FM% (Fig. 2b). This finding was confirmed in women and men separately stratified by sex-specific tertile of total NEFA and FM% (ESM Figs 2 and 3). In bivariate analyses, PO% positively correlated with M/I in the whole cohort (r = 0.20, p < 0.0001) and in women and men separately (r = 0.18, p < 0.0001 and r = 0.09, p = 0.03, respectively), while there was no significant relationship between absolute palmitoleate concentration and M/I (p = 0.99). The association between PO% and M/I remained significant after adjustment for age, sex, BMI, FM% and total NEFA (Model 1: std. β = 0.14, p < 0.0001) (Fig. 3) and when all other major NEFA were added to the model (Model 2: std. β = 0.16, p < 0.0001). PO% was also associated with the liver insulin resistance index both in bivariate analysis (r = −0.06, p = 0.04) and multiple adjusted analyses (Model 1: std. β = −0.08, p < 0.0001; Model 2: std. β = −0.14, p < 0.0001). Furthermore, high palmitoleate concentrations attenuated the expected decrease in insulin sensitivity associated with elevated palmitate levels (palmitate by palmitoleate interaction effect: p = 0.0001) (Fig. 4). With regard to other NEFA, oleate enrichment was not correlated with insulin sensitivity in adjusted models (p = 0.14); this was also the case for the two SFAs, palmitate and stearate (p > 0.10). PUFA showed a positive correlation with insulin sensitivity, though weaker compared with palmitoleate (linoleate: std. β = 0.06, p = 0.04; linolenate: std. β = 0.08, p = 0.006).
Insulin sensitivity (a, b), beta cell glucose sensitivity (c, d), potentiation (e, f) and 2 h plasma glucose (g, h) in non-diabetic individuals stratified by tertile of plasma NEFA (a, c, e, g) or FM% (b, d, f, h) and by tertile of PO% (n = 1234). Whole-body insulin sensitivity was measured by a hyperinsulinaemic–euglycaemic clamp (M/I). Beta cell function variables and glucose tolerance were assessed during an OGTT. Data are means ± SEM. Differences were tested using ANOVA or Kruskal–Wallis test followed by post hoc pairwise comparisons. *p < 0.05 vs PO% tertile I, †p < 0.05 vs PO% tertile II
Multivariable linear regression analyses of the relation between PO% and measures of insulin sensitivity, beta cell function and glucose tolerance in non-diabetic individuals (n = 1234). The effect of PO% remained significant after adjustment for sex, age, BMI, FM% and total NEFA. Standardised coefficients and 95% CI are shown
Association between plasma palmitate concentration and clamp-derived insulin sensitivity in non-diabetic individuals stratified by tertile of plasma palmitoleate (n = 1234). High palmitoleate levels prevented the reduction in insulin sensitivity induced by excess palmitate (palmitate by palmitoleate interaction effect in multivariable regression analysis: p = 0.0001)
Among beta cell function variables, beta cell glucose sensitivity and potentiation were significantly enhanced in individuals with higher PO% after stratification by total NEFA or FM% (Fig. 2c−f). In subgroup analyses, the effect of PO% on beta cell function appeared more consistent in women than in men (ESM Figs 2 and 3). In bivariate analyses in the whole cohort, beta cell glucose sensitivity and potentiation correlated with PO% (r = 0.08, p = 0.008 and r = 0.15, p < 0.0001, respectively), but not with absolute palmitoleate concentration (p = 0.72 and p = 0.12, respectively). The associations between beta cell function variables and PO% remained statistically significant after adjustment for age, sex, BMI, FM% and total NEFA (Model 1: std. β = 0.07, p = 0.01 and std. β = 0.10, p = 0.001, respectively) (Fig. 3) and, only for potentiation, after accounting for all measured NEFA (Model 2: std. β = 0.08, p = 0.045). Among other major NEFA, only linoleate enrichment was associated with beta cell glucose sensitivity (std. β = 0.08, p = 0.007), while no other NEFA was associated with potentiation besides palmitoleate.
Given the positive correlation of PO% with both insulin sensitivity and beta cell function, we tested the association with glucose tolerance assessed by the 2 h plasma glucose levels during the OGTT. As expected, 2 h plasma glucose was lower in individuals with higher PO% (Fig. 2g−h; ESM Figs 2 and 3). The two variables were negatively correlated in bivariate analysis (r = −0.08, p = 0.008), and after adjustments for potential confounders (Model 1: std. β= −0.12, p < 0.0001; Model 2: std. β = −0.24, p < 0.0001) (Fig. 3).
