Abstract
Quantitative data on the ecology of the main plant communities along an altitudinal gradient in northeastern Mexico were obtained with the aim of identifying the most important environmental variables that affect plant distribution and composition. The main threats to these communities were also investigated. Importance value index (IVi) of the 39 most important species and 16 environmental variables were recorded at 35 altitudinal gradients each spaced at intervals of at least 100-m altitude. Classification and ordination of vegetation showed six well-differentiated but overlapping plant communities: alpine meadow, cold conifer forest, mesic mixed forest, xeric scrub, Tamaulipan piedmont scrub, and halophytic grassland. Altitude, minimum and average temperatures, and organic matter content are the main variables affecting the plant distribution in northeastern Mexico. Urban growth, mechanized agriculture, and changes in land use are the main threats in the short and medium term to plant communities in this area. Climate change also seems to be having an impact at present or in the near future as shown by the presence of exotic shrubs from warmer areas in mesic and temperate areas inhabited by oak and oak-pine forest.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Identifying the factors controlling the distribution and diversity of species in ecological communities is one of the main issues in community ecology (Ohmann and Spies 1998). Environmental heterogeneity significantly influences the structure of ecological communities (Vivian-Smith 1997). Plant life forms and structure of the vegetation, therefore, reflect adaptations to multifactorial environments (Körner 1994), such as latitude (Willig et al. 2003), altitude (Akira et al. 2013), physiography (Encina-Domínguez et al. 2013), soil (Abella and Covington 2006), pH (Huerta-Martínez et al. 2004), soil texture (Burton et al. 2011), and temperature (Trivedi et al. 2008).
Over the last few decades, spatial scale (local or regional) changes in plant communities along physical environmental gradients have been used to quantify and predict how local or regional environments affect plant diversity (Willis and Whittaker 2002) and ecological relationships (Ohmann and Spies 1998). Mexico has one of the most rugged reliefs on the planet, less than 35 % of its surface has an altitude of less than 500 m, and more than half of its territory lies at altitudes of 1000 m (Rzedowski 1978). Mexico, along with Peru and India, is home to almost all the natural vegetal communities recognized by the world (Rzedowski 1978). The northeastern part of Mexico is no exception; it shows varied, contrasting relief and altitude levels (INEGI 1986a), ranging from 0 (low plains) to 3650 m (high mountains) (CONABIO 1997). Such contrasts in relief and climate have led to the formation of different environments characterized by particular climates (INEGI 1986a), soil types (Meyer et al. 1992), plant communities (González-Medrano 2003), and species diversity (Villarreal-Quintanilla and Estrada-Castillón 2008; Estrada-Castillón et al. 2012).
In spite of its rich ecosystem diversity and heterogeneous physical environments, no detailed studies have been published on a regional scale of plant-environment relationships in northeastern Mexico. Most quantitative studies published so far have been limited to ecology and classification of certain scrublands, grasslands and forest communities, or some species of regional ecological importance (González-Medrano 2003; Villarreal-Quintanilla and Estrada-Castillón 2008; Estrada-Castillón et al. 2012). However, none of these studies have included physical and biotic variables together with multivariate analysis to examine the influence of regional environmental gradients, which affect the structure, composition, distribution, and diversity of species. Therefore, this study carried out a quantitative analysis of the main plant communities along an altitudinal gradient spanning various types of scrubs, oak forest, mixed forest, pine forest, alpine meadow, and halophytic grassland to explore the influence of physical environmental factors on these communities.
The objective of the study was to identify the environmental factors associated with plant composition on a regional and local scale. Our hypothesis is that altitude, temperature, and precipitation are the most significant environmental factors that structure species composition, while soil type is a local second-order factor.
If climate variables are revealed as the most important determinants in species composition, this means that these plant communities will be susceptible to intense changes in the future due to predicted global warming (Christensen et al. 2007).
Material and methods
Study site
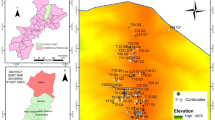
The study area comprised the westernmost region and the central portion of the states of Tamaulipas and Nuevo Leon, respectively (Fig. 1), comprising three heterogeneous physiographic provinces, the Gulf Coastal Plain, the Sierra Madre Oriental, and the Great Plains of North America (INEGI 1986a), characterized by low and high plains and high peaks, ranging in altitude from 250 to 3650 m (CONABIO 1997; Arriaga et al. 2000). The geology of the plains is mainly composed of sedimentary rocks of marine origin belonging to Mesozoic clastic deposits (INEGI 1986a); the high plains, highest peaks, and intermountain valleys consist essentially of litosol, yermosol, and gypsic soils (INEGI 1986a). There are two predominant types of climate, arid, BS (low and high plains), and temperate, C (mountains) (García 1973; INEGI 1986b).
