Abstract
Purpose
This study investigated the clinical outcomes and follow-up results of venous sinus stenting (VSS) for constrictive dural sinus restoration in patients with intracranial dural arteriovenous fistulas (DAVFs). We hypothesized that this treatment would have durable benefits in such patients.
Methods
Patients who underwent VSS for DAVFs with cortical venous reflux between January 2008 and June 2020 were identified after a retrospective review (n =18). Clinical and endovascular treatment data and follow-up information were reviewed.
Results
The mean age of the 18 patients was 59.9 years. Stents were implanted in 10 previously occluded sinuses and 9 stenotic sinuses in addition to endovascular embolization. One patient received bilateral VSS. Subdural hemorrhage occurred in one patient after recanalization for embolization, followed by uneventful stenting. In 17 patients with clinical follow-up, the median follow-up time was 59.5 months (interquartile range 18 to 84 months). Of these, sixteen patients had follow-up vascular imaging, revealing AVF obliteration in 6 patients (38%) and stent patency in 11 (69%). Retreatment was performed for 8 (50%) patients. The mean follow-up modified Rankin scale score was 1.28. All patients had longstanding symptomatic improvement.
Conclusion
Restoration of sinus flow in DAVFs with cortical venous reflux through VSS has an acceptable complication rate and long-term symptomatic control; however, retreatment is often required, and stent occlusion is not uncommon.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Intracranial dural arteriovenous fistulas (DAVFs) are cerebrovascular malformations characterized by an abnormal connection between the dural arteries and venous sinus or cortical veins [1]. Progression of DAVF results in excess cerebral venous flow, reactive dural sinus constriction, and impaired cerebral venous drainage [2]. Therefore, outflow restriction and venous hypertension are major factors for hemorrhagic complications [3]. Endovascular treatment is effective against intracranial DAVFs because it immediately curtails the shunting flow into the cerebral venous system. Nevertheless, a sinus occlusive approach, particularly that administered via the transvenous route, is crucial for obtaining angiographic cure and achieving optimal embolization [4]; however, normal cerebral drainage function can be affected after treatment. Therefore, a sinus-preserving approach is a more attractive choice than the preceding methods because it enables the physiological venous drainage of brain tissue.
Dural sinus angioplasty and stenting have been proposed as a method for restoring venous outflow after sinus-preserving embolization for high-grade DAVFs with sinus stenosis or occlusion [5]. Moreover, in some cases stenting can help reduce the shunting flow [6]; however, dural sinus preservation occasionally reduces the likelihood of achieving complete DAVF obliteration. Therefore, despite the technical feasibility of sinus stenting for DAVF with sinus constriction, its safety, efficacy, and long-term outcomes are largely unknown. We hypothesized that a sinus-preserving approach involving venous sinus stenting (VSS) would be effective in treating DAVFs with sinus restriction.
Accordingly, this paper presents our single-center experience of a sinus-preserving approach involving dural sinus stenting in addition to embolization for the treatment of DAVFs.
Material and Methods
Study Population
The study was approved by our institutional review board, and individual patient informed consent was waived. A total of 322 patients with intracranial DAVFs were treated through endovascular therapy between January 2008 and June 2020. After a retrospective review of patient records, those who received dural sinus stents for downstream sinus stenosis or occlusion were identified and their DAVFs were classified according to the Cognard classification system [7]. Because restoration of normal venous outflow function may be potentially beneficial for brain circulation, adjunct VSS was considered to treat those with downstream sinus constriction. At our institute, stenting is applied to restore a diseased dural sinus if the following criteria are met: 1) a lateral sinus DAVF involving the dominant sinus and having a size >5 mm in normal segment and 2) a superior sagittal sinus or straight sinus DAVF with at least one normal downstream lateral sinus; however, stent implantation would not be considered if the following clinical conditions are observed: 1) massive venous infarct and large intraparenchymal hematoma and 2) failed guiding catheter placement into the dural sinus. On the basis of these criteria, our final analysis included 18 patients.