Longitudinal analyses
Clinical and metabolic characteristics of study participants at baseline and at follow-up (n =924) are shown in Table 2.
During the 3 year observational follow-up, PO% decreased by 0.3% (range −3.7% to 5.3%, p < 0.0001). Percentage changes at follow-up in PO% were not associated with percentage changes in FM%, waist/hip ratio or hip circumference (p > 0.50 for both) and showed only a marginally significant correlation with percentage changes in BMI (r = 0.06, p = 0.05). Furthermore, we did not observe associations of PO% changes with either beta cell glucose sensitivity (p = 0.79) or potentiation (p = 0.43).
At follow-up, insulin sensitivity assessed by the OGIS decreased on average by 10 ml min−1 m−2 (range −283 to 533 ml min−1 m−2, p < 0.0001). The spontaneous decline in OGIS was gradually attenuated across tertiles of changes in PO% (p = 0.04), approaching the null value in individuals whose PO% increased (tertile III) (Fig. 5a). Consistently, insulin sensitivity at follow-up was lower in people who had a decrease in PO% (n = 630) compared with those whose PO% increased (n = 294, p = 0.01), despite no differences in baseline OGIS nor in changes in BMI, FM% and total NEFA between the two subgroups. Furthermore, a positive relationship was observed between percentage changes in PO% and insulin sensitivity (OGIS: r = 0.07, p = 0.05). This association remained significant after adjustment for baseline OGIS and percentage changes in BMI, FM% and total NEFA (std. β = 0.07, p = 0.03) (Fig. 5b). It also remained significant after further adjustment for sex, age, follow-up duration and an interaction factor between percentage changes in PO% and baseline OGIS (std. β = 0.07, p = 0.04). The last factor showed no significant effect (p = 0.49). The effect of PO% changes on insulin sensitivity was similar when accounting for baseline M/I instead of baseline OGIS in the fully adjusted model (std. β = 0.07, p = 0.049).
(a) Percentage change in OGIS (Δ% OGIS ) after a 3 year observational follow-up in non-diabetic individuals stratified by spontaneous percentage change in palmitoleate enrichment (Δ% PO%) (n = 924). Data are means ± SEM. Differences were tested using Kruskal–Wallis test followed by post hoc pairwise comparisons. *p < 0.05 vs Δ% PO% tertile I. (b) Multivariable linear regression analysis of the relationship between Δ% PO% and Δ% OGIS in the whole cohort, adjusted for baseline OGIS and for percentage changes in BMI (Δ% BMI), FM% (Δ% FM%) and total NEFA (Δ% NEFA). Standardised coefficients and 95% CI are shown
Discussion
In a large cohort of non-diabetic individuals, we demonstrated that the relative abundance of circulating palmitoleate, which increases proportionally with adiposity and total NEFA concentration, is associated with enhanced whole-body and liver insulin sensitivity, beta cell function and glucose tolerance. Furthermore, we observed that increased palmitoleate enrichment attenuates the negative effects of excess palmitate and ageing on insulin sensitivity. Our findings suggest that palmitoleate participates in the crosstalk between adipose tissue and other metabolically active organs/tissues, such as liver and beta cells, and support its protective role against glucose intolerance under physiological conditions.
