Abstract
Purpose
In high-risk patients with acute calculous cholecystitis (ACC), percutaneous cholecystostomy (PC) can serve as a bridging option to cholecystectomy [laparoscopic cholecystectomy (LC)] or as definitive treatment. The purpose of this study was to identify predictors of the need for permanent PC.
Methods
Data from 257 PCs performed for ACC (mean age 67.3 ± 14) was collected for a 10-year period. Demographic and clinical characteristics at initial admission, co-morbidities were analyzed. Patients who underwent interval LC were defined as the surgery group (SG; n = 163, 63.4 %) and the remaining patients as the non-SG (NSG; n = 94, 36.6 %).
Results
Patients in the SG were significantly younger and had a shorter length of hospital stay (p < 0.01). The rate of coronary artery disease (CAD; 63.2 vs. 20.2 %), chronic renal failure (14.9 vs. 6.1 %), and the mean number of co-morbidities (2.2 vs. 1.4) were significantly higher in the NSG. Sepsis at admission was more common in the NSG (19.1 vs. 4.9 %, p < 0.001). 56 patients (34.4 %) in the SG and 24 patients (25.5 %) in the NSG developed tube-related complications. In hospital mortality was similar between the groups. Multivariate analysis showed that age ≥75, increased alkaline phosphatase (ALK-P), history of CAD, were predictors of PC as a definite treatment in this high-risk group of patients with ACC.
Conclusions
High operative risk due to older age and CAD preclude LC in more than one-third of patients following PC especially presenting with sepsis and elevated ALK-P. This study suggests that PC could be a safe treatment option in this select group of high-risk patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The literature about the best treatment option of for acute calculous cholecystitis (ACC) has been recently examined in several consensus papers [1–6]. The spectrum of disease presentation ranges from a mild, self-limiting one to a potentially life-threatening condition. Age-related co-morbidities strongly influence the severity of patient condition [7]. Percutaneous tube cholecystostomy (PC) either as a bridge to cholecystectomy or as a definite procedure is reported in the literature as an alternative for the emergency treatment in septic, high-risk patients [8, 9]. The Tokyo guidelines consider PC as mandatory in the severe grade of ACC and also suggest its use in the moderate grade, in order to decrease the technical difficulties during laparoscopic cholecystectomy (LC) [10].
PC is considered an effective treatment option in critically ill patients, especially in elderly patients and in patients with complications; however, no randomized controlled trials are available on this issue [11, 12]. Also, reports which analyzed PC results rarely take into account patients who could not have tolerated any surgery, and this limitation has to be considered.
The primary objective of this work was to describe and analyze our experience with PC over a 10-year period. The secondary objective was to identify predictors of the need for definitive or permanent cholecystostomy.
Methods
The study was reviewed and approved by the Institutional Review Board at the Hadassah-Hebrew University Hospital in Jerusalem before launch of data collection. We retrospectively reviewed the records of all consecutive patients who were admitted with the diagnosis of ACC to the Ein Kerem and Mount Scopus Campuses from 1 January 2003 to 31 December 2012. Patients with the diagnosis of acute acalculous cholecystitis were excluded from the study.
The charts and records of all patients admitted to the hospital with the diagnosis of ACC and who underwent PC were reviewed for demographic data, co-morbid conditions, clinical presentation, laboratory and imaging findings at index admission, tube complications, and outcome. Laboratory parameters included white blood cell (WBC) count, hemoglobin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma-glutamyl transpeptidase (GGTP), total bilirubin (TB), alkaline phosphatase (ALK-P), C-reactive protein (CRP) and albumin.
The presence of co-morbid conditions was retrieved from hospital records. Co-morbid conditions were defined according to Table 1. Sepsis was defined as infection causing hypotension and warranting catecholamine support, and/or one or more dysfunctional organs.
Broad-spectrum antibiotics and bowel rest was our general policy for patients with ACC. The decision to perform a PC was at the discretion of the attending surgeon and generally based on either the presence of co-morbid conditions and/or lack of clinical improvement. All cholecystostomy tubes were inserted under either sonographic (US) or computerized tomography (CT) guidance according to the preferences of the attending radiologist.
