Abstract
Volatile odors from flowers play an important role in plant-pollinator interaction. The honeybee is an important generalist pollinator of many plants. Here, we explored whether any components of the odors of a range of honeybee-pollinated plants are commonly involved in the interaction between plants and honeybees. We used a needle trap system to collect floral odors, and GC–MS analysis revealed nonanal was the only component scent detected in 12 different honeybee-pollinated flowers and not present in anemophilous plant species. For Ligustrum compactum, blooming flowers released significantly more nonanal than buds and faded flowers. For Sapium sebiferum, nonanal release through the day correlated with nectar secretion. Experimentally increasing nectar load in flowers of Sapium sebiferum, Ligustrum compactum, and Castanea henryi increased nonanal levels also. Nonanal was also detected in flower nectar and honeys from experimental colonies. Electroantennogram recordings and behavioral observations showed that untrained honeybees could detect and were strongly attracted to nonanal. We argue that nonanal persists in both honey and nectar odors facilitating a learned association between nonanal and food reward in honeybees.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Most flowering plant species rely on animals for pollination and have developed a range of mechanisms to attract pollinators to them, commonly offering a form of food reward to the animals (Kiester et al. 1984). Many plant species use floral scents or flower colors as a cue to attract their pollinators, improving successful pollen transfer between flowers and pollination (Chittka and Raine 2006; Belsare et al. 2009; Wright and Schiestl 2009; Leonard et al. 2010; Zhang et al. 2012), particularly in habitats with high diversities of pollinators and plants (Pauw et al. 2009). A variety of floral odors have been shown to attract pollinators or repel enemies, facilitating the identification of host flowers for pollinators and improvement of pollination efficiency for plants (Schiestl 2010; Chapurlat et al. 2019) . For instance, volatile odor phenylacetaldehyde from flowers attracts bumble bees (Bombus terrestris) (Knauer and Schiestl 2015), and benzenoids produced by Dianthus inoxianus attract their pollinating hawkmoths (Hyles livornica) (Balao et al. 2011).
The honeybee (Apis mellifera) is an important insect pollinator with excellent intelligence, and also an extreme floral generalist (Menzel and Muller 1995; Klein et al. 2007). Many agriculturally important flower species are visited and pollinated by honeybees, including over 85% of crop plants (Klein et al. 2007). Here, we explored whether there was any evidence for a general reliable signal of floral nectar reward status produced by honeybee-pollinated flowers.
Nectar in most plants is colorless and concealed within the flower (Rodriguez-Girones and Santamaria 2005, 2006). Floral scents, however, play an important role in attracting bee pollinators to flowers from a distance (Daumer 1956, 1958; Glaettli and Barrett 2008; Wright and Schiestl 2009; Schiestl 2010; Krishna and Keasar 2018). Honeybees have excellent olfactory acuity and outstanding olfactory learning ability and sensitivity (Menzel and Muller 1995; Joerges et al. 1997; Laska et al. 1999; Galizia et al. 1999; Komischke et al. 2002; Scheiner et al. 2005; Galán et al. 2014). Therefore, we hypothesize that floral odors might be an important cue for honeybees to detect flower nectar status. Honeybees are also excellent learners and possessed of robust long-term memory (Müller 2002; Hourcade et al. 2009; Eisenhardt 2014). They rapidly learn to identify the most rewarding flowers (Bhagavan and Smith 1997; Laska et al. 1999; Chittka and Raine 2006; Wright et al. 2002, 2009). As a highly eusocial species, bees develop inside a colony feeding on honey which has been generated from nectars gathered by previous generations of foragers (Seeley 1985). If a nectar odor was able to persist in honey stored within a colony, bees may learn to associate the components of honey odors with feeding even before they begin to forage. This could predispose forager bees toward flowers possessed of that odor in their nectar. Hence, the social structure of the hive could facilitate transgenerational fidelity of a plant-pollinator relationship via long-lasting odor cues in nectars.
To explore this hypothesis, we examined whether there were any specific odors that were common to honeybee-pollinated flowers and that were also present in stored honey, and how honeybees reacted to those odors. We collected floral scents from 12 honeybee-pollinated plant species and 3 anemophilous species using a needle trap and a gas chromatography-mass spectrometry (GC–MS) system to investigate whether there were any common components to the odors of in honeybee-visited plants. We investigated the odor profiles of buds, blooming flowers and faded flowers, and related flower odor profiles to time of day and nectar amount. We also sampled honey odors. Finally, we tested the electroantennogram response of honeybee antennae to candidate odor cues. We also tested their behavioral response to these candidate odor cues.
Materials and methods
Insects and plants
Honeybees (Apis mellifera) were sourced from five colonies at Jiangxi Agricultural University, Nanchang, China (28.46uN, 115.49uE). Each colony contained a mated queen and 9 frames with approximately 30,000 workers.
Plant species were selected for this study from the list of known honeybee-pollinated species described in “China nectar and pollen plants” (Xu 1992). Litchi chinensis and Mangifera indica were sourced from Fuzhou city, Fujian province, China. All other plant species were sourced from Nanchang city, Jiangxi province, China.