Previous animal and cell culture studies have shown that palmitoleate can improve insulin signalling and enhance beta cell function [7, 14,15,16,17,18,19, 34, 35], in addition to its potential anti-obesity, anti-inflammatory, anti-oxidative and anti-atherosclerotic properties [11, 12]. Our novel findings support an insulin-sensitising effect of palmitoleate in humans. In multiple cross-sectional analyses, we described a positive relationship between palmitoleate levels and whole-body insulin sensitivity, determined by a gold standard clamp procedure. Furthermore, circulating palmitoleate showed a negative correlation with hepatic insulin resistance assessed by a validated index [27]. Palmitoleate was able to predict insulin sensitivity after stratification for adiposity and total NEFA as well as after accounting for potential confounders. Consistently, longitudinal analyses confirmed that changes in palmitoleate concentration are independent determinants of changes in insulin sensitivity. A relevant finding is that high palmitoleate concentrations appear to prevent the decrease in insulin sensitivity associated with its saturated counterpart palmitate, the most abundant SFA [33] (Fig. 4). Given that palmitate is the precursor of palmitoleate and the concentrations of the two NEFA are closely associated (r = 0.79, p < 0.0001), it is possible that increased palmitoleate synthesis in response to high palmitate levels can partly compensate for the detrimental effect of excess palmitate on insulin signalling. In contrast to SFA, circulating and dietary MUFA have been associated with increased insulin sensitivity and lower risk of type 2 diabetes [36]. However, the beneficial effect of palmitoleate on insulin signalling is unlikely to be explained solely by its belonging to the class of MUFA. In fact, we observed that oleate, by far the most abundant MUFA in human blood and in the diet [36], was not correlated with insulin sensitivity in adjusted models. Together, these results are in line with previous observations by Cao et al [7], who reported improved whole-body glucose disposal during palmitoleate infusion in mice, and with mechanistic studies in isolated skeletal muscle cells [15] and adipocytes [17]. The association of palmitoleate with hepatic insulin resistance, though measured by a surrogate index, is also in agreement with previous studies in rodents, in which palmitoleate administration improved hepatic insulin sensitivity [18] and prevented the accumulation of triacylglycerol in the liver [14].
Molecular mechanisms by which palmitoleate may exert its insulin-sensitising action include an enhancement in basal and insulin-stimulated glucose uptake due to increased GLUT1 and GLUT4 expression, activation of glucose flux through aerobic and anaerobic glycolysis, stimulation of glycogen synthesis and inhibition of lipogenesis [15, 17, 18]. The modulation of these mechanisms has been attributed to the activation of AMP-activated protein kinase (AMPK) by palmitoleate [15, 17, 18]. In humans, previous evidence on the effect of palmitoleate on insulin signalling is, however, limited and conflicting. In fact, the relationship between palmitoleate and insulin sensitivity has been described as either positive [8,9,10], not significant [4] or even negative [20,21,22]. The literature prior to the present study has been hampered by inadequate study samples, lack of appropriate correction for potential confounders (e.g. parallel changes in palmitoleate and total NEFA levels), use of surrogate markers of insulin sensitivity and lack of longitudinal data. Moreover, measures of palmitoleate in lipid compartments other than NEFA, such as serum cholesteryl esters [20] or phospholipids [22], may not accurately reflect adipocyte synthesis of palmitoleate for signalling purposes.
The present analysis describes, for the first time, a cross-sectional positive relationship between palmitoleate and beta cell function in humans, with potential differences between men and women. This finding is consistent with previous studies in pancreatic islets and isolated beta cells, which exhibited increased basal and glucose-stimulated insulin secretion [35] and were protected from apoptosis induced by glucotoxicity or lipotoxicity [19] when exposed to palmitoleate. Despite this evidence, the lack of a significant association between changes in palmitoleate and beta cell function over time, in agreement with previous observations [4], warrants cautious interpretation of this finding and confirmation by intervention studies.
Given its low and tightly regulated concentration, circulating palmitoleate is an ideal candidate to act as a lipokine. In the RISC and other cohorts [8, 10, 23, 37], palmitoleate represents up to 6–7% of total serum NEFA. Unlike that of most other fatty acids, dietary intake of palmitoleate is very low in Western diets [38, 39] and dietary supplementation with palmitoleate-rich food is unable to significantly increase its plasma concentration [40,41,42,43,44]. On the contrary, circulating palmitoleate levels in NEFA closely reflect its endogenous synthesis in adipocytes, which occurs through desaturation of palmitate by the enzyme stearoyl-CoA desaturase-1 (SCD-1) [45]. SCD-1 activity is enhanced by insulin and influenced by dietary intake of SFAs, sucrose, fructose and alcohol [45]. Thus, the synthesis of palmitoleate in the adipocyte can be modulated in response to different metabolic states and dietary habits. Remarkably, we observed differences between palmitoleate and other abundant NEFA in relation to measures of adiposity, in that the relative concentration of palmitoleate was increased proportionally with the expansion of adipose tissue and the parallel increase in total NEFA level, regardless of sex, while other major NEFA were decreased or remained stable. Given the plausible beneficial effects of palmitoleate on insulin sensitivity and secretion, we can speculate that increased palmitoleate synthesis may be an adaptive mechanism through which the adipocyte can counterbalance (at least in part) the detrimental effect of fat tissue expansion and increased NEFA concentrations on systemic glucose metabolism.
