Abstract
While most ant species carry burdens solitarily, a few have evolved impressive skills at cooperative transport that are important to their ecological success. We examined one of these species, Novomessor cockerelli, with the goals of better understanding how transport groups are coordinated and how cooperation influences the efficiency of load transport. Ants were induced to carry standard artificial loads over a smooth horizontal surface, both in groups and as solitary individuals. Porters in groups were non-randomly distributed around the load, with most pulling from the front while walking backward. Porters were more persistent at the leading edge than at the rear, where high rates of leaving and joining were observed. Solitary porters also pulled the load, but they usually first attempted to walk forward and push, until their interactions with the unwieldy load rotated them into a pulling position. Cooperative transport entailed a loss of efficiency, in that solitary porters moved faster and on straighter paths than groups, even though carrying the same per capita weight. This led to a higher prey delivery rate for individuals than for groups. These findings contradict earlier claims of ‘superefficient’ transport for this species, but are consistent with the overall pattern across the ants, in which only a few swarm-raiding species show higher transport efficiency by groups than individuals. We interpret cooperative transport by N. cockerelli and many other species as the product of selection for competitor avoidance, in contrast to swarm raiders, where the special need for efficient prey retrieval has shaped distinctive transport tactics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cooperative transport is one of the more distinctive collective behaviours of ants (Moffett 1988; Czaczkes and Ratnieks 2013; McCreery and Breed 2014). Groups can carry food items too large for a single porter, helping colonies enhance foraging efficiency (Franks 1986; Moffett 1988; Franks et al. 1999) or prevent interference competition, especially by other ants (Hölldobler et al. 1978; Traniello 1983; Traniello and Beshers 1991; Yamamoto et al. 2009). Cooperative transport has evolved independently in a broad array of ant genera, but it is not universal: many species either do not exploit large items or they do so by dissecting them in place and retrieving the parts via individual porters (Hölldobler and Wilson 1990; Detrain and Deneubourg 1997; Yamamoto et al. 2009; Moffett 2010). For those species that are proficient at collective retrieval, understanding the underlying behavioural mechanisms is relevant not only to the foraging ecology of ants, but also to larger questions about the emergence of complex collective phenomena in highly decentralized societies (Bonabeau et al. 1999; Kube and Bonabeau 2000; Camazine et al. 2001).
Successful group transport requires many elements, including deciding when cooperation is necessary (Pratt 1989; Detrain and Deneubourg 1997; Daly-Schveitzer et al. 2007; Amor et al. 2009), recruiting porters (Hölldobler et al. 1978, 1995; Markl and Hölldobler 1978; Traniello 1983; Czaczkes and Ratnieks 2012), matching group size to prey size (Robson and Traniello 1998; Franks et al. 2001), reaching consensus on the direction of travel, and negotiating obstacles and rough terrain. A central but still poorly understood task is the distribution and coordination of porters around the load. The most detailed work to date is on the highly adept prey retrieval of the army ants Eciton burchelli and Dorylus wilverthi (Franks 1986; Franks et al. 1999, 2001). Their transport teams can carry more weight together than the summed efforts of each ant working alone. This “superefficiency” has been argued to derive from how the ants are positioned: all porters straddle the load and face forward, allowing them to run in a common direction and in their normal posture (Franks 1986). The front ant is usually from a specialist porter caste whose larger size makes them more efficient at carrying heavy loads; one or more smaller ants behind keep the load from dragging and stabilize it by countering rotational forces (Franks et al. 2001).
Army ants, however, are unusual: most group-transporting species encircle the load rather than straddling it, so that some are walking backward, some forward, and some sideways (Moffett 1988; Czaczkes and Ratnieks 2013). This variety of postures makes it harder to determine the role of each ant, whether pulling, pushing, lifting, stabilizing against rotations, or some other task. If some positions do not allow an ant to contribute effectively to moving the load, it is possible that the encircling mode of transport makes it harder to attain the superefficiency seen in army ants.
