Abstract
Heat shock proteins are constitutively expressed chaperones induced by cellular stress, such as changes in temperature, pH, and osmolarity. These proteins, present in all organisms, are highly conserved and are recruited for the assembly of protein complexes, transport, and compartmentalization of molecules. In fungi, these proteins are related to their adaptation to the environment, their evolutionary success in acquiring new hosts, and regulation of virulence and resistance factors. These characteristics are interesting for assessment of the host adaptability and ecological transitions, given the emergence of infections by these microorganisms. Based on phylogenetic inferences, we compared the sequences of HSP9, HSP12, HSP30, HSP40, HSP70, HSP90, and HSP110 to elucidate the evolutionary relationships of different fungal organisms to suggest evolutionary patterns employing the maximum likelihood method. By the different reconstructions, our inference supports the hypothesis that these classes of proteins are associated with pathogenic gains against endothermic hosts, as well as adaptations for phytopathogenic fungi.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fungi are crucial ecosystem components and play essential roles in environmental processes, such as biotransformation and maintaining nutrient and CO2 cycling (Case et al. 2022) They have been shown to inhabit diverse niches, having direct or indirect involvement in the health disease processes of all organisms, acting as both pathogen and protector during infections (Zhang et al. 2020). However, this conquest scenario is commonly overlooked.
Since the appearance of the ancestors of these organisms about 1200 million years ago in aquatic bodies, there has been an improvement in characteristics that helped the predatory, parasitoid, symbiont, and commensal lifestyles of fungi. Their terrestrialization boosted the development of morphological structures that allowed their dispersion over land in search of substrates and a protein arsenal that allowed them to obtain nutrients (Nagy et al. 2017). The successful mastery of these environments was essential for their propagation, as the selective pressure of the coevolution of plants, invertebrates, and vertebrates boosted their ability to infect different organisms, mainly by the clades Glomeromycota, Mucoromycota, and Dikarya (Naranjo-Ortiz and Gabaldón 2019).
Millions of years later, the fungi, adapted to sauropodomorph, arthropod, and plant parasitism Woodruff et al. 2022), underwent a severe adaptive process. With the cataclysm of the Cretaceous-Paleogene, which caused the extinction of the extensive megafauna of the period, there was a global modification of the post-calamity environment, with a decrease in temperature and luminosity, and an increase in humidity and decomposing substrates, allowing greater development of the funga (Vajda and McLoughlin 2004). However, these organisms, which were associated with ectomorphic vertebrates, would face the challenge of transitioning to the animals that would come to rise and dominate the new environment: mammals. The endothermy and immunity of mammals, which allowed the survival of animals in the face of environmental adversities, also became a barrier to the development of pathogens of the time. This characteristic boosted the development of mechanisms already present in fungi, such as the production of toxins, immune system evasion, and mycelium adaptation to survive in these hosts. This proposal is known as the “fungal infection-mammalian selection” (FIMS) hypothesis (Casadevall 2005; Casadevall and Damman 2020).
Heat shock proteins (HSPs) stand out among the mechanisms that helped adapt fungi to new hosts, responding to cellular stress, thermotolerance, and changes in pH and osmolarity (Burnie et al. 2006). These proteins are ubiquitous, highly conserved, and play a fundamental role for both parasites and hosts. Alongside the agent, they stimulate the growth phase, change in dimorphic microorganisms, and increase resistance and pathogenicity (Cleare et al. 2017; Martinez-Rossi et al. 2016). On the other hand, in the host, HSPs allow the activation of several defense cells through toll-like receptors and play a role in the activation by presenting antigens in infective diseases. Due to these features, we can characterize them as both targets and therapeutic agents (Fang et al. 2011; Jacob et al. 2015). Other functions include chaperone activity, assembly of protein complexes, and transport and compartmentalization of molecules (Table 1) (Bolhassani and Agi 2019).
The HSPs seem to be related to diverse responses in morphological features of fungi. In Candida albicans, a yeast-like fungus involved in both saprophytic and invasive lifestyles, the modulation of heat shock factors (HSFs) plays a role in the regulation of the transition to yeast from hyphae and virulence. Via multiple pathways, particularly in temperature, osmolarity, and CO2 changes, the overexpression of the HSP70 impacts the filamentation of C. albicans and its invasiveness (Robbins and Cowen 2023; Tiwari and Shankar 2018).
These chaperones can be classified according to their molecular weight ranging from 10 to more than 100 kDa. The main known HSPs are HSP10, HSP12, HSP26, HSP30, HSP40, HSP60, HSP70, HSP78, HSP90, and HSP110, and are allocated in families according to their function and location (Lindquist & Craig, 1988; Zara et al. 2002). Their structure generally comprises two subunits: an amino-terminal nucleotide-biding domain (NBD) with ATPase function and a carboxy-terminal substrate-binding domain (SBD). A high percentage of identity between sequences of these proteins is estimated among distantly related eukaryotes (> 50%), with a higher percentage among fungi (> 80%) (Burnie et al. 2006; Ramos et al. 2008). However, there is no data about the evolutionary relationship between fungal organisms, their lifestyle, and these proteins specifically.
Given the increasing adaptation of fungi to different niches and hosts, it is necessary to investigate the evolutionary patterns of the family of HSPs among the fungi that help them adapt to new habitats and select new organisms to infect. Here, we sought to elucidate the evolutionary relationship of these chaperones from different fungal clades with their ecological transition, according to their interaction with other hosts.
