Abstract
Bioenergetics models for fishes are useful for understanding ecological processes (e.g., survivorship, growth, and reproduction) and can also inform fisheries management. Yet, current bioenergetics models are unable to ascertain direct energetic costs associated with standard and active metabolism for wild, free-swimming fishes. The use of telemetry with accelerometer sensors, calibrated in the laboratory using swim tunnel respirometers, have made it possible to estimate field metabolic activity in wild fish. Our objectives were to determine seasonal thermal habitat use and habitat-dependent metabolic costs associated with standard, active, and maximum metabolism in a ~1400 ha multibasin lake in Québec, Canada. We implanted 47 wild, free-swimming Lake Trout (Salvelinus namaycush) with either acoustic transmitters equipped with temperature and depth sensors or an acceleration sensor. Three sets of water temperature loggers (one set for each basin) were deployed at 2, 4, 6, 10, and 18 or 20 m to measure seasonally available thermal habitat. Thermal profiles of lake water temperature varied among basins with the thermocline being ~5 m in the smallest basin (north basin) and ~7.5 m in the largest basin (east basin). Thermal habitat used by Lake Trout varied seasonally, coupled with seasonal and basin differences in standard and maximum metabolism. Daily active metabolism loosely followed seasonal changes in thermal habitat use but was largely unaffected by differences in thermal habitat use among capture basins. The theoretical scope-for-activity followed seasonal trends and was estimated to range between 47% and 74% of theoretical aerobic scope. Our observations suggest that available thermal habitats influence Lake Trout thermal habitat use, and thus metabolic costs associated with swimming. These changes in thermal habitats could have metabolic consequences for individuals in a population, resulting in altered fitness metrics (i.e., survival, growth, and/or reproduction). Reductions in the volume and availability of optimal thermal habitats for Lake Trout are likely to occur under climate change scenarios. Our study indicates that the species can adjust metabolic costs throughout the year even when thermal habitats appear to be limiting, which may inform future evidence-based management decisions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding why animals move provides insight into ecological processes that are focal points of conservation and resource management (e.g., predator–prey dynamics, energetics, resource partitioning, and habitat selection; Hugie and Dill 1994; Morris 2003; Nathan et al. 2008). The energy an individual consumes, retains, and excretes alters its capacity for movement that in turn impacts its survival, growth, and reproductive success (i.e., fitness), thereby affecting population-level processes for the species (Tytler and Calow 1985). Understanding how energy is transferred between an ecosystem and an organism is the study of bioenergetics and is of fundamental interest to ecology, conservation, and resource management (Kitchell 1983; Hansen et al. 1993; Chipps and Wahl 2008). Bioenergetics models are energy-balance equations based on the first law of thermodynamics where energy is consumed and transformed for various uses such as metabolism, growth, and waste, with active metabolism considered energetically costly (Kitchell et al. 1977; Kitchell 1983; Hansen et al. 1993).
Temperature, for ectothermic organisms such as fishes, has a substantial influence on energy use, and is one of the most important drivers of biological processes due to its effects on the physiological mechanisms that dictate movement, survival, growth, and reproduction (Fry 1947). Many fishes are stenothermal, relying on thermal habitats defined by a narrow range of temperatures for survival. Thermal habitats of many aquatic environments are currently being altered by human-induced climate change and other anthropogenic stressors (Reid et al. 2019; Jane et al. 2021; Kraemer et al. 2021). In the face of thermal habitat loss, fishes have three potential options: (1) endure warming thermal habitats, (2) seek the next best thermal habitat by seasonally shifting habitat use which might increase energy demands and the potential for a mismatch between predator and prey (Thackeray et al. 2010), or (3) if available, use deeper, cooler water, that might be unsuitable in other ways (e.g., having limited dissolved oxygen or light conditions; Hansen 2021). Each of these options will have different consequences for fish movement and energetics, therefore understanding how movement within and among thermal habitats influences metabolism and theoretical aerobic scope (AS; i.e., the difference between standard and maximum metabolic rates) in the wild is important for ongoing conservation and management of climate-affected species.