Location of the 61 sites sampled (black dots, sampled between May and November 2013–2014); they include Tamaulipan piedmont scrubs from lower plains and lower hills (right side), mixed mesic forest (central), xeric scrubland and halophytic grasslands on higher plains (left side, lower), and cold conifer forest chaparral on the highest peaks (left side upper). ArcGis was used to create the map
The Cerro El Potosí, the tallest peak (3650 m) in northern Mexico, climatically has been included in type C (García 1973; INEGI 1986b), in the semicold-subhumid type. However, from 2800 to 3650 m in altitude, the mean temperatures range from −2 to 5 °C and the Cerro El Potosi minimum temperatures range from −5 to −11 °C much lower than the ones occurring in the oak-pine forest (below these altitudes), which are recognized as the cold climate type described by García (1973). Annual rainfall varies with altitude from 400 to 700 mm on low and high plains and from 600 to 800 mm in the mountains. The annual average temperatures on the low plains range from 20 to 23 °C; in contrast, in mountain areas, they range from 12 to 18 °C (INEGI 1986a, b; García 1973). Ten main vegetation types have been recorded along the longitudinal and altitudinal gradient (Estrada-Castillón et al. 2010, 2012) which are the following: Tamaulipan thornscrub composed mainly of legume thorny species; piedmont scrub, very similar to Tamaulipan thornscrub, but differing in tall plant composition and physiognomy, predominating non-thorny species of genera such as Fraxinus, Helietta, and Forestiera along with thorny species Acacia and Zanthoxylum; oak forest (Quercus spp.); mixed forest (oak-pine, pine-oak forest); xeric scrubland, mainly composed of Larrea tridentata and rosetophyllous genera such as Agave, Nolina, and Yucca; oak scrubland (dwarf oak scrubland or chaparral); halophytic grasslands, with Muhlenbergia, Frankenia, Nerysirenia, and Euphorbia as the dominant genera; pine forest (Pinus spp.); conifer forest (Abies, Pseudotsuga, Juniperus, and Cupressus); and alpine meadow, composed mainly of herbaceous species and including Pinus culminicola and Juniperus zanonii, also called Pinus scrubland, the only Pinus bush existing in Mexico.
Field survey
Vegetation and land use layer series IV (INEGI 2011) was used to identify the different plant communities. In order to represent the total environmental and physiographic variation, 35 altitudinal (from 300 to 3600 m) belts, which included all plant communities and their main plant species, each spaced at intervals of at least 100-m altitude were set. Within each belt, areas with no or low perceptive disturbance were selected and randomly sampled. In each belt, five (20 × 20 m) sampling plots were set within 30-m horizontal distance of each other in order to avoid border effect with the contiguous plots. Seventeen environmental factors were selected for the study: elevation, exposed bedrock, bare soil, litter thickness layer, rockiness, slope, aspect, soil depth, pH, organic matter content, sand, silt, clay, average annual precipitation, maxim annual temperature, mean annual temperature, and minimum annual temperature.
Plot position and elevation were measured using a global position system (GPS; Etrex, Garmin Ltd., Olathe, KS). Topographic factors measured were slope inclination, obtained by clinometer; exposed bedrock (%); litter thickness layer (%); and rockiness (% of rocks between 2 (cobble) and 12 (boulders) cm in diameter were considered). According to main gradient (eastern or western), only E (1) and W (0) aspects were included; thus, when several plots lay on NE, ENE, SE, and ESE, these were included in the E aspect and those NW, WNW, SW, and WSW were included in the W aspect (using a compass). Climatic factors considered were average annual precipitation, maximum annual temperature, mean annual temperature, and minimum annual temperature; the quantitative information was taken from database records from CONAGUA (2014).
Chemical and physical factors were based on three of the five plots sampled (extremes and central plot); soil samples of about 1 kg were taken from 0 to 30 cm depth; they were mixed, dried, and sieved through a 2 mm sieve; finally, debris and stones were eliminated from the soil. Texture (sand, clay silt) was determined by bouyoucos hydrometer; organic matter content was determined by the ignition technique, and pH was measured in a soil-water ratio of 1:5 extract.
The biotic information was an average value of the five plots for each species, since the main aim was to describe the gradient community with respect to environmental variables.
An average value for the aerial cover and density for each species of the five plots at each altitudinal belt was calculated. For each individual species, the aerial cover (AC) was calculated using Mueller-Dumbois and Ellenberg (1974) for each plot (from 1 to 5). Density (D) was calculated by counting all the individuals of all species in each of the five plots. The frequency (F) was calculated according to the number of altitude gradients where each species occurred (35 altitudinal belts). Density, aerial cover, and frequency data of each species were transformed into their relative values (IVi = RD + RAC + RF) (Mueller-Dumbois and Ellenberg 1974; Brower et al. 1997), and an IVi average for the species in the five plots at each altitude gradient was calculated (as a standardized value from 0 to 100).
