Abstract
IL-17A and IL-17F share the highest sequence homology of the IL-17 family and signal via the same IL-17RA/RC receptor heterodimer. To better explore the expression of these two cytokines, we used a double reporter mouse strain (IL-17DR mice), where IL-17A expressing cells are marked by enhanced green fluorescent protein (eGFP) while red fluorescence protein (RFP) reports the expression of IL-17F. In steady state, we found that Th17 and γδ T cells only expressed IL-17A, while IL-17F expression was restricted to CD8 T cells (Tc17) and innate lymphoid cells (ILC type 3) of the gut. In experimental autoimmune encephalomyelitis, the vast majority of CNS-infiltrating Th17 cells expressed IL-17A but not IL-17F. In contrast, anti-CD3-induced, TGF-β-driven Th17 cells in the gut expressed both of these IL-17 cytokines. In line with this, in vitro differentiation of Th17 cells in the presence of IL-1β led primarily to IL-17A expressing T cells, while TGF-β induced IL-17F co-expressing Th17 cells. Our results suggest that expression of IL-17F is associated with non-pathogenic T cells, pointing to a differential function of IL-17A versus IL-17F.
Key messages
-
Naïve mice: CD4+ T cells and γδ T cells express IL-17A, and Tc17 cells express IL-17F. Gut ILC3 show differential expression of IL17A and F.
-
Th17 differentiation with TGF-β1 induces IL-17A and F, whereas IL-1β induced cells expressing IL-17A.
-
Th17 cells in EAE in CNS express IL-17A only.
-
Gut Th17 cells induced by anti-CD3 express IL-17A and F together as skin γδ T cells of IMQ-treated mice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The IL-17 cytokine family consists of the six members (IL-17A-F) [1, 2]. Out of those six cytokines, IL-17A was identified first after being cloned from a murine cytotoxic T cell hybridoma cell line. Therefore, it was initially termed cytotoxic T-lymphocyte antigen-8 (CTLA-8) [3]. The individual family members share homology in their amino acid sequence to IL-17A, with IL-17F showing with about 50% the highest similarity. Furthermore, in human and mice, the Il17f gene locus is in close proximity to Il17a, indicating that the different members of the IL-17 family of cytokines may originate from gene duplication.
Since the initial discovery of IL-17A, it became clear that this cytokine is mainly expressed by a distinct T helper cell subset, termed Th17 cells [4]. However, subsequently other cell types, or subpopulations of those, have been found to express IL-17A, including type 3 innate lymphoid cells (ILC3s) [5], natural killer T cells [6, 7], γδ T cells [8, 9], and cytotoxic CD8+ T (Tc17) cells [10, 11]. It is known that Th17 cell differentiation is induced in the presence of TGF-β and IL-6 or IL-21 [12,13,14], which induce the expression of the transcription factors RORγt and RORα [4, 15]. In contrast to naïve T helper cells, Th17 cells express the receptor for IL-23 (IL-23R), which is critical for their proper maintenance and expansion. Recently, it was shown that IL-1β also plays an important role in the expansion of Th17 cells in the context of neuroinflammation [16,17,18,19]. In the steady state, IL-17 is crucial for the host homeostatic relationship with commensal and pathogenic bacteria and fungi, which is achieved by inducing proinflammatory cytokines, chemokines, and antimicrobial peptides, especially at mucosal surfaces. However, a role for IL-17 in the development of different autoimmune diseases, including multiple sclerosis and psoriasis, was shown. Accordingly, different approaches were attempted to target either the differentiation of Th17 cells or to neutralize IL-17A and IL-17F [20].
Biological activity of IL-17A and IL-17F is mediated by binding to their cognate heterodimeric receptor complex consisting of IL-17RA and IL-17RC. So far, experiments analyzing the differential expression of these two cytokines were conducted either by studying mRNA levels or via flow cytometric analysis using antibodies specific to IL-17A or IL-17F [21,22,23,24]. Herein we used a genetic model to study the differential expression of these two related cytokines in steady state and in different disease models. To this end, we crossed IL-17A-IRES-enhanced green fluorescent protein (eGFP) mice [25] to IL-17F-IRES-RFP [26] mice to generate IL-17AF double reporter mice (IL-17DR). By using this new tool, we found that Th17 cells and γδ T cells in naïve mice primarily express IL-17A. Polarization of Th17 cells under different in vitro conditions and in vivo inflammation models point towards a TGF-β-driven expression of IL-17F and an IL-1β-driven expression of IL-17A.
Results
IL-17A is often considered to be co-expressed with IL-17F [27]. However, analysis of the expression of both cytokines was mostly performed by qRT-PCR or ELISA using bulk cell populations. Alternatively, intracellular staining with cell-permeabilizing conditions was used. The IL-17DR mice now enabled us to study the expression profile of IL-17A and IL-17F also on the single cell level with high accuracy using gentle surface marker staining conditions.