Experiment 1: qualitative analysis on floral scents
All experiments were performed on sunny days (temperature: 20–30 °C, wind speed < 5 m/s). Prior to sampling, needles from the needle trap system (fatty acid type, PAS technology, Germany) were inserted into the GC–MS at 250 °C for 2 h to desorb any chemical residues. Female or hermaphroditic flowers from all plant species were wrapped in a plastic bag (25 cm * 35 cm) for 15 min. Five to hundreds of flowers were bagged depending on the flower size and structure for each species. A needle was used to adsorb floral scents. Needles were inserted into the plastic bag surrounding the flower bract to extract 50 mL of air with an airflow speed of 15 mL/min to sample floral odors. Three independent samples were taken for each plant species, each sample from a different bag of flowers.
The needle with adsorbed floral scent was injected into a GC–MS system (5977B-7890B, Agilent Technologies) with a DB-5MS chromatographic column (30 m, 0.25 mm, 0.25 μm film thickness, 112–5532, Agilent Technologies). GC–MS analysis methods are fully described in He et al. (2016). Briefly, needles were connected to a sterile syringe (PAS technology). The syringe drove 1 mL pure helium through the needle to desorb floral scents into the injection port of the GC–MS system at 250 °C. The column temperature profile was 35 °C for 2 min, then increasing from 35 to 240 °C at 5 °C /min. Then the column temperature was held at 240 °C for 5 min. The helium pressure was 6.7776 psi and the electron impact ion source (EI) was 70 eV. GC–MS data were mapped to the NIST 17.0 database. Gas from plastic bags without flowers was also sampled (replicated three times) as controls.
Experiment 2: quantitative analysis of nonanal from flowers
Data from experiment 1 indicated nonanal as the only odor common to all bee-pollinated flowers sampled. To measure the daily variation in amount of nonanal from Sapium sebiferum, flowers were sampled at 6:00 a.m. (temperature: 25 °C), 9:00 a.m. (27 °C), 12:00 a.m. (29 °C), 3:00 p.m. (32 °C), and 6:00 p.m. (29 °C) on 19 June 2019. In experiment 2, each sample contained 12 flowers, which were placed into a 250-mL glass bottle containing 2 uL 1-octanol (2 mL/L, purity > 99.8%, Xilong Science, China) as an internal standard. Flowers in bottles were kept under room temperature (25 °C) for 15 min. A needle was used to extract 50 mL gas from the bottle at an airflow speed of 15 mL/min. And floral scents were injected into GC–MS system as in experiment 1 for quantitative analysis, but here, we used a different temperature profile for the column to save the retention time. The column temperature was programmed as follows: 40 °C for 2 min, then rising to 150 °C at a rate of 8 °C/min, and then rising to 250 °C at a rate of 16 °C/min, finally held constant at 250 °C for 5 min. Other GC–MS procedures were same as above, and are detailed in He et al. (2016).
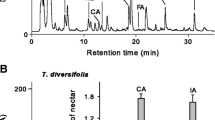
To test whether the release of nonanal is consistent with states of flowers, we also compared the amount of nonanal released from blooming flowers (250 flowers per sample), buds (250), and faded flowers (250) of Ligustrum compactum (Fig. 1B) using the method as above. This study was repeated three times.
A Nonanal amount from Sapium sebiferum flowers at five time points on a sunny day. Each bar means mean ± SE of nonanal from samples of 12 cluster of flowers. Each group had three biological replicates. Data were normalized by square-root transformation and were analyzed by ANOVA test followed by Fisher’s PLSD test. “*” represents significant difference (p < 0.05). B Nonanal amounts from buds, blooming, and faded flowers of Ligustrum compactum. Each bar shows mean ± SE of nonanal amount from samples of 250 flowers, and each group had three biological replicates. Data were normalized by square-root transformation and were analyzed by ANOVA test followed by Fisher’s PLSD test. “*” represents significant difference (p < 0.05), and no “*” represents no significant difference
A standard curve was established for quantitative analysis of nonanal. Two standard chemicals nonanal (purity: 95%) and 1-octanol were purchased from Sigma-Aldrich (the United States of America) and Xilong Science (China). One μL 1-octanol injections and six levels of nonanal (0 μL, 0.01 μL, 0.1 μL, 1 μL, 10 μL, and 100 μL) were added into 5 mL ethanol (purity > 99.8%, Xilong Science). Selected ion monitoring chromatograms (SIM) were reconstructed for nonanal and the internal standard 1-octanol. These generated major peaks at the mass-to-charge ratio (m/z) 56 and 57 respectively. The correlation coefficient of the standard curve was 0.9998 (Fig. S1). By reference to the standard curve, we were able to quantify the amount of nonanal in floral odor samples.
Experiment 3: measurement of nonanal and nectar from flowers
To explore the relationship between amount of nectar and amount of nonanal in flowers, for two plant species (Sapium sebiferum and Ligustrum compactum), we manipulated nectar amount and measured nonanal amount. Branches of flowers were wrapped with plastic mesh net (mesh size 5 mm2) for 2 h to exclude insect visits. This prevented pollinators to visit the wrapped flowers but allowed airflow across the mesh net. Control branches were not wrapped. Twelve Sapium sebiferum and 250 Ligustrum compactum flowers were cut and placed into 250-mL glass bottles with 2 μL 1-octanol internal standard as in experiment 2. Air from these samples was collected and analyzed by needle trap and GC–MS systems as in experiment 2. Three measures were made for each treatment of each species.