The gluteo-femoral subcutaneous adipose tissue has been proposed as the principal site of palmitoleate production, storage and release [9]. This observation may contribute to explain the beneficial metabolic properties of lower-body subcutaneous adipose tissue compared with abdominal subcutaneous and visceral adipose tissue [46,47,48,49]. A key role of gluteo-femoral adipose tissue on palmitoleate secretion is supported by our cross-sectional analysis, in which individuals with increased lower-body circumference feature higher palmitoleate levels. The lack of association between changes in hip circumference and palmitoleate enrichment in longitudinal analysis, however, suggests that the lower-body fat mass is only one of a complex interplay of factors regulating palmitoleate release. Still, differences in palmitoleate enrichment might aid understanding of the wide inter-individual variability in the negative impact of obesity on glucose metabolism [50].
This is the largest study to date that has investigated the role of palmitoleate on glucose homeostasis in humans. We analysed data from a thoroughly characterised cohort of non-diabetic individuals with evaluation of NEFA concentration and composition and accurate measures of insulin sensitivity and beta cell function. Whether these findings extend to individuals with diabetes remains to be examined; however, animal studies suggest that this may be the case [14]. Metabolic assessments were repeated after a follow-up that allowed the evaluation of concomitant changes of palmitoleate and metabolic variables over time. All significant relationships described in bivariate analyses were confirmed in multivariable models, accounting for important potential confounders as appropriate for the large sample.
Nonetheless, there are some limitations to our study. We determined the concentration of the six most prevalent NEFA, representative of the three classes of SFA, MUFA and PUFA, while the effect of other, less abundant, NEFA was not evaluated. The use of a validated surrogate index of liver insulin resistance allowed us to explore the association between palmitoleate and hepatic insulin sensitivity; however, this relationship should be further tested using direct measures of liver insulin resistance. In keeping with this, the lack of a direct measure of insulin sensitivity at follow-up is also a limitation of our study. Moreover, although causality is supported by the above-mentioned previous experimental evidence, intervention studies are needed to confirm the causal role of endogenous palmitoleate in preserving insulin sensitivity and/or beta cell secretion in humans.
In conclusion, our novel data suggest that palmitoleate has beneficial metabolic properties on insulin signalling, beta cell function and glucose tolerance in humans and support its key role in the endocrine network between adipose tissue and other metabolically active organs/tissues.
Data availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
Abbreviations
- FFM:
-
Fat-free mass
- FM%:
-
Percentage fat mass
- IFG:
-
Impaired fasting glucose
- IGT:
-
Impaired glucose tolerance
- MUFA:
-
Monounsaturated fatty acid
- NGT:
-
Normal glucose tolerance
- OGIS:
-
Oral glucose insulin sensitivity index
- PO%:
-
Percentage palmitoleate enrichment in the total NEFA pool
- PUFA:
-
Polyunsaturated fatty acid
- RISC:
-
Relationship between Insulin Sensitivity and Cardiovascular disease study
- SCD-1:
-
Stearoyl-CoA desaturase-1
- SFA:
-
Saturated fatty acid
- std. β:
-
Standardised regression coefficient
References
Opie LH, Walfish PG (1963) Plasma free fatty acid concentrations in obesity. N Engl J Med 268:757–760. https://doi.org/10.1056/NEJM196304042681404