Expression of IL-17A and F in naïve mice
Analysis of naïve IL-17DR mice by flow cytometry showed that CD4+ T cells and γδ T cells in the spleen and lymph nodes (LNs) express IL-17A but very little IL-17F (Fig. 1a, b). Interestingly, when we analyzed CD8+ T cells, we observed only very few IL-17A expressing cells but a sizeable population of cells that was positive for IL-17F-RFP. Furthermore, co-expression of both cytokines was absent in these cells (Fig. 1a, b). ILC3s were shown to express IL-17 and to play an important role in the maintenance of gut homeostasis [28, 29]. Among ILC3s, IL-17 is selectively expressed by T-bet− CCR6+ lymphoid tissue inducer (LTi) cells [30]. However, in most studies, expression of RORγt is used to identify this specific ILC subset and little is known about the differential expression of IL-17A and IL-17F in them. We therefore isolated ILCs from the colon and small intestine of naïve mice. To specifically analyze ILC3, we used the surface markers CD127 and c-Kit to identify them among the lineage-negative cells [31]. These cells were further separated into CCR6lo/− and CCR6+ subsets. As previously described [28, 30], CCR6lo/− ILC3 did not express IL-17A or IL-17F (data not shown). We further did not observe differences in IL-17 production between CD4+ and CD4− LTi cells (data not shown). Interestingly, we found differential patterns of expression of IL-17A-eGFP and IL-17F-RFP when comparing CCR6+ ILC3s isolated from the small intestine versus the colon of naïve mice (Fig. 1c, d). In the small intestine, these cells mainly expressed IL-17F-RFP and only few IL-17A-eGFP positive or double positive cells could be detected (Fig. 1c, d). In contrast, in the colon, the majority of these cells expressed IL-17A-eGFP either alone or together with IL-17F-RFP (Fig. 1c, d).
Expression of IL-17A and F in naïve mice. a Flow cytometric analysis of T cells in the spleen and lymph nodes of naïve IL-17DR mice. Cells were activated with PMA and Ionomycin for 4 h prior to analysis and gated for CD90.2+ γδTCR− CD4+, CD90.2+ γδTCR− CD8+, or CD90.2+ γδTCR+ live cells. b Graphical display of the data shown in a of IL-17A-eGFP+, IL-17F-RFP+, and double positive (DP) cells in respective organs. Shown is mean with SD (n = 4). Data is representative of at least three independent experiments. c, d ILCs were isolated from the small intestine and colon of naïve IL-17DR mice. c Flow cytometric analysis of IL-17A-eGFP and IL-17F-RFP expression in ILC3s in indicated parts of the gut. CCR6+ ILC3s were defined as CD45+/Lineage−/CD127+/c-kit+/ CCR6+ live cells. d Graphical display of the data shown in c of IL-17A-eGFP+, IL-17F-RFP+, and double positive (DP) ILC3s in indicated parts of the gut. Shown is mean with SD (n = 3). Data is representative of two independent experiments
In vitro differentiation conditions determine the extent of IL-17F co-expression with IL-17A
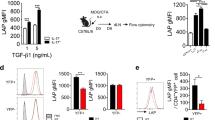
As we found that in naïve mice Th17 cells express mainly IL-17A but not IL-17F, we determined the expression profile of both cytokines after in vitro differentiation. For that, we induced Th17 differentiation by addition of either TGF-β1 or IL-1β in combination with IL-6 and IL-23 to otherwise polyclonally activated naïve T cells. We found that most Th17 cells cultured with TGF-β1 expressed IL-17F-RFP with a large proportion of IL-17AF double positive cells and only few IL-17A-eGFP single positive cells (Fig. 2a, b). Moreover, upon addition of IL-23, the majority of Th17 cells expressed both IL-17 cytokines and we observed a slight but significant increase in the number of IL-17A-eGFP single-expressing cells (Fig. 2a, b). In contrast, Th17 cells differentiated with IL-1β and IL-6 mainly expressed IL-17A-eGFP, which was augmented by addition of IL-23. Under these conditions, only a minority of cells were IL-17AF double positive, but we found a significantly increased population of IL-17F-RFP positive cells when IL-23 was added (Fig. 2a, b).
Differential expression of IL-17A and F after in vitro T cell differentiation. a, b Purified CD4+ T cells from the spleen and lymph nodes of IL-17DR mice were differentiated in vitro using indicated cytokines and antibodies for 5 days. a Flow cytometric analysis of IL-17A-eGFP and IL-17F-RFP expression in helper T cells polarized using indicated conditions. b Graphical display of the data shown in a of IL-17A-eGFP+, IL-17F-RFP+, and double positive (DP) cells after 5 days of culture. Shown is mean with SD (differentiation and analysis was performed in triplicates). Data is representative of three independent experiments
Dominant expression of IL-17A in Th17 cells during CNS inflammation
Next, we turned to study the pattern of expression of IL-17A and IL-17F in experimental autoimmune encephalomyelitis (EAE), a disease model with high presence of IL-17 producing T cells [18, 32,33,34] which is highly dependent on the presence of IL-23 [35, 36] and IL-1β [18, 37]. We subjected the IL-17DR mice to MOG35–55-induced active EAE and analyzed lymphocytic infiltrates in the CNS and lymph node cells using flow cytometry. We found that the majority of CD4+ T cells in the CNS of mice with EAE (clinical score of 1.5–2) expressed IL-17A-eGFP alone, with only few IL-17AF double positive cells (Fig. 3a, b). In contrast, Th17 cells in the lymph nodes of diseased mice showed equal distribution of IL-17A-eGFP, IL-17F-RFP single positive, and IL-17AF double positive cells (Fig. 3a, b). The here found difference of CNS Th17 cells to LN Th17 cells is in line with either an enrichment of strongly activated, encephalitogenic T cells in the CNS or a highly proinflammatory IL-1β-dominated environment which infiltrating T cells encounter in the inflamed CNS in EAE. Similarly, we previously demonstrated changes of encephalitogenic T cells in LN vs. CNS in respect to IL-17A and IFNγ expression [38] and in respect to EBI2 expression [39].