To confirm whether the nectar volume in Ligustrum compactum flowers would increase after branches enveloped by plastic mesh net for 2 h, 30 flowers were picked at random in the mesh net and control branches and the nectaries were imaged with a dissecting microscope (Nanjing Jiangnan Novel Optics Co., Ltd, SE2200). Each flower has one nectary. We scored how many of the 30 nectaries had nectar droplets. Each group had 6 replicates. Sapium sebiferum flowers have many nectaries. For this species, we randomly sampled 30 nectaries from a flower and scored how many nectaries had visible nectar droplets. Each group had 30 replicates. Since the nectar drops in flowers are not stable, a small sample size was used and flowers were rapidly measured in this experiment.
Previous studies indicated that the nonanal exists in many parts of flowers (Kaškonienė et al. 2015; Uzun et al. 2017; Liu et al. 2020); therefore, this experiment aimed to test whether nonanal is directly from nectar droplets. We selected Castanea henryi flowers (this plant had the highest proportion of nonanal in floral scents, see Table 1) as experimental materials. Blooming flowers of the experimental group were wrapped by plastic mesh net as in experiment 2 for 2 h from 7:00 a.m. to 9:00 a.m., and nectar droplets were collected using precalibrated pipettes (5μL; inner diameter: 0.4 mm; A. Hartenstein GmbH, Germany). Nectar from 5 clusters of flowers was collected as one sample and 7 biological replicates were sampled. Nectar from unwrapped flowers from same trees was sampled as control. Nectar droplets were blown into H2O (5 mL) with N2 gas, added with 2 μL 1-octanol as internal standard. The floral scents in nectar were extracted with CH2Cl2 (5 mL) and shaken for 10 min using an oscillator (200 r/min). The CH2Cl2 layer was transferred into a new vial and concentrated under nitrogen flow to about 100 μL. A 1 μL sample was injected into the GC/MS. The GC–MS method (SIM method) was the same as experiment 2. A standard curve for this analysis was established and the correlation coefficient was 0.9989. Using the same methods, we also examined nonanal in Brassica campestris nectar droplets.
Experiment 4: measurement of nonanal from honeys
Honey samples were collected from 5 research hives. For each sample, we extracted 5 mL honey into a 20-mL headspace vial. These vials were placed in a water bath (Tianjin Laiyuenage Co. Ltd.) under 80 °C for 15 min. A needle was used to extract 10 mL of air with an airflow speed of 2 mL/min from a honey sample and were injected into GC–MS system for qualitative analysis same as experiment 1. The column temperature profile used here was 40 °C for 2 min, then 40 to 250 °C at 8 °C /min, and then 250 °C for 5 min. Honey odors were then identified by the GC–MS system. From each hive, we took three honey samples. In total, 15 samples were collected.
Experiment 5: honeybee antennal response to nonanal
To determine whether honeybees could detect nonanal, pollen forager honeybees were captured at the hive entrance, and the left antenna was cut for electrophysiological recording by electroantennogram (using a Syntech EAG platform, Germany). The five odors used in experiment 4 were presented to each antenna: nonanal, benzaldehyde diethyl acetal (purity > 98%, Tokyo Chemical Industry), benzaldehyde (purity > 99%, Tokyo Chemical Industry), linalool (purity ≥ 95%, Sigma-Aldrich), and phenylacetaldehyde (purity ≥ 95%, Sigma-Aldrich). Chemicals were dissolved in ethanol (Brockmann et al. 1998), each at five different concentrations (0.08 mL/L, 0.4 mL/L, 2 mL/L, 10 mL/L, and 50 mL/L). Ethanol alone served as a control. Ten microliters of an odor solution was loaded onto a filter paper strip and placed in a 1-cm Pasteur pipette (15 cm). The honeybee antenna was connected to the detection electrodes and placed 0.5 cm away from the tip of the pipette in a high humidity continuous airflow at 10 mL/s into which a stimulus pulse of the odor was added for 2 s at 5 mL/s. Two antennal responses to each odor solution were recorded, and five different antennae were used for each chemical. In total, 25 antennae were used.
Since antennae had the strongest responses to 2 mL/L concentration of the odorants (Fig. 4A–E), we repeated the study focusing on this dose only using further 10 antennae. Each honeybee antenna was tested with five compounds, as above, and each compound repeated twice.
Experiment 6: behavioral responses of honeybees to floral odors
To assess honeybee preferences for different floral odors, color marked honeybee foragers were trained to feed on sucrose solution (30–50% depending to the weather and temperature) from an artificial flower placed in the center of a 50-cm-diameter circular platform. Artificial flowers were similar to those used by Andrew et al. (2014): an inverted orange bottle cap (diameter: 34.4 mm) with four holes (diameter: 4.4 mm). Sucrose could be drunk from a shallow well in the middle of each cap, and odors dispersed from 10 μL droplets of odor solution placed beneath each cap.
Forager bees were caught randomly at the entrance of one of the same five research hives from which we sampled honey (experiment 4) and relocated to the artificial flower on the circular platform. Once the focal bee had returned by themselves to the platform, they were marked with colored paint on the thorax. For an odor test, five artificial flowers were placed equidistant around the edge of the platform. All artificial flowers offered sucrose, but each artificial flower offered a different odor: nonanal, benzaldehyde diethyl acetal, benzaldehyde, linalool, and phenylacetaldehyde. These were the main floral compounds from honeybee-visiting flowers (Table S1). Odors were diluted in paraffin oil (Spectrum pure, Sigma-Aldrich) at a concentration of 10%, following Andrew et al. (2014).
