Abstract
Background
Pericardial effusion (PE) is a common clinical condition that can develop as a result of systemic or cardiac diseases. Here, we report the results of cytology for patients who underwent pericardiocentesis for PE.
Methods
The study comprised 283 patients who underwent primary percutaneous pericardiocentesis between 2007 and 2016. The mean age of the patients was 60.0 ± 16.6 years; 162 (57.2%) were male and 121 (42.8%) were female. The presence of reactive mesothelial cells, acute and chronic inflammatory cells, and/or blood without evidence of malignant cells was considered as benign. The presence of malignant cells with/without reactive mesothelial cells, inflammatory cells, and/or blood was considered as malignant.
Results
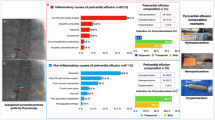
The vast majority of PE specimens (219 cases; 77.4%) were classified as benign. Only 20 cases (7.1%) were classified as atypical, and malignant cells were present in the PE specimens of 44 cases (15.5%). The most common diagnosis was benign PE. The most commonly encountered malignancy was lung cancer. The rate of malignancy was 1.9% in the serous group and 24% in the hemorrhagic group, which was statistically significant.
Conclusion
Benign PE was the most frequent cytological diagnosis in our study. Chronic nonspecific pericarditis was the most frequent type of pericarditis in the benign PE group, while lung adenocarcinoma was the most frequent malignancy in the malignant PE group. The rate of malignancy was significantly higher in the hemorrhagic group than in the serous group.
Zusammenfassung
Hintergrund
Ein Perikarderguss (PE) ist ein häufiges klinisches Ereignis, dass infolge systemischer oder kardialer Erkrankungen auftreten kann. Im vorliegenden Beitrag wird über die zytologischen Ergebnisse von Patienten berichtet, bei denen wegen eines PE eine Perikardpunktion erfolgte.
Methoden
Die Studie umfasste 283 Patienten, bei denen zwischen 2007 und 2016 eine primäre Perikardpunktion durchgeführt wurde. Das Durchschnittsalter der Patienten lag bei 60,0 ± 16,6 Jahren; 162 (57,2 %) Männer und 121 (42,8 %) Frauen. Das Vorliegen reaktiver Mesothelialzellen, akuter und chronischer Entzündungszellen und/oder von Blut ohne Anhalt für maligne Zellen wurde als benigne eingestuft. Das Vorliegen maligner Zellen mit/ohne reaktive Mesothelialzellen, Entzündungszellen und/oder Blut wurde als maligne eingestuft.
Ergebnisse
Die überwiegende Mehrheit der PE-Punktate (219 Fälle; 77,4 %) wurde als benigne klassifiziert. Nur 20 Fälle (7,1 %) wurden als atypisch eingestuft, und in den PE-Punktaten von 44 Fällen (15,5 %) lagen maligne Zellen vor. Die häufigste Diagnose war ein benigner PE. Häufigstes Malignom war das Bronchialkarzinom. Die Malignomrate betrug 1,9 % in der Gruppe mit serösem PE, aber 24 % in der Gruppe mit hämorrhagischem PE, was statistisch signifikant war.
Schlussfolgerung
Ein benigner PE war die häufigste zytologische Diagnose in der vorliegenden Studie. In der Gruppe mit benignem PE kam als häufigster Typ der Perikarditis die chronische nichtspezifische Form vor, während in der Gruppe mit malignem PE das Bronchialkarzinom das häufigste Malignom darstellte. Die Malignomrate war in der Gruppe mit hämorrhagischem PE signifikant höher als in der Gruppe mit serösem PE.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Pericardial effusion (PE) is a common clinical condition that can develop as a result of systemic or cardiac diseases. PE has a broad spectrum starting from mild asymptomatic effusion to tamponade. It may develop slowly or suddenly [1,2,3] with infectious or noninfectious etiology [4].
Any condition that creates an inflammatory process causes increased production of pericardial fluid and may lead to the formation of exudative fluid. Other alternative mechanisms such as increased systemic venous pressure secondary to congestive heart failure and pulmonary hypertension lead to decreased reabsorption and thereby transudative effusion [5]. The most common causes of PE are infections (viral, bacterial, e. g., tuberculosis), cancer, connective tissue diseases, pericardial injury syndromes (postpericardiotomy syndromes, posttraumatic pericarditis, postmyocardial infarction effusions), metabolic causes (hypothyroidism, renal insufficiency), and myopericardial diseases. Moreover, PE may develop as a result of the opening of an aortic dissection into the pericardium or the use of drugs such as minoxidil. PE may occur after hypoalbuminemia, which develops secondary to hydropericardium, noninflammatory transudative PE, heart failure, cardiac cirrhosis, or nephrotic syndrome [5].