Boden G, Shulman GI (2002) Free fatty acids in obesity and type 2 diabetes: defining their role in the development of insulin resistance and beta-cell dysfunction. Eur J Clin Invest 32(Suppl 3):14–23
Boden G (2002) Interaction between free fatty acids and glucose metabolism. Curr Opin Clin Nutr Metab Care 5(5):545–549
Johnston LW, Harris SB, Retnakaran R et al (2018) Association of NEFA composition with insulin sensitivity and beta cell function in the Prospective Metabolism and Islet Cell Evaluation (PROMISE) cohort. Diabetologia 61(4):821–830. https://doi.org/10.1007/s00125-017-4534-6
Seghieri M, Trico D, Natali A (2017) The impact of triglycerides on glucose tolerance: Lipotoxicity revisited. Diabetes Metab 43(4):314–322. https://doi.org/10.1016/j.diabet.2017.04.010
Hodson L, Skeaff CM, Fielding BA (2008) Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog Lipid Res 47(5):348–380. https://doi.org/10.1016/j.plipres.2008.03.003
Cao H, Gerhold K, Mayers JR, Wiest MM, Watkins SM, Hotamisligil GS (2008) Identification of a lipokine, a lipid hormone linking adipose tissue to systemic metabolism. Cell 134(6):933–944. https://doi.org/10.1016/j.cell.2008.07.048
Stefan N, Kantartzis K, Celebi N et al (2010) Circulating palmitoleate strongly and independently predicts insulin sensitivity in humans. Diabetes Care 33(2):405–407. https://doi.org/10.2337/dc09-0544
Pinnick KE, Neville MJ, Fielding BA, Frayn KN, Karpe F, Hodson L (2012) Gluteofemoral adipose tissue plays a major role in production of the lipokine palmitoleate in humans. Diabetes 61(6):1399–1403. https://doi.org/10.2337/db11-1810
Bergman BC, Howard D, Schauer IE et al (2013) The importance of palmitoleic acid to adipocyte insulin resistance and whole-body insulin sensitivity in type 1 diabetes. J Clin Endocrinol Metab 98(1):E40–E50. https://doi.org/10.1210/jc.2012-2892
Nunes EA, Rafacho A (2017) Implications of palmitoleic acid (palmitoleate) on glucose homeostasis, insulin resistance and diabetes. Curr Drug Targets 18(6):619–628. https://doi.org/10.2174/1389450117666151209120345
de Souza CO, Vannice GK, Rosa Neto JC, Calder PC (2018) Is palmitoleic acid a plausible nonpharmacological strategy to prevent or control chronic metabolic and inflammatory disorders? Mol Nutr Food Res 62(1). https://doi.org/10.1002/mnfr.201700504
Mozaffarian D, Cao H, King IB et al (2010) Trans-palmitoleic acid, metabolic risk factors, and new-onset diabetes in U.S. adults: a cohort study. Ann Intern Med 153(12):790–799. https://doi.org/10.7326/0003-4819-153-12-201012210-00005
Yang ZH, Miyahara H, Hatanaka A (2011) Chronic administration of palmitoleic acid reduces insulin resistance and hepatic lipid accumulation in KK-Ay mice with genetic type 2 diabetes. Lipids Health Dis 10:120. https://doi.org/10.1186/1476-511X-10-120
Dimopoulos N, Watson M, Sakamoto K, Hundal HS (2006) Differential effects of palmitate and palmitoleate on insulin action and glucose utilization in rat L6 skeletal muscle cells. Biochem J 399(3):473–481. https://doi.org/10.1042/BJ20060244
Matsuzaka T, Shimano H, Yahagi N et al (2007) Crucial role of a long-chain fatty acid elongase, Elovl6, in obesity-induced insulin resistance. Nat Med 13(10):1193–1202. https://doi.org/10.1038/nm1662
Bolsoni-Lopes A, Festuccia WT, Chimin P et al (2014) Palmitoleic acid (n-7) increases white adipocytes GLUT4 content and glucose uptake in association with AMPK activation. Lipids Health Dis 13:199. https://doi.org/10.1186/1476-511X-13-199