Expression of IL-17A and F in inflammation. a, b Active EAE induction in IL-17DR mice. Animals were sacrificed with clinical score of 1.5–2. Cells were activated with PMA and Ionomycin for 4 h prior to analysis. a Flow cytometric analysis of IL-17A-eGFP and IL-17F-RFP expression in CD3+ CD4+ in the lymph nodes and CNS. b Graphical display of the data shown in a of IL-17A-eGFP+, IL-17F-RFP+, and double positive (DP) cells of indicated cells in respective organs. Shown is the mean with SD (n = 3). c, d Adoptive transfer EAE of Th17 cells from IL-17DR mice in RAG1-deficient hosts. Mice were sacrificed with clinical score of 1.5–2. Cells were activated with PMA and Ionomycin for 4 h prior to analysis. c Flow cytometric analysis of IL-17A-eGFP and IL-17F-RFP expression in transferred CD3+ CD4+ T cells and in the CNS and lymph nodes. d Graphical display of the data shown in c of IL-17A-eGFP+, IL-17F-RFP+, and double positive (DP) cells of indicated cells in respective organs. Shown is mean with SD (n = 3). Data is representative of two independent experiments. e, f IL-17DR mice were injected i.p. with anti-CD3 antibodies or PBS. Cells were activated for 4 h with PMA and Ionomycin. e Flow cytometric analysis of IL-17A-eGFP and IL-17F-RFP expression in CD3+ CD4+ cells in the lamina propria of PBS and anti-CD3-treated animals. f Graphical display of the data shown in e of IL-17A-eGFP+, IL-17F-RFP+, and double positive (DP) cells in the lamina propria of indicated experimental groups. Shown is mean with SD (n = 3). Data is representative of two independent experiments. g, h Back skin and ears of IL-17DR mice were treated with Aldara or sham cream for five consecutive days. g Flow cytometric analysis of IL-17A-eGFP and IL-17F-RFP expression in γδ T cells in the lymph nodes and ears of indicated experimental groups. For lymph nodes, cells were gated as CD3+/γδTCR+ live cells and for ears as CD45+/CD11b−/CD3+/γδTCRint live cells. h Graphical display of the data shown in g of IL-17A-eGFP+, IL-17F-RFP+, and double positive γδ T cells after 5 days of treatment. Shown is mean with SD (n = 3). Data is representative of three independent experiments
In the course of EAE, Th17 cells display a high degree of plasticity and switch from IL-17 to IFNγ production [38, 40]. To study whether IL-17A and IL-17F production in Th17 cells also change in the course of the disease, we transferred IL-23 expanded encephalitogenic T cells into RAG1 deficient mice and analyzed expression of IL-17A vs. IL-17F before and after transfer in the course of EAE. After 4 days of in vitro culture, we found that the majority of cells that were IL-17 positive expressed IL-17A-eGFP, with (ca. 40%) or without (ca. 60%) IL-17F-RFP expression, but only few cells that expressed IL-17F-RFP only (Fig. 3c). Interestingly, when we recovered the transferred cells from the CNS of diseased recipient mice, we found that most of the Th17 cells expressed IL-17A but only very few were double positive (Fig. 3c, d). Similar as in the active EAE, this may be due either to an enrichment of IL-17A expressing T cells in the CNS or change of the expression by individual T cells under the influence of IL-1β in the CNS. Together, our data show that IL-17A expression in inflammatory settings in the context of EAE is dominant over that of IL-17F, and mirror our in vitro data, where we found that differentiation of Th17 cells in the presence of IL-1β, known to preferentially lead to the generation of pathogenic T cells [18, 37], also leads to the generation of IL-17A expressing Th17 cells.
High proportion of IL-17AF co-expressing Th17 cells in the gut
It was previously shown that Th17 cells are present in the lamina propria of naïve mice and it was suggested that these cells rather display a regulatory phenotype with high expression of the anti-inflammatory cytokine IL-10 [25]. Recent studies have demonstrated that systemic in vivo application of anti-CD3 antibodies leads to the generation and expansion of this so called regulatory Th17 cell subset via secretion of TGF-β1 [25]. As we found that in vitro differentiated Th17 cells placed under TGF-β1 conditions do co-express IL-17F-RFP, we were interested to analyze the differential expression of IL-17A and IL-17F in the gut upon anti-CD3 treatment. Therefore, we isolated lymphocytes from the lamina propria of anti-CD3-treated mice or PBS-injected control mice. As expected, Th17 cells strongly expanded in the guts of anti-CD3-treated mice. Interestingly, we found a distinct population of IL-17AF double positive cells in the lamina propria of PBS-treated naïve control mice, which strongly expanded upon anti-CD3 treatment (Fig. 3e, f).
Induction of IL-17F expression in γδ T cells in the skin upon Imiquimod-induced psoriasis-like dermatitis
IL-17A was shown to be highly relevant for the pathogenesis of psoriasis in mice and man [41,42,43,44,45,46]. Several approaches were made to interfere with IL-17 in autoimmune disease by either neutralizing IL-17A (Secukinumab and Ixekizumab), targeting of the IL-17 receptor (Brodalumab), or inhibition of Th17 differentiation by neutralizing the p40 subunit of IL-12/23 (Ustekinumab) [20]. As Secukinumab and Ixekizumab target IL-17A only, we were interested to investigate the expression of IL-17F in a psoriasis-like disease setting. The Imiquimod model in mice induces a psoriasis-like dermatitis and is at first line driven by γδ T cells [42, 47]. After treating mice with Imiquimod, we found, in the lymph nodes, that the γδ T cells expressed mainly IL-17A-eGFP (Fig. 3g, h). However, IL-17F-RFP was co-expressed in some of these cells as well. Interestingly, a significant proportion of dermal γδ-TCRint T cells in the ear skin of sham-treated mice expressed IL-17F-RFP alone or together with IL-17A-eGFP. Strikingly, the majority of γδ T cells in the inflamed skin co-expressed IL-17F-RFP together with IL-17A-eGFP (Fig. 3g, h).