Our policy was to leave the PC drain in place until delayed cholecystectomy (DC) was performed, usually 6 weeks later. Before discharge a routine cholecystogram was performed in order demonstrate the biliary system and passage of contrast to the duodenum. In cases of normal flow through the biliary tree to the duodenum the patient was sent home with the drain closed. In cases of cystic duct obstruction the drain was left open. In cases of suspected biliary tree stones, patients were referred for ambulatory ERCP prior to cholecystectomy. Following discharge from index admission, patients were referred to our outpatient clinic. Patients were then scheduled for DC depending on their age and overall general condition. PC tubes were left in place indefinitely in patients who were not considered to be candidates for surgery, almost universally due to associated co-morbid conditions and old age.
Major complications were defined as any event requiring either radiological or emergency intervention, including repeat hospitalization. Mortality was defined as any death occurring during the index admission and up to 30 days thereafter.
Statistical analysis
Data are presented as mean plus minus standard deviation (±SD). The χ 2 test and Fisher’s exact test were used to compare proportions and the Mann–Whitney U test was used to compare continuous variables. Multivariate logistic regression analysis was performed to analyze predictors of a permanent PC. The adjusted odds ratio (aOR) as well as its 95 % confidence interval (CI) was calculated for each parameter. A p value of 0.05 or less was considered statistically significant. Statistical analysis was performed using SPSS version 11.5 (Statistical Package for Social Science, Chicago, IL).
Results
Patient population
From 1 January 2003 to 31 December 2012 we admitted 1093 patients with the diagnosis of ACC to both Hadassah campuses in Jerusalem, Israel. A total of 257 patients (23.5 %) underwent PC due to either co-morbid conditions and/or lack of clinical improvement. Of those, DC was eventually performed for 163 patients (63.4 %) who were defined as the DC group. The median time to elective cholecystectomy was 67 days (range 27–222 days). The remaining 94 patients (36.67 %) were defined as the non-surgery group (NC).
Patients in the NC group were older and more often presented with fever and septic shock compared with the DC group (p < 0.01 for all; Table 2). Bacteremia and a positive bile culture were more common in the NC group compared with the DC group, but this difference did not reach statistical significance [19 patients (20.2 %) vs. 19 patients (11.7 %), p = 0.07, and 54 patients (57.4 %) vs. 75 patients (46 %), p = 0.09, respectively].
Co-morbid conditions
The most common co-morbid condition in the cohort was hypertension (HTN; n = 134, 52.1 %) followed by diabetes mellitus (DM; n = 74, 28.8 %) and coronary artery disease (CAD; n = 67, 26.1 %). HTN, CAD and chronic renal failure (CRF) were significantly more common in the NC group compared with the DC group (Table 3). The mean number of co-morbid conditions was higher in the NC group and there were significantly more patients in the NC group who had three or more co-morbid conditions (Table 3).
Laboratory and imaging findings
WBC at admission was significantly higher in the NC group. Cholestatic and hepato-cellular enzymes and TB tended to be higher in the NC group and this difference reached statistical significance for four of five parameters (Table 4).
The presence of sonographic findings of ACC such as wall thickening and peri-cholecystic fluid was not different between the groups. Intra or extra-hepatic biliary dilatation was more common in the NC group but this difference did not reach statistical significance (14 patients [14.9 %] vs. 12 patients [7.4 %], respectively, p = 0.08). CT was performed for 46 patients (48.9 %) in the NC group compared with 66 patients (40.5 %) in the DC group (p = NS).
Tube complications
Overall 56 patients (34.4 %) in the DC group and 24 patients (25.5 %) in the NC group developed tube-related complications (p = NS; Table 5). Re-admission for tube complications was significantly more common in the DC group compared with the NC group [31 patients (19 %) vs. 8 patients (8.5 %), p = 0.03]. 14 patients (8.6 %) in the DC group and 12 patients (12.8) in the NC required re-insertion of the tubes (p = NS).
Outcome
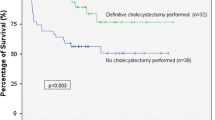
Improvement of abdominal pain and resolution of fever at 24 h occurred similarly between the groups. No patient required urgent or immediate surgery following PC. Length of index hospital stay (LOS) was significantly longer in the NC group compared with the DC group (15.0 ± 12.4 vs. 10.4 ± 6.3 days, respectively, p = 0.002). More patients in the NC group required ICU admission but this difference did not reach statistical significance (14 patients [14.9 %] vs. 13 patients [8 %], respectively, p = 0.09). Three patients (1.8 %) in the DC group and 4 patients (4.3 %) in the NC group died (p = NS).