The cause of PE is found to be idiopathic in nearly 50% of cases in developed countries [6]. The other observed causes are: 10–25%, cancer; 15–30%, pericarditis and infectious causes; 15–20%, iatrogenic causes; and 5–15%, connective tissue diseases [6]. More than 60% of PE is caused by tuberculosis in developing countries and in areas where tuberculosis is endemic [6]. In the case of pericarditis with PE, malignant or infectious causes vary between 15 and 50% [7].
Data on the pericardial cytology of PE are limited. We therefore aimed to report the results of pericardial cytology in patients who underwent pericardiocentesis owing to PE.
Patients and methods
Patients who underwent primary percutaneous pericardiocentesis between 2007 and 2016 were enrolled in the study retrospectively. Patients in whom surgical pericardial window placement was performed were excluded from the study as were patients with iatrogenic PE. All samples were sent to the pathology laboratory for cytological examination.
Pathology
Fresh pericardial fluid specimens were delivered to the pathology laboratory. The specimens were centrifuged at 1,500 rpm for 15 min. One drop of sediment was placed in the cytospin chamber. Two cytospin slides and a cell block were prepared for each specimen. Of the two cytospin slides, one was air-dried and the other was fixed in 95% alcohol. The air-dried slide was stained using May-Grünwald–Giemsa (MGG) staining and the 95% alcohol-fixed slide with Papanicolaou (PAP) staining. Immunohistochemical staining was performed on the cell block in selected cases. The cytological microscopic interpretation was divided into three general categories: “benign,” “malignant,” and “atypical.” The presence of reactive mesothelial cells, acute and chronic inflammatory cells, and/or blood without evidence of malignant cells was considered as benign. The benign category included nonspecific (neutrophilic and lymphocytic) inflammation and specific (rheumatoid) inflammation. The presence of malignant cells with/without reactive mesothelial cells, inflammatory cells, and/or blood was considered as malignant. For the malignant cases, tumor type and/or favored primary tumor site were stated based on cytomorphologic characteristics and immunohistochemical staining performed on the cell block. Specimens that included rare cells with atypical cytological features, and when a definitive diagnosis (benign versus malignant) could not be made, were considered as atypical.
Statistical analyses
Continuous variables are presented as mean ± standard deviation or median (interquartile range), while categorical variables are presented as numbers and percentages. Categorical variables were compared using chi-square or Fisher’s exact tests when appropriate. A two-sided p value of less than 0.05 was considered significant in all analyses. The collection and statistical analysis of data were performed using SPSS 11.5 (SPSS Inc., Chicago, Ill.).
Results
The study comprised 283 patients whose mean age was 60.0 ± 16.6 years; 162 (57.2%) patients were male and 121 (42.8%) were female. The cytological diagnoses of the patients are listed in Table 1. The vast majority of PE specimens (219 cases; 77.4%) were classified as benign. Only 20 cases (7.1%) were classified as “atypical” in terms of cytology, and malignant cells were present in the PE specimens of 44 cases (15.5%). The most common diagnosis was benign PE.
The cytological diagnoses of benign PE are shown in Table 2. Of 219 benign PE cases, 52 (23.7%) were acute nonspecific pericarditis, 166 (75.7%) were chronic nonspecific pericarditis, and one (0.45%) was cholesterol pericarditis (rheumatoid inflammation) according to the clinical, radiologic, biochemical, immunologic, and microbiologic background of the patient. The most prevalent type of pericarditis was chronic nonspecific pericarditis in the benign group.
The cytological diagnoses of malignant PE are shown in Table 3. Of 44 malignant PE cases, four (9.1%) were mesothelioma and 40 (90.1%) were metastasis (two hematologic malignancy and 38 carcinomas). Examining the primary origins of metastatic carcinomas indicated that 24 originated from the lung (24 adenocarcinoma), three from the upper gastrointestinal tract (one signet ring gastric carcinoma and two cases of esophageal cancer), one from the breast, one from the cervix, and one from the kidney. The primary origins of the other eight cases were indefinite. The most commonly encountered malignancy was lung cancer. Adenocarcinoma was detected in all patients with lung cancer. The second most common malignancy was cancer of unknown primary origin and the third was mesothelioma.