In summary, our data show that IL-17A and IL-17F are often unevenly regulated and expressed. Whereas IL-17A expression coincides with an IL-1β driven profile, IL-17F rather seems to be induced under TGF-β containing differentiation conditions.
Discussion
It is speculated that IL-17A is co-expressed with IL-17F in different cell types. However, detailed studies using genetic models to verify this hypothesis are missing. By crossing IL-17A-IRES-GFP to IL-17F-IRES-RFP mice, we obtained IL-17AF double reporter mice (IL-17DR mice), which enabled us a systematic expression analyses of these two cytokines on the single cell level. We found that IL-17A and F are only partially co-expressed and that highly inflammatory settings like the CNS milieu in EAE favor expression of IL-17A over F whereas TGF-β driven polarization led to IL-17F co-expression.
To define the cytokine milieu for T cell polarization under controlled settings, we differentiated Th17 cells in vitro using IL-6 and either TGF-β1 or IL-1β, respectively, in the presence or absence of IL-23. Interestingly, we observed that differentiation of Th17 cells in the presence of TGF-β1 led to higher frequencies of IL-17F-RFP positive cells as compared to differentiation with IL-1β, which favored the differentiation of IL-17A expressing Th17 cells. In both cases, addition of IL-23 increased the number of the respective Th17 cells. It was previously shown that in vitro differentiated Th17 cells using TGF-β1 are not encephalitogenic, in contrast to IL-1β differentiated Th17 cells. In both pathways, addition of IL-23 increases their pathogenicity [32]. Moreover, IL-1β and IL-23 signaling have been shown to be mandatory for disease pathogenesis [18, 32, 33]. In agreement, we found that the majority of Th17 cells in the CNS of mice with EAE expressed only IL-17A-eGFP.
Our in vitro differentiation data together with our findings that T cells in the CNS of EAE mice rather express IL-17A and not IL-17F let us conclude that expression of IL-17A and not IL-17F is associated with encephalitogenic Th17 cells. In contrast, differentiation of Th17 cells in the presence of TGF-β1 favored the generation of IL-17F expressing cells, cells also found in vivo in the gut after anti-CD3 treatment. As both of these conditions were shown to lead to the differentiation of regulatory Th17 cells, our data suggests that expression of IL-17F is associated with these regulatory cells, rather than with pathogenic Th17 cells. In line with our findings, differentiation of CD4+ T cells with TGF-β1 alone to induce iTreg cells was previously demonstrated to induce a transient wave of IL-17F expression in a subpopulation of the cells [26].
IL-17 has been shown to play a pivotal role in the gut of naïve mice where it acts on epithelial cells to maintain integrity of the epithelium. Moreover, it was demonstrated that systemic application of anti-CD3 antibodies triggers the generation of so called regulatory Th17 cells in the lamina propria. These cells differ from “classical” Th17 cells in cytokine expression and may inhibit the progression of EAE when transferred into disease-induced recipient mice [25]. These cells have been shown to be dependent on TGF-β1 signaling [25] and therefore may rather represent the IL-17A/F double positive Th17 cells found in the TGF-β1/IL-6 differentiation protocol. Therefore, we were curious to analyze Th17 cells isolated from the lamina propria of PBS and anti-CD3 injected IL-17DR mice. In PBS-injected mice, we could already detect a significant proportion of T helper cells being double positive for IL-17A and F. This population became predominant after treatment with anti-CD3 antibodies. Nevertheless, in both treated and untreated mice, we also found a high number of IL-17A-eGFP single positive cells. In summary, using this model being dependent on TGF-β1 signaling, we found high expression of IL-17F-RFP in gut Th17 cells, which is in line with our results obtained by in vitro differentiation of Th17 cells using TGF-β1 and IL-6. These data are in accordance with a specific expression of IL-17F in a rather immunosuppressive (TGF-β dominated) environment. Whether IL-17F may have a specific role under such conditions needs to be investigated in the future.
Antibodies neutralizing IL-17A are used for treatment of psoriasis and may bind either to IL-17A homodimers or IL-17A/F heterodimers. However, up to now, no antibody solely targeting IL-17F is available. We found that in lymph nodes of naïve mice γδ T cells express only IL-17A-eGFP in contrast to γδ T cells in the ears, which also secrete IL-17F. Interestingly, upon induction of psoriasis-like disease, the majority of γδ T cells in the ears co-expressed both cytokines. Recent findings indicate that a specific subset of γδ T cells establishes long-lived memory upon Imiquimod-induced dermatitis. These memory cells express high levels of IL-17A and F after reactivation and are highly pathogenic [48]. In contrast to the EAE model, IL-17F seems to be constantly expressed by γδ T cells during inflammation. It has been shown that IL-17F may trigger IL-6 production from epidermal keratinocytes and acts as neutrophil chemoattractant, thus playing a pathogenic role. However, further studies using IL-17F deficient mice are necessary to analyze its role in this model.