Bees were tested individually. The first artificial flower each marked forager landed on was recorded. Artificial flowers were replaced after each visit to avoid any marking pheromones from previous foragers. The platform was randomly rotated to avoid any possible direction preference of foragers. Foragers were captured after their first landing so each visit scored was from a different bee, and bees could not influence each other’s choices.
Data analysis
Nonanal amounts and numbers of flowers or nectaries with nectar drops (Figs. 2 and 3) were analyzed with one-way ANOVA (Statview 5.01 package, SAS Institute Inc., the United States of America). Electroantennogram (EAG) response data (Figs. 4 and S2) were analyzed by ANOVA test followed by Fisher’s PLSD test (Statview 5.01). Artificial flower preferences of bees (Fig. 5) were analyzed with chi-square tests (SPSS 17.0 package, IBM, the United States of America). Nonanal amounts (Fig. 1) were normalized by square-root transformation and were analyzed by ANOVA test followed by Fisher’s PLSD test (Statview 5.01).
A Nectar drop on Sapium sebiferum nectaries viewed under microscope. Flowers were magnified 40 times. B A nectar drop in a Ligustrum compactum flower under microscope. Flowers were magnified 40 times also. C Number of flowers or nectaries of Sapium sebiferum and Ligustrum compactum with nectar drops from samples of 30 nectaries studied. Each group in Sapium sebiferum and Ligustrum compactum flowers had 30 and 6 replicates respectively. D Nonanal amounts from netted and control flowers. Twelve cluster of Sapium sebiferum flowers and 250 Ligustrum compactum flowers of the netted and control groups were sampled and analyzed by needle trap and GC–MS. Each group had three biological replicates. Each bar shows the mean ± SE. Data were analyzed using one-way ANOVA test. “*” represents significant difference (p < 0.05) between two groups
Nectar and nonanal amounts from Castanea henryi flowers. The left bars were nectar amounts from 5 clusters of flowers and the right bars were nonanal amounts from the relative nectar droplets. Each bar shows the mean ± SE. Data were analyzed using one-way ANOVA test. “*” represents significant difference (p < 0.05) between two groups
EAG response of honeybees to 5 floral components. Antenna response to the 2 mL/L concentration of each of the five compounds. Each bar shows the mean ± SE of EAG response. Data were analyzed using ANOVA tests followed with Fisher’s PLSD test. Different letters represent significant difference (p < 0.05), and same letters represent no significant difference
The attractiveness of 5 floral odors to honeybees using mimic flowers. Each bar shows the number of foragers that landed on each of the five odors in the preference test expressed as a percentage of the 193 foragers tested in this study. Choice data were compared to the nonanal group with a chi-square test. “*” represents significant difference (p < 0.05) between nonanal and other four groups
Results
Nonanal was the unique floral scent that commonly existed in all 12 honeybee-visited flower species
Qualitative analysis of flower scents of 12 honeybee-visited plant species indicated that nonanal was the unique component commonly in floral odors of all 12 honeybee-visited plant species, and not identified in the three anemophilous flowers, which are not insect pollinated (Tables 1 and S1).
Nonanal levels peaked at 9:00 a.m. and 15:00 p.m. and in blooming flowers
For Sapium sebiferum, nonanal release peaked at 9:00 a.m. and 3:00 p.m. Nonanal at 3:00 p.m. was significantly higher than that at 6:00 p.m. (Fig. 1A). Open flowers of Ligustrum compactum released significantly more nonanal than either buds or faded flowers (Fig. 1B).
Netted flowers released significantly more nonanal
For Sapium sebiferum and Ligustrum compactum, flowers wrapped with a mesh net for 2 h had significantly more nectar droplets than unwrapped controls (Fig. 2C). Nonanal amounts were higher in wrapped flowers than unwrapped flowers for both species (Fig. 2D). Furthermore, nonanal was directly detected from Castanea henryi nectar droplets, and experimentally manipulated flowers had significantly higher nectar and nonanal amounts compared to the control (Fig. 3). These results suggest that nonanal was correlated with nectar status of flowers.
Nonanal detected in honey odor and flower nectar
In total, 13 volatile components were identified from honey samples (Table S1). The largest component was phenylacetaldehyde (27.32 ± 4.79, mean ± SE, percentage composition). Nonanal was detected from all samples and ranked as the seventh most abundant component of the odors (6.24 ± 0.93, mean ± SE, %). In addition, nonanal also existed in Brassica campestris nectar droplets and was 6.95 ± 0.58 μg/mL (Table 1).
Honeybee antennae are sensitive to nonanal
EAG responses to nonanal and benzaldehyde were greatest at the 2 mL/L odor concentration (Fig. S2). EAG responses to nonanal were greater than to four other common floral odors and the control (Fig. 4).
Nonanal was the most attractive odor to honeybees
Five main floral components were selected for a preference test with honeybee foragers. Artificial flowers scented with nonanal were more attractive to foragers than four other common floral scents (Fig. 5).