Material and methods
Library construction
The nucleotide sequences corresponding to the different HSPs of different fungal organisms were obtained from NCBI Protein, and the coding regions were identified based on the most recent annotation of the genomes. The organisms were selected based on origin/host (saprophytes/symbionts, phytopathogens, animal pathogens, human pathogens, mycoparasites, and toxin producers) and morphology (yeasts, filamentous, and dimorphic). The selected HSPs were based on their molecular weight and classification: small HSPs (sHSPs)—HSP9 kDa, HSP12 kDa, HSP30 kDa; cochaperone HSP40 kDa; and classical HSP70 kDa, HSP90 kDa, and HSP110 kDa. The corresponding amino acid sequences were employed for phylogenetic inference. Protein Data Bank entries and correlated species are shown on Supplementary Table 1.
Phylogenetic inference
Amino acid sequences were aligned using the multiple sequence alignment program MAFFT (Katoh et al. 2017) filtered for hypervariable regions with GUIDANCE2 (Sela et al. 2015) using default parameters of the online platform. The following thresholds were employed for each set: HSP9/HSP12 (0.8349), HSP30 (0.6166), HSP40 (0.8446), HSP70 (0.5681), HSP90 (0.5506), and HSP110 (0.9617). The resulting alignments were used for phylogenetic inference, employing the maximum likelihood method via default parameters of the online suite PhyML (Guindon et al. 2010), choosing the best evolutionary model being with Smart Model Selection (SMS) (Lefort et al. 2017). The statistical support for the ramifications was estimated by the approximate likelihood-ratio test (aLRT), requiring a minimum confidence threshold of ≥ 60% (Anisimova and Gascuel 2006).
Results and discussion
HSP9 and HSP12
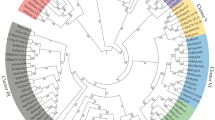
This analysis evaluated 54 NCBI Protein fungal sequences corresponding to HSP9/HSP12. The sequences containing at least one of these two proteins were annotated, and the tree was constructed using both. Figure 1 shows the phylogenetic inference of these sHSPs in fungi of different families.
Phylogenetic reconstruction based on the heat shock proteins 9 kDa and 12 kDa (HSP9/HSP12), using the maximum likelihood method (Q.pfam model). Confidence scores ≥ 60% are shown along each branch. Classification is shown following each corresponding OTU, according to the colors: saprophytes/ubiquitous (gray), plant pathogens (green), animal pathogens (yellow), human pathogens (orange), mycoparasites (purple), dimorphic fungi (blue), and toxin producers (asterisk)
All phylogenies have identifying icons based on the literature for the following correspondences: saprophytes/ubiquitous (gray), plant pathogens (green), animal pathogens (yellow), human pathogens (orange), mycoparasites (purple), dimorphic fungi (blue), and toxin producers (asterisk). The evolutionary history was inferred by using the maximum likelihood method based on the Q.pfam model (Minh et al. 2021).
From the maximum likelihood analysis, it was possible to observe several monophyletic groups with their particularities in the consensus tree. The first group, formed by fungi present in nature, diversifies into 2 clades with specific adaptations: the group of environmental fungi of the Herpotrichiellaceae family known for having pleomorfism and etiological agents of chromoblastomycosis (Fonsecaea pedrosoi, Cladophialophora spp., Exophiala dermatitidis, and Rhinocladiella mackenziei) and the group formed by those who cause diseases in plant hosts (Fusarium nygamai, Botrytis spp., Alternaria spp., Bipolaris maydis).
Heat shock proteins of less than 43 kDa are known as sHSPs. Their structure contains α/β-crystalline domains and a conserved site at the C-terminal up to 100 amino acids (Gusev et al. 2002). The 9 kDa and 12 kDa HSPs are present in the cytoplasm and membrane of the fungal cell and are involved, in addition to the temperature variance, with barotolerance and response to stress associated with nutrients, being demonstrated their role in the biofilm formation in Saccharomyces cerevisiae and oxidative stress protection in Candida albicans (de Groot et al. 2000; Zara et al. 2002; Chauhan et al. 2003).
The other group is made up of primary human pathogens known to cause disease in other animals. This clade is composed of the monophyletic group of dermatophytes, adapted for infection with keratinolytic enzymes (Trichophyton spp., Microsporum canis, Nannizzia gypsea) as well as the paraphyletic group of endemic dimorphic fungi, adapted to cross the endothermy barrier of mammals (Histoplasma spp., Blastomyces spp., Coccidioides spp.). This could be related to the role of these sHSPs in C. albicans in yeast-to-hyphal transition and tissue invasion (Fu et al. 2012)
In addition, we can observe that the sHSPs of the species Cryptococcus floricola and C. gattii are found in different branches of the tree. While the former is in an external clade group, along with the plant pathogen Neofusicoccum parvum, the latter is more closely related to the human and animal pathogenic species Trichosporon asahii. In C. neoformans, the homologous protein HSP12 is involved in cell survival in the face of increased antifungal temperatures via cyclic AMP (cAMP) pathway (Maeng et al. 2010). This could suggest that these proteins may be involved in the ecological adaptation of this species to hosts with higher body temperatures.
HSP30
Unlike previous sHSPs, we performed the analysis with 48 sequences, due to the fewer availability compared to classic HSPs. Despite this, we were able to obtain a consensus tree with the same diversity of fungi in relation to their hosts (Fig. 2). The evolutionary history was inferred by using the maximum likelihood method based on the Q.pfam model (Minh et al. 2021).
Phylogenetic reconstruction based on the heat shock protein 30 kDa (HSP30), using the maximum likelihood method (Q.pfam model). Confidence scores > 60% are shown along each branch. Classification is shown following each corresponding OTU, according to the colors: saprophytes/ubiquitous (gray), plant pathogens (green), animal pathogens (yellow), human pathogens (orange), mycoparasites (purple), dimorphic fungi (blue), and toxin producers (asterisk)
Differently from HSP9/HSP12, in this analysis, we observed a large clade representative of the primary and opportunistic pathogens of animals and humans, having an ancestral sequence in common with other phytopathogenic fungi (Botrytis spp. and Cercospora beticola). Within this clade, we have a paraphyletic group of dimorphic fungi and a monophyletic group of the Trichocomaceae family, which contains the well-known molds of the genera Aspergillus and Penicillium.