Expression of IL-17A/F by dermal γδ T cells in the skin resembled very much the expression of those cytokines recovered from the gut of Th17 cells after treatment with anti-CD3. This may indicate that T cells at epithelial borders experience a similar cytokine profile, which may contain TGF-β in addition to other locally induced cytokines. The finding in naïve mice that Tc17 cells rather expressed IL-17F whereas Th17 and γδ T cells of the latter animals expressed rather IL-17A alone was surprising and needs further investigation.
Interesting functional differences have been delineated for IL-17A vs. IL-17F using mice deficient for either of the cytokines subjected to different disease models [24]. In light of our data, it is interesting to see that EAE depends on IL-17A expression while colitis on IL-17F expression [24]. Whether our differential expression profiles of IL-17A and IL-17F in different cell types, organs and disease models are indeed of functional relevance in the investigated circumstances need to be further addressed in the future.
Materials and methods
Mice
IL-17A-IRES-eGFP (MGI nomenclature: Il17atm1.1Flv) and IL-17F-IRES-RFP (MGI nomenclature: Il17ftm1Cdon) mice were generously provided by the group of Richard Flavell and Chen Dong respectively and generated as described [24, 25]. Homozygous mice of both strains were crossed to each other to obtain heterozygous IL-17AF double reporter mice. Mice of both sexes were used at an age of 8–12 weeks of age and were housed under SPF conditions.
Organ preparation
Single cell suspensions of LNs and spleen were prepared non-enzymatically in Dulbecco’s phosphate-buffered saline supplemented with 2% fetal calf serum. For isolation of lymphocytes from the CNS, brain and spinal cord were cut and digested for 20 min at 37 °C with 1 mg/ml collagenase II (Sigma) and 40 μg/ml DNase I (Roche) followed by centrifugation in a Percoll gradient [49]. Lymphocytes from the ears of mice were isolated by digestion in DPBS (+/+) with 0.25 mg/ml Liberase and 40 μg/ml DNase I for 1 h at 37 °C. Preparation of lymphocytes from the small intestine and colon of mice was performed as described [50].
In vitro T cell differentiation
CD4+ T cells were isolated from spleen and LNs by MACS purification (Miltenyi Biotec) according to the manufacturer’s protocol. Cells were cultured at 1 × 105 cells/well in 200 μl T cell medium (TCM) (RPMI with 10% FCS, 2 mM L-Gln, 100 U/ml penicillin, 100 mg/ml streptomycin, 1 mM sodium pyruvate, 50 mM 2-mercaptoethanol, 10 mM HEPES, and 1% non-essential amino acids (MEM)) in 96-well plates. For Th17 differentiation, 1 μg/ml α-CD3, 6 ng/ml α-CD28, and 10 μg/ml α-IFNγ antibodies (BioXCell) with 2 ng/ml TGF-β1 (R&D) or 50 ng/ml IL-1β (R&D), 5 ng/ml IL-6 (Promocell), and 20 ng/ml IL-23 (Miltenyi) were used. For each condition, technical replicates were used. Cells were cultured at 37 °C with 5% CO2 for 5 days.
Active EAE induction
Mice were immunized subcutaneously at the base of the tail with 100 μg MOG35–55 peptide emulsified in CFA supplemented with 1.1 mg heat-inactivated Mycobacterium tuberculosis. Along with immunization and at day 2, mice were injected intraperitoneally (i.p.) with 200 ng of pertussis toxin (Ptx) (Sigma-Aldrich) in PBS. Mice were scored for signs of EAE as described [51].
Adoptive transfer EAE
Mice were immunized as described above except administration of Ptx. Ten days after immunization, spleen and lymph node cells were prepared and cultured in T cell medium with 50 μg/ml MOG35–55, 10 μg/ml α-IFNγ antibodies (BioXCell), and 10 ng/ml IL-23 (Miltenyi). Cells were cultured for 4 days at 37 °C at 5% CO2. Afterwards, 5 × 106 blasting cells were injected intravenously into RAG1−/− mice. Ptx (200 ng) was administered i.p. along with transfer and 2 days post-transfer.
Anti-CD3 treatment
IL-17DR mice were treated i.p. with 20 μg anti-CD3 antibodies in PBS for four consecutive days or received PBS without antibodies as described (Esplugues E et al., 2011, Nature). The mice were sacrificed and cells from the lamina propria were isolated as described [52] and analyzed via flow cytometry after treatment.
Induction of psoriasis-like dermatitis
Psoriasis-like dermatitis was induced by application of 5 mg Aldara cream containing 5% Imiquimod on the ears for five consecutive days. Similarly, the back skin of mice was treated with 50 mg of Aldara cream. After treatment, mice were sacrificed and cells from the lymph nodes and ear skin were isolated as described and analyzed via flow cytometry.