Discussion
Here, we explored whether honeybee-pollinated plants have a common odor that may play a role in plant and honeybee interaction. We analyzed all floral scents from 12 honeybee-pollinated plant flowers. We found nonanal was the only component common to the floral bouquets of all 12 honeybee-pollinated plants measured (Tables 1 and S1). Nonanal is common in floral scents of honeybee-visited plants (Jorgensen et al. 2000; Alissandrakis et al. 2011; Twidle et al. 2018). We noted a correlation between amount of nectar offered by different plant species and the proportion of nonanal in their floral scents (Tables 1 and S1).
For Sapium sebiferum, we noted two peaks of nonanal release—09:00 a.m. and 3:00 a.m. (Fig. 1A)—which coincided with the recorded peak nectar secretion for this species (Xu 1992). Moreover, we found that experimentally increasing the amount of nectar in flowers of both Sapium sebiferum and Ligustrum compactum increased amounts of nonanal (Fig. 2). Nonanal was directly detected from Castanea henryi nectar, and experimentally manipulating flowers significantly increased both nectar and nonanal amounts (Fig. 3). Therefore, in both within and between honeybee-pollinated flower species, we documented a relationship between the amount of nonanal in floral odors and nectar availability. Nonanal elicited a robust EAG response (Fig. 4). A similar EAG result was previously reported for Apis cerana that is more sensitive to nonanal than other floral odors such as linalool and decanal (Luo et al. 2013). Nonanal was also found at significant levels in honey odors (Tables 1 and S1) and it was found to be highly attractive to untrained honeybees sampled from these colonies (Fig. 5). Therefore, we are confident that honeybees can perceive nonanal and respond behaviorally to it.
Previously, nonanal has been found in both flowers of Castanea sativa and Eucalyptus globulus and the honeys produced from these flowers (Alissandrakis et al. 2011). Naef et al. (2004) showed that nonanal exists in both nectar and the content of the bee honey stomach. Here, we examined nonanal in flowers and honeybee colonies simultaneously, and our results showed that nonanal existed both in nectar droplets (Fig. 3 and Table 1) and honey samples (Table 1). Therefore, these indicate that nonanal is present within nectar and persists in honey and may be involved into the plant-honeybee interaction.
Interestingly, nonanal was not the highest component of honey odor by any means: phenylacetaldehyde (Table S1). Even so, nonanal was more attractive to untrained forager honeybees than phenylacetaldehyde (Figs. 4 and 5). Phenylacetaldehyde was detected in the floral bouquet of just 2 of the 12 flower species we sampled, whereas nonanal was common to all (Table S1). It appears that a range of diverse honeybee-pollinated plants advertise their current nectar status by the same volatile chemical that is highly attractive to honeybees. Therefore, nonanal possibly play as a general cue of nectar availability for honeybees.
Nonanal is a very common volatile substance existing in the world, and it can be produced by plants and microorganisms, etc. (Abanda-Nkpwatt et al. 2006; Baldwin 2010; Wu et al. 2014). Our results detected nonanal in nectar droplets (Fig. 3 and Table 1) and honey samples but not in blank control and flower samples from anemophilous plant species (Table 1). Nonanal has been detected in dozens of flowers and honey samples (Jorgensen et al. 2000; Schade et al. 2001; Alissandrakis et al. 2007, 2011; Jerkovi et al. 2009; Bianchi et al. 2011; Bayraktar and Ur 2011; Twidle et al. 2018). Nonanal has been found in both flowers of Castanea sativa and Eucalyptus globulus and the honeys produced from these flowers (Alissandrakis et al. 2011). Naef et al (2004) showed that nonanal exists in both nectar and the content of the bee honey stomach, suggesting that nonanal can be transferred from nectar into honey in beehives. In addition, nonanal can also be found in indoor air but the concentration is lower than 100 μg/m3 (Daisey and Hopke 1991; Iwashita and Hibino 2011; Iwashita and Tokunaga 2012). The concentration of nonanal in nectar (6.95 ± 0.58 μg/mL) is dramatically higher than that in an indoor air pollution, suggesting that nonanal originated from honey samples rather from air pollution. Therefore, we are confident that nonanal is present within nectar, and persists in honey rather an environmental pollutant.
Honeybees are excellent olfactory learners and can learn both complex odor blends and components contained therein (Bhagavan and Smith 1997; Laska et al. 1999; Wright et al. 2002, 2009). With this capacity, bees could certainly learn features of specific flower types to maintain floral constancy (Wright and Schiestl 2009). A generalizable cue associated with food information that operated across flower types could increase the speed of food collection at which a bee could maximize foraging efficiency on a new flower species. This would benefit the forager bee, and also improve pollination by enhancing fidelity to the new flower type (Arenas and Kohlmaier 2019; Wright and Schiestl 2009). Honeybees also share nectar after foraging and recruit to rewarding flowers via their dance language (Farina et al. 2007). Moreover, nurse bees deliver characteristic information of food to young larvae and young bees in the whole colony. New bees thereby obtain this food characteristic information by excellent learning and memory abilities before foraging (Bhagavan and Smith 1997; Laska et al. 1999; Wright et al. 2002, 2009). Individually, distinctive flower odors would allow new recruits to identify the advertised flower type, but a generalizable cue of nectar availability would facilitate the new recruits to obtain reward from the flowers. In this case, the response of foragers to nonanal could be facilitated by the shared social environment of successive generations of forager bees, and their excellent learning abilities. Since nonanal persists in honey odors, it is possible that forager bees have already learned to associate nonanal with feeding in the colony before foraging. The ubiquitous association of nonanal and honeybee-pollinated flower species would consistently reinforce that association.