The cytological diagnoses of serous and hemorrhagic effusion are shown in Table 4. Chronic nonspecific pericarditis was identified as the most common cause in both the serous and hemorrhagic groups. The rate of malignancy was found to be 1.9% in the serous group and 24% in the hemorrhagic group (p < 0.001).
Discussion
In our study, benign PE was the most frequently observed cytological diagnosis of PE. Chronic nonspecific pericarditis was detected as the most frequent pericarditis in the benign group. Lung adenocarcinoma was the most frequently detected malignancy in the malignant PE group. The rate of malignancy was significantly higher in the hemorrhagic group than in the serous group.
Guidelines on pericardial disease differentiate between acute and chronic pericarditis according to the clinical duration, starting from the onset of disease [8]. In our study, the benign PE group was divided into three groups according to cytology findings as: (a) acute nonspecific pericarditis, (b) chronic nonspecific pericarditis, and (c) specific cholesterol pericarditis (rheumatoid inflammation). We think that nonspecific pericarditis may include all of the diseases mentioned in the guidelines in both groups. However, in our study, we did not specify the clinical features because our main aim was to report on the cytological data. There are different reports in the literature regarding the cytology study results of PE. In the pericardial cytology study of Dragoescu and Liu, 74.2% of patients had benign PE [9]. In a single-center study with 6‑year pericardial cytology data of 116 patients with PE, the rate of malignancy was higher [10]. In our study, 77.4% of the effusions were detected as benign. There was one case of cholesterol pericarditis in our study [11]. Cholesterol pericarditis is an uncommon condition, characterized by increased levels of cholesterol in the pericardial fluid [12, 13]. Although it is usually idiopathic, it may develop secondary to some systemic diseases such as rheumatoid arthritis, tuberculosis, and hypothyroidism [14].
Malignant invasion of the pericardium can be seen in about 10% of all cancer patients, and one third of these patients die as a result of this involvement. Primary tumors of the pericardium are very rare, whereas secondary or metastatic tumors of the pericardium are quite common; lung cancer, breast cancer, melanoma, and hematological malignancies constitute the majority of the latter [15,16,17,18]. Dragoescu and Liu indicated that pericardial fluid cytology is better than pericardial biopsy for detecting malignancy [9]. The diagnosis of malignant PE is made on the basis of positive results from cytological examinations of pericardial fluid. Although pericardial fluid cytology is specific, its sensitivity varies. Despite the frequent examination of tumor markers after pericardiocentesis, their diagnostic use has not been well established [19]. In a study including 140 Chinese patients undergoing pericardiocentesis, malignancy and tuberculosis were detected as the most common cause of moderate and large effusion. Lung cancer was the most common cause among the malignancies [20].
In a series including 322 patients with moderate and severe PE, idiopathic pericarditis was found in 20%, iatrogenic causes in 16%, cancer in 13%, and chronic idiopathic PE in 9% of the patients. In 192 (60%) of the patients, the cause of the effusion was a known medical condition [21]. In a single-center study examining the 6‑year pericardial cytology data of 116 patients with PE, 74 patients had malignancy clinically, 39 patients did not have malignancy, and in three patients no diagnosis could be made. Lung cancer was the most common cause among the patients with malignancy [10]. In a pericardial cytology study conducted by Dragoescu and Liu that included 128 patients, 95 (74.2%) patients had benign PE, 31 (24.2%) patients had malignant PE, and two patients had severe atypical cells. The benign etiology included 23.1% of cases of neoplasia, 19% of cases with an idiopathic cause, 14.7% of cases with an infectious cause, and 12.6% of cases of connective tissue disease. The most commonly detected cancer was lung adenocarcinoma. Lung cancer was the most commonly seen malignancy in both genders, 75% of men and 52.2% of women. In women, breast cancer (39.1%) was the second most common tumor [9]. In this study, the main point to be emphasized was that the differentiation of benign and malignant could be made easily via cytological examination in 98.4% of the specimens. Sometimes cytological examination cannot distinguish malignant mesothelial cells from reactive mesothelial cells or metastatic cells. These different diagnoses may include primary tumors such as mesothelioma, tumors such as angiosarcoma or rhabdomyosarcoma that originate from the pericardium, mediastinal tumors, chronic inflammation such as tuberculosis, or metastatic tumors [22,23,24,25,26]. In our study, in 7.1% of the patients, the differentiation between benign and malignant could not be made with cytological methods. Moreover, the primary origin of the tumor could not be determined by cytology in 18.1% of patients with malignant PE in our study.