HSP30s are proteins with hydrophobic characteristics present in the plasma membrane, and it is suggested that the major inducers of the production of these proteins are factors that affect the fluidity of the membrane, such as high temperatures and substances that cause a change in solubility in the lipid bilayer, such as ethanol (Seymour and Piper 1999). Those chaperones also assist in the stabilization of unfolded proteins in yeasts, forming a complex with HSP80 and HSP70 (Tiwari et al. 2015). Since HSP30s are modulated with the alteration of the pH of the environment and their expression is induced by extracellular acidification, we can insinuate their involvement in the invasion of epithelial tissue in dermatophytosis and dermatomycosis (Behzadi et al. 2009; Freitas et al. 2011).
HSP40
Present in the cytosol, plasma membrane, and endoplasmic reticulum of cells, HSP40s act together with HSP104 as cochaperones to HSP70, binding to unfolded peptides to be delivered to HSP70 (Burnie et al. 2005). Due to their molecular composition, these cochaperones are classified into two categories: class I, containing three conserved domains (cysteine-rich, zinc finger, and carboxy-terminal domains), and class II, containing two conserved domains (glycine-rich and phenylalanine-rich domains). Also, their sequences have similarities with the J domain of the Escherichia coli DnaJ, modulating the conformation of the HSP70 in order to facilitate its ATP-hydrolytic activity (Lee et al. 2002, Burnie et al. 2005).
Here, we evaluated the sequence of 68 fungal organisms, as illustrated in the phylogeny in Fig. 3. The homologous sequences mainly involved the proteins Caj1, Sis1, Psi1, Xdj1, and bpa. As with sHSPs, we can suggest the presence of a common ancestor for the ecological transition of animal pathogenic fungi. In addition, we can observe a divergence between the sequences of yeast, dimorphic, and filamentous fungi. SMS (Lefort et al. 2017) suggested the Q.pfam model (Minh et al. 2021) to account for evolutionary rate differences among sites (four categories, +G), considering some sites to be evolutionarily invariable (+I).
Phylogenetic reconstruction based on the heat shock protein 40 kDa (HSP40), using the maximum likelihood method (Q.pfam model, +G, +I). Confidence scores > 60% are shown along each branch. Classification is shown following each corresponding OTU, according to the colors: saprophytes/ubiquitous (gray), plant pathogens (green), animal pathogens (yellow), human pathogens (orange), mycoparasites (purple), dimorphic fungi (blue), and toxin producers (asterisk)
It is possible to observe a divergence between the species of the genus Fusarium. This genus is highly disseminated and known for its transkingdom infections, affecting plants, animals, and humans, and can even produce toxins known as fumonisins. In our analysis, Fusarium oxysporum and Fusarium nygamai species form a polyphyletic group along with the phytopathogens of the genus Colletotrichum. In the species F. oxysporum, it has been shown that post-translational modifications involving the HSP40-HSP70 complexes modulate the formation of conidia, which are crucial for their dispersal and reproduction (Lv et al. 2022).
HSP70
The results of the phylogenetic analysis of 68 sequences of HSP70 are shown in Fig. 4. SMS (Lefort et al. 2017) suggested the Legascuel (LG) (Le and Gascuel 2008) considering rate variation (+R).
Phylogenetic reconstruction based on the heat shock protein 70 kDa (HSP70), using the maximum likelihood method (LG+R). Confidence scores > 60% are shown along each branch. Classification is shown following each corresponding OTU, according to the colors: saprophytes/ubiquitous (gray), plant pathogens (green), animal pathogens (yellow), human pathogens (orange), mycoparasites (purple), dimorphic fungi (blue), and toxin producers (asterisk)
Both the genera Aspergillus and Fusarium are known to produce mycotoxins linked to grain cultivation. Aflatoxins, produced by species of the genus Aspergillus, as an example of the section Flavus of this genus, are a worrying group of compounds capable of poisoning animals and humans. Aflatoxins may be related to about 30% of hepatocellular cancers, given their great hepatotoxic and carcinogenic potential. Here, we can observe the formation of a polyphyletic group involving the toxin-producing fungi linked to agriculture between the heat shock protein sequences. This relationship is particularly interesting since the HSP70 seems to be involved in aflatoxin production at different temperatures in both toxigenic and atoxigenic isolates. The increase in temperature in toxigenic isolates of A. flavus decreased the expression of HSP70, impairing the production of aflatoxins. On the other hand, in non-toxigenic isolates, the overexpression of HSP70 seems to be related to the inhibition of aflatoxin biosynthesis (Thakur et al., 2016; Ting et al. 2020).
Interestingly, we can observe that the species of the genus Cryptococcus are present in distinct sister clades, with C. floricola present in a clade together with fungi that do not have pathogenic potential for animals. In addition, we can suggest an adaptation in relation to the proteins of the sister clades of environmental fungi in two groups: (i) dimorphic clade that infects vertebrates and (ii) clade that composes the species of the genus Trichoderma, known to be mycoparasites, widely used in biological control.
The 70-kDa heat shock proteins are among the HSPs most induced in relation to temperature increase, being directed to several compartments: cytoplasm, cell membrane, endoplasmic reticulum, and mitochondria. It is estimated that these proteins have more than 60% similarity between members of the Eukarya domain (Boorstein et al. 1994). In fungi, different homologous proteins are expressed constitutively, such as the Ssa1 and Ssa2 proteins, as well as facultative proteins, such as the Ssa3 and Ssa4 proteins, produced in response to stress.