Endovascular Procedure
The endovascular procedure was performed by a board certified neurointerventionalist (C-W L) and at least one of two other neurointerventionalists (H-M L and Y‑H L) in the neuroangiography suite by using a biplane angiography machine with the patient under general anesthesia. Loading doses of dual antiplatelet agents, including 300 mg aspirin and 300 mg clopidogrel, were administered before or at the start of the procedure. Maintenance doses of 100 mg aspirin and 75 mg clopidogrel were continued for 6 months, followed by single agent indefinitely. Heparinization was not applied. Angioplasty was performed first with a monorail angioplasty balloon after successful wire crossing of the involved dural sinus to perform sinus stenting. For an occluded sinus, a 0.035-inch guidewire or 0.027-inch microcatheter-microwire kit (Progreat, Terumo, Tokyo, Japan) was used along with diagnostic or guiding catheters applied in a coaxial or triaxial manner. After angioplasty, stents of variable sizes and lengths were used; such stents were primarily self-expandable nitinol stents (Precise, Cordis, Santa Clara, CA, USA and Zilver Flex, Cook Medical, Bloomington, IN, USA) and occasionally balloon-expandable stent grafts (GraftMaster, Abbott, Chicago, IL, USA). Moreover, a guiding sheath was placed into the sinus if feasible to facilitate stent placement. In addition to stent placement, endovascular embolization was performed either before or after stent placement. The goal was to sufficiently reduce the flow of AVF. Notably, the embolization process was performed through either a transarterial or a transvenous route using various embolic materials, including coils, n‑butyl-2-cyanoacrylate (Histoacryl®, B. Braun, Melsungen, Germany), and ethylene-vinyl alcohol (Onyx™, Medtronic, Dublin, Ireland). Balloon-assisted techniques were not used during Onyx injection. The procedures were occasionally performed in stages, with the stenting and embolization performed at different sessions.
Clinical Assessment
Clinical data were retrieved from the electronic medical records of each patient for analysis. Presenting symptoms were classified as aggressive or nonaggressive symptoms [8]. Aggressive symptoms included focal neurological deficits, seizures, rapidly progressive dementia and hemorrhagic presentation; nonaggressive symptoms included ocular symptoms, pulsatile tinnitus, headache, dizziness, and vertigo. Radiological investigations, including catheter angiography and magnetic resonance imaging (MRI), were conducted routinely before the procedure. After the procedure, patients were instructed to undergo follow-up examination in clinics. Noninvasive radiological follow-up was ordered according to physicians’ requests. In principle, MRI was arranged 6 months after the procedure and annually thereafter. If there was evidence of disease progression on MRI, catheter angiography would be considered. All imaging studies were re-evaluated by two experienced neuroradiologists (C.W.L and Y.H.L with 19 and 11 years of experience, respectively). The conditions of presenting symptoms were investigated. In addition, any retreatment during the follow-up period was recorded. The clinical functional status was assessed using the modified Rankin scale (mRS).
Statistical Analysis
Herein, descriptive statistics are presented for demographic, angiographic, and clinical treatment data, and individual treatment details are provided. In addition, the follow-up results are presented. Because of the limited sample size, other inference statistics were not generated. All statistical analyses were performed using SAS software version 9.4 (SAS Institute, Cary, NC, USA).
Results
Study Patients
The mean age of the 18 study patients was 59.9 years (standard deviation 14.2 years) and 8 patients (44%) were male. Angiographic studies revealed that all patients had cortical venous reflux. Cortical venous reflux from an isolated sinus was noted in four patients, and the corresponding DAVFs were classified as Cognard type IV fistulas. The remaining patients had a visible flow in the sinus, and the corresponding DAVFs were classified as Cognard type IIB or IIA + B fistulas. In 16 patients, the fistulas were located in the lateral sinus, comprising the sigmoid, transverse sinus, and torcular region. One fistula was located in the posterior superior sagittal sinus, and one was in the falcotentorial region. Aggressive symptoms were observed in 13 patients (72%). The remaining five patients (28%) had headache, dizziness, tinnitus, and chemosis. Intracranial hemorrhage occurred in one patient (patient 2) at the time of diagnosis. Table 1 shows individual patient data.
Endovascular Treatment Strategy and Initial Angiographic Result
Because VSS was used as adjunct therapy, endovascular embolization, either via transarterial, transvenous, or combined routes, were planned in single or staged fashion. In 11 patients, embolization and stent placement were performed simultaneously. A patient (patient 5) only had minimal residual AVF after stent implantation and thus did not require further embolization. In patients with staged endovascular treatment, VSS was performed before embolization in four patients; in these patients, the interval between embolization and stenting ranged from 3 to 183 days and 4 patients received planned staged embolization after stenting within 1 month.