Lake Trout (Salvelinus namaycush) are a stenothermal, cold-water fish native to oligotrophic aquatic ecosystems within northern North America (Shuter et al. 1998; Muir et al. 2016; Muir et al. 2021; Gunn and Louste-Fillion 2022). Access to cold water is vital for the species, with reports of the species using temperatures ranging from 4–18 °C (Martin and Olver 1980; Morbey et al. 2006), with metabolic optimum documented between 8–12 °C (Stewart et al. 1983; Rottier 1993; Evans et al. 2007), and multiple studies indicating that the species often uses thermal habitats ≤ 15 °C (Evans 2007; Plumb et al. 2009; Guzzo et al. 2017). Climatic changes to thermal habitats (e.g., warmer water extending deeper, and/or longer stratified periods) could impact Lake Trout populations in a variety of ways including by forcing the species to shift seasonal habitat use, resulting in increased metabolic costs and reduced growth rates (Schindler et al. 1996; Sharma et al. 2007; Plumb et al. 2014; Guzzo et al. 2017). Understanding how Lake Trout metabolism (i.e., standard, SMR; active, RMR; and maximum metabolic rates, MMR) will change in response to changing thermal habitats will provide scientists, fisheries managers, and conservation organizations more information to sustainably conserve and manage the species (Zimmerman and Krueger 2009).
Bioenergetics models for Lake Trout were developed in the 1980s to assist in population rehabilitation and inform management (Stewart et al. 1983; Hansen et al. 1993). Subsequent studies used bioenergetics to explore contaminant assimilation (Madenjian et al. 2000), sexual differences and/or the influence thermal habitat has on prey consumption and growth (McDonald et al. 1996; Madenjian and O’Connor 1999; Madenjian et al. 2000, 2010; Kao et al. 2015), food web structure and trophic dynamics (Pazzia et al. 2002; Sherwood et al. 2002; Harvey et al. 2003), morphotype differences (Kepler et al. 2014), and growth and metabolism (Evans 2007; Guzzo et al. 2019). These studies have provided novel information but have relied on laboratory-derived measurements of metabolism, or do not directly assess metabolic costs for wild, free-swimming populations (Madenjian et al. 2013). These limitations inevitably constrain the ability to accurately understand how anthropogenic stressors will impact the metabolic costs of Lake Trout and the availability of suitable thermal habitats for the species (Plumb et al. 2014). Recent work though has provided insight on metabolic costs for wild, free-swimming fish, by using a combination of respirometry and acoustic telemetry (Cruz-Font et al. 2016). Our study furthers this work by providing context on how a complex freshwater ecosystem influences metabolic costs, including theoretical SMR, MMR, AS, and estimated RMR and scope-for-activity (i.e., the difference between theoretical MMR and estimated RMR) for Lake Trout throughout an entire year.
The objective of this study was to determine whether thermal habitats influence theoretical SMR, MMR, AS, and estimated RMR and scope-for-activity for wild Lake Trout as inferred by variation in locomotor activity and thermal habitat use. We hypothesized that within a multibasin lake, seasonal thermal habitats would be influenced by basin size resulting in changes in Lake Trout thermal habitat use, theoretical SMR and MMR, and estimated RMR and scope-for-activity, and thus influence metabolic costs for the species. We predicted that fish that primarily use smaller basins with limited thermal habitat (volume of water ≤ 12 °C) would exhibit reduced activity and metabolic costs because movement and access to preferred resources (e.g., habitat and/or prey) is limited compared with fish that use larger basins.