Other open questions concern the distribution of ants around the load and how it is achieved. There is some evidence that an ant’s grasping location depends on specific rules. For example, porters may prefer to walk backward, pulling the load from its leading edge (Sudd 1960; Czaczkes et al. 2011) or to grasp a load at a sharp corner, perhaps to gain more room for manoeuvre (Czaczkes et al. 2011). On the other hand, porters often change their direction of pulling or their grasping location, especially when the load is not moving (Sudd 1960, 1965; Chauvin 1968; Gelblum et al. 2015). Even if random, such changes might lead to the emergence of coordination if ants stabilize their behaviour once they happen upon a configuration that moves the load effectively in the correct direction (Sudd 1960). After transport is underway, further changes in team membership can help to maintain coordination. In Paratrechina longicornis, for example, the load is steered by transiently informed individuals who join the team after detecting the homing direction along a pheromone trail, but later give way to better-informed successors (Gelblum et al. 2015). Ant behaviour can also interact with the physics of the load to produce a well-coordinated outcome—for example, rotating the load into a stable orientation that minimizes drag (Czaczkes and Ratnieks 2011).
In this study we compile a detailed description of the distribution, orientation, and dynamics of porters in a species skilled at the encircling mode of transport. We also compare the efficiency of group transport with that of solitary porters carrying the same per capita weight, in order to determine whether cooperation improves efficiency in ants that encircle their loads. Novomessor cockerelli [recently re-named from Aphaenogaster cockerelli (Demarco and Cognato 2015)] transports heavy food items in order to evade competitors whose mass recruitment and chemical defences would otherwise overwhelm them (Hölldobler et al. 1978). Their use of local and long-distance recruitment to assemble a transport team has been well described (Hölldobler et al. 1995, 1978; Markl and Hölldobler 1978), but how the assembled ants coordinate to move the item to the nest is much less well understood (Berman et al. 2011). We thoroughly characterized the behaviour of groups and individuals carrying standard artificial loads over a smooth surface, thus minimizing variation due to peculiarities of object or terrain.
Methods
Group transport
Cooperative transport was studied in 16 colonies of N. cockerelli located in South Mountain Park in Phoenix, AZ (33.335°N, 112.027°W). Experiments were carried out during the ants’ activity peaks in the early morning (06:00–08:30) and late afternoon (17:00–19:00) in May 2012. Temperature varied between 21 and 27 °C.
A single group transport episode was observed for each of the 16 colonies. Ants were induced to transport across a Plexiglas platform (61 × 46 × 0.5 cm) placed south of the main nest entrance, with its long axis in a north–south orientation and its proximate edge 50 cm from the entrance. The platform was adjusted to a horizontal position (as determined by a spirit level) and covered with a single sheet of white paper (75.2 g/m2).
To lure ants onto the platform, a dried fig was placed at its distal edge. Foragers found the fig and recruited nestmates; once ten workers were simultaneously gripping the fig, it was removed and replaced with a standard load made from a United States ten-cent coin (1.35 mm thick and 9 mm radius) glued (with ethyl-2-cyanoacrylate) to an ethylene vinyl acetate (EVA) foam disk (0.2 mm thick and 10 mm radius). The coin was centred on the EVA disk to create a 1-mm fringe of foam that could be easily gripped by the ants. The total mass of the load was 2.4 g. To make the load attractive to the ants, it was lightly rubbed against the surface of a dried fig before each experiment. Ants that had been recruited to the fig readily grasped the load and transported it across the platform toward the nest entrance (Fig. 1).