Predictors of the need for permanent cholecystostomy
Multivariate regression analysis was performed in order to identify predictors of the need for a permanent PC. Parameters which were found significant on univariate analysis were entered into a forward stepwise regression model. Older age (aOR 1.1, 95 % CI 1–1.1, p < 0.001), presence of CAD (aOR 3.0, CI 1.3–7.0, p = 0.013), elevated ALK-P (aOR 1.01, CI 1.00–1.01, p < 0.01), and the presence of septic shock at admission (aOR 9.9, CI 1.5–66.7, p = 0.02) were found to be independent predictors of a permanent PC. In order to quantify for age and level of ALK-P, we entered age ≥75 and ALK-P >135 (75th percentile, and upper border of normal in our laboratory) as categorical parameters. Then, age ≥75 and ALK-P >135 were found to be independent predictors of the need for permanent PC (aOR 3.6, CI 1.5–8.2, p < 0.01, and aOR 8.63, CI 3.3–22.5, p < 0.01, respectively).
Discussion
In common, data on the clinical course of patients managed with PC are limited and come from single-institution studies [13–16]. Cholecystectomy is the only definitive treatment for ACC. The place of PC versus cholecystectomy in high-risk patients remains poorly defined. Currently, there are no clear guidelines for choosing who may benefit from PC over urgent cholecystectomy, and the decision relies mainly on clinical experience and judgement. To further elucidate this subject, a prospective randomized study was initiated in 2012 to compare PC–LC in high-risk patients and results are still pending [17].
There is controversy regarding whether cholecystectomy should be performed after PC in elderly high-risk patients. Multiple reports have demonstrated that PC can temporize a patient with multiple comorbid conditions with ACC. Potential reasons for this include increased compliance with current treatment guidelines for ACC, and improvements in radiological interventional techniques.
An average of 40 % cholecystectomy rate was achieved and a 30-day mortality of 15.4 % was reported following cholecystostomy in one systematic review of 53 studies comprising 1918 patients [9]. The highest reported mortality rate after cholecystectomy in elderly patients with comorbidities was 19 %, while the average mortality rate was 4.5 % [9, 18]. Furthermore, while the majority of patients did not undergo cholecystectomy, the risk of a subsequent gallstone-related ED visit or re-admission was almost 50 % within 1 year of index discharge. We performed delayed LC in nearly two-thirds of patients following PC. Our overall mortality was 2.7 %, possibly justifying our management strategy.
In a study of 24 elderly patients, Griniatsos et al. reported control of ACC in 90 % of patients with cholecystostomy tubes alone and recommended against interval cholecystectomy [19]. In another recent article the authors reported that ACC resolved in 98.6 % of patients treated with PC [20]. Drainage-related morbidity and mortality rates were 4.1 and 1.4 %, respectively. No patient underwent cholecystectomy after PC and the recurrence rate of ACC was 4.1 %. However, advocates for interval cholecystectomy quote a 46 % recurrence rate at 3 years for patients treated by cholecystostomy drainage alone [21]. Our results show that PC is extremely helpful in temporizing the infectious complications associated with ACC (100 %). It is unlikely that one best management strategy will apply for all patients, and only sound clinical judgement and a fully informed consent on the risks of surgery versus non-operative management will remain essential.
Our results show that tube-related complications occur in up to one-third of patients following PC and may require re-admission in up to one-fifth of patients. Tube-related complications were surprisingly more common in the DC group. Perhaps this finding is due to the more mobile status of these patients who are generally younger and suffer from fewer co-morbid conditions.
Our study also shows that identification of the majority of patients with ACC who cannot have cholecystectomy and require permanent PC can be achieved utilizing certain admission parameters. Older age, CAD, elevated ALK-P and septic shock at admission are all predictors of the need for a long term or indefinite PC. These patients should be differentiated upon admission and drained earlier thus minimizing the septic complications associated with ongoing biliary infection.