Methods
Study site
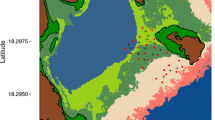
Lake Papineau (45.815120° N, 74.770875° W) lies within the boundaries of a privately owned 26,305 ha fish and game reserve named Kenauk Nature X Limited Partnership Landholdings (hereafter referred to as Kenauk Nature), near Montebello, Québec, Canada. Lake Papineau is a 1,357-ha, oligotrophic lake with three basins: the east basin is 714 ha, maximum depth ~90 m; the west basin is 409 ha, maximum depth ~27 m; and the north basin is 234 ha, max depth ~50 m (Fig. 1). The basins are connected by narrow, shallow channels (100–250 m wide, ≤ 10 m deep) that may act as physical (e.g., ice) and thermal barriers preventing fish movement between basins (Fig. 1). Each basin has a large hypolimnetic volume during summer stratification (east basin, 169.9 billion liters or 72.3% of east basin; west basin, 18.9 billion liters or 33.5% of west basin; and north basin, 22.3 billion liters or 54.3% of north basin), providing thermal refugia for Lake Trout.
Acoustic telemetry
An acoustic telemetry array was deployed in Lake Papineau from fall 2017 to summer 2021 (Fig. 1). Seventeen acoustic receivers (VR2W—69 kHZ, Innovasea Systems Inc., Halifax, NS, CA) were deployed in September 2017 among the three basins: east (n = 8), west (n = 5), and north basin (n = 4; Fig. 1). An additional four receivers were deployed in July 2018, two in the east basin (ntotal = 10) and two in the west basin (ntotal = 7). In June 2019, one additional receiver was deployed in the southern portion of the west basin (ntotal = 8). That receiver was relocated in November 2019, to a Lake Trout spawning reef in the east basin (ntotal = 11) as part of a separate study on spawning movements. Receivers were moored 3–5 m off the lakebed using two 30 kg sandbags with a single floating line running to a sub-surface buoy (2–3 m below the surface). Detection range and efficiency were determined by using one representative receiver in each basin (Supplementary Fig. 1). Sentinel transmitters (V13; 840–960 s delays, Innovasea Systems Inc., Halifax, NS, CA) were deployed at a distance where approximately 50% of detections within 24 h would be heard (Brownscombe et al. 2020). The distance at which 50% of detections were heard varied in each basin between 270 m and 340 m. Thermal habitats were measured in Lake Papineau by deploying five temperature loggers (Vemco Mini-logger II, Innovasea Systems Inc., Halifax, NS, CA) in July 2018 to the mooring line of one receiver in the east and west basins (Supplementary Fig. 1) at ~2, 4, 6, 10, and 20 m and one receiver in the north basin at ~2, 4, 6, 10, and 18 m. Loggers recorded temperature every 10 min to determine the timing of spring and fall turnover, and the thermocline depth during summer stratification. Data were downloaded from receivers and temperature loggers in June or July of each year.
Animal care protocols, in accordance with the Animal Care Council of Canada guidelines as administered by Carleton University, were followed throughout this study. All animal collection was done under provincial scientific collection permits administered by the Québec Ministère des Forêts, de la Faune et des Parcs. Forty-eight Lake Trout (Table 1) were captured across all three basins by rod and reel or multipaneled monofilament gill nets (64 m × 1.8 m; 8 m panels; stretch mesh sizes of 57, 64, 70, 76, 89, 102, 114, and 127 mm). Gill nets were used in either spring or fall with a soak duration of 1–2 h. Fish were immobilized for surgery using electronarcosis (Vandergoot et al. 2011). All instruments were sterilized prior to surgery using betadine (povidone iodine, 10%). A small mid-ventral incision (~25 mm) was made to implant an acoustic transmitter into the body cavity. An acoustic transmitter with accelerometer sensor was internally tethered with Vicryl sutures (Ethicon VCP423, 3–0 FS-2 cutting) near the incision site following implant protocols described in Cruz-Font et al. (2016) and Brownscombe et al. (2014). The incision was closed by two or three Vicryl sutures (Ethicon VCP423, 3–0 FS-2 cutting) tied with a double surgeon’s knot. The transmitter-to-body weight ratio was < 2% for all fish (Winter 1996). All fish were measured (total length; TL, mm) and weighed (g). For fish where weight was unattainable because of issues with scale stability, weight was derived from length–weight relationship calculated from regressing log10 transformed length and weight of Lake Papineau Lake Trout (Piccolo et al. 1993). An external T-bar anchor tag (Floy Tag and Mfg., Inc.) was inserted into the fish musculature behind the dorsal fin as an external identifier for researchers and recreational anglers. After surgery, all fish were placed in a recovery tank and monitored until they regained equilibrium. Each procedure lasted 2–4 min and all fish were released within 10 min of the surgery near the site of capture within each basin (n = 16 east basin; n = 24 west basin; and n = 8 north basin; Table 1).