Statistical analysis
The classification was based on the IVi values of the species at the different altitudinal gradients represented by the average values of the five plots. The vegetation assemblages were classified using WinTWINS Cluster Analysis (TWINSPAN for Windows) version 2.3 (Hill and Šmilauer P 2005). This is a tool to classify samples and can be used to obtain a classification of the species according to their ecological preferences. Both classifications were used to obtain an ordered two-way table, which shows species synecological relations (Hill and Šmilauer P 2005). These relations can be used as ecological indicators of community assemblages, environmental changes, and habitat conditions. A dendrogram with characteristic or indicator species was constructed based on eigenvalues and class ranges according to five cutoff levels: 0, 2, 5, 10, and 20, with similar weighting to avoid the influence of rare species. Owing to high heterogeneity in landscapes and physical variables along the altitudinal gradient, vegetation and plant diversity changed dramatically, even in adjacent sampled sites, leading to the appearance of several unclear misclassifications. To characterize plant communities, we chose to select the most common species, those with IVi values higher than 4.5. These 39 final species were then used to classify and ordinate the 61 sites at each altitudinal belt with satisfactory ecological significance.
For ordination, CCA (CANOCO version 4.5 software (ter Braak and Šmilauer 2002) analysis was used to examine the distribution and relationship between species and edaphic and topographic factors (ter Braak 1986, 1987). The analysis was based on the IVi values and environmental variables, which were log transformed before statistical analysis to ensure homogeneity of variance (Lepš and Šmilauer 2003).
To test the significance of the eigenvalue of the first axis and to select the environmental variables explaining species composition, manual forward selection through a Monte Carlo permutation test with 499 permutations (P < 0.05 significance) was performed (Blanchet et al. 2008 warn that some unimportant variables can be selected when there is a very large set of variable, what it is not our case). The manual forward selection was performed by selecting the variables that explained the most inertia (ordered by CANOCO) of the total inertia of the CCA axis 1. This was once their individual significance that had been tested in the total inertia (Lepš and Šmilauer 2003). Although we do not include spatial autocorrelation in our analyses, some studies suggests that in linear models, the effect of the environmental variables can be reverse (Kühn 2007). However, we used a direct unimodal model (CCA), and also, we designed the sampling for this specific analysis. Because of that, we consider that results of these analyses will be consistent.
Results
The total flora recorded at the 61 sites sampled included 265 species, 168 genera, and 53 families of vascular plants. Asteraceae (19 genera, 33 species), Fabaceae (Leguminosae, 12, 16), Rosaceae (8, 8), Euphorbiaceae (6, 9), Cactaceae (5, 11), Agavaceae (4, 8), Pinaceae (3, 9), Rutaceae (3, 3), Verbenaceae (3, 3), Rhamnaceae (2, 2), Celastraceae (2, 2), and Anacardiaceae (2, 3) contain the highest diversity in both genera and species, respectively. Quercus (11 species), Pinus (7), Agave (6), Opuntia (5), and Juniperus (4) are the most diversified genera. The three main biological forms recorded at the sampled sites included herbaceous (10 %), shrubs (67 %), and trees (23 %). The 39 most important species according to their IVi value are shown in Table 1.
Based on TWINSPAN, six interpretable groups of plant communities were discriminated (Fig. 2). The plant communities were separated into three main groups: (a) scrublands, including the Tamaulipan piedmont scrub and the xeric scrub; (b) forest, including oak and conifer vegetation which includes mixed forest, cold conifer forest chaparral, and alpine meadow (pine bush vegetation); and (c) halophytic grasslands. As indicated by high eigenvalue (1.00), the first branch of the dendrogram shows that group VI (halophytic grasslands) differs notably from the rest. Clustering shows two evident gradients: (1) altitudinal (from low lands (360 m) to high peaks (2933 m to 3506 m) and (2) climatic, from low minimal temperatures (−12 to −6 °C) and mesic (average) temperatures (17 °C) to hot (average) temperatures (24 to 26 °C) (Table 2). The first division easily recognizes and separates two clusters based on structure and plant composition, scrublands and forests, and herbaceous grasslands. Second division segregates cold forest from mesic mixed forest and shrub vegetation, and the third division completes the separation between mesic mixed forest and scrublands (Fig. 2).
Canonical correspondence analysis (CCA) was performed for the 39 species on 61 sites and the 17 environmental variables to understand the composition of plant communities. Only 10 of the 17 initial environmental variables analyzed were selected by forward selection (P < 0.05); they were elevation, minimum temperature, average temperature, pH, soil depth, organic matter content, sand, rockiness, and aspect. The first two axes accounted for 7.7 and 14.9 %, respectively, of the total explained variation, and their eigenvalues were 0.917 and 0.856, respectively. Considering the correlations of environmental variables with axes, the first CCA axis was positively and strongly correlated with elevation (r = 0.9211) and negatively and strongly correlated with minimum temperature (r = −0.9590) and average temperature (r = −0.9367), suggesting a topographic-temperature gradient from low plains with high temperatures to high mountains with low temperatures. The second axis was positively correlated with pH (r = 0.6701) and soil depth (r = 0.5856) reflecting chemical and edaphic gradients, separating the arid areas with deep soils from mesic and cold areas with shallow soils (all these correlations for a P < 0.05). The test of significance for the first canonical axis was significant (P < 0.002).