Transport was recorded with a 720p high definition video camera (Canon G12) positioned above the centre of the platform on a tripod (Manfrotto 055 Series). The field of view was shaded using an umbrella. Recording began once the ants had moved the load 1 cm and stopped once the load was carried across an imaginary line 40 cm from the start point and perpendicular to the long axis of the platform. A sample recording is available as Online Resource 1. From the recordings, we used ImageJ software (Rasband 1997) with the Mtrack plug-in (Meijering et al. 2012) to extract the following data: position of the centre of the load, number of porters, angular position of each porter around the load perimeter, and orientation of each porter with respect to the load. Angular positions were measured counter clockwise from the direction of movement of the load. This information was obtained from single frames at 5-s intervals. We also recorded the load’s position when a porter let go or a new porter joined the group, and the angular position on the load where the change occurred.
To track load movement, we measured the straight line distance between successive positions recorded at 5 s intervals. We estimated total distance transported as the sum of these interval lengths. Because all transports covered the same displacement in the direction toward the nest (40 cm) the distance travelled was an indicator of path straightness: a perfectly straight path would have a distance of 40 cm, and longer distances reflect decreasing straightness. The average speed was taken as the total distance divided by the duration of transport. To account for variation in speed over the course of transport, we also measured maximal speed by finding for each trial the 10-s interval with the largest distance moved.
Individual transport
To compare the efficiency of group and individual transport, we recorded lone porters carrying loads of 300 mg, the average per capita weight carried by members of group transport teams (as described below in the “Results”). Individual transport was observed in March 2013 at the same field site used for group transport, but with three colonies not used in the group experiments. The experimental setup was identical to that described above except for the design of the load, which was made from a lump of solder (Bernzomatic 60 % tin, 40 % lead) glued to an EVA foam disk rubbed against a fig. A total of 25, 21 and 18 individual transports were filmed for the three colonies, respectively. Transport was video-recorded as for groups, and similar data were extracted from the recordings. A sample recording is available as Online Resource 2. Because we detected no colony effect on individual speed (Kruskal–Wallis K = 1, p = 0.6) or displacement (Kruskal–Wallis K = 2, p = 0.34), all individual data were combined when comparing these variables between individuals and groups.
Statistical analysis
For each measurement we report average and standard deviation, except for variables that showed evidence of non-normality (one-sample Kolmogorov–Smirnov test, α = 0.05), in which case we report the median and the 25th and 75th percentiles. All statistical analysis was done using Matlab. Load headings, ant body orientations, and grasping positions were analysed with the circular analysis toolbox CircStat (Berens 2009).
Results
Group size
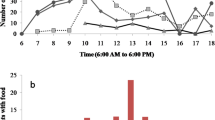
The average number of porters increased during transport from 4 (3–7) at the beginning to 8 (7–11) at the end of the 40 cm displacement (Fig. 2). Team composition was dynamic, with, on average, 63 (50–126) joining events and 60 (47–127) leaving events over the 4.9 min (2.9–9.6) taken to move the load 40 cm. Average leaving rate [10.8/min (8.1–16.9)] and joining rate [10.8/min (8.4–15.2)] were very similar. Most changes, whether joining or leaving, occurred at the back of the load [mean event angle, joining 179.9° (174.2–185.1), leaving 180.5° (175.3–185.6); Fig. 3]. Team size was not affected by ambient temperature (Spearman’s r = 0.36, p = 0.16).
Change in number of porters over the course of cooperative retrieval of a standardized load. Boxplots show the distribution of group size at 1 cm intervals (n = 16 retrievals). In each boxplot the horizontal line shows the median, boxes delimit the 1st and 3rd quartiles, whiskers show the range, and crosses show outliers
Histogram showing the distribution of leaving and joining events around the perimeter of the load. Radial positions are indicated relative to the load’s direction of travel, set at zero degrees. Joining occurs when an ant seizes the load with her mandibles (grey bars n = 458). Leaving occurs when a porter lets go of the load (black-outlined bars n = 419). Events are pooled over 16 independent retrievals
Distribution and orientation of porters around the load
In both individual and group transport, porters tended to pull the load from the front while walking backwards (Fig. 4). Porters in groups were not distributed uniformly around the load, but instead tended to cluster at the front (Fig. 4a). During individual transport, ant body orientation was correlated with the direction of transport (Fig. 4b), but there was no detectable effect of position on speed of transport (circular correlation r = 0.13, p = 0.55; n = 65). Individual transporters rarely walked forward while carrying the load (5 out of 65 trials). However, they nearly always initiated transport by attempting to push the load ahead of them toward the nest. For light enough loads, this behaviour ends with the ant lifting the load and walking forward toward the nest. The 300 mg load used in this experiment was too heavy to be lifted, hence an ant’s efforts ended by rotating her into a backward-facing position in which she pulled the load toward the nest. This stereotypical sequence was observed in almost every trial.