Stents were implanted in 10 previous angiographically occluded sinuses and 9 stenotic sinuses. Fig. 1 shows an illustrative case with occluded sinus (patient 5). Patient 10 received bilateral lateral sinus stents (Fig. 2). Angiographic complete obliteration was observed immediately in three patients (17%), subtotal obliteration (trace amount of residual Cognard type 1 flow without significant early venous drainage) in six patients (33%), and significant residual flow (substantial Cognard type 1 and type 2A flow) in nine patients (50%). During the procedure, acute stent occlusion occurred in two patients (patients 1 and 6), and thrombectomy was performed successfully in them. Patient 18 had subdural hemorrhage immediately after embolization, and underwent decompressive craniectomy. This patient received stent placement 6 months after embolization uneventfully. No new hemorrhage, venous infarcts, or focal neurological deficits occurred after stent implantation in any of the 18 patients.
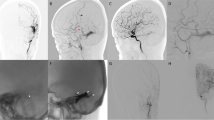
Illustrative case of stent placement in an isolated occluded right lateral sinus (patient 5). This 55-year-old man presented with hemianopia. a, b Frontal and lateral view arterial phase of right external carotid artery reveals a right sigmoid sinus Cognard type 4 dural arteriovenous fistula with an isolated right lateral sinus with cortical reflux into the vein of Labbé (asterisk). White arrows indicate locations of stenosis and occlusion at right transverse and sigmoid sinus. c A self-expandable stent (Zilver flex, Cook) was placed to right sigmoid sinus after angioplasty. The locations of stent edges are indicated by the black arrows. Transarterial embolization by Onyx was then performed after stent placement. d, e Lateral view arterial phase image after stent placement and embolization reveals nearly complete obliteration of fistula with residual trace amount of shunting flow at jugular bulb (dashed circle). Venous phase image shows antegrade flow of vein of Labbé draining into sigmoid sinus (asterisk). e Contrast-enhanced MR venogram 20 months after endovascular treatment reveals enhancement within stent, suggestive of patency. No evidence of fistula progression was found on MR angiography (not shown). The neurological symptom improved clinically
Patient 11 with bilateral sinus placements. This 80-year-old woman had left side weakness and consciousness disturbance at time of diagnosis. a, b Frontal view arterial phase of bilateral common carotid artery injection reveal dural arteriovenous fistula at bilateral lateral sinus and torcular area. There are right transverse sinus focal stenosis and left transverse sinus occlusion, as well as extensive venous reflux into the superior sagittal sinus and cortical veins (asterisk), suggestive of Cognard type 2A + B disease. c, d After the guiding catheter was navigated across the right jugular bulb, angioplasty was performed on the occluded segment of bilateral lateral sinuses. The stents were deployed thereafter sequentially in the same session (arrows). Combined transarterial and transvenous embolization was performed after stent placement. e, f Frontal view of bilateral common carotid artery injection after embolization reveals substantially decreased shunting flow but residual type 2A disease at torcular region with retrograde flow into superior sagittal sinus. g After 45 months later, follow-up MRI shows engorged cortical and medullary vein in the bilateral cerebral hemispheres, suggestive of disease progression and venous congestion. h Left common carotid angiography reveals recurrent fistula with superior sagittal sinus and cortical venous reflux (asterisks) and occlusion of previous deployed stents (arrow). i Salvage transvenous dural sinus occlusion by coils was performed. j, k Bilateral common carotid artery shows obliteration of fistula after embolization. l Follow-up MRI 79 months after initial diagnosis shows normalization of engorged cortical and medullary veins. Besides occasional dizziness, the patient was in independent functional status
Follow-Up Results and Retreatment
One patient was lost to follow-up after 2 months, and the remaining patients had at least 5 months of follow-up (median 59.5 months; interquartile range, IQR 18–84 months). Among the 17 patients who received adequate clinical follow-up, 16 underwent vascular imaging studies (median 25 months; IQR 10–51 months). Among patients who underwent follow-up vascular imaging 5 (31%) had AVF obliteration and 11 (69%) had stent patency. On the other hand, eight (50%) had progressive recurrent AVF as observed through radiological assessment, including one (patient 1) with previous complete obliteration. Cortical venous reflux occurred in five patients (63%) with progressive recurrent AVF. The remaining three patients (19%) had stable disease status. In four patients with Cognard type IV DAVFs (isolated sinus), AVF obliteration was persistent in all patients during follow-up; patent stents were noted in three patients (75%).