The same six groups (61 sites) clustered by TWINSPAN were superimposed on the CCA along with the 10 significant environmental variables (Fig. 3). The plots of the same community are enclosed in a polygon. The first axis discriminated from mixed forest to alpine meadow with the climatic variables (MITEM, AVTEM, and AVPRE) being responsible for this gradient. There is a turnover of species from Acacia amentacea, Fraxinus greggii, Quercus laceyi, Pinus teocote, Pinus pseudostrobus, Quercus rysophylla to P. culminicola, Lupinus cacuminis, Hymenoxys insignis, and J. zanonii. Axis 2 discriminated halophytic grasslands from xeric scrub (more variable) based on pH and soil depth, with species L. tridentata and Yucca filifera in the xeric scrub and Euphorbia stictosphora, Frankenia gypsophila, and Nerisyrenia linearifolia from the halophytic grasslands.
Canonical correspondence analysis (CCA) of 61 sites (six groups clustered by TWINSPAN) and 39 species and the 10 most significant environmental variables. Polygons enclose the sites originated with TWINSPAN and selected in Fig. 2. Abbreviations of the species use the first four letters of the genus and the four first letters of the species name (see acronyms in the Table 1). ELEV elevation, ASPECT aspect, SAND % soil sand, pH pH, ROCK rockiness, SOIDEP soil depth, ORGMA organic matter, AVTEM average temperature, MITEM minimum temperature, AVPRE average precipitation. The two axes accounted for 7.7 and 14.9 %, respectively, of the total explained inertia of the data set, and their eigenvalues were 0.917 and 0.856, respectively. However, using the inertia restricted to the environmental variables, both axes explain more than 70 %, indicating that the distribution of species is explained by the used environmental variables in this percentage
The clustering analysis identified six well-differentiated, but without discrete limits, plant communities: Tamaulipan piedmont scrub, mixed forest, xeric scrub, halophytic grasslands, cold conifer forest chaparral, and alpine meadow. Except for halophytic grasslands, the rest of the plant communities share species with the adjacent ones as the environmental gradient changes, thus constituting transition zones.
Discussion
Most sites showed a clear distribution according to altitude, temperature, and precipitation vectors, as revealed by the CCA analysis (Fig. 1). Axis 1 explains, from right to left, not only the distribution of the main plant communities in most ecosystems of northeastern Mexico, where a strong altitudinal gradient from 360 to 1600 m exists, but also the main life forms associated with them. These are influenced mainly by three factors: altitude, temperature, and precipitation. There are scrublands, inhabiting low altitudes with warmer conditions; mixed forest, located at medium altitudes with mesic weather conditions, and cold conifer forest, distributed at high altitudes with cold weather. Higher average temperatures and higher minimum temperatures are present at lower altitudes; both these variables were highly and positively correlated to the distribution of mixed forest (oak-pine) and Tamaulipan piedmont scrub (as can be seen along axis 1, while the pH variable was highly and positively correlated to axis 2). The variable separates the arid environments with alkaline soils, especially those on high plains dominated by xeric scrublands and halophytic grasslands from the mixed forest and most of alpine meadow and cold conifer forest chaparral sites, which have the most acidic soils. Our results support the hypothesis of the primary importance of elevation, temperature, and precipitation and the secondary role of soil type in controlling the distribution of regional plant communities. Species distribution across altitudinal gradients on a regional scale were closely associated with moisture and temperature variables or of zonal type; however, on a smaller scale, edaphic factors play a key role in the zonal distribution.
The three environmental variables: elevation, minimum temperature, and average temperature separated the alpine meadow and the cold conifer forest chaparral from the rest of the plant communities. Both sites are characterized by other significant variables, but ones such as organic matter content and aspect only play a minor role. Species that were strongly positively correlated on the right side of axis 1 included the ones inhabiting the highest elevations in northeastern Mexico such as Abies vejarii, J. zanonii, H. insignis, L. cacuminis, P. culminicola, Pinus flexilis, and Pinus hartwegii. These were located on northern, wet aspects with high organic matter contents.
Alpine meadow constitutes a small area of about 25–30 ha (García-Aranda et al. 1999; Jiménez et al. 2005) between 3600 and 3650 m altitude, dominated in shrub strata by P. culminicola and J. zanonii on the summit of Cerro El Potosí, which is the only 0.4–2.5 m tall pine bush in Mexico. Adjacent to and immediately below it, the cold conifer forest chaparral constitutes the main vegetation, an association of A. vejarii, P. hartwegii, and P. flexilis in the upper strata and a dense 1–1.5 m tall shrub community composed almost entirely of Quercus greggii and scattered individuals of Ceanothus fendleri, inhabiting shallow and rocky soils. These different growth forms at different elevation gradients have been reported by Whittaker (1956, 1960), representing different plant growth forms over gradient changes. Elevation influences precipitation and temperature, and these directly affect plant growth (Aguado-Santacruz and García-Moya 1998). Ordination indicates that cold conifer forest chaparral and alpine meadow are influenced mainly by elevation and, to a minor degree, by aspect and rockiness. Similar results have been reported for this vegetation from central Mexico, where Pinus and Abies forest are the dominant elements at high altitudes in the Trans volcanic belt in the south of Mexico (Nieto de Pascual 1995). The dwarf oak (Q. greggii) vegetation associated with cold forest has also been recorded in the south of Mexico (Valiente-Banuet et al. 1998), inhabiting intermediate areas between arid vegetation and subhumid forest (Rzedowski 1978). This vegetation exists partly not only due to arid conditions but also as a consequence of cyclic fires on the summit of this area (Jiménez et al. 2005). Several studies of mountains in the USA have also found that humidity and temperature are strongly correlated to altitude in controlling plant communities’ distribution along with other variables such as sun radiation, soil depth, organic matter content, bare soil, fire, pH (Allen and Peet 1990; Barton 1993), slope, and aspect (Birkland 1984).