Seventeen of the 47 Lake Trout captured in September 2017 were implanted with Innovasea transmitters: V13TP (80–160 s delays, n = 15), and V13A (90–180 s delays, n = 2, swim activity rate of 5 HZ, i.e., five samples of acceleration per second, with 25-s sampling duration). An additional ten Lake Trout were tagged in spring 2018 with V13TP (80–160 s delays) and 20 Lake Trout in spring 2019 with V13A (320–640 s delays, tailbeat sampled at a rate of 10 HZ, i.e., ten samples of acceleration per second, with a 45-s sampling duration). One Lake Trout tagged in spring 2018 with an V13TP was lethally captured in spring 2019 and its transmitter was implanted into another fish in spring 2019 (Table 1). All transmitters (emitting) and receivers (recording) were set to a frequency of 69 kHZ.
Data analysis
Acoustic telemetry detection data were cleaned by first removing detections from unknown transmitter identifiers (IDs). A minimum lag filter (i.e., time between detections) was then applied to each transmitter ID, because the delays for each transmitter varied (Pincock 2012; see Supplementary Material 1.1). Data were further inspected visually using abacus plots to identify any additional suspicious detections that were subsequently removed. A minimum of three detections were needed every hour for a fish to be considered present and the data valid (Papastamatiou et al. 2010). A total of 10 out of 48 tagged fish were never detected, with the remaining 38 individuals detected throughout the study time period.
We measured thermal habitat use of Lake Trout by calculating daily mean fish temperature (°C) obtained from 26 acoustically implanted Lake Trout. Daily metabolic costs (SMR, MMR, and RMR; mg O2 h−1 kg−1) were calculated for each individual Lake Trout. Standard, maximum, and active metabolism were estimated using formulae described in the Supplementary Material sections. 1.2.1, 1.2.2, and 1.2.3 using individual temperature or acceleration detections, weight of each Lake Trout, and the mean laboratory weight of Lake Trout used by Cruz-Font et al. (2016). Raw acceleration detections from accelerometer acoustic transmitters (0–255 arbitrary units; a.u.) were used in calculations of active metabolism, following a protocol in Cruz-Font et al. (2016). These raw values capture varying tail beat frequency of the fish and are linearly related to the acceleration [\(accleation\left(m\bullet {s}^{-2}\right)=0+0.01922(a.u.)\)] experienced by the transmitter in the two directions perpendicular to the anterior–posterior axis of the fish. Detections that produced an a.u. value ≤ 8 a.u. were removed because they were considered “inactive” based on laboratory measurements and visual observations from Cruz-Font et al. (2016) that indicated fish were at rest. Daily theoretical AS for 26 wild free-swimming Lake Trout was estimated by taking the difference between daily mean MMR and daily mean SMR (AS; mg O2 h−1 kg−1; for formula see Supplementary Material 1.2.4). The remaining daily scope-for-activity was calculated by taking the difference between daily mean MMR and daily mean RMR (mg O2 h−1 kg−1; for formula see Supplementary Materials 1.2.5).
Statistical analysis
Data cleaning, visualization, mapping, and statistical analyses were conducted in R v4.2.3. Distributions and heteroscedasticity were assessed using {moments}, {fitdistrplus}, and {cars} packages.