In conclusion, this study demonstrated that the floral scent nonanal is correlated with nectar status across a range of honeybee-pollinated plants. The attraction of bees to nonanal appears to be facilitated by nonanal persisting in the odors of honeybees feed on in the hive. Nonanal seems to play an important role in the plant-honeybee interaction, but this interaction is far more complex and poorly understood. Not only floral scents but also flower shapes, colors, and ultraviolet signal (Moyroud et al. 2017) may also involve in this complex plant-pollinator interaction, which needs deeper investigations.
References
Abanda-Nkpwatt D, Krimm U, Coiner HA, Schreiber L, Schwab W (2006) Plant volatiles can minimize the growth suppression of epiphytic bacteria by the phytopathogenic fungus Botrytis cinerea in co-culture experiments. Environ Exp Bot 56:108–119. https://doi.org/10.1016/j.envexpbot.2005.01.010
Alissandrakis E, Tarantilis PA, Harizanis PC, Polissiou M (2007) Comparison of the volatile composition in thyme honeys from several origins in Greece. J Agric Food Chem 55:8152–8157. https://doi.org/10.1021/jf071442y
Alissandrakis E, Tarantilis PA, Pappas C, Harizanis PC, Polissiou M (2011) Investigation of organic extractives from unifloral chestnut (Castanea sativa L.) and eucalyptus (Eucalyptus globulus Labill.) honeys and flowers to identification of botanical marker compounds. LWT-Food Sci Technol 44:1051
Andrew SC, Perry CJ, Barron AB, Berthon K, Peralta V, Cheng K (2014) Peak shift in honey bee olfactory learning. Anim Cogn 17:1177–1186. https://doi.org/10.1007/s10071-014-0750-3
Arenas A, Kohlmaier MG (2019) Nectar source profitability influences individual foraging preferences for pollen and pollen-foraging activity of honeybee colonies. Behav Ecol Sociobiol 73:34. https://doi.org/10.1007/s00114-006-0176-0
Balao F, Herrera J, Talavera S, Dotterl S (2011) Spatial and temporal patterns of floral scent emission in Dianthus inoxianus and electroantennographic responses of its hawkmoth pollinator. Phytochem 72:601–609. https://doi.org/10.1016/j.phytochem.2011.02.001
Baldwin IT (2010) Plant volatiles. Curr Biol 20:392–397. https://doi.org/10.1016/j.cub.2010.02.052
Bayraktar D, Ur TAO (2011) Investigation of the aroma impact volatiles in Turkish pine honey samples produced in Marmaris, Data and Fethiye regions by SPME/GC/MS technique. Int J Food Sci Tech 46:1060–1065. https://doi.org/10.1111/j.1365-2621.2011.02588.x
Belsare PV, Sriram B, Watve MG (2009) The co-optimization of floral display and nectar reward. J Biosci 34:963–967. https://doi.org/10.1007/s12038-009-0110-7
Bhagavan S, Smith BH (1997) Olfactory conditioning in the honey bee, Apis mellifera: effects of odor intensity. Physiol Behav 61:1–117. https://doi.org/10.1016/s0031-9384(96)00357-5
Bianchi F, Mangia A, Mattarozzi M, Musci M (2011) Characterization of the volatile profile of thistle honey using headspace solid-phase microextraction and gas chromatography–mass spectrometry. Food Chem 129:1030–1036. https://doi.org/10.1016/j.foodchem.2011.05.070
Brockmann A, Brückner D, Crewe RM (1998) The EAG response spectra of workers and drones to queen honeybee mandibular gland components: the evolution of a social signal. Naturwissenschaften 85:283–285. https://doi.org/10.1007/s001140050500
Chapurlat E, Ågren J, Anderson J, Friberg M, Sletvold N (2019) Conflicting selection on floral scent emission in the orchid Gymnadenia conopsea. New phytologist 222:2009–2022. https://doi.org/10.1111/nph.15747
Chittka L, Raine NE (2006) Recognition of flowers by pollinators. Curr Opin Plant Biol 9:428–435. https://doi.org/10.1016/j.pbi.2006.05.002
Daisey JM, Hopke PK (1991) Potential for ion-induced nucleation of volatile organic compounds by radon decay in indoor environments. Aerosol Sci Tech 19:80–93. https://doi.org/10.1080/02786829308959623
Daumer K (1956) Reizmetrische Untersuchung des Farbensehens der Bienen. Z Vgl Physiol 38:413–478. https://doi.org/10.1007/BF00340456
Daumer K (1958) Blumenfarben, wie sie die Bienen sehen. Z Vgl Physiol 41:49–110. https://doi.org/10.1007/BF00340242
Eisenhardt D (2014) Molecular mechanisms underlying formation of long-term reward memories and extinction memories in the honeybee (Apis mellifera). Learn Memory 21:534–542. https://doi.org/10.1101/lm.033118.113
Farina WM, Gruter C, Acosta L, Mc CS (2007) Honeybees learn floral odors while receiving nectar from foragers within the hive. Naturwissenschaften 94:55–60. https://doi.org/10.1007/s00114-006-0157-3