The mixed mesic forest, which occurs at medium and low altitudes, is also strongly and highly positively correlated with climatic variables, temperature, and precipitation. Mixed forests (oak-pine/pine-oak) are the main plants associated with the Mexican mountains (Rzedowski 1978), occupying 21 % of their surface, harboring almost 7000 plant species, with around 70 % of them being endemic (Rzedowski 1991). These forests are also the most important in terms of economic value (Challenger 2003).
Tamaulipan piedmont scrub is distributed at lower elevations with warmer conditions and higher precipitation. The dominant species in these scrublands include two common legumes (Estrada-Castillón et al. 2014): A. amentacea and Havardia pallens associated with F. greggii. Some species found in this plant community such as Rhus virens, Malacomeles denticulata, Agave lecheguilla, and Dasylirion berlandieri are generally widespread at the other altitudinal gradients and in other plant communities (Villarreal-Quintanilla and Estrada-Castillón 2008). In favorable wet conditions, piedmont scrub can reach 1800 m (Rzedowski 1978); hence, sometimes, some of its species form ecotones with oak and mixed forests, mainly with Quercus polymorpha, Q. laceyi, and Quercus canbyi (Estrada-Castillón et al. 2012). Piedmont scrub is composed of almost 185 species, dominated by thorny, non-thorny, and succulent species of Neotropical origin (Estrada-Castillón et al. 2012).
Tamaulipan thorn scrub belongs to the Mexican xerophytic region (Rzedowski 1978) and along with piedmont scrub has been described as matorral mediano subinerme (semithorn medium scrubland) in the northern region of northeastern Mexico (Briones and Villarreal 2001). It is mainly composed of Leucophyllum frutescens-A. amentacea community. However, the variability of environmental factors where it inhabits, and the species associated with it results in different physiognomic types, though it is always distributed on hills, plateaus, and valleys in alluvial fans of the area, associated with warm and humid conditions (Briones and Villarreal 2001), and positively related to shallow soils, steeper slopes, and sedimentary substrate (Huerta-Martínez et al. 2004). In northeastern Mexico, piedmont scrub follows the same direction (NE-SW) as the mountains in a narrow belt about 270 km long (Estrada-Castillón et al. 2012). Its southern distribution limit reaches San Luis Potosí (Giménez-de Azcárate and González-Costilla 2011). The main species recorded along this belt of vegetation are A. amentacea, Acacia berlandieri, A. lecheguilla, Bernardia myricifolia, Cordia boissieri, Diospyros texana, F. greggii, Gochnatia hypoleuca, H. pallens, Helietta parvifolia, L. frutescens, Neopringlea integrifolia, and Zanthoxylum fagara (Rzedowski 1978; Briones and Villarreal 2001; Giménez-de Azcárate and González-Costilla 2011). Both Tamaulipan thorn and piedmont scrub communities mainly not only develop on silty clay but are also associated with other factors such as rough topography, microenvironments, deep soil, and slope (Estrada-Castillón et al. 2012; Salinas-Rodríguez et al. 2013). Piedmont scrub is distributed between 100 and 2200 m altitude, and as an ecotonal community between xeric scrublands and temperate forests, it has also been recorded in the semiarid region of Querétaro (Martorell and Ezcurra 2002).
Halophytic grasslands are characterized by E. stictosphora, F. gypsophila, Heteropogon contortus, Lepidium montanum, Muhlenbergia villiflora, and N. linearifolia, including two common shrubby species of the xeric scrubland, L. tridentata and Yucca carnerosana, and two conifer forest species adapted to dry conditions and calcareous soils, Juniperus angosturana (Eckenwalder 2009) and Pinus cembroides (Rzedowski 1978; Romero-Manzanares et al. 2012). Halophytic grasslands and xeric scrub are found at intermediate altitudes, 1400 to 1600 m; they are restricted to deeper soils with lower pH than the other plant communities and are negatively correlated to organic matter contents. L. tridentata and Y. carnerosana were also highly positively correlated on axis 2, showing high scores and also determined by pH and soil deep vectors. However, the role of pH and soil depth in explaining the other xeric scrub species declines as sand content increases, clustering a small group of species such as J. angosturana, Lindleya mespilioides, Mortonia greggii, P. cembroides, and Quercus sideroxyla associated with deep soils with higher sand content. Halophytic grasslands are distributed in patches in northeastern Mexico (Scott-Morales et al. 2004) and are commonly surrounded by xeric scrublands (L. tridentata) on high plains and by conifer forest (J. angosturana) in high mountains (Estrada-Castillón et al. 2010). Studies in the Chihuahuan Desert in Mexico have documented the influence of elevation, slope angle, rockiness, and edaphic variables on plant distribution in these arid environments; in fact, L. tridentata and Y. filifera correlate positively to soil depth, pH, and salt contents (Huerta-Martínez et al. 2004; Henrickson 1974; Henrickson and Johnston 1997).