In clinical assessment six patients were asymptomatic during follow-up (35%), particularly patients with Cognard type IIA + B or IIB DAVFs (5 patients, 83%). In other patients, the presenting symptoms improved, but they still had minor symptoms or pre-existing neurological deficits. As for functional status, the mean mRS score at follow-up was 1.28 (SD: 1.45). Patient 4 had a mRS score of 3 and was lost to follow-up because of a psychiatric disorder. Patient 16 had left M1 middle cerebral artery large vessel occlusion stroke 3 years after DAVF treatment, with a final mRS score of 5 after the event; however, other patients had favorable functional outcomes (mRS score ≤2).
Retreatment was conducted in all of 8 patients with progressive AVF. In patients 11 and 17 the diseased sinuses with permanent stent occlusion were expunged to achieve complete AVF obliteration. In patient 10 cerebral congestion was noted 4 years after stenting. The angiographic study revealed stent occlusion, and angioplasty was performed on the stent in addition to AVF embolization. In patient 13 Onyx embolization via puncturing to skull intraosseus vascular channel was performed due to limited access route. The remaining patients all received standard endovascular embolization. Angiographic complete obliteration was observed in three patients (38%), subtotal obliteration (trace amount of residual Cognard type 1 flow without significant early venous drainage) in four patients (50%), and significant residual flow (type 2A flow) in one patient (12%). No periprocedural complication was found. The clinical symptoms and functional status were stable during further follow-up.
Discussion
Patients with high-grade intracranial DAVFs often experience an unfavorable natural history if untreated. Our results revealed that the dural sinus could be restored through stent implantation with acceptable complication rates. During the long-term follow-up in this study, stent patency was noted to be maintained in 69% of the patients; however, only 38% of the patients could achieve angiographic AVF obliteration, and up to 50% of the patients needed retreatment. Notably, all patients had symptomatic improvement compared with the preoperative condition. Therefore, we believe that adjunct dural sinus stenting is a reasonable treatment approach when carefully planned, especially in previously occluded sinuses; however, this approach would require long-term follow-up.
In DAVFs with cortical venous reflux, the annual hemorrhage rate is 6% in patients with Borden type II and 10% in those with Borden type III fistulas [9, 10]. Therefore, in such patients, treatment is mandatory with an endovascular or surgical approach being the preferred method for immediate risk reduction. The worst complication of endovascular treatment is hemorrhage, which typically occurs because of venous hemorrhage from compromised cerebral venous drainage. Japanese registry data revealed the occurrence of complications in 7.7% of cases [11]. Therefore, sinus restoration could be a more physiological method in treating this disease than sinus sacrifice. In our study, only one patient had intracranial hemorrhage after embolization without stenting. Because the hematoma was located in the subdural space after initial embolization, it was considered a procedure-related complication of transvenous embolization. Overall, the complication rate is comparable to that in a large cohort study; however, prudency related to technical issues is warranted in this approach, especially with high-grade stenosis or occlusion.
Nevertheless, the relatively low angiographic cure rate based on our results is a concern for this approach. Ertl et al. revealed that a sinus-preserving approach had a lower cure rate than a sinus occlusive approach (71% vs. 93%) [12]. The likelihood of angiographic cure rate by sinus-preserving embolization can be enhanced by dedicated techniques, such as transarterial dual-lumen scepter balloon or transvenous balloon-assisted Onyx injection [13, 14]. A study by Vollherbst et al. reported that combined venous sinus balloon-assisted protection and transarterial Onyx injection can achieve cure rate of 86.4% [15]. Our results reveal a relatively low angiographic cure rate. The possible reason for this inconsistency could be because we did not use supplementary technique during liquid embolic agent injection. Notably, a recurrence after initial complete angiographic occlusion during follow-up is not uncommon. Ambekar et al. reported that asymptomatic recurrence could occur in 12.3% of patients during follow-up after angiographic cure with Onyx [16]. Hence, clinical follow-up is always required. In our patients, the decision for retreatment was based on imaging findings, and we did not wait until symptom progression. There is possibility that some recurrence might be occult on MRI; but they are usually clinically silent; however, no patient needed emergency retreatment. Nonetheless, although the presenting symptoms seldom disappeared, patients generally experienced symptomatic improvement and had excellent functional status unless burdened by other unrelated diseases. Our results suggest that this treatment approach could maintain long-term efficacy despite the low initial cure rate.