Galán RF, Weidert M, Menzel R, Herz AV, Galizia CG (2014) Sensory memory for odors is encoded in spontaneous correlated activity between olfactory glomeruli. Neural Comput 18:10–25. https://doi.org/10.1162/089976606774841558
Galizia CG, Sachse S, Rappert A, Menzel R (1999) The glomerular code for odor representation is species specific in the honeybee Apis mellifera. Nat Neurosci 2:473–478. https://doi.org/10.1038/8144
Glaettli M, Barrett SC (2008) Pollinator responses to variation in floral display and flower size in dioecious Sagittaria latifolia (Alismataceae). New Phytol 179:1193–1201. https://doi.org/10.1111/j.1469-8137.2008.02532.x
He XJ, Zhang XC, Jiang WJ, Barron AB, Zhang JH, Zeng ZJ (2016) Starving honey bee (Apis mellifera) larvae signal pheromonally to worker bees. Sci Rep 6:22359. https://doi.org/10.1038/srep22359
Hourcade B, Perisse E, Devaud JM, Sandoz JC (2009) Long-term memory shapes the primary olfactory center of an insect brain. Learn Memory 16:607–615. https://doi.org/10.1101/lm.1445609
Iwashita G, Hibino T (2011) Assessment on odor intensity of bioeffluents by VOC concentration perceived air pollution caused by human bioeffluents (part 2). J Environ Eng AIJ 76:539–545. https://doi.org/10.3130/aije.76.539
Iwashita G, Tokunaga N (2012) Discussion on the index of body odor pollution based on VOC concentration in classrooms perceived air pollution caused by human Bioeffluents (part 3). J Environ Eng AIJ 77:65–70. https://doi.org/10.3130/aije.77.65
Jerkovi I, Tuberoso CIG, Marijanovi Z, Jeli M, Kasum A (2009) Headspace, volatile and semi-volatile patterns of Paliurus spina-christi unifloral honey as markers of botanical origin. Food Chem 112:239–245. https://doi.org/10.1016/j.foodchem.2008.05.080
Joerges J, Küttner A, Galizia CG, Menzel R (1997) Representations of odours and odour mixtures visualized in the honeybee brain. Nature 387:285–287. https://doi.org/10.1038/387285a0
Jorgensen U, Hansen M, Christensen LP, Jensen K, Kaack K (2000) Olfactory and quantitative analysis of aroma compounds in elder flower (Sambucus nigra L.) drink processed from five cultivars. J Agric Food Chem 48:2376–2383. https://doi.org/10.1021/jf000005f
Kaškonienė V, Ruočkuvienė G, Kaškonas P, Akuneca I, Maruška A (2015) Chemometric analysis of bee pollen based on volatile and phenolic compound compositions and antioxidant properties. Food Anal Method 8:1150–1163. https://doi.org/10.1007/s12161-014-9996-2
Kiester AR, Lande R, Schemske D (1984) Models of coevolution and speciation in plants and their pollinators. Am Nat 2:220–243. https://doi.org/10.1086/284265
Klein AM, Vaissiere BE, Cane JH, Steffan-Dewenter I, Cunningham SA, Kremen C, Tscharntke T (2007) Importance of pollinators in changing landscapes for world crops. Proc Biol Sci 274:303–313. https://doi.org/10.1098/rspb.2006.3721
Knauer AC, Schiestl FP (2015) Bees use honest floral signals as indicators of reward when visiting flowers. Ecol Lett 18:135–143. https://doi.org/10.1111/ele.12386
Komischke B, Giurfa M, Lachnit H, Malun D (2002) Successive olfactory reversal learning in honeybees. Learn Memory 9:122. https://doi.org/10.1101/lm.44602
Krishna S, Keasar T (2018) Morphological Complexity as a Floral Signal: From Perception by Insect Pollinators to Co-Evolutionary Implications. Int J Mol Sci 19:1681. https://doi.org/10.3390/ijms19061681
Laska M, Galizia CG, Giurfa M, Menzel R (1999) Olfactory discrimination ability and odor structure-activity relationships in honeybees. Chem Senses 24:429–438. https://doi.org/10.1093/chemse/24.4.429
Leonard AS, Dornhaus A, Papaj DR (2010) Flowers help bees cope with uncertainty: signal detection and the function of floral complexity. J Exp Biol 214:113–121. https://doi.org/10.1242/jeb.047407
Liu JH, Liu W, Qing ZX, Zeng ZY, Zhong XH, Zeng JG (2020) Analysis of volatile compositions in different flower parts of two varieties of Hemerocallis citrina Baroni by HS-SPME-GC-MS. Nat Prod Res Dev 32:269–377, 304 (in Chinese). https://doi.org/10.16333/j.1001-6880.2020.2.011
Luo C, Huang ZY, Li K, Chen X, Chen Y, Sun Y (2013) EAG responses of Apis cerana to floral compounds of a biodiesel plant, Jatropha curcas (Euphorbiaceae). J Econ Entomol 106:1653–1658. https://doi.org/10.1603/EC12458
Menzel R, Muller U (1995) Learning and memory in honeybees: from behavior to neural substrates. Annual Annu Rev Neurosci 19:379–404. https://doi.org/10.1146/annurev.ne.19.030196.002115
Moyroud E, Wenzel T, Middleton R, Rudall PJ, Banks H, Reed A, Mellers G, Killoran P, Westwood MM, Steiner U, Vignolini S, Glover BJ (2017) Disorder in convergent floral nanostructures enhances signalling to bees. Nature 550:469–474. https://doi.org/10.1038/nature24285