Distribution of porter orientation and position during retrieval by a groups and b individuals. Disks represent snapshots taken at 10 cm intervals. The outer dot histograms show the gripping locations of porters during cooperative retrieval at 12° intervals (pooled data from 16 trials). Radial positions are indicated relative to the load’s direction of travel (directly to the right). The ants are non-randomly distributed around the load in all five snapshots (Rayleigh test Z > 22, p < 0.001). The inner rose histograms show the porters’ body orientation relative to the direction of travel. An orientation of zero (directly to the right) means that the ant’s body axis is parallel to the direction of travel. During cooperative retrieval, orientations are randomly distributed at the first snapshot (Raleigh test Z = 0.89, p = 0.41), but non-random after that (Raleigh test Z > 4, p < 0.02). During individual retrieval, orientations are non-randomly distributed in all five snapshots (Rayleigh test Z > 28, p < 0.005, N = 65). For clarity, histogram gridlines and polar positions are labelled only on the leftmost disks
Transport speed and efficiency
Groups moved more slowly than individuals, even though they carried the same per capita load (i.e., the individual load of 300 mg was equal to the group load of 2.4 g divided by the average group size of eight ants). The average transport speed (distance covered/time) for individuals was 5.3 mm/s (4.1–6.5) whereas groups moved at a speed of 2.5 mm/s (1.6–2.9) (Fig. 5). Maximal speed (measured by selecting for each trial the 10-s interval during which the greatest distance was travelled) showed the same pattern (individuals 8.6 mm/s (6.8–9.8); groups 3.9 mm/s (2.4–5.1); Mann–Whitney U 65, 16 = 34, p < 0.001). Average speed was not correlated with group size (Pearson’s r = −0.05, p = 0.83).
Individuals were also more efficient than groups when scored by the prey delivery rate, a metric that integrates load mass and transport velocity (Moffett 1988; Detrain 1990; Traniello and Beshers 1991). The prey delivery rate is calculated as the product of the load mass and transport velocity divided by the group size. For individual porters it was 1590 mg mm/s (1230–1950) while for groups it was only 710 mg mm/s (390–1010) per porter.
Both individuals and groups moved at a steady speed through most of the observation period, but groups showed a brief initial phase of acceleration not seen in individuals (Fig. 6). Speed was not affected by ambient temperature (Spearman’s r = 0.29, p = 0.26).
Path straightness
Travel paths were relatively straight, and somewhat more so for individuals than for groups (Fig. 7). The average distance travelled by each group was 48.1 cm (43.5–49.6, n = 16), while that for individuals was 44.3 cm (42.7–46.2, n = 65). For the same displacement of 40 cm, individuals travelled a significantly smaller distance than groups (Mann–Whitney U 65, 16 = 308, p = 0.01; Fig. 8). Distance travelled was independent of group size (Pearson’s r = 0.13, p = 0.62).
Discussion
We observed great variation in the number of transporters (from 5 to 13), but no correlation between this number and transport speed. This contrasts with earlier studies of Paratrechina longicornis (Gelblum et al. 2015) and Pheidole oxyops (Czaczkes et al. 2011), both of which showed increases in speed with group size. However, the latter study also found that speed levelled off at around 5–7 ants, and argued that crowding may limit the value of additional porters once ants have occupied the most efficient grasping positions (i.e., the corners of square loads). We can speculate that analogous crowding effects in N. cockerelli may have prevented larger groups from moving any faster than smaller ones.