de Souza CO, Teixeira AAS, Biondo LA, Lima Junior EA, Batatinha HAP, Rosa Neto JC (2017) Palmitoleic acid improves metabolic functions in fatty liver by PPARα-dependent AMPK activation. J Cell Physiol 232(8): 2168-2177. https://doi.org/10.1002/jcp.25715
Maedler K, Oberholzer J, Bucher P, Spinas GA, Donath MY (2003) Monounsaturated fatty acids prevent the deleterious effects of palmitate and high glucose on human pancreatic beta-cell turnover and function. Diabetes 52(3):726–733
Vessby B, Tengblad S, Lithell H (1994) Insulin sensitivity is related to the fatty acid composition of serum lipids and skeletal muscle phospholipids in 70-year-old men. Diabetologia 37(10):1044–1050
Kusunoki M, Tsutsumi K, Nakayama M et al (2007) Relationship between serum concentrations of saturated fatty acids and unsaturated fatty acids and the homeostasis model insulin resistance index in Japanese patients with type 2 diabetes mellitus. J Med Invest 54(3-4):243–247
Mozaffarian D, Cao H, King IB et al (2010) Circulating palmitoleic acid and risk of metabolic abnormalities and new-onset diabetes. Am J Clin Nutr 92(6):1350–1358. https://doi.org/10.3945/ajcn.110.003970
Fabbrini E, Magkos F, Su X et al (2011) Insulin sensitivity is not associated with palmitoleate availability in obese humans. J Lipid Res 52(4):808–812. https://doi.org/10.1194/jlr.M013599
Hills SA, Balkau B, Coppack SW et al (2004) The EGIR-RISC Study (The European Group for the study of Insulin Resistance: Relationship between Insulin Sensitivity and Cardiovascular disease risk): I. Methodology and objectives. Diabetologia 47(3):566–570. https://doi.org/10.1007/s00125-004-1335-5
American Diabetes Association (2019) Classification and diagnosis of diabetes: standards of medical care in diabetes–2019. Diabetes Care 42(Suppl 1):S13–S28. https://doi.org/10.2337/dc19-S002
Mari A, Pacini G, Murphy E, Ludvik B, Nolan JJ (2001) A model-based method for assessing insulin sensitivity from the oral glucose tolerance test. Diabetes Care 24(3):539–548
Vangipurapu J, Stancakova A, Kuulasmaa T et al (2011) A novel surrogate index for hepatic insulin resistance. Diabetologia 54(3):540–543. https://doi.org/10.1007/s00125-010-1966-7
Van Cauter E, Mestrez F, Sturis J, Polonsky KS (1992) Estimation of insulin secretion rates from C-peptide levels. Comparison of individual and standard kinetic parameters for C-peptide clearance. Diabetes 41(3):368–377
Mari A, Ferrannini E (2008) Beta-cell function assessment from modelling of oral tests: an effective approach. Diabetes Obesity Meta 10(Suppl 4):77–87. https://doi.org/10.1111/j.1463-1326.2008.00946.x
Mari A, Tura A, Natali A et al (2010) Impaired beta cell glucose sensitivity rather than inadequate compensation for insulin resistance is the dominant defect in glucose intolerance. Diabetologia 53(4):749–756
Trico D, Filice E, Baldi S, Frascerra S, Mari A, Natali A (2016) Sustained effects of a protein and lipid preload on glucose tolerance in type 2 diabetes patients. Diabetes Metab 42(4):242–248. https://doi.org/10.1016/j.diabet.2016.03.004
Gall WE, Beebe K, Lawton KA et al (2010) Alpha-hydroxybutyrate is an early biomarker of insulin resistance and glucose intolerance in a nondiabetic population. PLoS One 5(5):e10883. https://doi.org/10.1371/journal.pone.0010883
Carta G, Murru E, Banni S, Manca C (2017) Palmitic acid: physiological role, metabolism and nutritional implications. Front Physiol 8:902. https://doi.org/10.3389/fphys.2017.00902
Maeda K, Cao H, Kono K et al (2005) Adipocyte/macrophage fatty acid binding proteins control integrated metabolic responses in obesity and diabetes. Cell Metab 1(2):107–119. https://doi.org/10.1016/j.cmet.2004.12.008
Gravena C, Mathias PC, Ashcroft SJ (2002) Acute effects of fatty acids on insulin secretion from rat and human islets of Langerhans. J Endocrinol 173(1):73–80