Plant communities in northeastern Mexico are under high anthropogenic impact; population growth has mainly affected Tamaulipan and piedmont scrubs. Large areas of these plant communities are removed year after year because of urban development despite them being home to a rich and specialized flora not found in other areas, such as the Agave albopilosa, Agave bracteosa, Agave victoriae-reginae, and Myrospermum sousanum (Estrada-Castillón et al. 2012). Furthermore, in spite of the wide distribution of oak forest in the mountains in northeastern Mexico, this forest has also been highly impacted by change in land use, often being replaced by temporary crop areas (mainly corn) and subsequently abandoned, as well as being affected by excessive logging by illegal loggers. This has allowed the invasion of exotic plants such as Melia azederach and Ligustrum lucidum in protected areas such as the Cumbres de Monterrey National Park (pers. obs.).
Cold conifer forest chaparral is also threatened by illegal logging and by recurrent fires. The alpine meadow has lost almost 60 % of its surface due to fires, and this loss is worsened by browsing and trampling by livestock. However, the most dramatic loss of an ecosystem has occurred in the plant community with the least surface area (almost 400 km2) in northeastern Mexico, the halophytic grasslands, which has lost nearly 80 % of its area in the last 50 years due to mechanized agriculture (potatoes and alfalfa). This loss has affected the habitat of the endemic prairie dog (Cynomys mexicanus) (Scott-Morales et al. 2004) and 6 % of the grasslands’ endemic flora (Estrada-Castillón et al. 2010). Hence, the halophytic grasslands are the most vulnerable plant communities to human impact in northeastern Mexico.
Increases in minimum temperatures have been shown to be an important variable to discriminate plant communities. In other studies related to altitudinal gradients, minimum temperatures also appear as good predictors of the displacement of trees or other species lines at higher altitudes (Dukes and Mooney 1999). We expected that these temperature variations (minimum) would play a more significant role in the changes in species composition and richness over the coming decades. We consider that temperature variations, as long as they follow intergovernmental panel on climate change (IPCC) predictions, will become an important driver of change in plant communities in the future. In fact, climatic changes predicted by the IPCC are likely to lead to significant changes in species forest composition. Even the moderate scenarios of the IPCC predict a severe decrease in precipitations and a rise of 3–4 °C in average temperatures (Christensen et al. 2007). Specifically, modifications in rainfall and runoff and a replacement of semiarid vegetation by arid vegetation are expected in northern Mexico due to the effects of land use and climatic change (Bates et al. 2008).
The state of Nuevo León has 23 state natural protected areas, one national monument, one urban park, and three natural protected areas (Secretaría de Desarrollo Urbano y Obras Públicas 2000), which in total comprise 150,931 ha. All plant communities studied in this work are included in varying degrees within these protected areas, which, in principle, could mitigate the impact and preserve the presence of these valuable natural resources. This study confirms not only the importance of the diversity of ecosystems along altitudinal gradients on a regional scale but also the importance of the soil composition as the main factor in defining unique plant communities. However, the main threat facing all plant communities is their continuing depletion by human intervention, thus making the protection of these communities even more urgent.