In its structure, HSP70 has an NBD domain of 44 kDa and an SBD domain of 18 kDa. Thus, like HSP40, its cochaperone is similar to the DnaK protein of Escherichia coli. Ssa1, the most studied cytosolic protein in Saccharomyces cerevisiae, is induced by the expression of HSP40 Sis1, whose activity involves the direction of proteins in the assembly process for conformation modification by the ATPase domain of Ssa1 (Qian et al. 2001; Burnie et al. 2006).
The signaling and induction pathway between these proteins is well established and involves different mechanisms, including pH change and phosphate levels. This pathway involves heat shock factor 1 (HSF1), heat shock elements (HSEs), a species-dependent variable set of genes. Among the transcription factors involved with the highest homology is PacC, which responds to increased temperature in the physiological environment by the pal genes cascade, modulating the expression of HSP70 genes and increasing glycosylation and enzyme secretion. In addition, these factors are related to a higher gene expression in dermatophyte cultures in response to heat and the presence of keratin (Nozawa et al. 2003; Ferreira-Nozawa et al. 2006; Martinez-Rossi et al. 2016).
In dimorphic fungi, it is known that this class of proteins is responsible for cellular adaptation during the change from filamentous to yeast-like stage in Histoplasma capsulatum. Its heat-inducive response may involve this adaptation, hitting its maximum expression at higher temperatures in more virulent strains (Caruso et al. 1987; Allendoerfer et al. 1996). The same was observed for Paracoccidioides brasiliensis HSP80, with about 90% of identity in relation to H. capsulatum. In this case, it had a lower expression when transitioning to the mycelial state (Díez et al. 2002).
HSP90
The tree reconstruction of 68 sequences corresponding to HSP90 of fungal organisms suggests an involvement of these proteins in the morphology of the species (Fig. 5). The evolutionary history was inferred by using the maximum likelihood method based on the LG (Le and Gascuel 2008) + R model.
Phylogenetic reconstruction based on the heat shock protein 90 kDa (HSP90), using the maximum likelihood method (LG+R). Confidence scores > 60% are shown along each branch. Classification is shown following each corresponding OTU, according to the colors: saprophytes/ubiquitous (gray), plant pathogens (green), animal pathogens (yellow), human pathogens (orange), mycoparasites (purple), dimorphic fungi (blue), and toxin producers (asterisk)
HSP90 chaperones are the largest representatives of this class, as they correspond to about 2% of cytosolic proteins in different eukaryotes. These proteins have a structural difference in relation to the other HSPs, since they have a dimerization domain associated with the NBD and SBD domains. This is reflected in their activity, being recruited to aid in assembling proteins that are difficult to fold, as well as in preventing aggregate formation and intermediate formations (Finkelstein and Strausberg 1983; Taipale et al. 2010).
Among pathogenic fungi, its modulation is related to the gain of invasive characteristics. In the dermatophyte T. rubrum, its expression is related to increased keratinolytic activity at human body temperature (37 °C) via metabolic pathways after alternative splicing (Neves-da-Rocha et al. 2019). It is also possible that these proteins are acting in the adaptive process to gain resistance to azoles and echinocandins in C. albicans (Cowen and Lindquist 2005). The role of HSP90 in the virulence of C. albicans was suggested by the overexpression of its homologous in S. cerevisiae (84% identity) in a mice model (Hodgetts et al. 1996). This protein is also present in the cell wall of C. albicans and plays a key role in hyphal elongation and protein secretion (Hube and Naglik 2001).
Our inference supports the hypothesis that this class of proteins is associated with pathogenic gains against endothermic hosts. What reinforces this suggestion is the presence of four sister groups of pathogens with a common ancestral sequence, especially with the presence of primary pathogens and endemic dimorphic fungi of different origins.
Following this perspective, we can justify the presence of an exclusive clade of phytopathogenic fungi. The crucial role of HSP90 in the modulation of genes involved in vegetative growth, virulence, and conidiation of Fusarium graminearum has been demonstrated. The repression of this gene prevents the formation of phialides, and most of the conidia are produced directly by the hyphae, modifying the phenotype (Bui et al. 2016).
HSP110
HSP110s are highly similar to HSP70s and have been suggested as a subgroup of them. However, they have an additional holdase activity and act like cochaperones, facilitating their activity as nucleotide exchange factors (Oh et al. 1997; Andréasson et al. 2008; Polier et al. 2008).
One of the members of this class, HSP104, which has a role in the recovery of denatured proteins, is known for having two NBDs. This is relevant in a scenario of temperature increase, in which it is expressed for the maintenance of the stationary stage (Hattendorf 2002).
As in previous reconstructions, there is the formation of monophyletic groups related to endothermic and plant hosts. The evolutionary transition to warm hosts may be associated with a change in mycelial to yeast structures (Fig. 6). SMS (Lefort et al. 2017) suggested the Q.yeast model to account for evolutionary rate differences among sites (four categories, +G), considering some sites to be evolutionarily invariable (+I) (Minh et al. 2021). Nevertheless, further studies are needed to evaluate the role of this HSP in the infective process.
Phylogenetic reconstruction based on the heat shock protein 110 kDa (HSP110), using the maximum likelihood method (Q.yeast model, +G, +I). Confidence scores > 60% are shown along each branch. Classification is shown following each corresponding OTU, according to the colors: saprophytes/ubiquitous (gray), plant pathogens (green), animal pathogens (yellow), human pathogens (orange), mycoparasites (purple), dimorphic fungi (blue), and toxin producers (asterisk)
The investigation of HSPs and their role in host-pathogen interaction, morphological features, and production of compounds can provide new perspectives in health, agriculture, and industry (Tiwari and Shankar 2018). The application of these proteins as molecular targets to new vaccines and treatments could improve the assistance in invasive fungemia and another systemic mycosis (De Souza et al. 2019; Baltazar et al. 2021; Hu et al. 2023). In addition, this could prevent crop loss due to anthracnosis and benefit production via biological control development (Montero-Barrientos et al. 2010; Dong et al. 2023).