Palomer X, Pizarro-Delgado J, Barroso E, Vazquez-Carrera M (2018) Palmitic and oleic acid: the yin and yang of fatty acids in type 2 diabetes mellitus. Trends Endocrinol Metab 29(3):178–190. https://doi.org/10.1016/j.tem.2017.11.009
Walker CG, Browning LM, Stecher L et al (2015) Fatty acid profile of plasma NEFA does not reflect adipose tissue fatty acid profile. Br J Nutr 114(5):756–762. https://doi.org/10.1017/S0007114515002251
Knutsen SF, Fraser GE, Beeson WL, Lindsted KD, Shavlik DJ (2003) Comparison of adipose tissue fatty acids with dietary fatty acids as measured by 24-hour recall and food frequency questionnaire in Black and White Adventists: the Adventist Health Study. Ann Epidemiol 13(2):119–127
Hodge AM, Simpson JA, Gibson RA et al (2007) Plasma phospholipid fatty acid composition as a biomarker of habitual dietary fat intake in an ethnically diverse cohort. Nutr Metab Cardiovasc Dis 17(6):415–426. https://doi.org/10.1016/j.numecd.2006.04.005
Nestel P, Clifton P, Noakes M (1994) Effects of increasing dietary palmitoleic acid compared with palmitic and oleic acids on plasma lipids of hypercholesterolemic men. J Lipid Res 35(4):656–662
Curb JD, Wergowske G, Dobbs JC, Abbott RD, Huang B (2000) Serum lipid effects of a high-monounsaturated fat diet based on macadamia nuts. Arch Intern Med 160(8):1154–1158
Garg ML, Blake RJ, Wills RB (2003) Macadamia nut consumption lowers plasma total and LDL cholesterol levels in hypercholesterolemic men. J Nutr 133(4):1060–1063. https://doi.org/10.1093/jn/133.4.1060
Hiraoka-Yamamoto J, Ikeda K, Negishi H et al (2004) Serum lipid effects of a monounsaturated (palmitoleic) fatty acid-rich diet based on macadamia nuts in healthy, young Japanese women. Clin Exp Pharmacol Physiol 31(Suppl 2):S37–S38. https://doi.org/10.1111/j.1440-1681.2004.04121.x
Griel AE, Cao Y, Bagshaw DD, Cifelli AM, Holub B, Kris-Etherton PM (2008) A macadamia nut-rich diet reduces total and LDL-cholesterol in mildly hypercholesterolemic men and women. J Nutr 138(4):761–767. https://doi.org/10.1093/jn/138.4.761
Hodson L, Fielding BA (2013) Stearoyl-CoA desaturase: rogue or innocent bystander? Prog Lipid Res 52(1):15–42. https://doi.org/10.1016/j.plipres.2012.08.002
Canoy D, Luben R, Welch A et al (2004) Fat distribution, body mass index and blood pressure in 22,090 men and women in the Norfolk cohort of the European Prospective Investigation into Cancer and Nutrition (EPIC-Norfolk) study. J Hypertens 22(11):2067–2074
Yusuf S, Hawken S, Ounpuu S et al (2005) Obesity and the risk of myocardial infarction in 27,000 participants from 52 countries: a case-control study. Lancet 366(9497):1640–1649. https://doi.org/10.1016/S0140-6736(05)67663-5
Venturi E, Pinnola S, Morizzo C et al (2016) Clinical phenotype and microvascular dynamics of subjects with endothelial dysfunction as assessed by peripheral tonometry. Microcirculation 23(3):230–239. https://doi.org/10.1111/micc.12268
Umano GR, Shabanova V, Pierpont B et al (2019) A low visceral fat proportion, independent of total body fat mass, protects obese adolescent girls against fatty liver and glucose dysregulation: a longitudinal study. Int J Obes (Lond) 43(4):673–682. https://doi.org/10.1038/s41366-018-0227-6
Ferrannini E, Natali A, Bell P, Cavallo-Perin P, Lalic N, Mingrone G (1997) Insulin resistance and hypersecretion in obesity. European Group for the Study of Insulin Resistance (EGIR). J Clin Invest 100(5):1166–1173. https://doi.org/10.1172/JCI119628
Acknowledgements
Preliminary study findings were presented as an abstract at the 55th EASD Annual Meeting in Barcelona, Spain, in 2019.