Flow cytometry and cell sorting
Antibodies were purchased from eBioscience, BD, and BioLegend. For some experiments, cells were activated for 4 h in TCM with 50 ng/ml phorbol-12-myristate-13-acetate (PMA), 500 ng/ml ionomycin at 37 °C, and 5% CO2. All incubations were performed on ice or at 4 °C in the dark in PBS containing 0.5% BSA and 0.02% NaN3. Prior to surface staining, cells were pretreated with 5 μg/ml FC-Block (BioXCell). Staining of ILCs was performed using following panel:
Antigen | Fluorochrome | Dilution | Clone | Company |
|---|---|---|---|---|
IL-17A | eGFP | – | – | – |
IL-17F | RFP | – | – | – |
IL-7Rα | PerCpCy5.5 | 1/100 | A7R34 | eBio |
CD4 | PeCy7 | 1/400 | GK1.5 | eBio |
c-Kit | APC | 1/500 | ACK2 | eBio |
CD45 | APCCy7 | 1/200 | 104 | eBio |
CCR6 | BV421 | 1/100 | 29-2L17 | BioLegend |
Lineage CD3 GR1 CD19 CD5 Nk1.1 KLRG1 CD11c B220 FcεRI F4/80 CD64 TCRγδ TCRβ | SA-V500 Biotin Biotin Biotin Biotin Biotin Biotin Biotin Biotin Biotin Biotin Biotin Biotin Biotin | 1/200 1/600 1/600 1/100 1/1000 1/200 1/600 1/100 1/1000 1/300 1/300 1/300 1/400 1/200 | – 145-2C11 RB6-8C5 MB19-1 53-7.3 PK136 2F1 HL3 RA3-6B2 MAR-1 BM8 X54-5/7.1 GL3 H57-597 | BD eBio eBio eBio eBio eBio eBio eBio eBio eBio BioLegend BioLegend BD eBio |
Nkp46 | BV605 | 1/100 | 29A1.4 | BioLegend |
Statistics
Statistical analysis was performed using GraphPad Prism. Statistical significance was calculated using the unpaired two-tailed t test for two independent parameters. Differences within paired values (ratios) between organs, such as, e.g., spleen and LNs in the same animals, were detected using paired two-tailed t tests. For comparative analyses of three groups, we used one-way ANOVA and performed post hoc tests with Bonferroni correction. Distributions are visualized by box plots. Based on the small sample sizes, the band inside the box denotes the mean instead of the generally depicted median. A p value ≤ 0.05 was considered as statistically significant. Due to the number of hypotheses to be tested, an adjustment for multiplicity was not feasible and this study is to be considered as purely explorative. Therefore, statistical tests and resulting p values can only be interpreted descriptively and have no confirmatory value.
References
Waisman A, Hauptmann J, Regen T (2015) The role of IL-17 in CNS diseases. Acta Neuropathol 129:625–637
Kolls JK, Linden A (2004) Interleukin-17 family members and inflammation. Immunity 21:467–476
Rouvier E, Luciani MF, Mattei MG, Denizot F, Golstein P (1993) CTLA-8, cloned from an activated T cell, bearing AU-rich messenger RNA instability sequences, and homologous to a herpesvirus saimiri gene. J Immunol 150:5445–5456
Yang XO, Pappu BP, Nurieva R, Akimzhanov A, Kang HS, Chung Y, Ma L, Shah B, Panopoulos AD, Schluns KS, Watowich SS, Tian Q, Jetten AM, Dong C (2008) T helper 17 lineage differentiation is programmed by orphan nuclear receptors ROR alpha and ROR gamma. Immunity 28:29–39
Takatori H, Kanno Y, Watford WT, Tato CM, Weiss G, Ivanov II, Littman DR, O'Shea JJ (2009) Lymphoid tissue inducer-like cells are an innate source of IL-17 and IL-22. J Exp Med 206:35–41
Michel ML, Keller AC, Paget C, Fujio M, Trottein F, Savage PB, Wong CH, Schneider E, Dy M, Leite-de-Moraes MC (2007) Identification of an IL-17-producing NK1.1 (neg) iNKT cell population involved in airway neutrophilia. J Exp Med 204:995–1001
Michel ML, Mendes-da-Cruz D, Keller AC, Lochner M, Schneider E, Dy M, Eberl G, Leite-de-Moraes MC (2008) Critical role of ROR-gammat in a new thymic pathway leading to IL-17-producing invariant NKT cell differentiation. Proc Natl Acad Sci U S A 105:19845–19850
Sutton CE, Lalor SJ, Sweeney CM, Brereton CF, Lavelle EC, Mills KH (2009) Interleukin-1 and IL-23 induce innate IL-17 production from gammadelta T cells , amplifying Th17 responses and autoimmunity. Immunity 31:331–341
Roark CL, French JD, Taylor MA, Bendele AM, Born WK, O'Brien RL (2007) Exacerbation of collagen-induced arthritis by oligoclonal, IL-17-producing gamma delta T cells. J Immunol 179:5576–5583
Ortega C, Fernandez AS, Carrillo JM, Romero P, Molina IJ, Moreno JC, Santamaria M (2009) IL-17-producing CD8+ T lymphocytes from psoriasis skin plaques are cytotoxic effector cells that secrete Th17-related cytokines. J Leukoc Biol 86:435–443
Huber M, Heink S, Grothe H, Guralnik A, Reinhard K, Elflein K, Hunig T, Mittrucker HW, Brustle A, Kamradt T, Lohoff M (2009) A Th17-like developmental process leads to CD8(+) Tc17 cells with reduced cytotoxic activity. Eur J Immunol 39:1716–1725
Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, Weiner HL, Kuchroo VK (2006) Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441:235–238
Korn T, Bettelli E, Gao W, Awasthi A, Jager A, Strom TB, Oukka M, Kuchroo VK (2007) IL-21 initiates an alternative pathway to induce proinflammatory T(H)17 cells. Nature 448:484–487