Conclusion
In this article, we evaluated by phylogenetic inferences the hypothesis that the heat shock proteins may be involved in the fungal host range by its temperature and ecological adaptation. Since these proteins seem to be related to different mechanisms of protection against unfavorable environments, it is possible that it led to transitions in order to predate under specific substrates, such as keratin and cellulose.
This taxonomically diverse investigation of HSP evolutionary relationships highlights the relevance of these stress-related proteins for fungal environment and host plasticity. Regardless of their high sequence conservation, it is tempting to consider them as suitable targets for novel antifungal treatments. Still, further studies could be able to determine if the post-translational changes promoted by HSPs aided in adapting to the new hosts, pinpointing how these chaperones were able to influence fungal dispersion and environmental conquests.
Abbreviations
- HSP:
-
Heat shock protein
- HSF:
-
Heat shock factors
- NBD:
-
Nucleotide-biding domain
- SBD:
-
Substrate-binding domain
- sHSPs:
-
Small HSPs
- cAMP:
-
Cyclic AMP
References
Allendoerfer R, Maresca B, Deepe GS (1996) Cellular immune responses to recombinant heat shock protein 70 from Histoplasma capsulatum. Infect Immun 64(10):4123–4128. https://doi.org/10.1128/iai.64.10.4123-4128.1996
Andréasson C, Fiaux J, Rampelt H, Mayer MP, Bukau B (2008) HSP110 is a nucleotide-activated exchange factor for HSP70. J Biol Chem 283(14):8877–8884. https://doi.org/10.1074/jbc.m710063200
Anisimova M, Gascuel O (2006) Approximate likelihood-ratio test for branches: a fast, accurate, and powerful alternative. Syst Biol 55(4):539–552. https://doi.org/10.1080/10635150600755453
Baltazar LM, Ribeiro GF, De Freitas GJC, Queiroz-Júnior CM, Fagundes CT, Chaves-Olórtegui C, Teixeira MM, Souza DG (2021) Protective response in experimental paracoccidioidomycosis elicited by extracellular vesicles containing antigens of Paracoccidioides brasiliensis. Cells 10(7):1813. https://doi.org/10.3390/cells10071813
Behzadi E, Behzadi P, Sirmatel F (2009) Identification of 30 kDa heat shock protein gene in Trichophyton rubrum. Mycoses 52(3):234–238. https://doi.org/10.1111/j.1439-0507.2008.01561.x
Bolhassani A, Agi E (2019) Heat shock proteins in infection. Clinica Chimica Acta 498:90–100. https://doi.org/10.1016/j.cca.2019.08.015
Boorstein WR, Ziegelhoffer T, Craig EA (1994) Molecular evolution of the HSP70 multigene family. J Mol Evol 38(1). https://doi.org/10.1007/BF00175490
Bui D, Lee Y, Lim JY, Fu M, Kim JC, Choi GJ, Son H, Lee YW (2016) Heat shock protein 90 is required for sexual and asexual development, virulence, and heat shock response in Fusarium graminearum. Sci Rep 6(1). https://doi.org/10.1038/srep28154
Burnie JP, Carter TL, Hodgetts SJ, Matthews RC (2006) Fungal heat-shock proteins in human disease. FEMS Microbiol Rev 30(1):53–88. https://doi.org/10.1111/j.1574-6976.2005.00001.x
Caruso M, Sacco M, Medoff G, Maresca B (1987) Heat shock 70 gene is differentially expressed in histoplasma capsulatum strains with different levels of thermotolerance and pathogenicity. Mol Microbiol 1(2):151–158. https://doi.org/10.1111/j.1365-2958.1987.tb00507.x
Casadevall A (2005) Fungal virulence, vertebrate endothermy, and dinosaur extinction: Is there a connection? Fungal Genet Biol 42(2):98–106. https://doi.org/10.1016/j.fgb.2004.11.008
Casadevall A, Damman, CJ (2020) Updating the fungal infection-mammalian selection hypothesis at the end of the Cretaceous Period. PLOS Pathogens 16(7):e1008451. https://doi.org/10.1371/journal.ppat.1008451
Case NT, Berman J, Blehert DS, Cramer RA, Cuomo C, Currie CR, Ene IV, Fisher MC, Fritz-Laylin LK, Gerstein AC, Glass NL, Gow NA, Gurr SJ, Hittinger CT, Hohl TM, Iliev ID, James TY, Jin H, Klein BS et al (2022) The future of fungi: Threats and opportunities. G3 Genes|Genomes|Genetics 12(11). https://doi.org/10.1093/g3journal/jkac224
Chauhan N, Inglis D, Roman E, Pla J, Li D, Calera JA, Calderone R (2003) Candida albicans response regulator gene ssk1 regulates a subset of genes whose functions are associated with cell wall biosynthesis and adaptation to oxidative stress. Eukaryot Cell 2(5):1018–1024. https://doi.org/10.1128/ec.2.5.1018-1024.2003
Cleare LG, Zamith-Miranda D, Nosanchuk JD (2017) Heat shock proteins in histoplasma and paracoccidioides. Clin Vaccine Immunol 24(11). https://doi.org/10.1128/cvi.00221-17
Cowen LE, Lindquist S (2005) Hsp90 potentiates the rapid evolution of new traits: drug resistance in diverse fungi. Science 309(5744):2185–2189. https://doi.org/10.1126/science.1118370
de Groot E, Bebelman J, Mager WH, Planta RJ (2000) Very low amounts of glucose cause repression of the stress-responsive gene HSP12 in saccharomyces cerevisiae. Microbiol 146(2):367–375. https://doi.org/10.1099/00221287-146-2-367