To determine the relationship that day-of-year (DOY) and lake basin have on lake water temperature profiles, daily mean water temperature (°C) at each depth interval (2, 4, 6, 8, and 18 or 20 m) for each basin from July 2018 to July 2021 were analyzed using generalized additive mixed effects models (GAMMs) from the {mgcv} package. Models used a gamma error distribution with the link function set to log. Interpolated temperature values for each basin were determined using the model for every ~0.2 m from 0 to 20 m. The thermocline was identified by estimating depth that 15 °C isothermal occured throughout periods of stratification (June–October) of each year. We choose to use 15 °C isothermal because multiple studies have documented the importance of thermal habitats ≤ 15 °C for Lake Trout (Evans et al. 2007; Plumb et al. 2009; and Guzzo et al. 2017).
To understand the relationship that day-of-year and capture basin have on daily fish temperature use and RMR, we used GAMMs with a Gamma error distribution with either a log or identity link function. The relationship that day-of-year has on daily mean RMR, regardless of basin, was also analyzed using a GAMM with a Gaussian error distribution with a log link function. To obtain multiple comparisons, linear predictors (seasons, e.g., spring defined as March, April, and May and capture basin), and random effects from GAMMs for daily fish temperature, SMR, and RMR, were further tested using generalized linear mixed effects models (GLMMs) from the {glmmTMB} package. Models used Gaussian or gamma error distributions with a log link function.
To explain the relationship that day-of-year has on the daily mean theoretical SMR, MMR, and AS, we used GAMMs with a gamma error distribution with either an identity, inverse, or log link function. To illustrate the relationship that day-of-year has on the remaining daily mean scope-for-activity, we used GAMMs with an inverse link function.
Multiple comparison for GLMMs were conducted using {emmeans} with a Tukey multiplicity adjustment to evaluate capture basin differences within a season and seasonal differences for each capture basin (Supplementary Materials Table 1). To compensate for autocorrelation, autocorrelation structures were added to the models using the package {itsadug}or {glmmTMB}. For all response variables, several models were fitted and the model that retained the predictors of interest, fitted the data appropriately, and produced the lowest AIC value (evaluated using the function simulateResiduals() from {DHARMa} or mgcv::gam.check(); Supplementary Materials Table 2) were selected and are reported below.
Results
Lake Papineau temperature profiles
Thermal stratification occurred each year (2018–2021), starting in late-May to mid-June and ending in mid- to late-October. Thermocline depth (i.e., 15 °C) in all basins started shallow (2 m) in early summer (late-May to mid-June), and increased in depth through late summer to between approximately 5 m to 7.5 m in August and September (smoothers of DOY interacting with depth by basin—F = 331.2–336.3, effective degrees of freedom (edf) = 47.6–49.0, reference degrees of freedom (ref. df) = 65, p ≤ 0.001). The maximum thermocline depth was estimated from the models to occur late-September and be 5.41 m in the north basin, 6.95 m in the west basin, and 7.53 m in the east basin (Fig. 2; GAMM depth—F = 357, df = 1, p ≤ 0.001; basin—F = 10.43, df = 2, p = 0.005). Model selection resulted in the following model: mean daily water temperature at a given depth was regressed against lake basin and depth as linear predictors and four smoothers, with year as a random effect [Akaike information criterion (AIC) = 17,942.8, Supplementary Material Table 2]. The lake started to destratify in mid- to late-October, becoming isothermal (8 °C) by early- to mid-November.