Heink S, Yogev N, Garbers C, Herwerth M, Aly L, Gasperi C, Husterer V, Croxford AL, Moller-Hackbarth K, Bartsch HS, Sotlar K, Krebs S, Regen T, Blum H, Hemmer B, Misgeld T, Wunderlich TF, Hidalgo J, Oukka M, Rose-John S, Schmidt-Supprian M, Waisman A, Korn T (2017) Trans-presentation of IL-6 by dendritic cells is required for the priming of pathogenic TH17 cells. Nat Immunol 18:74–85
Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, Cua DJ, Littman DR (2006) The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126:1121–1133
Acosta-Rodriguez EV, Napolitani G, Lanzavecchia A, Sallusto F (2007) Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells. Nat Immunol 8:942–949
Ghoreschi K, Laurence A, Yang XP, Tato CM, McGeachy MJ, Konkel JE, Ramos HL, Wei L, Davidson TS, Bouladoux N, Grainger JR, Chen Q, Kanno Y, Watford WT, Sun HW, Eberl G, Shevach EM, Belkaid Y, Cua DJ, Chen W, O'Shea JJ (2010) Generation of pathogenic T(H)17 cells in the absence of TGF-beta signalling. Nature 467:967–971
Mufazalov IA, Schelmbauer C, Regen T, Kuschmann J, Wanke F, Gabriel LA, Hauptmann J, Muller W, Pinteaux E, Kurschus FC, Waisman A (2017) IL-1 signaling is critical for expansion but not generation of autoreactive GM-CSF+ Th17 cells. EMBO J 36:102–115
Chung Y, Chang SH, Martinez GJ, Yang XO, Nurieva R, Kang HS, Ma L, Watowich SS, Jetten AM, Tian Q, Dong C (2009) Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity 30:576–587
Kurschus FC, Moos S (2017) IL-17 for therapy. J Dermatol Sci 87:221–227
Suto A, Kashiwakuma D, Kagami S, Hirose K, Watanabe N, Yokote K, Saito Y, Nakayama T, Grusby MJ, Iwamoto I, Nakajima H (2008) Development and characterization of IL-21-producing CD4+ T cells. J Exp Med 205:1369–1379
Simonian PL, Wehrmann F, Roark CL, Born WK, O'Brien RL, Fontenot AP (2010) Gammadelta T cells protect against lung fibrosis via IL-22. J Exp Med 207:2239–2253
Kim JS, Smith-Garvin JE, Koretzky GA, Jordan MS (2011) The requirements for natural Th17 cell development are distinct from those of conventional Th17 cells. J Exp Med 208:2201–2207
Yang XO, Chang SH, Park H, Nurieva R, Shah B, Acero L, Wang YH, Schluns KS, Broaddus RR, Zhu Z, Dong C (2008) Regulation of inflammatory responses by IL-17F. J Exp Med 205:1063–1075
Esplugues E, Huber S, Gagliani N, Hauser AE, Town T, Wan YY, O'Connor W Jr, Rongvaux A, Van Rooijen N, Haberman AM, Iwakura Y, Kuchroo VK, Kolls JK, Bluestone JA, Herold KC, Flavell RA (2011) Control of TH17 cells occurs in the small intestine. Nature 475:514–518
Yang XO, Nurieva R, Martinez GJ, Kang HS, Chung Y, Pappu BP, Shah B, Chang SH, Schluns KS, Watowich SS, Feng XH, Jetten AM, Dong C (2008) Molecular antagonism and plasticity of regulatory and inflammatory T cell programs. Immunity 29:44–56
Chang SH, Dong C (2009) IL-17F: regulation, signaling and function in inflammation. Cytokine 46:7–11
Satoh-Takayama N, Vosshenrich CA, Lesjean-Pottier S, Sawa S, Lochner M, Rattis F, Mention JJ, Thiam K, Cerf-Bensussan N, Mandelboim O, Eberl G, Di Santo JP (2008) Microbial flora drives interleukin 22 production in intestinal NKp46+ cells that provide innate mucosal immune defense. Immunity 29:958–970
Geremia A, Arancibia-Carcamo CV, Fleming MP, Rust N, Singh B, Mortensen NJ, Travis SP, Powrie F (2011) IL-23-responsive innate lymphoid cells are increased in inflammatory bowel disease. J Exp Med 208:1127–1133
Klose CS, Kiss EA, Schwierzeck V, Ebert K, Hoyler T, d'Hargues Y, Goppert N, Croxford AL, Waisman A, Tanriver Y, Diefenbach A (2013) A T-bet gradient controls the fate and function of CCR6-RORgammat+ innate lymphoid cells. Nature 494:261–265
Kiss EA, Vonarbourg C, Kopfmann S, Hobeika E, Finke D, Esser C, Diefenbach A (2011) Natural aryl hydrocarbon receptor ligands control organogenesis of intestinal lymphoid follicles. Science 334:1561–1565
Lee Y, Awasthi A, Yosef N, Quintana FJ, Xiao S, Peters A, Wu C, Kleinewietfeld M, Kunder S, Hafler DA, Sobel RA, Regev A, Kuchroo VK (2012) Induction and molecular signature of pathogenic T(H)17 cells. Nat Immunol 13:991–999
Becher B, Durell BG, Noelle RJ (2002) Experimental autoimmune encephalitis and inflammation in the absence of interleukin-12. J Clin Invest 110:493–497
Kurschus FC (2015) T cell mediated pathogenesis in EAE: molecular mechanisms. Biom J 38:183–193
Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B, Lucian L, To W, Kwan S, Churakova T, Zurawski S, Wiekowski M, Lira SA, Gorman D, Kastelein RA, Sedgwick JD (2003) Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421:744–748
Becher B, Durell BG, Noelle RJ (2003) IL-23 produced by CNS-resident cells controls T cell encephalitogenicity during the effector phase of experimental autoimmune encephalomyelitis. J Clin Invest 112:1186–1191