De Aguiar Cordeiro R, De Jesus Evangelista AJ, Serpa R, De Farias Marques FJ, De Melo CVS, De Oliveira JS, Da Silva Franco J, De Alencar LP, Bandeira DJPG, Brilhante RSN, Sidrim JJC, Rocha MFG (2016) Inhibition of heat-shock protein 90 enhances the susceptibility to antifungals and reduces the virulence of Cryptococcus neoformans/Cryptococcus gattii species complex. Microbiol 162(2):309–317. https://doi.org/10.1099/mic.0.000222
De Souza IEL, Fernandes FF, Schiavoni MCL, Silva CL, Panunto-Castelo A (2019) Therapeutic effect of DNA vaccine encoding the 60-kDa-heat shock protein from Paracoccidoides brasiliensis on experimental paracoccidioidomycosis in mice. Vaccine 37(37):5607–5613. https://doi.org/10.1016/j.vaccine.2019.07.090
De Souza AF, Tomazett MV, Silva KSFE, De Curcio JS, Pereira CA, Baeza LC, Paccez JD, Gonçales RA, Rodrigues F, Pereira M, De Almeida Soares CM (2021) Interacting with Hemoglobin: Paracoccidioides spp. Recruits hsp30 on Its Cell Surface for Enhanced Ability to Use This Iron Source. J Fungi 7(1):21. https://doi.org/10.3390/jof7010021
Díez S, Gómez BL, Restrepo A, Hay RJ, Hamilton AJ (2002) Paracoccidioides brasiliensis 87-kilodalton antigen, a heat shock protein useful in diagnosis: Characterization, purification, and detection in biopsy material via immunohistochemistry. J Clin Microbiol 40(2):359–365. https://doi.org/10.1128/jcm.40.2.359-365.2002
Dong D, Huang R, Hu Y, Yang X, Xu D, Jiang Z (2023) Assessment of candidate reference genes for gene expression studies using RT-qPCR in Colletotrichum fructicola from litchi. Genes (Basel) 14(12):2216. https://doi.org/10.3390/genes14122216
Dragovic Z, Broadley SA, Shomura Y, Bracher A, Hartl FU (2006) Molecular chaperones of the Hsp110 family act as nucleotide exchange factors of Hsp70s. EMBO J 25(11):2519–2528. https://doi.org/10.1038/sj.emboj.7601138
Eroles P, Sentandreu M, Elorza MV, Sentandreu R (1997) The highly immunogenic enolase and Hsp70p are adventitious Candida albicans cell wall proteins. Microbiol 143(2):313–320. https://doi.org/10.1099/00221287-143-2-313
Fang H, Wu Y, Huang X, Wang W, Ang B, Cao X, Wan T (2011) Toll-like receptor 4 (TLR4) is essential for hsp70-like protein 1 (HSP70L1) to activate dendritic cells and induce th1 response. J Biol Chem 286(35):30393–30400. https://doi.org/10.1074/jbc.m111.266528
Feder ME, Hofmann GE (1999) Heat-shock proteins, molecular chaperones, and the stress response: evolutionary and ecological physiology. Annu Rev Physiol 61(1):243–282. https://doi.org/10.1146/annurev.physiol.61.1.243
Ferreira-Nozawa MS, Silveira HC, Ono CJ, Fachin AL, Rossi A, Martinez-Rossi NM (2006) The ph signaling transcription factor PACC mediates the growth of Trichophyton rubrum on human nail in vitro. Med Mycol 44(7):641–645. https://doi.org/10.1080/13693780600876553
Finkelstein DB, Strausberg S (1983) Identification and expression of a cloned yeast heat shock gene. J Biol Chem 258(3):1908–1913. https://doi.org/10.1016/s0021-9258(18)33075-8
Freitas JS, Silva EM, Leal J, Gras DE, Martinez-Rossi NM, dos Santos LD, Palma MS, Rossi A (2011) Transcription of the HSP30, HSP70, and hsp90 heat shock protein genes is modulated by the Pala protein in response to acid ph-sensing in the fungus aspergillus nidulans. Cell Stress Chaperones 16(5):565–572. https://doi.org/10.1007/s12192-011-0267-5
Fu M-S, De Sordi L, Mühlschlegel FA (2012) Functional characterization of the small heat shock protein hsp12p from candida albicans. PLoS ONE 7(8). https://doi.org/10.1371/journal.pone.0042894
Guindon S, Dufayard J-F, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of phyml 3.0. Syst Biol 59(3):307–321. https://doi.org/10.1093/sysbio/syq010
Gusev NB, Bogatcheva NV, Marston SB (2002) Biochem Mosc 67(5):511–519. https://doi.org/10.1023/a:1015549725819
Hattendorf DA (2002) Cooperative kinetics of both HSP104 atpase domains and interdomain communication revealed by AAA sensor-1 mutants. EMBO J 21(1):12–21. https://doi.org/10.1093/emboj/21.1.12
Hodgetts S, Matthews R, Morrissey G, Mitsutake K, Piper P, Burnie J (1996) Over-expression of Saccharomyces cerevisiae hsp90 enhances the virulence of this yeast in mice. FEMS Immunol Med Microbiol 16(3–4):229–234. https://doi.org/10.1111/j.1574-695x.1996.tb00140.x
Hu L, Sun C, Kidd J, Han J, Fang X, Li H, Liu Q, May AE, Li Q, Liu Z, Liu Q (2023) A first-in-class inhibitor of Hsp110 molecular chaperones of pathogenic fungi. Nat Commun 14(1). https://doi.org/10.1038/s41467-023-38220-2
Hube B, Naglik J (2001) Candida albicans proteinases: resolving the mystery of a gene family. Microbiol 147(8):1997–2005. https://doi.org/10.1099/00221287-147-8-1997