Because teams were often larger than necessary to achieve maximum speed, this could indicate poor regulation of group size by these ants. However, we do not think this conclusion is warranted. First, speed is not the only factor expected to influence optimal group size. In Formica pallidefulva (formerly F. schaufussi), which also shows no correlation between group size and retrieval velocity, larger groups were more effective at defending medium-sized food items from ant competitors (Traniello and Beshers 1991). N. cockerelli also faces competition for rich food items, and so may benefit from recruiting for defense as well as speedy movement (Hölldobler et al. 1978). Second, our methods may have artificially enhanced group size. By using a large fig as bait to attract foragers to the platform, we may have stimulated stronger recruitment than would the load itself. Some other ants are known to regulate porter numbers by recruiting nestmates only until enough ants are present to move the load (Detrain 1990). If this is also true of N. cockerelli, our large bait may have disrupted group size regulation.
We found that ants predominantly pulled the load, rather than pushing it, in both individual and cooperative transport. Pulling behaviour was also more stable, with lower turnover of group members at the leading edge of the load, where porters walked backward and presumably pulled, than at the trailing edge. A similar distribution of porters has been described in Pheidole crassinoda (Sudd 1960), P. oxyops (Czaczkes and Ratnieks 2011) and Paratrechina longicornis (Gelblum et al. 2015). In P. oxyops, for example, the first porters grasp the load at the front, with later ants successively joining at the rear and then side positions, as more preferred spots become occupied (Czaczkes et al. 2011). However, it is not necessarily the case that N. cockerelli porters prefer pulling over pushing. In nearly all of our individual trials, ants began by attempting to lift the load and to hold it in front of them while walking forward. Their interactions with the heavy load eventually rotated them into a backward position in which they could successful move the load. Similar behaviour has been reported in P. crassinoda (Sudd 1960) and Myrmica rubra (Sudd 1965). Thus, the predominance of pulling may emerge from physical interactions between the ants’ behaviour and the load, rather than reflecting a preferred tactic. In cooperative transport, the high readiness of porters to join at the back may result from a similar preference for pushing the load. On the other hand, it may be a simple result of there being more open positions at the rear, due to high leaving rates relative to ants at the front. We speculate that the greater persistence of ants at the front is due to advantage of dragging vs. pushing in minimizing friction, with the ants following a rule of persisting in any behaviour that successfully moves the load.
Individual transporters of N. cockerelli moved more rapidly and in straighter paths than did groups, even though they carried the same per capita weight. This may reflect coordination costs of group transport, where porters counteract one another’s efforts to the degree that the forces they exert on the load are not perfectly aligned. Similarly, a lone porter does not need to compromise on the precise path to take to the nest, and she will not be diverted from her trajectory by the efforts of other ants. In group transport, each worker may steer the load towards the nest according to idiosyncratic navigation cues, requiring constant adjustments by each participant.
The slower movement of groups than individuals contradicts earlier claims that cooperative transport by these ants is ‘superefficient’ (Franks et al. 1999; McCreery and Breed 2014). These papers cite Hölldobler et al. (1978), which itself did not claim superefficiency but simply noted that group transport of large prey gains more energy for the colony than solo transport of small items, despite the loss of speed. Later claims of superefficiency appear to stem from the higher prey delivery rate observed for groups than for individuals (Table 1), but the data of Hölldobler et al. (1978) do not support this inference. The loads given to solo transporters were very light and so did not measure the limits of their transport efficiency. Superefficiency is properly shown by evidence that a group working together can carry more total weight than would its members working separately (Franks 1986). This was not the case in our experiments: had our standard load of 2.4 g been divided into 300 mg pieces, then the eight ants in an average-sized group could have carried it more quickly as separate individuals than they did as a group.