Contribution statement
DT contributed to study design, data analysis and interpretation and writing the manuscript. AMe, LN, MH, RGS, TK, KL and NML contributed to data collection and edited the manuscript. AM undertook the mathematical modelling of insulin secretion and beta cell function variables, contributed to data interpretation and edited the manuscript. AN contributed to study design and supervision, securing of funding, and data collection, analysis and interpretation, and edited and critically revised the manuscript. All authors read and approved the final submitted version of the manuscript. DT and AN are the guarantors of this work and, as such, have full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Funding
The RISC study was partly supported by European Union grant QLG1-CT-2001-01252. DT is funded by the European Foundation for the Study of Diabetes (EFSD) Mentorship Programme.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
The authors declare that there is no duality of interest associated with this manuscript.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM Figures
(PDF 168 kb)
Appendix
Appendix
EGIR-RISC Investigators
Further information on the RISC project and participating centres can be found at http://www.egir.org
EGIR-RISC recruiting centres
Amsterdam, the Netherlands: RJ Heine, J Dekker, S de Rooij, G Nijpels, W Boorsma
Athens, Greece: A Mitrakou, S Tournis, K Kyriakopoulou, P Thomakos
Belgrade, Serbia: N Lalic, K Lalic, A Jotic, L Lukic, M Civcic
Dublin, Ireland: J Nolan, TP Yeow, M Murphy, C DeLong, G Neary, MP Colgan, M Hatunic
Frankfurt, Germany: T Konrad, H Böhles, S Fuellert, F Baer, H Zuchhold
Geneva, Switzerland: A Golay, E Harsch Bobbioni,V Barthassat, V Makoundou, TNO Lehmann, T Merminod
Glasgow, Scotland, UK: JR Petrie, C Perry, F Neary, C MacDougall, K Shields, L Malcolm
Kuopio, Finland: M Laakso, U Salmenniemi, A Aura, R Raisanen, U Ruotsalainen, T Sistonen, M Laitinen, H Saloranta
London, England, UK: SW Coppack, N McIntosh, J Ross, L Pettersson, P Khadobaksh
Lyon, France: M Laville, F Bonnet (now Rennes), A Brac de la Perriere, C Louche-Pelissier, C Maitrepierre, J Peyrat, S Beltran, A Serusclat
Madrid, Spain: R Gabriel, EM Sánchez, R Carraro, A Friera, B Novella
Malmö, Sweden (1): P Nilsson, M Persson, G Östling, (2): O Melander, P Burri
Milan, Italy: PM Piatti, LD Monti, E Setola, E Galluccio, F Minicucci, A Colleluori
Newcastle-upon-Tyne, England, UK: M Walker, IM Ibrahim, M Jayapaul, D Carman, C Ryan, K Short, Y McGrady, D Richardson
Odense, Denmark: H Beck-Nielsen, P Staehr, K Højlund, V Vestergaard, C Olsen, L Hansen
Perugia, Italy: GB Bolli, F Porcellati, C Fanelli, P Lucidi, F Calcinaro, A Saturni
Pisa, Italy: E Ferrannini, A Natali, D Tricò, E Muscelli, S Pinnola, M Kozakova, A Casolaro, BD Astiarraga
Rome, Italy: G Mingrone, C Guidone, A Favuzzi, P Di Rocco
Vienna, Austria: C Anderwald, M Bischof, M Promintzer, M Krebs, M Mandl, A Hofer, A Luger, W Waldhäusl, M Roden
Project Management Board: B Balkau (Villejuif, France), F Bonnet (Rennes, France), SW Coppack (London, England, UK), JM Dekker (Amsterdam, the Netherlands), E Ferrannini (Pisa, Italy), A Mari (Padova, Italy), A Natali (Pisa, Italy), J Petrie (Glasgow, Scotland, UK), M Walker (Newcastle, England, UK)
Core laboratories and reading centres
Lipids: Dublin, Ireland: P Gaffney, J Nolan, G Boran
Hormones: Odense, Denmark: C Olsen, L Hansen, H Beck-Nielsen
Albumin:creatinine: Amsterdam, the Netherlands: A Kok, J Dekker
Genetics: Newcastle-upon-Tyne, England, UK: S Patel, M Walker
Stable isotope laboratory: Pisa, Italy: A Gastaldelli, D Ciociaro
Ultrasound reading centre: Pisa, Italy: M Kozakova
ECG reading: Villejuif, France: MT Guillanneuf
Actigraph: Villejuif, France: B Balkau, L Mhamdi
Data Management: Villejuif, France, Padova, and Pisa, Italy: B Balkau, A Mari, L Mhamdi, L Landucci, S Hills, L Mota
Mathematical modelling and website management: Padova, Italy: A Mari, G Pacini, C Cavaggion, A Tura
Coordinating office: Pisa, Italy: SA Hills, L Landucci, L Mota
Rights and permissions
About this article
Cite this article
Tricò, D., Mengozzi, A., Nesti, L. et al. Circulating palmitoleic acid is an independent determinant of insulin sensitivity, beta cell function and glucose tolerance in non-diabetic individuals: a longitudinal analysis. Diabetologia 63, 206–218 (2020). https://doi.org/10.1007/s00125-019-05013-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-019-05013-6