References
Abella SR, Covington WW (2006) Vegetation environment relationships and eco-logical species groups of an Arizona Pinus ponderosa landscape. Plant Ecol 185:225–268
Aguado-Santacruz GA, García-Moya E (1998) Environmental factors and community dynamics at the southernmost part of the North American Graminetum. Plant Ecol 135:13–29
Akira SM, Shiono T, Koide D, Kitagawa R, Ota AT, Mizumachi E (2013) Community assembly processes shape an altitudinal gradient of forest biodiversity. Glob Ecol Biogeogr 22:878–888
Allen RB, Peet PK (1990) Gradient analysis of forests of the Sangre de Cristo Range, Colorado. Can J Bot 68:193–201
Arriaga L, Espinoza JM, Aguilar C, Martínez E, Gómez L (2000) Regiones terrestres prioritarias. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México
Barton AM (1993) Factors controlling plant distributions: drought, competition, and fire in montane pines in Arizona. Ecol Monogr 63:367–397
Bates BC, Kundzewicz ZW, Wu S, Palutikof JP (2008) Climate change and water. Technical paper of the intergovernmental panel on climate change. IPCC Secretariat, Geneva
Birkland PW (1984) Soils and geomorphology. Oxford University Press, Oxford
Blanchet FG, Legendre P, Borcard D (2008) Forward selection of explanatory variables. Ecology 89:2623–2632
Briones O, Villarreal JA (2001) Vegetación y flora de un ecotono entre las provincias del Altiplano y la Planicie Costera del noreste de México. Acta Bot Mex 55:39–67
Brower JE, Zar JH, von Ende CN (1997) Field and laboratory methods for general ecology. The McGraw-Hill Companies, Massachusetts
Burton J, Mladenoff DJ, Clayton MK, Forrester JA (2011) The roles of environmental filtering and colonization in the fine-scale spatial patterning of ground-layer plant communities in north temperate deciduous forests. J Ecol 99:764–776
Challenger A (2003) Conceptos generales acerca de los ecosistemas templados de montaña de México y su estado de conservación. In: Sánchez O, Vega E, Peters E, Monroy-Vilchis O (eds) Conservación de ecosistemas templados de montaña en México. Instituto Nacional de Ecología, México, pp 7–42
Christensen JH, Hewitson B, Busuioc A, Chen A, Gao X, Held I, Jones R, Kolli RK, Kwon WT, Laprise R, Magaña-Rueda V, Mearns L, Menéndez CG, Räisänen J, Rinke A, Sarr A, Whetton P (2007) Regional climate projections. In: Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Climate change 2007: the physical science basis, Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Comisión Nacional para el Conocimiento y Uso de la Biodiversidad (CONABIO) (1997) Provincias biogeográficas de México. Escala 1:4 000 000. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México
CONAGUA (2014) Bases de datos de la CONAGUA/SIGA. Subgerencia de información geográfica del agua. www.siga.cna.gob.mx (consulted March 2014). Accessed May 2014
Dukes JS, Mooney HA (1999) Does global change increase the success of biological invaders? Trends Ecol Evol 99:135–139
Eckenwalder JE (2009) Conifers of the world. Oregon. Timber Press, Portland
Encina-Domínguez JA, Meave JA, Zárate-Lupercio A (2013) Structure and woody species diversity of the Dasylirion cedrosanum (Nolinaceae) rosette scrub of central and southern Coahuila State, Mexico. Bot Sci 91:335–347
Estrada-Castillón E, Scott-Morales L, Villarreal-Quintanilla J, Jurado-Ybarra E, Cotera-Correa M, Cantú-Ayala C, García-Pérez J (2010) Clasificación de los pastizales halófilos del noreste de México asociados con perrito de las praderas (Cynomys mexicanus): diversidad y endemismo de especies. Rev Mex Biodivers 81:401–416
Estrada-Castillón E, Villarreal-Quintanilla JA, Jurado-Ybarra E, Cantú-Ayala C, García-Aranda MA, Sánchez-Salas J, Jiménez-Pérez J, Pando-Moreno M (2012) Clasificación, estructura y diversidad del matorral submontano adyacente a la planicie costera del Golfo Norte en el Noreste de México. Bot Sci 90:37–52
Estrada-Castillón E, Delgado Salinas A, Villarreal Quintanilla JA (2014) Leguminosas de Nuevo León. Instituto de Biología, Universidad Nacional Autónoma de México, México
García E (1973) Modificaciones al sistema de clasificación climática de Köppen, 2ath edn. Instituto de Geografía. Universidad Nacional Autónoma de México, México
García-Aranda M, Treviño-Garza EJ, Cantú-Ayala CM, González-Saldívar FN (1999) Zonificación ecológica del cerro “El Potosí”, Galeana, Nuevo León, México. Investig Geogr 38:31–40
Giménez-de Azcárate J, González-Costilla O (2011) Pisos de vegetación de la Sierra de Catorce y territorios circundantes (San Luís Potosí, México). Acta Bot Mex 94:91–123
González-Medrano F (2003) Las comunidades vegetales de México. Propuesta para la unificación de la clasificación y nomenclatura de la vegetación de México. INE, Semarnat, México
Henrickson J (1974) Saline habitats and halophytic vegetation of the Chihuahuan Desert region. In: Wauer RH, Riskind DH (eds) Transactions of the symposium of the biological resources of the Chihuahuan Desert region. United States and Mexico, vol 3, U S. Department of Interior, National Park Service Transaction and Proceeding Series. U. S. Government Printing Office, Washington DC, pp 289–314