A survey of published studies shows that N. cockerelli’s lack of superefficiency is a common pattern in cooperatively transporting ants. Table 1 summarizes team size, load weight, and transport velocity in all species for which such data are available. The table also reports prey delivery rate, a general measure of transport efficiency that can be compared among groups of different size and speed carrying loads of different weight. For most species, this rate is lower for groups than for individuals, meaning that cooperation is less efficient than solo transport. The exception is the swarm-raiding species Pheidologeton diversus (Moffett 1988). In addition, two other swarm raiders, D. wilverthi and E. burchelli (for which prey delivery rate has not been measured), show superefficiency in that larger teams carry disproportionately heavy items without any loss in speed (Franks 1986; Franks et al. 1999).
The high transport efficiency of swarm-raiders may be a product of their distinctive foraging strategy. In order to gather the large amounts of food needed to maintain their intense brood-rearing and high populations, colonies exploit broad foraging areas via long raiding columns. Their transport tactics on these columns, at least for Dorylus and Eciton, share a suite of distinctive features that contribute to efficient prey movement. First, they are exceptionally fast. Teams of D. wilverthi travel at 30 mm/s and those of E. burchelli at 80 mm/s (Franks 1986; Franks et al. 1999). No other species has been recorded to move this fast while transporting heavy items, and most show speeds well below 10 mm/s (Table 1). Second, these ants show a remarkable constancy of speed, with all porters on the trail, regardless of load, moving at the same high velocity, a feature that may help them avoid congestion on their crowded trails (Franks et al. 1999). Third, porters straddle their loads and walk in their normal forward-facing posture, improving stability of the load and avoiding potentially inefficient sideways or backward orientations (Franks 1986; Czaczkes and Ratnieks 2013). Fourth, and related to their posture and high speed, these ants carry small burdens relative to those reported in other cooperative transporters. Extensive sampling of natural prey items found only a handful exceeding a dry weight of 20 mg, with most less than 10 mg (Franks et al. 1999). Even assuming that three-fourths of the original load was water weight [a generous estimate for arthropod prey (Studier and Sevick 1992)], then the heaviest items, at about 80 mg, are much smaller than the heaviest loads carried by non-swarm-raiding cooperative transporters (Table 1).
In contrast to swarm raiders, other cooperative transporters may face greater pressure to avoid competition than they do to move prey at maximum efficiency (Traniello 1989; Traniello and Beshers 1991). N. cockerelli, in particular, face competition from effective mass recruiters that can readily dislodge them using chemical weapons or stings that N. cockerelli does not possess (Hölldobler et al. 1978). More generally, Yamamoto et al. (2009) found that cooperative transport was much more common in terrestrial species that likely encounter a large array of other ants, compared to arboreal species that often dominate their habitat and so face less competitive pressure (as well as a risk of falling if they attempt cooperative transport). Competitors can be avoided if foragers initiate transport as soon as possible, even for an item so large that it could more efficiently be moved were it first cut into smaller pieces. This contrasts with swarm raiders, whose aggressive and numerous foragers can safely spend time dissecting captured prey before swiftly carrying the pieces to the nest. While small pieces can be straddled, large ones must instead be encircled. This arrangement can reduce efficiency, if ants must adopt suboptimal orientations with respect to the load and the direction of travel (e.g., walking sideways). Ants in different orientations may also exert forces that are not parallel with the direction of transport or with one another, leading to wasted effort or even cancelling out one another’s contributions. In Pheidole crassinoda, for example, the resultant force exerted by a team increased sublinearly with team size, perhaps because of workers cancelling out one another’s efforts (Sudd 1960). In pairs of F. lugubris, the resultant force was correlated with the degree of alignment of individual forces (Sudd 1965).