Notably, dural sinus stenting has several applications in the treatment of cerebrovascular disease [17, 18]. In DAVFs, a sinus stent is used for diseases occurring at the lateral sinus [5, 19]; however, several reports have indicated the application of this approach at various locations, including the torcular, superior sagittal, and straight sinuses [20,21,22]. A series by Levrier et al. reported poststent insertion outcomes in 10 patients, and all patients achieved cure or symptomatic improvement [23]. In their series, all diseases were located in the lateral sinus, and only 30% of patients had cortical venous reflux; the mean follow-up period was 21.1 months. They concluded that stent placement is a promising technique for DAVF treatment. Furthermore, Liebig et al. reported four low-grade lateral sinus DAVFs treated using only angioplasty and stent deployment, achieving satisfactory results [6]. Our results are concordant with their conclusions and we think that stent treatment could be efficient even in patients with cortical venous reflux during a longer follow-up period. Notably, stent placement in chronic stenotic or occluded sinuses is technically demanding in some cases. Nonetheless, crossing a chronically occluded sinus is frequently achievable, which is the foundation of transvenous embolization [24]. Advancements in neurovascular guiding catheters and peripheral or carotid self-expandable stents have facilitated the tracking of a stent to cross the tortuous dural sinus.
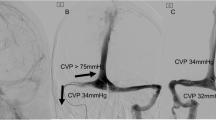
Notably, only one patient in our series had an initial presentation of intracranial hemorrhage. In patients who already had a severe hemorrhage, neurosurgical decompression was often required immediately, thereby creating a concern for the use of dual antiplatelet agents after stenting; however, whether patients with hemorrhage are suitable for sinus stents is still inconclusive. Nevertheless, a stepwise approach, wherein AVF is embolized first and the stenting is delayed, might prove to be a feasible strategy for long-term sinus restoration. We do not consider hemorrhage to be an absolute contraindication for dural sinus stent placement, and in certain circumstances acute dural sinus stenting might be helpful [25]; however, there is requirement for further investigation on criteria for sinus stent placement on the basis of morphological and technical parameters. Evidence of impaired venous collateral routes might help in the selection of the appropriate treatment strategy [26]. Therefore, quantitative measurements, such as digital subtraction angiography or sinus pressure gradient measurement, delineating the severity of sinus restriction could be a useful tool in patient selection and follow-up [27, 28]. Nevertheless, further investigation is required to refine the criteria for patient selection.
Our study had some limitations. First, the retrospective nature of this study could have exerted a risk of selection bias; however, because of the relatively low prevalence of DAVFs and complex anatomical variations, a prospective design was not feasible. Second, our study cohort sample size was small. Nevertheless, the number of patients in our series is larger than those previously reported. Third, relatively heterogeneous treatment strategies were applied to our patients. Because the period was relatively long, the experience and device advancement during embolization treatment might have changed over time. We believe that embolization outcomes have improved in more recent cases. Fourth, the criteria for sinus stent placement were somehow subjective. Nevertheless, all procedures were performed by the same neurointerventionalist, thereby minimizing the variability between patients.
In conclusion, the use of stenting in addition to embolization for treating DAVFs with outflow restriction is safe and durable in most cases; however, the angiographic cure rate is low, and AVF progression is common. Moreover, long-term surveillance is required, and any retreatment can be performed accordingly.
References
Miller TR, Gandhi D. Intracranial Dural Arteriovenous Fistulae: Clinical Presentation and Management Strategies. Stroke. 2015;46:2017–25.
Cognard C, Casasco A, Toevi M, Houdart E, Chiras J, Merland JJ. Dural arteriovenous fistulas as a cause of intracranial hypertension due to impairment of cranial venous outflow. J Neurol Neurosurg Psychiatry. 1998;65:308–16.