Henrickson J, Johnston MC (1997) A flora of the Chihuahuan Desert region, vol 2, 12th edn. Henrickson J, Los Angeles
Hill MO, Šmilauer P (2005) TWINSPAN for Windows version 2.3. Centre for Ecology and Hydrology & University of South Boehmia, Huntingdon and Ceske Budejovice
Huerta-Martínez FM, Vázquez-García JA, García-Moya E, López-Mata L, Vaquera-Huerta H (2004) Vegetation ordination at the southern Chihuahuan Desert (San Luis Potosi, Mexico). Plant Ecol 174:79–87
INEGI (1986a) Síntesis geográfica de Nuevo León. Secretaría de Programación y Presupuesto, México
INEGI (1986b) Carta de climas, Monterrey, escala 1: 1,000 000. Secretaría de Programación y Presupuesto, México
INEGI (2011) Carta de uso del suelo y vegetación, escala 1:250,000, vol V. Instituto Nacional de Estadística y Geografía, México
Jiménez J, Jurado E, Aguirre O, Estrada E (2005) Effect of grazing on restoration of endemic dwarf pine (Pinus culminicola Andresen et Beaman) populations in northeastern Mexico. Restor Ecol 13:103–107
Körner C (1994) Scaling from species to vegetation: the usefulness of functional groups. In: Schulze E, Mooney HA (eds) Biodiversity and ecosystem function. Springer, Berlin, pp 117–140
Kühn I (2007) Incorporating spatial autocorrelation may invert observed patterns. Divers Distrib 13:66–69
Lepš L, Šmilauer P (2003) multivariate analysis of ecological data using CANOCO. Cambridge University Press, New York
Martorell C, Ezcurra E (2002) Rosette scrub occurrence and fog availability in arid mountains of Mexico. J Veg Sci 13:651–662
Meyer SE, García-Moya E, Lagunes-Espinoza L (1992) Topographic and soil surfaces effects on gypsophile plant community patterns in central Mexico. J Veg Sci 3:429–438
Mueller-Dumbois D, Ellenberg H (1974) Aims and methods of vegetation ecology. Wiley, New York
Nieto de Pascual PC (1995) Estudio sinecológico del bosque de oyamel de la cañada de Contreras, Distrito Federal. Rev Cien For Méx 20:3–34
Ohmann JL, Spies TA (1998) Regional gradient analysis and spatial pattern of woody plant communities of Oregon forests. Ecol Monogr 68:151–182
Romero-Manzanares A, Flores-Flores JL, Luna-Cavazos L, García-Moya E (2012) Effect of slope and aspect on the associated flora of pinyon pines in central Mexico. Southwest Nat 57:452–456
Rzedowski J (1978) La vegetación de México. Limusa, México
Rzedowski J (1991) Diversidad y orígenes de la flora fanerogámica de México. Acta Bot Mex 14:3–21
Salinas-Rodríguez M, Estrada-Castillón E, Villarreal-Quintanilla JA (2013) Flora and phytogeography of the Cañón de Iturbide, Nuevo León, México. J Bot Res Inst Texas 7:803–819
Scott-Morales L, Estrada-Castillón E, Chávez-Ramírez F, Cotera M (2004) Continued decline in geographic distribution of the Mexican prairie dog (Cynomys mexicanus). J Mammal 85:1095–1101
Secretaría de Desarrollo Urbano y Obras Públicas (2000) Áreas Naturales para la Conservación Ecológica en el Estado de Nuevo León. Secretaría de Desarrollo Urbano y Obras Públicas. Subsecretaria de Ecología, Monterrey
ter Braak CJE (1986) Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67:1167–1179
ter Braak CJE (1987) The analysis of vegetation-environment relationships by canonical correspondence analysis. Vegetatio 69:69–77
ter Braak CJF, Šmilauer P (2002) CANOCO reference manual and CanoDraw for windows user’s guide: software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca
Trivedi MR, Berry PM, Morecroftz MD, Dawson TP (2008) Spatial scale affects bioclimate model projections of climate change impacts on mountain plants. Glob Chang Biol 14:1089–1103
Valiente-Banuet A, Flores-Hernández N, Verdú M, Dávila P (1998) The chaparral vegetation in Mexico under nonmediterranean climate: the convergence and Madrean-Tethyan hypothesis reconsidered. Am J Bot 85:1398–1408
Villarreal-Quintanilla JA, Estrada-Castillón E (2008) Flora de Nuevo León. Listados Florísticos de México XXIV. Instituto de Biología, Universidad Nacional Autónoma de México, México
Vivian-Smith G (1997) Microtopographic heterogeneity and floristic diversity in experimental wetland communities. J Ecol 85:71–82
Whittaker RH (1956) Vegetation of the great smoky mountains. Ecol Monogr 26:1–80
Whittaker RH (1960) Vegetation of the Siskiyou Mountains, Oregon and California. Ecol Monogr 30:279–338
Willig MR, Kaufman DM, Stevens RD (2003) Latitudinal gradients of biodiversity: pattern, process, scale, and synthesis. Annu Rev Ecol Evol Syst 34:273–309
Willis KJ, Whittaker RJ (2002) Species diversity scale matters. Science 295:1245–1248
Acknowledgments
The authors thank Carmen Yen and Josué Estrada for field assistance and the Programa Interinstitucional para el Fomento a la Investigación (PIFI) of the Autonomous University of Nuevo León for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Beverley Glover
Rights and permissions
About this article
Cite this article
Castillón, E.E., Arévalo, J.R., Quintanilla, J.Á.V. et al. Classification and ordination of main plant communities along an altitudinal gradient in the arid and temperate climates of northeastern Mexico. Sci Nat 102, 59 (2015). https://doi.org/10.1007/s00114-015-1306-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-015-1306-3