Older age, history of co-morbidities such as CAD, and the presence of sepsis at admission are intuitively factors that influence the attending surgeon’s inclination to perform PC early in the course of ACC. Our results show that elevated hepatocellular and cholestatic enzymes and biliary dilatation are associated with a permanent PC. Moreover, elevated levels of ALK-P, especially abnormal levels of ALK-P, are predictors of the need for a permanent PC. These suggest that a different disease process, perhaps with more pronounced biliary injury, is involved in patients who require a definitive PC. These patients should probably be identified early in their hospital course and drained. The remaining patients should probably undergo early cholecystectomy during their initial admission.
The main strength of the present study is the large sample size. Limitations of this study include the retrospective nature of data collection and the lack of a specified organizational protocol in the management of ACC. First, an obvious selection bias for elderly comorbid patients being placed in the PC group exists. Second, the length of time the cholecystostomy drain remained in place is unknown. Third, we should assume that several patients in the NC group were lost to follow-up and could potentially influence the results.
Conclusions
Early percutaneous gallbladder drainage is safe and effective in critically ill patients in the acute phase of ACC, with a high rate of clinical improvement. Almost two-thirds of patients who are initially treated with PC at our institution eventually undergo DC. The majority of these patients will probably benefit from early surgery.
Elderly high-risk patients probably have limited benefit from cholecystectomy due to their inherent surgical risk and an expected low recurrence rate of ACC. We thus suggest that PC can be performed as definitive treatment in this select group of patients above 75 year old and presenting with septic shock and elevated ALK-P.
References
Agresta F, Ansaloni L, Baiocchi GL, Bergamini C, Campanile FC, Carlucci M, Cocorullo G, Corradi A, Franzato B, Lupo M, Mandalà V, Mirabella A, Pernazza G, Piccoli M, Staudacher C, Vettoretto N, Zago M, Lettieri E, Levati A, Pietrini D, Scaglione M, De Masi S, De Placido G, Francucci M, Rasi M, Fingerhut A, Uranüs S, Garattini S. Laparoscopic approach to acute abdomen from the Consensus Development Conference of the Società Italiana di Chirurgia Endoscopica e nuove tecnologie (SICE), Associazione Chirurghi Ospedalieri Italiani (ACOI), Società Italiana di Chirurgia (SIC), Società Italiana di Chirurgia d’Urgenza e del Trauma (SICUT), Società Italiana di Chirurgia nell’Ospedalità Privata (SICOP), and the European Association for Endoscopic Surgery (EAES). Surg Endosc. 2012;26:2134–64.
Yamashita Y, Takada T, Kawarada Y, Nimura Y, Hirota M, Miura F, Mayumi T, Yoshida M, Strasberg S, Pitt HA, de Santibanes E, Belghiti J, Büchler MW, Gouma DJ, Fan ST, Hilvano SC, Lau JW, Kim SW, Belli G, Windsor JA, Liau KH, Sachakul V. Surgical treatment of patients with acute cholecystitis: Tokyo guidelines. J Hepatobiliary Pancreat Surg. 2007;14:91–7.
Yamashita Y, Takada T, Strasberg SM, Pitt HA, Gouma DJ, Garden OJ, Büchler MW, Gomi H, Dervenis C, Windsor JA, Kim S-W, de Santibanes E, Padbury R, Chen X-P, Chan ACW, Fan S-T, Jagannath P, Mayumi T, Yoshida M, Miura F, Tsuyuguchi T, Itoi T, Supe AN. TG13 surgical management of acute cholecystitis. J Hepatobiliary Pancreat Sci. 2013;20:89–96.
Lo CM, Liu CL, Fan ST, Lai EC, Wong J. Prospective randomized study of early versus delayed laparoscopic cholecystectomy for acute cholecystitis. Ann Surg. 1998;227:461–7.
Chandler CF, Lane JS, Ferguson P, Thompson JE, Ashley SW. Prospective evaluation of early versus delayed laparoscopic cholecystectomy for treatment of acute cholecystitis. Am Surg. 2000;66:896–900.
Johansson M, Thune A, Blomqvist A, Nelvin L, Lundell L. Management of acute cholecystitis in the laparoscopic era: results of a prospective, randomized clinical trial. J Gastrointest Surg. 2003;7:642–5.
Borzellino G, Sauerland S, Minicozzi AM, Verlato G, Di Pietrantonj C, de Manzoni G, Cordiano C. Laparoscopic cholecystectomy for severe acute cholecystitis. A meta-analysis of results. Surg Endosc. 2008;22:8–15.