Hu YS, Lin CJ, Wu HM, Guo WY, Luo CB, Wu CC, Chung WY, Liu KD, Yang HC, Lee CC. Lateral Sinus Dural Arteriovenous Fistulas: Sinovenous Outflow Restriction Outweighs Cortical Venous Reflux as a Parameter Associated with Hemorrhage. Radiology. 2017;285:528–35.
Carlson AP, Alaraj A, Amin-Hanjani S, Charbel FT, Aletich V. Endovascular approach and technique for treatment of transverse-sigmoid dural arteriovenous fistula with cortical reflux: the importance of venous sinus sacrifice. J Neurointerv Surg. 2013;5:566–72.
Murphy KJ, Gailloud P, Venbrux A, Deramond H, Hanley D, Rigamonti D. Endovascular treatment of a grade IV transverse sinus dural arteriovenous fistula by sinus recanalization, angioplasty, and stent placement: technical case report. Neurosurgery. 2000;46:497–500; discussion 500–1.
Liebig T, Henkes H, Brew S, Miloslavski E, Kirsch M, Kühne D. Reconstructive treatment of dural arteriovenous fistulas of the transverse and sigmoid sinus: transvenous angioplasty and stent deployment. Neuroradiology. 2005;47:543–51.
Cognard C, Gobin YP, Pierot L, Bailly AL, Houdart E, Casasco A, Chiras J, Merland JJ. Cerebral dural arteriovenous fistulas: clinical and angiographic correlation with a revised classification of venous drainage. Radiology. 1995;194:671–80.
Lasjaunias P, Chiu M, ter Brugge K, Tolia A, Hurth M, Bernstein M. Neurological manifestations of intracranial dural arteriovenous malformations. J Neurosurg. 1986;64:724–30.
Gross BA, Du R. The natural history of cerebral dural arteriovenous fistulae. Neurosurgery. 2012;71:594-602; discussion 602–3.
Borden JA, Wu JK, Shucart WA. A proposed classification for spinal and cranial dural arteriovenous fistulous malformations and implications for treatment. J Neurosurg. 1995;82:166–79.
Hiramatsu M, Sugiu K, Hishikawa T, Nishihiro S, Kidani N, Takahashi Y, Murai S, Date I, Kuwayama N, Satow T, Iihara K, Sakai N. Results of 1940 embolizations for dural arteriovenous fistulas: Japanese Registry of Neuroendovascular Therapy (JR-NET3). J Neurosurg. 2019. https://doi.org/10.3171/2019.4.JNS183458. Epub ahead of print.
Ertl L, Brückmann H, Kunz M, Crispin A, Fesl G. Endovascular therapy of low- and intermediate-grade intracranial lateral dural arteriovenous fistulas: a detailed analysis of primary success rates, complication rates, and long-term follow-up of different technical approaches. J Neurosurg. 2017;126:360–7.
Jang CK, Kim BM, Park KY, Lee JW, Kim DJ, Chung J, Kim JH. Scepter dual-lumen balloon catheter for Onyx embolization for dural arteriovenous fistula. BMC Neurol. 2021;21:31.
Kerolus MG, Chung J, Munich SA, Matsuda Y, Okada H, Lopes DK. An Onyx tunnel: reconstructive transvenous balloon-assisted Onyx embolization for dural arteriovenous fistula of the transverse-sigmoid sinus. J Neurosurg. 2018;129:922–7.
Vollherbst DF, Ulfert C, Neuberger U, Herweh C, Laible M, Nagel S, Bendszus M, Möhlenbruch MA. Endovascular Treatment of Dural Arteriovenous Fistulas Using Transarterial Liquid Embolization in Combination with Transvenous Balloon-Assisted Protection of the Venous Sinus. AJNR Am J Neuroradiol. 2018;39:1296–302.
Ambekar S, Gaynor BG, Peterson EC, Elhammady MS. Long-term angiographic results of endovascularly “cured” intracranial dural arteriovenous fistulas. J Neurosurg. 2016;124:1123–7.
Levitt MR, Albuquerque FC, Gross BA, Moon K, Jadhav AP, Ducruet AF, Crowley RW. Venous sinus stenting in patients without idiopathic intracranial hypertension. J Neurointerv Surg. 2017;9:512–5.
Xu K, Yu T, Yuan Y, Yu J. Current Status of the Application of Intracranial Venous Sinus Stenting. Int J Med Sci. 2015;12:780–9.