Available data support the idea that cooperative transport in N. cockerelli is driven more by the need to escape competition than to move loads with maximum efficiency. However, this scenario still needs further exploration. For one thing, the encircling mode of transport does not exclude the possibility of superefficient transport, as shown by the example of Pheidologeton diversus. These ants encircle their loads but show a steady increase in prey delivery rate with number of transporters, up to a team size of about ten (Moffett 1988). It is possible that other species, including N. cockerelli, can also do so, at least over some range of load weights. We tested only a single, relatively heavy load that may have stretched the limits of these ants’ transport capacities. Future work should examine a broader range of load sizes. More importantly, efficiency is ultimately not best measured by prey delivery rate, but by energetic costs and benefits. Direct measurement of metabolic rate during each phase of cooperative retrieval, combined with measures of the energetic benefits of natural food items that require group transport, will ultimately give a more revealing picture of the pressures shaping retrieval tactics.
References
Amor F, Ortega P, Cerdá X, Boulay R (2009) Cooperative prey-retrieving in the ant Cataglyphis floricola: an unusual short-distance recruitment. Insect Soc 57:91–94
Berens P (2009) CircStat: a MATLAB toolbox for circular statistics. J Stat Softw 31:1–21
Berman SM, Lindsey Q, Sakar MS et al (2011) Experimental study and modeling of group retrieval in ants as an approach to collective transport in swarm robotic systems. Proc IEEE 99:1470–1481
Bonabeau E, Dorigo M, Theraulaz G (1999) Swarm intelligence. Oxford University Press, New York
Camazine S, Deneubourg J-L, Franks NR et al (2001) Self-organization in biological systems. Princeton University Press, Princeton
Cerdá X, Angulo E, Boulay R, Lenoir A (2009) Individual and collective foraging decisions: A field study of worker recruitment in the gypsy ant Aphaenogaster senilis. Behav Ecol Sociobiol 63:551–562
Chauvin R (1950) Le transport des proies chez les fourmis. Y-a-t-il entr’aide? Behaviour 2:249–256
Chauvin R (1968) Collective prey transport of Formica polyctena. Insect Soc 15:193–200
Czaczkes TJ, Ratnieks FLW (2011) Simple rules result in the adaptive turning of food items to reduce drag during cooperative food transport in the ant Pheidole oxyops. Insect Soc 58:91–96
Czaczkes TJ, Ratnieks FLW (2012) Pheromone trails in the Brazilian ant Pheidole oxyops: extreme properties and dual recruitment action. Behav Ecol Sociobiol 66:1149–1156
Czaczkes TJ, Ratnieks FLW (2013) Cooperative transport in ants (Hymenoptera: Formicidae) and elsewhere. Myrmecol News 18:1–11
Czaczkes TJ, Nouvellet P, Ratnieks FLW (2011) Cooperative food transport in the neotropical ant, Pheidole oxyops. Insect Soc 58:153–161
Daly-Schveitzer S, Beugnon G, Lachaud J-P (2007) Prey weight and overwhelming difficulty impact the choice of retrieval strategy in the neotropical ant Gnamptogenys sulcata (F. Smith). Insect Soc 54:319–328
Demarco BB, Cognato AI (2015) Phylogenetic analysis of Aphaenogaster supports the resurrection of Novomessor (Hymenoptera: Formicidae). Ann Entomol Soc Am 108:201–210
Detrain C (1990) Field study on foraging by the polymorphic ant species, Pheidole pallidula. Insect Soc 37:315–332
Detrain C, Deneubourg J-L (1997) Scavenging by Pheidole pallidula: a key for understanding decision-making systems in ants. Anim Behav 53:537–547
Franks NR (1986) Teams in social insects: group retrieval of prey by army ants (Eciton burchelli, Hymenoptera, Formicidae). Behav Ecol Sociobiol 18:425–429
Franks NR, Sendova-Franks AB, Simmons J, Mogie M (1999) Convergent evolution, superefficient teams and tempo in Old and New World army ants. Proc R Soc B Biol Sci 266:1697–1701