PE formation from malignancies is a multifactorial process and can also be caused by treatment modalities, especially radiotherapy treatment, direct proliferation of the tumor in the pericardium, hematogenous or lymphatic pericardial spread, or processes such as uremia or thrombocytopenia [27]. Similar to reports to the literature, lung cancer was detected as the most frequent malignancy in our study. All of the lung cancer cases were adenocarcinoma. This finding is consistent with previous studies in the literature.
Mesothelioma is a malignancy originating from the epithelial cells of the mesothelium. Primary malignant pericardial mesotheliomas are very rare, and their incidence is 0.0022% [28]. It commonly affects men and develops in the fifth and seventh decades of life [29]. Primary malignant pericardial mesotheliomas make up 0.7% of all mesotheliomas, and primary malignant pericardial mesothelioma is the most common cause of primary pericardial tumors, accounting for nearly half of them [30]. Four patients were found to have mesothelioma in our study. This higher rate compared with the literature can be attributed to asbestos, which is still widely used in our country.
Patients with hemorrhagic PE are more likely to have malignancy as the etiologic cause compared with those with serous PE. In a study conducted by Meyers et al. of 175 patients, it was found that PE was hemorrhagic in 80% of patients with malignancy and this was statistically significant [31]. In another study by Colombo et al., it was found that neoplasia (36%), idiopathic pericarditis (32%), and uremia (20%) were the most frequent etiological causes in 25 patients with severe PE, and all cases of PE in the neoplastic group were hemorrhagic [32]. In a study conducted by Zayas et al. of 100 patients with acute pericarditis, 27 patients underwent pericardiocentesis, and benign causes were significantly higher in the serous pericardial fluid group [33]. In line with the literature, we also found that the malignancy rate was significantly higher in the hemorrhagic group than in the serous group.
Limitations
The main limitation of our study is that it is single-center study and was not carried out as a randomized and prospective study. The absence of clinical characteristics and follow-up data of the patients are other limiting factors.
Conclusion
In our study, benign PE was the most frequent cytological diagnosis of PE. Chronic nonspecific pericarditis was detected as the most frequent type of pericarditis in the benign group. Lung adenocarcinoma was the most frequently detected malignancy in the malignant PE group. The rate of malignancy was significantly higher in the hemorrhagic group than in the serous group. Cytological investigation cannot alone determine the primary origin of a tumor in patients with malignant PE. In the stetting of malignant PE, a multidisciplinary team involving cardiologists, oncologists, radiologists, and possibly gynecologists is essential for an early diagnosis and for appropriate management.
Change history
20 July 2017
An erratum to this article has been published.
References
Shabetai R (2004) Pericardial effusion: haemodynamic spectrum. Heart 90:255–256
Shabetai R (1999) Function of the normal pericardium. Clin Cardiol 22:I4–5
Spodick DH (2003) Acute cardiac tamponade. N Engl J Med 349:684–690
Braunwald E (1996) Pericardial disease. In: Braunwald E, Fauci AS, Kasper DL, Hauser SL, Lohr DC, Jameson JL (eds) Principles of Internal Medicine, 14th edn. McGraw-Hill, New York, pp 1334–1341
Imazio M, Adler Y (2013) Management of pericardial effusion. Eur Heart J 34:1186–1197
Syed FF, Ntsekhe M, Mayosi BM (2010) Tailoring diagnosis and management of pericardial disease to the epidemiological setting. Mayo Clin Proc 85:866 (author reply 866)
Imazio M, Cecchi E, Demichelis B et al (2007) Indicators of poor prognosis of acute pericarditis. Circulation 115:2739–2744
Adler Y, Charron P, Imazio M et al (2015) ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2015(36):2921–2964