Choi BJ, Lee TH, Kim CW, Choi CH. Reconstructive treatment using a stent graft for a dural arteriovenous fistula of the transverse sinus in the case of hypoplasia of the contralateral venous sinuses: technical case report. Neurosurgery. 2009;65:E994–6; discussion E996.
Takada S, Isaka F, Nakakuki T, Mitsuno Y, Kaneko T. Torcular dural arteriovenous fistula treated via stent placement and angioplasty in the affected straight and transverse sinuses: case report. J Neurosurg. 2015;122:1208-13.
Ohara N, Toyota S, Kobayashi M, Wakayama A. Superior sagittal sinus dural arteriovenous fistulas treated by stent placement for an occluded sinus and transarterial embolization. A case report. Interv Neuroradiol. 2012;18:333–40.
Troffkin NA, Graham CB 3rd, Berkmen T, Wakhloo AK. Combined transvenous and transarterial embolization of a tentorial-incisural dural arteriovenous malformation followed by primary stent placement in the associated stenotic straight sinus. Case report. J Neurosurg. 2003;99:579–83.
Levrier O, Métellus P, Fuentes S, Manera L, Dufour H, Donnet A, Grisoli F, Bartoli JM, Girard N. Use of a self-expanding stent with balloon angioplasty in the treatment of dural arteriovenous fistulas involving the transverse and/or sigmoid sinus: functional and neuroimaging-based outcome in 10 patients. J Neurosurg. 2006;104:254–63.
Lekkhong E, Pongpech S, Ter Brugge K, Jiarakongmun P, Willinsky R, Geibprasert S, Krings T. Transvenous embolization of intracranial dural arteriovenous shunts through occluded venous segments: experience in 51 Patients. AJNR Am J Neuroradiol. 2011;32:1738–44.
Chen KW, Lin YH, Lee CW. Acute Posttraumatic Cerebral Venous Sinus Thrombosis-Induced Malignant Increased Intracranial Pressure Treated with Endovascular Dural Sinus Thrombectomy and Stenting. World Neurosurg. 2019;128:393–7.
Lin YH, Lee CW, Wang YF, Lu CJ, Chen YF, Liu HM. Engorged medullary vein on CT angiography in patients with dural arteriovenous fistula: prevalence, types, and comparison between regional and extensive types. J Neurointerv Surg. 2018;10:1114–9.
Guo WY, Lee CJ, Lin CJ, Yang HC, Wu HM, Wu CC, Chung WY, Liu KD. Quantifying the Cerebral Hemodynamics of Dural Arteriovenous Fistula in Transverse Sigmoid Sinus Complicated by Sinus Stenosis: A Retrospective Cohort Study. AJNR Am J Neuroradiol. 2017;38:132–8.
Rivera-Rivera LA, Johnson KM, Turski PA, Wieben O. Pressure Mapping and Hemodynamic Assessment of Intracranial Dural Sinuses and Dural Arteriovenous Fistulas with 4D Flow MRI. AJNR Am J Neuroradiol. 2018;39:485–7.
Acknowledgements
This manuscript was edited by Wallace Academic Editing.
Funding
This study was supported by grant from Ministry of Science and Technology, Taiwan (MOST 109-2314-B-002-097-).
Author information
Authors and Affiliations
Contributions
All authors made (1) substantial contributions to the conception or design of the work or the acquisition, analysis or interpretation of data for the work; (2) drafting of the work or revising it critically for important intellectual content; (3) final approval of the version to be published; and (4) are in agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
Y.-H. Lin, C.-W. Lee and H.-M. Liu declare that they have no competing interests.
Ethical standards
The work was approved by research ethics committee of National Taiwan University Hospital (Approval number: 202010071RIND). Individual informed consent was waived.
Additional information
Availability of Data and Material
Data can be made available by contacting the corresponding author via email under reasonable request.
Code Availability
The SAS code can be made available by contacting the corresponding author via email under reasonable request.
Rights and permissions
About this article
Cite this article
Lin, YH., Lee, CW. & Liu, HM. Clinical outcomes of adjunct sinus stenting in dural arteriovenous fistulas. Clin Neuroradiol 32, 455–464 (2022). https://doi.org/10.1007/s00062-021-01031-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-021-01031-3