Jacob TR, Peres NT, Martins MP, Lang EA, Sanches PR, Rossi A, Martinez-Rossi NM (2015) Heat shock protein 90 (HSP90) as a molecular target for the development of novel drugs against the Dermatophyte trichophyton rubrum. Front Microbiol 6. https://doi.org/10.3389/fmicb.2015.01241
Jang YJ, Park SK, Yoo HS (1996) Isolation of an HSP12-homologous gene of Schizosaccharomyces pombe suppressing a temperature-sensitive mutant allele of cdc4. Gene 172(1):125–129. https://doi.org/10.1016/0378-1119(96)00058-3
Jia L, Krüger T, Blango MG, Von Eggeling F, Kniemeyer O, Brakhage AA (2020) Biotinylated surfome profiling identifies potential biomarkers for diagnosis and therapy of Aspergillus fumigatus infection. mSphere 5(4). https://doi.org/10.1128/msphere.00535-20
Jung KW, Kang HA, Bahn Y (2013) Essential roles of the Kar2/BiP molecular chaperone downstream of the UPR pathway in Cryptococcus neoformans. Plos One 8(3):e58956. https://doi.org/10.1371/journal.pone.0058956
Katoh K, Rozewicki J, Yamada KD (2017) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20(4):1160–1166. https://doi.org/10.1093/bib/bbx108
Le SQ, Gascuel O (2008) An improved general amino acid replacement matrix. Mol Biol Evol 25(7):1307–1320. https://doi.org/10.1093/molbev/msn067
Lee S, Fan CY, Younger JM, Ren H, Cyr DM (2002) Identification of essential residues in the type II hsp40 SIS1 that function in polypeptide binding. J Biol Chem 277(24):21675–21682. https://doi.org/10.1074/jbc.m111075200
Lefort V, Longueville J-E, Gascuel O (2017) SMS: smart model selection in phyml. Mol Biol Evol 34(9):2422–2424. https://doi.org/10.1093/molbev/msx149
Li H, Hu L, Cuffee CW, Mohamed M, Li Q, Liu Q, Liu Z, Liu Q (2021) Interdomain interactions dictate the function of the Candida albicans Hsp110 protein Msi3. J Biol Chem 297(3):101082. https://doi.org/10.1016/j.jbc.2021.101082
López-Matas MÁ, Núñez PM, Soto Á, Allona I, Casado R, Collada C, Guevara MÁ, Aragoncillo C, Gómez L (2004) Protein cryoprotective activity of a cytosolic small heat shock protein that accumulates constitutively in chestnut stems and is Up-Regulated by low and high temperatures. Plant Physiol 134(4):1708–1717. https://doi.org/10.1104/pp.103.035857
Lv F, Xu Y, Gabriel DW, Wang X, Zhang N, Liang W (2022) Quantitative proteomic analysis reveals important roles of the acetylation of ER-resident molecular chaperones for conidiation in fusarium oxysporum. Mol Cell Proteomics 21(5):100231. https://doi.org/10.1016/j.mcpro.2022.100231
Martinez-Rossi NM, Jacob TR, Sanches PR, Peres NTA, Lang EAS, Martins MP, Rossi A (2016) Heat shock proteins in dermatophytes: current advances and perspectives. Curr Genomics 17(2):99–111. https://doi.org/10.2174/1389202917666151116212437
Maeng S, Ko Y-J, Kim G-B, Jung K-W, Floyd A, Heitman J, Bahn Y-S (2010) Comparative transcriptome analysis reveals novel roles of the Ras and cyclic AMP signaling pathways in environmental stress response and antifungal drug sensitivity in Cryptococcus neoformans. Eukaryot Cell 9(3):360–378. https://doi.org/10.1128/ec.00309-09
Minh BQ, Dang CC, Vinh LS, Lanfear R (2021) QMaker: Fast and accurate method to estimate empirical models of protein evolution. Syst Biol 70(5):1046–1060. https://doi.org/10.1093/sysbio/syab010
Mitra A, Bhakta K, Kar A, Roy A, Mohid SA, Ghosh A, Ghosh A (2023) Insight into the biochemical and cell biological function of an intrinsically unstructured heat shock protein, Hsp12 of Ustilago maydis. Mol Plant Pathol 24(9):1063–1077. https://doi.org/10.1111/mpp.13350
Montero-Barrientos M, Hermosa R, Cardoza RE, Gutiérrez S, Nicolás C, Monte E (2010) Transgenic expression of the Trichoderma harzianum hsp70 gene increases Arabidopsis resistance to heat and other abiotic stresses. J Plant Physiol 167(8):659–665. https://doi.org/10.1016/j.jplph.2009.11.012
Motta H, Reuwsaat JCV, Squizani ED, Da Silva Camargo M, Garcia AWA, Schrank A, Vainstein MH, Staats CC, Kmetzsch L (2023) The small heat shock protein Hsp12.1 has a major role in the stress response and virulence of Cryptococcus gattii. Fungal Genet Biol 165:103780. https://doi.org/10.1016/j.fgb.2023.103780
Nagy LG, Tóth R, Kiss E, Slot J, Gácser A, Kovács GM (2017) Six key traits of fungi: Their evolutionary origins and genetic bases. Microbiol Spectrum 5(4). https://doi.org/10.1128/microbiolspec.funk-0036-2016
Naranjo-Ortiz MA, Gabaldón T (2019) Fungal evolution: major ecological adaptations and evolutionary transitions. Biol Rev 94(4):1443–1476. https://doi.org/10.1111/brv.12510
Neves-da-Rocha B, Oliveira S, Rossi, & Martinez-Rossi. (2019) Alternative splicing in heat shock protein transcripts as a mechanism of cell adaptation in Trichophyton Rubrum. Cells 8(10):1206. https://doi.org/10.3390/cells8101206
Nozawa SR, Ferreira-Nozawa MS, Martinez-Rossi NM, Rossi A (2003) The PH-induced glycosylation of secreted phosphatases is mediated in aspergillus nidulans by the regulatory gene PACC-dependent pathway. Fungal Genet Biol 39(3):286–295. https://doi.org/10.1016/s1087-1845(03)00051-3