Franks NR, Sendova-Franks AB, Anderson C (2001) Division of labour within teams of New World and Old World army ants. Anim Behav 62:635–642
Gelblum A, Pinkoviezky I, Fonio E et al (2015) Ant groups optimally amplify the effect of transiently informed individuals. Nat Commun 6:7729
Hölldobler B, Wilson EO (1990) The ants. Harvard University Press, Cambridge
Hölldobler B, Stanton RC, Markl H (1978) Recruitment and food-retrieving behavior in Novomessor (Formicidae, Hymenoptera) I. Chemical signals. Behav Ecol Sociobiol 4:163–181
Hölldobler B, Oldham NJ, Morgan ED, König WA (1995) Recruitment pheromones in the ants Aphaenogaster albisetosus and A. cockerelli (Hymenoptera: Formicidae). J Insect Physiol 41:739–744
Kube C, Bonabeau E (2000) Cooperative transport by ants and robots. Robot Auton Syst 30:85–101
Markl H, Hölldobler B (1978) Recruitment and food-retrieving behavior in Novomessor (Formicidae, Hymenoptera) II. Vibration signals. Behav Ecol Sociobiol 4:183–216
McCreery HF, Breed MD (2014) Cooperative transport in ants: a review of proximate mechanisms. Insect Soc 61:99–110
Meijering E, Dzyubachyk O, Smal I (2012) Methods for cell and particle tracking. In: Conn PM (ed) Methods in enzymology. Academic Press, New York, pp 183–200
Moffett MW (1988) Cooperative food transport by an Asiatic ant. Natl Geogr Res 4:386–394
Moffett MW (2010) Adventures among ants. University of California Press, Berkeley
Pratt SC (1989) Recruitment and other communication behavior in the ponerine ant Ectatomma ruidum. Ethology 81:313–331
Rasband WS (1997) ImageJ. U.S. National Institutes of Health, Bethesda
Robson S, Traniello JFA (1998) Resource assessment, recruitment behavior, and organization of cooperative prey retrieval in the ant Formica schaufussi (Hymenoptera: Formicidae). J Insect Behav 11:1–22
Studier EH, Sevick SH (1992) Live mass, water content, nitrogen and mineral levels in some insects from south-central lower Michigan. Comp Biochem Physiol 103A:579–595
Sudd J (1960) The transport of prey by an ant, Pheidole crassinoda Em. Behaviour 16:295–308
Sudd J (1965) The transport of prey by ants. Behaviour 25:234–271
Tibbetts EA, Levy S, Donajkowski K (2011) Reproductive plasticity in Polistes paper wasp workers and the evolutionary origins of sociality. J Insect Physiol 57:995–999. doi:10.1016/j.jinsphys.2011.04.016
Traniello JFA (1983) Social organization and foraging success in Lasius neoniger (Hymenoptera: Formicidae): Behavioral and ecological aspects of recruitment communication. Oecologia 59:94–100
Traniello JF (1989) Foraging strategies of ants. Ann Rev Entomol 34:191–210
Traniello JFA, Beshers SN (1991) Maximization of foraging efficiency and resource defense by group retrieval in the ant Formica schaufussi. Behav Ecol Sociobiol 29:283–289
Yamamoto A, Ishihara S, Ito F (2009) Fragmentation or transportation: mode of large-prey retrieval in arboreal and ground nesting ants. J Insect Behav 22:1–11
Acknowledgments
This research was supported by the United States Office of Naval Research (Award N00014-08-1-0696) and the United States National Science Foundation (Award CCF-1012029). AB was supported by the Wallonie-Bruxelles International and the Belgian American Education Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online resource 1: Video of a group of N. cockerelli ants carrying a 2.4 g artificial standard load made of a dime attached to a foam disk (MOV 221273 kb)
Online resource 2: Video of a single N. cockerelli ant carrying an artificial standard load consisting of a 0.3 g piece of solder (MOV 283446 kb)
Rights and permissions
About this article
Cite this article
Buffin, A., Pratt, S.C. Cooperative transport by the ant Novomessor cockerelli . Insect. Soc. 63, 429–438 (2016). https://doi.org/10.1007/s00040-016-0486-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-016-0486-y