Papineau Lake temperature profiles interpolated for each basin generated from generalized additive mixed effects models (GAMMs) for July 2018 to July 2021. The thermocline was identified using 15 °C isothermal throughout periods of stratification (June to October) of each year. We choose to use 15 °C isothermal because multiple studies have documented the importance of thermal habitats ≤ 15 °C for Lake Trout (Evans et al. 2007; Plumb et al. 2009; and Guzzo et al. 2017)
Lake Trout thermal habitat use
Daily thermal habitat use was more variable for each capture basin in summer and fall compared with spring and winter (Fig. 3a; GAMM DOY by capture basin smoothers—F = 1133.8–6089.8, edf = 12.8–12.9, ref. df = 13, p ≤ 0.001). The best candidate model included daily thermal habitat use as a response, with capture basin as a linear predictor, two smoothers, and fish ID and year as random effects (AIC = 55,781; Supplementary Material Table 2). In spring, the thermal habitat used was warmer than in winter but cooler than in summer and fall. Summer and fall thermal habitat use differed for the east and west basins, while in fish from the north basin, thermal habitat use did not differ between summer and fall. Winter thermal habitat use was the least variable when compared to other seasons. North basin fish used warmer temperatures in winter compared with fish in the east and west basins (Fig. 3b; GLMM season—χ2 = 24,253, df = 3, p ≤ 0.001; season *capture basin—χ2 = 546, df = 6, p ≤ 0.001; Supplementary Material Table 1).
Generalized additive mixed effects models and their 95% confidence intervals (a) were used to determine differences in mean daily thermal habitat use (colored points) for each basin in Lake Papineau by Lake Trout. Violin plots (b) represent the seasonal distribution of daily thermal habitat use for each basin by Lake Trout. Black circles denote the seasonal mean for each basin with error bars representing ± SEM. Statistical differences were determined by generalized linear mixed effects models and are denoted by upper case letters. Color denotes basin while shading in a denotes the available thermal habitat throughout the lake regardless of basin. The y-axis has been limited to 13 °C for scaling and interpretation purposes. Dotted boxes in a denote summer and winter. Data were collected from September 2017 to October 2020
Lake trout active metabolism
Daily RMR increased during warmer months and decreased during cooler months, with no observable differences between capture basins (Fig. 4a; GAMM DOY by capture basin smoothers—F = 315.7–3940.2, edf = 13.6–17.2, df = 18, p ≤ 0.001; Fig. 5a; GAMM DOY smoother—F = 1271.51, edf = 12.0, df = 13, p ≤ 0.001). Model selection indicated the best model contained RMR as a response, with capture basin as a linear predictor, two smoothers, and fish ID as random effect (AIC = 24,530; Supplementary Material Table 2). Lake Trout were more active in spring than in winter, but activity in spring was lower than in summer and fall. Fish in the north basin did not differ in RMR between spring and fall, while levels of RMR in summer regardless of basin were the highest compared with every other season. Levels of activity of fish in the east basin, however, did not differ between summer and fall (Fig. 4b; GLMM season—χ2 = 878.2, df = 2, p ≤ 0.001; season *capture basin—χ2 = 121.7, df = 6, p ≤ 0.001; Supplementary Material Table 1).
Generalized additive mixed effects models and their 95% confidence intervals (a) were used to determine differences in mean daily active metabolism (colored points) for Lake Trout. Violin plots (b) represent the seasonal distribution of daily active metabolism for each basin by Lake Trout. Black circles denote the seasonal mean for each basin with error bars representing ± SEM. Statistical differences were determined by generalized linear mixed effects models and are denoted by upper case letters. Color denotes basin while the lack of shading in a denotes spring and fall. Data were collected from May 2019 to April 2020
Generalized additive mixed effects models and their 95% confidence intervals (a) were used to determine daily mean standard, active, and maximum metabolism (b) and aerobic scope and scope-for-activity for Lake Trout in Lake Papineau. The daily mean regardless of basin is denoted by the colored points for each metric. Unshaded regions represent spring and fall. Data were collected from May 2019 to April 2020
Lake trout standard and maximum metabolism
Theoretical daily SMR and MMR for Lake Trout were influenced across months and seasons, with warmer months exhibiting greater estimates of SMR and MMR than cooler months (Fig. 5a; SMR—GAMM DOY smoothers—F = 12,054.23, edf = 15.8, df = 16, p ≤ 0.001; MMR—GAMM DOY smoothers—F = 9939, edf = 14.8, df = 15, p ≤ 0.001). The best model described daily SMR or MMR as the response, one smoother, and fish ID and year as random effects (SMR—AIC = 96,491; MMR—AIC = 142,434; Supplementary Material Table 2). Lake Trout had greater SMR and MMR in spring than in winter, but SMR and MMR in spring was lower than in summer and fall. Daily SMR and MMR were greater in summer compared to any other season but at times were comparable to fall levels. Winter SMR and MMR estimates were the lowest compared with every other season (Fig. 5a).