Cherng N, Witkowski ET, Sneider EB, Wiseman JT, Lewis J, Litwin DE, Santry HP, Cahan M, Shah SA. Use of cholecystostomy tubes in the management of patients with primary diagnosis of acute cholecystitis. J Am Coll Surg. 2012;214:196–201.
Winbladh A, Gullstrand P, Svanvik J, Sandström P. Systematic review of cholecystostomy as a treatment option in acute cholecystitis. HPB (Oxf). 2009;11:183–93.
Gomi H, Solomkin JS, Takada T, Strasberg SM, Pitt HA, Yoshida M, Kusachi S, Mayumi T, Miura F, Kiriyama S, Yokoe M, Kimura Y, Higuchi R, Windsor JA, Dervenis C, Liau K-H, Kim M-H. Tokyo Guideline Revision Committee: TG13 antimicrobial therapy for acute cholangitis and cholecystitis. J Hepatobiliary Pancreat Sci. 2013;20:60–70.
Gurusamy KS, Rossi M, Davidson BR. Percutaneouscholecystostomy for high-risk surgical patients with acute calculous cholecystitis. Cochrane Database Syst Rev. 2013;8:CD007088.
Campanile FC, Catena F, Coccolini F, Lotti M, Piazzalunga D, Pisano M, Ansaloni L. The need for new “patient-related” guidelines for the treatment of acute cholecystitis. World J Emerg Surg. 2011;6:44–6.
Abi-Haidar Y, Sanchez V, Williams SA, Itani KM. Revisiting percutaneous cholecystostomy for acute cholecystitis based on a 10-year experience. Arch Surg. 2012;147:416–22.
Morse BC, Smith JB, Lawdahl RB, Roettger RH. Management of acute cholecystitis in critically ill patients: contemporary role for cholecystostomy and subsequent cholecystectomy. Am Surg. 2010;76:708–12.
Joseph T, Unver K, Hwang GL, Rosenberg J, Sze DY, Hashimi S, Kothary N, Louie JD, Kuo WT, Hofmann LV, Hovsepian DM. Percutaneous cholecystostomy for acute cholecystitis: ten-year experience. J Vasc Interv Radiol. 2012;23:83–8.
McKay A, Abulfaraj M, Lipschitz J. Short- and long-term outcomes following percutaneous cholecystostomy for acute cholecystitis in high-risk patients. Surg Endosc. 2012;26:1343–51.
Kortram K, van Ramshorst B, Bollen TL, Besselink MG, Gouma DJ, Karsten T, Kruyt PM, Nieuwenhuijzen GA, Kelder JC, Tromp E, Boerma D. Acute cholecystitis in high risk surgical patients: percutaneous cholecystostomy versus laparoscopic cholecystectomy (CHOCOLATE trial): study protocol for a randomized controlled trial. Trials. 2012;13:1–7.
Houghton PW, Jenkinson LR, Donaldson LA. Cholecystectomy in the elderly: a prospective study. Br J Surg. 1985;72:220–2.
Griniatsos J, Petrou A, Pappas P, Revenas K, Karavokyros I, Michail OP, Tsigris C, Giannopoulos A, Felekouras E. Percutaneous cholecystostomy without interval cholecystectomy as definitive treatment of acute cholecystitis in elderly and critically ill patients. South Med J. 2008;101:586–90.
Li M, Li N, Ji W, Quan Z, Wan X, Wu X, Li J. Percutaneous cholecystostomy is a definitive treatment for acute cholecystitis in elderly high-risk patients. Am Surg. 2013;79:524–7.
Ha JP, Tsui KK, Tang CN, Siu WT, Fung KH, Li MK. Cholecystectomy or not after percutaneous cholecystostomy for acute calculous cholecystitis in high-risk patients. Hepatogastroenterology. 2008;55:1497–502.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the Hadassah Medical Center Ethics Committee and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Conflict of interest
Miklosh Bala, Ido Mizrahi, Haggi Mazeh, Jonathan Yuval, Ahmed Eid and Gidon Almogy declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Bala, M., Mizrahi, I., Mazeh, H. et al. Percutaneous cholecystostomy is safe and effective option for acute calculous cholecystitis in select group of high-risk patients. Eur J Trauma Emerg Surg 42, 761–766 (2016). https://doi.org/10.1007/s00068-015-0601-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-015-0601-1