Müller U (2002) Learning in honeybees: from molecules to behaviour. Zoology 105:313–320. https://doi.org/10.1078/0944-2006-00075
Naef R, Jaquier A, Velluz A, Bachofen B (2004) From the linden flower to linden honey-volatile constituents of linden nectar, the extract of bee-stomach and ripe honey. Chem Biodivers 1:1870–1879. https://doi.org/10.1002/cbdv.200490143
Pauw A, Stofberg J, Waterman RJ (2009) Flies and flowers in Darwin’s race. Evolution 63:268–279. https://doi.org/10.1111/j.1558-5646.2008.00547.x
Rodriguez-Girones MA, Santamaria L (2005) Resource partitioning among flower visitors and evolution of nectar concealment in multi-species communities. Proc Biol Sci 272:187–192. https://doi.org/10.1098/rspb.2005.2936
Rodriguez-Girones MA, Santamaria L (2006) Models of optimum foraging and resource partitioning: deep corollas for long tongues. Behav Eco 17:905–910. https://doi.org/10.1093/beheco/arl024
Seeley TD (1985) Honeybee Ecology: A Study of Adaptation in Social Life. Princeton University Press, New Jersey
Schade F, Legge RL, Thompson JE (2001) Fragrance volatiles of developing and senescing carnation flowers. Phytochemistry 56:703–710. https://doi.org/10.1016/S0031-9422(00)00483-0
Scheiner R, Schnitt S, Erber J (2005) The functions of antennal mechanoreceptors and antennal joints in tactile discrimination of the honeybee (Apis mellifera L.). J Comp Physiol A Neuroethol 191:857–864. https://doi.org/10.1007/s00359-005-0009-1
Schiestl FP (2010) The evolution of floral scent and insect chemical communication. Ecol Lett 13:643–656. https://doi.org/10.1111/j.1461-0248.2010.01451.x
Twidle AM, Barker D, Seal AG, Fedrizzi B, Suckling DM (2018) Identification of Floral Volatiles and Pollinator Responses in Kiwifruit Cultivars, Actinidia chinensis var. chinensis. J Chem Ecol 44:406–415. https://doi.org/10.1007/s10886-018-0936-2
Uzun Y, Dalar A, Konczak L (2017) Sempervivum davisii: phytochemical composition, antioxidant and lipase-inhibitory activities. Pharm Biol 55:532–540. https://doi.org/10.1080/13880209.2016.1255979
Wright GA, Skinner BD, Smith BH (2002) Ability of honeybee, Apis mellifera, to detect and discriminate odors of varieties of canola (Brassica rapa and Brassica napus) and snapdragon flowers (Antirrhinum majus). J Chem Ecol 28:721–740. https://doi.org/10.1023/a:1015232608858
Wright GA, Carlton M, Smith BH (2009) A honeybee’s ability to learn, recognize, and discriminate odors depends upon odor sampling time and concentration. Behav Neurosci 123:36–43. https://doi.org/10.1037/a0014040
Wright GA, Schiestl FP (2009) The evolution of floral scent: the influence of olfactory learning by insect pollinators on the honest signalling of floral rewards. Funct Ecol 23:841–851. https://doi.org/10.1111/j.1365-2435.2009.01627.x
Wu Q, Ling J, Xu Y (2014) Starter culture selection for making Chinese sesame-flavored liquor based on microbial metabolic activity in mixed-culture fermentation. Appl Environ Microb 80:4450–4459. https://doi.org/10.1128/AEM.00905-14
Xu WL (1992) Nectar and pollen plants of China. Heilongjiang Science and Technology Press, Harbin
Zhang FP, Larson-Rabin Z, Li DZ, Wang H (2012) Colored nectar as an honest signal in plant-animal interactions. Plant Signal Behav 7:811–812. https://doi.org/10.4161/psb.20645
Acknowledgements
We thank Dr. Guangli Wang for suggesting on EAG experimental design, Dr. Shengliang Liao for help in GC-MS experiments, and Dr. Xinjian Xu for help in floral scent collection.
Funding
This work was supported by the National Natural Science Foundation of China (no. 31702193), Natural Science Foundation of Jiangxi province (no. 20171BAB214018), Key Research and Development Project of Jiangxi province (no. 20181BBF60019), and the Earmarked Fund for China Agriculture Research System (no. CARS-44-KXJ15).
Author information
Authors and Affiliations
Contributions
XJH and ZJZ designed the experiments; YBL and XJH conducted the experiments; YM, YZH, JFL, ZL, and WYY helped in performing the experiments; XJH, YBL, ABB, and ZJZ wrote and revised the paper.
Corresponding author
Ethics declarations
Ethics approval
The study was performed in accordance with the relevant legal requirements in China.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by John A. Byers
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Y.B., Zeng, Z.J., Barron, A.B. et al. The involvement of a floral scent in plant-honeybee interaction. Sci Nat 109, 30 (2022). https://doi.org/10.1007/s00114-022-01802-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-022-01802-z