Sutton C, Brereton C, Keogh B, Mills KH, Lavelle EC (2006) A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J Exp Med 203:1685–1691
Kurschus FC, Croxford AL, Heinen AP, Wortge S, Ielo D, Waisman A (2010) Genetic proof for the transient nature of the Th17 phenotype. Eur J Immunol 40:3336–3346
Wanke F, Moos S, Croxford AL, Heinen AP, Graf S, Kalt B, Tischner D, Zhang J, Christen I, Bruttger J, Yogev N, Tang Y, Zayoud M, Israel N, Karram K, Reissig S, Lacher SM, Reichhold C, Mufazalov IA, Ben-Nun A, Kuhlmann T, Wettschureck N, Sailer AW, Rajewsky K, Casola S, Waisman A, Kurschus FC (2017) EBI2 is highly expressed in multiple sclerosis lesions and promotes early CNS migration of encephalitogenic CD4 T cells. Cell Rep 18:1270–1284
Hirota K, Duarte JH, Veldhoen M, Hornsby E, Li Y, Cua DJ, Ahlfors H, Wilhelm C, Tolaini M, Menzel U, Garefalaki A, Potocnik AJ, Stockinger B (2011) Fate mapping of IL-17-producing T cells in inflammatory responses. Nat Immunol 12:255–263
Lowes MA, Kikuchi T, Fuentes-Duculan J, Cardinale I, Zaba LC, Haider AS, Bowman EP, Krueger JG (2008) Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. J Invest Dermatol 128:1207–1211
Cai Y, Shen X, Ding C, Qi C, Li K, Li X, Jala VR, Zhang HG, Wang T, Zheng J, Yan J (2011) Pivotal role of dermal IL-17-producing gammadelta T cells in skin inflammation. Immunity 35:596–610
El Malki K, Karbach SH, Huppert J, Zayoud M, Reissig S, Schuler R, Nikolaev A, Karram K, Munzel T, Kuhlmann CR, Luhmann HJ, von Stebut E, Wortge S, Kurschus FC, Waisman A (2013) An alternative pathway of imiquimod-induced psoriasis-like skin inflammation in the absence of interleukin-17 receptor a signaling. J Invest Dermatol 133:441–451
Waisman A (2012) To be 17 again—anti-interleukin-17 treatment for psoriasis. N Engl J Med 366:1251–1252
Croxford AL, Karbach S, Kurschus FC, Wortge S, Nikolaev A, Yogev N, Klebow S, Schuler R, Reissig S, Piotrowski C, Brylla E, Bechmann I, Scheller J, Rose-John S, Wunderlich FT, Munzel T, von Stebut E, Waisman A (2014) IL-6 regulates neutrophil microabscess formation in IL-17A-driven psoriasiform lesions. J Invest Dermatol 134:728–735
van der Fits L, Mourits S, Voerman JS, Kant M, Boon L, Laman JD, Cornelissen F, Mus AM, Florencia E, Prens EP, Lubberts E (2009) Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol 182:5836–5845
Pantelyushin S, Haak S, Ingold B, Kulig P, Heppner FL, Navarini AA, Becher B (2012) Rorgammat+ innate lymphocytes and gammadelta T cells initiate psoriasiform plaque formation in mice. J Clin Invest 122:2252–2256
Hartwig T, Pantelyushin S, Croxford AL, Kulig P, Becher B (2015) Dermal IL-17-producing gammadelta T cells establish long-lived memory in the skin. Eur J Immunol 45:3022–3033
Cardona AE, Huang D, Sasse ME, Ransohoff RM (2006) Isolation of murine microglial cells for RNA analysis or flow cytometry. Nat Protoc 1:1947–1951
Gronke K, Kofoed-Nielsen M, Diefenbach A (2017) Isolation and flow cytometry analysis of innate lymphoid cells from the intestinal lamina propria. Methods Mol Biol 1559:255–265
Huppert J, Closhen D, Croxford A, White R, Kulig P, Pietrowski E, Bechmann I, Becher B, Luhmann HJ, Waisman A, Kuhlmann CR (2010) Cellular mechanisms of IL-17-induced blood-brain barrier disruption. FASEB J 24: 1023–1034. DOI fj.09–141978 [pii]
Reissig S, Hackenbruch C, Hovelmeyer N (2014) Isolation of T cells from the gut. Methods Mol Biol 1193:21–25
Acknowledgements
We thank Bettina Kalt, Petra Adams, and Michaela Blanfeld for excellent technical assistance. We acknowledge Steffanie Bürger and Ina Schäfer from the Institute for Molecular Biology in Mainz for excellent assistance with the flow cytometry. We thank Svenja Schüler for help with statistical data representation. We are grateful to Richard Flavell and Chen Dong to share with us the reporter mouse lines used in this study. This work was supported by the Deutsche Forschungsgemeinschaft SFB/TR-128 to F.C.K. and A.W. and by SFB/TR-156 to A.W., A.D., and F.C.K.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All experiments with mice were carried out in accordance with the guidelines of the Central Animal Facility Institution of Mainz and in accordance with relevant laws and guidelines with permission by the state Rhineland-Palatinate (animal experimentation applications (TVA) nos. G13-1-099 and G12-1-057). The approval process contained an ethical committee meeting instated by the Landesuntersuchungsamt Rheinland-Pfalz.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wanke, F., Tang, Y., Gronke, K. et al. Expression of IL-17F is associated with non-pathogenic Th17 cells. J Mol Med 96, 819–829 (2018). https://doi.org/10.1007/s00109-018-1662-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-018-1662-5