Lake trout aerobic scope and scope-for-activity
Estimates of daily AS and scope-for-activity for Lake Trout varied among months and seasons with warmer months resulting in increases in aerobic scope than cooler months (Fig. 5b; AS—GAMM DOY—F = 3086.37, edf = 14.9, df = 15, p ≤ 0.001; scope-for-activity—GAMM DOY—F = 566.21, edf = 14.3, df = 15, p ≤ 0.001). The best model described daily AS or scope-for-activity as the response, one smoother, and year as random effect (AS—AIC = 134,887; scope-for-activity—AIC = 9549; Supplementary Material Table 2). Aerobic scope was greater during summer than during any other period, however, estimates of aerobic scope during fall were momentarily at levels comparable with estimates during summer (Fig. 5b). Scope-for-activity followed similar trajectories annually (i.e., greater during summer and lower during winter) but was greatest late fall when water temperature starts to cool, and Lake Trout become more active. The theoretical scope-for-activity was estimated to range between 47% and 74% of theoretical aerobic scope.
Discussion
Our observations indicated that thermal habitat availability (Fig. 2) dictated Lake Trout habitat use (Fig. 3), and in turn influenced their RMR, aerobic scope, and scope-for-activity (Fig. 4 and 5). We did not observe fish captured from the smaller basins to exhibit reduced activity and metabolic costs caused by a lack of suitable thermal habitats, and thus we reject our proposed hypothesis (Fig. 3). We did observe unexpected increases in scope-for-activity when least expected (i.e., fall and winter; Fig. 5b) that were likely related to changes in thermal habitat availability and thermal habitat use (Figs. 2 and 3). Scope-for-activity followed seasonal trends but at times was much closer to aerobic scope, thus potentially affecting the ability of Lake Trout to allocate energy towards other processes such as somatic and/or gonadal growth (Fig. 5b).
The influence of temperature on aerobic scope and scope-for-activity is well-established (Fry 1971), but in wild populations other factors can influence aerobic scope and scope-for-activity such as phenotypic variation, food availability, and hypoxia (Evans 2007; Enders and Boisclair 2016; Metcalfe et al. 2016). Our observations of theoretical SMR and MMR, and estimated RMR, indicated that other environmental factors might be influencing metabolic processes for Lake Trout in Lake Papineau (Fig. 5a). Food availability can cause salmonid species to exhibit phenotypic plasticity in metabolic processes, with individuals that have greater flexibility in SMR, MMR, and AS, having greater growth rates than less-flexible conspecifics (Norin and Malte 2012; Auer et al. 2015a, b). Prey diversity in Lake Papineau is quite limited for Lake Trout with only one pelagic prey fish, rainbow smelt Osmerus mordax, documented (Pers. Comm. Québec Ministère des Forêts, de la Faune et des Parcs). This limitation in prey could be contributing to changes in RMR and scope-for-activity for Lake Trout, particularly in winter when food availability might become even more limited. Changes in food availability has been shown to affect SMR and activity in salmonids, with individuals that experience reductions in food having lower SMR but exhibiting higher rates of activity and greater lipid reserves (Byström et al. 2006; Auer et al. 2016). Flexibility in aerobic scope in response to changing food resources can allow energy to be allocated to other processes, such as scope-for-activity, and thus Lake Trout in Lake Papineau might have greater flexibility in scope-for-activity (Auer et al. 2016). This flexibility might provide Lake Trout the ability to respond quickly to altered thermal habitats, changes in prey resources, and other anthropogenic disturbances.