Oh HJ, Chen X, Subjeck JR (1997) HSP110 protects heat-denatured proteins and confers cellular thermoresistance. J Biol Chem 272(50):31636–31640. https://doi.org/10.1074/jbc.272.50.31636
Polier S, Dragovic Z, Hartl FU, Bracher A (2008) Structural basis for the cooperation of HSP70 and HSP110 chaperones in protein folding. Cell 133(6):1068–1079. https://doi.org/10.1016/j.cell.2008.05.022
Qian X, Hou W, Zhengang L, Sha B (2001) Direct interactions between molecular chaperones heat-shock protein (HSP) 70 and HSP40: yeast hsp70 SSA1 binds the extreme C-terminal region of yeast HSP40 SIS1. Biochem J 361(1):27–34. https://doi.org/10.1042/bj3610027
Ramos CHI, Oliveira CLP, Fan C-Y, Torriani IL, Cyr DM (2008) Conserved central domains control the quaternary structure of type I and type II HSP40 molecular chaperones. J Mol Biol 383(1):155–166. https://doi.org/10.1016/j.jmb.2008.08.019
Raviol H, Sadlish H, Rodriguez F, Mayer M, Bukau B (2006) Chaperone network in the yeast cytosol: Hsp110 is revealed as an Hsp70 nucleotide exchange factor. EMBO J 25(11):2510–2518. https://doi.org/10.1038/sj.emboj.7601139
Robbins N, Cowen LE (2023) Roles of Hsp90 in Candida albicans morphogenesis and virulence. Curr Opin Microbiol 75:102351. https://doi.org/10.1016/j.mib.2023.102351
Rocha MC, Minari K, Fabri JHTM, Kerkaert JD, Gava LM, Da Cunha AF, Cramer RA, Borges JC, Malavazi I (2020) Aspergillus fumigatus Hsp90 interacts with the main components of the cell wall integrity pathway and cooperates in heat shock and cell wall stress adaptation. Cell Microbiol 23(2). https://doi.org/10.1111/cmi.13273
Sela I, Ashkenazy H, Katoh K, Pupko T (2015) Guidance2: accurate detection of unreliable alignment regions accounting for the uncertainty of multiple parameters. Nucleic Acids Res 43(W1). https://doi.org/10.1093/nar/gkv318
Seymour IJ, Piper PW (1999) Stress induction of HSP30, the plasma membrane heat shock protein gene of saccharomyces cerevisiae, appears not to use known stress-regulated transcription factors. Microbiol 145(1):231–239. https://doi.org/10.1099/13500872-145-1-231
Taipale M, Jarosz DF, Lindquist S (2010) Hsp90 at the hub of protein homeostasis: emerging mechanistic insights. Nat Rev Mol Cell Biol 11(7):515–528. https://doi.org/10.1038/nrm2918
Ting W, Chang C, Szonyi B, Gizachew D (2020) Growth and aflatoxin B1, B2, G1, and G2 production by Aspergillus flavus and Aspergillus parasiticus on ground flax seeds (Linum usitatissimum). J Food Prot 83(6):975–983. https://doi.org/10.4315/jfp-19-539
Tiwari S, Thakur R, Shankar J (2015) Role of heat-shock proteins in cellular function and in the biology of fungi. Biotechnol Res Int 2015:1–11. https://doi.org/10.1155/2015/132635
Tiwari S, Shankar J (2018) HSP70 in Fungi: evolution, function and vaccine candidate. In: Heat shock proteins, pp 381–400. https://doi.org/10.1007/978-3-319-89551-2_20
Vajda V, McLoughlin S (2004) Fungal proliferation at the cretaceous-tertiary boundary. Science 303(5663):1489–1489. https://doi.org/10.1126/science.1093807
Vanittanakom N, Pongpom M, Praparattanapan J, Cooper CR, Sirisanthana T (2009) Isolation and expression of heat shock protein 30 gene fromPenicillium marneffei. Med Mycol 47(5):521–526. https://doi.org/10.1080/13693780802566358
Woodruff DC, Wolff ED, Wedel MJ, Dennison S, Witmer LM (2022) The first occurrence of an avian-style respiratory infection in a non-avian dinosaur. Sci Rep 12(1). https://doi.org/10.1038/s41598-022-05761-3
Zara S, Antonio Farris G, Budroni M, Bakalinsky AT (2002) Hsp12 is essential for biofilm formation by a Sardinian wine strain OFS. Cerevisiae Yeast 19(3):269–276. https://doi.org/10.1002/yea.831
Zhang D, Wang Y, Shen S, Hou Y, Chen Y, Wang T (2020) The mycobiota of the human body: a spark can start a prairie fire. Gut Microbes 11(4):655–679. https://doi.org/10.1080/19490976.2020.1731287
Zhou X, Su L, Tang R, Dong Y, Wang F, Li R, Xie Q, Zhang X, Xiao G, Li H (2023) Genome-wide analysis of Hsp40 and Hsp70 gene family in four cotton species provides insights into their involvement in response to Verticillium dahliae and abiotic stress. Front Genet 14. https://doi.org/10.3389/fgene.2023.1120861
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by João Pedro Sagini and Rodrigo Ligabue-Braun. The first draft of the manuscript was written by João Pedro Sagini, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by: Volker Loeschke
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sagini, J.P.N., Ligabue-Braun, R. Fungal heat shock proteins: molecular phylogenetic insights into the host takeover. Sci Nat 111, 16 (2024). https://doi.org/10.1007/s00114-024-01903-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-024-01903-x