Dragoescu EA, Liu L (2013) Pericardial fluid cytology: an analysis of 128 specimens over a 6-year period. Cancer Cytopathol 121:242–251
He B, Yang Z, Zhao P et al (2017) Cytopathologic analysis of pericardial effusions in 116 cases: Implications for poor prognosis in lung cancer patients with positive interpretations. Diagn Cytopathol 45(4):287–293
Gecmen C, Gecmen GG, Kahyaoglu M et al (2016) Pericardial effusion due to cholesterol pericarditis in case of rheumatoid arthritis. Echocardiography 33:1614–1616
Alexander SJ (1919) A pericardial effusion of “gold paint” appearance due to presence of cholesterin. Br Med J 2:463
Brawley RK, Vasko JS, Monrow AG (1966) Cholesterol pericarditis: consideration of its pathogenesis and treatment. Am J Cardiol 41:235–248
Knobel B, Rosman P (2001) Cholesterol pericarditis associated with rheumatoid arthritis. Harefuah 140:10–12 (87)
Mukai K, Shinkai T, Tominaga K et al (1988) The incidence of secondary tumours of the heart and pericardium: a 10 year study. Jpn J Clin Oncol 18:195–201
Posner MR, Cohen GI, Skarin AT (1981) Pericardial disease in patients with cancer. Am J Med 71:407–413
Wilding G, Green HL, Longo DL et al (1988) Tumours of the heart and pericardium. Cancer Treat Rev 15:165–181
Maisch B, Seferovic PM, Ristic AD et al (2004) Task Force on the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology. Guidelines in the diagnosis and management of pericardial diseases. Eur Heart J 25:587–610
Karatolios K, Pankuweit S, Maisch B (2013) Diagnostic value of biochemical biomarkers in malignant and non-malignant pericardial effusion. Heart Fail Rev 18:337–344
Ma W, Liu J, Zeng Y et al (2012) Causes of moderate to large pericardial effusion requiring pericardiocentesis in 140 Han Chinese patients. Herz 37:183–187
Sagristà-Sauleda J, Mercé J, Permanyer-Miralda G et al (2000) Clinical clues to the causes of large pericardial effusions. Am J Med 109:95–101
Rizzardi C, Barresi E, Brollo A et al (2010) Primary pericardial mesothelioma in an asbestos-exposed patient with previous heart surgery. Anticancer Res 30:1323–1325
Kwon HY, Cho KI, Kim SM et al (2010) A rare case with primary undifferentiated carcinoma of pericardium. J Cardiovasc Ultrasound 18:104–107
Petcu DP, Petcu C, Popescu CF et al (2009) Clinical and cytological correlations in pericardial effusions with cardiac tamponade. Rom J Morphol Embryol 50:251–256
Koss GL, Melamed MR (2006) Effusions in the absence of cancer and effusions in the presence of cancer. In: Koss GL, Melamed MR (eds) Koss’ diagnostic cytology and its histopathologic bases, 5th edn. Lippincott Williams & Wilkins, Philadelphia
Reuter H, Burgess L, van Vuuren W et al (2006) Diagnosing tuberculous pericarditis. QJM 99:827–839
Reynen K, Köckeritz U, Strasser RH (2004) Metastases to the heart. Ann Oncol 15:375–381
Patel J, Sheppard MN (2011) Primary malignant mesothelioma of the pericardium. Cardiovasc Pathol 20:107–109
Thomason R, Schlegel W, Lucca M et al (1994) Primary malignant mesothelioma of the pericardium. Case report and literature review. Tex Heart Inst J 21:170–174
Hillerdal G (1983) Malignant mesothelioma 1982: review of 4710 published cases. Br J Dis Chest 77(4):321–343
Meyers DG, Meyers RE, Prendergast TW (1997) The usefulness of diagnostic tests on pericardial fluid. Chest 111:1213–1221
Colombo A, Olson HG, Egan J et al (1988) Etiology and prognostic implications of a large pericardial effusion in men. Clin Cardiol 11:389–394
Zayas R, Anguita M, Torres F et al (1995) Incidence of specific etiology and role of methods for specific etiologic diagnosis of primary acute pericarditis. Am J Cardiol 75:378–382
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
C. Gecmen, G.G. Gecmen, D. Ece, M. Kahyaoğlu, A. Kalayci, C.Y. Karabay, O. Candan, M.E. Isik, F. Yilmaz, O. Akgun, M. Celik, I.A. Izgi, C. Kirma, S. Keser declare that they have no competing interests.
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Gecmen, C., Gecmen, G.G., Ece, D. et al. Cytopathology of pericardial effusions. Herz 43, 543–547 (2018). https://doi.org/10.1007/s00059-017-4596-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00059-017-4596-8