Fish behaviorally thermoregulate by actively seeking preferred thermal habitats (Beitinger and Fitzpatrick 1979; Amat-Trigo et al. 2023). Metabolic thermal optimum (Topt) for Lake Trout has been reported within a narrow thermal range of 10 ± 2 °C (McCauley and Tait 1970; Stewart et al. 1983; Magnuson et al. 1990). Several studies, however, have shown wild populations of Lake Trout will occupy thermal habitats at or below Topt (Bergstedt et al. 2003; Mackenzie-Grieve and Post 2006; Blanchfield et al. 2009; Guzzo et al. 2016). Our observations of daily thermal habitat use were consistently below the historically reported Topt of 10 °C and ranged from 2.8 °C ± 0.01 in winter to 8.2 °C ± 0.05 in summer (Fig. 3). Our findings add to the growing evidence that wild populations of Lake Trout often use colder thermal habitats than previously hypothesized or observed (Elrod et al. 1996; Bergstedt et al. 2012; Raby et al. 2019). Cold-water fisheries management often relies on historically derived laboratory observations of thermal ranges for Lake Trout, yet those observations might be unsuitable as the basis of management decisions because they do not accurately capture thermal habitats used by wild populations (Plumb and Blanchfield 2009). If fish are behaviorally thermoregulating by actively seeking colder thermal habitats as our results suggest, then there might be increased energy expenditures resulting in an individual having less energy for growth and reproduction (Fig. 5). Our results are further supported by similar observations by others, particularly when thermal habitats become seasonally limited (e.g., summer; Plumb et al. 2014; Cruz-Font et al. 2019). This evidence suggests that as cold-water habitats are reduced by changes in climatic conditions and/or other anthropogenic stressors, populations of Lake Trout might experience unaccounted increases in energy expenditures (RMR), that could result in limited allocation of energy into survival, somatic growth, and reproductive potential.
In freshwater ecosystems with complex geomorphology (e.g., multibasin lakes; Fig. 1), thermal habitats can be discontinuous during parts of the year (e.g., summer; Fig. 2), and might affect the ability of Lake Trout (and other cold-water fishes) to behaviorally thermoregulate (Kraemer et al. 2015; Guzzo and Blanchfield 2017; Ridgway et al. 2022). In southern populations, thermal preferences typically result in the seasonal use of deep hypolimnetic zones (Dillon et al. 2003) or metalimnetic and shallow hypolimnetic zones (Christie and Regier 1988). Changes in access to thermal habitats within a lake, however, could cause an individual to expend more energy foraging and seeking habitats compared to in systems where prey and/or habitats are more concentrated (e.g., small basins and/or lakes with less complex geomorphology; Brett 1971; Tunney et al. 2012; McMeans et al. 2016; Rodrigues et al. 2022). Shifts in season duration (e.g., extended springs) have already been shown to change Lake Trout phenologies, causing reduced spatial occupancy of spring littoral habitats, influencing prey consumption and growth (Guzzo et al. 2017). Cold-water habitats are particularly susceptible to anthropogenic stressors that are vital for cold-water fishes (e.g., Lake Trout; Tunney et al. 2014; Hansen et al. 2022) and our study provides direct evidence of how important thermal habitats are in the context of bioenergetics and metabolism for this cold-water fish.
In the wild, aerobic scope and active metabolism are driven simultaneously by multiple factors, making them difficult to accurately predict when using laboratory-derived single variable models (Brownscombe et al. 2022). Our approach further extends bioenergetics modeling by measuring multiple metrics simultaneously in situ (i.e., temperature use and activity) to estimate metabolic costs for wild populations. This approach can provide more accurate and ecologically-relevant information on fish metabolism and bioenergetics to scientists and fisheries managers.
Our study estimated metabolic costs for wild Lake Trout throughout an entire year. The ability to estimate multiple parameters associated with metabolism for the species in the wild has been a gap in the literature and our work sheds light on how thermal habitats affect energy expenditures. In freshwater ecosystems of moderate size (surface area ≤ ~ 5,000 ha), where cold-water habitats will likely be reduced by climatic changes (Jane et al. 2021; Kraemer et al. 2021), metabolic costs for cold-water species will likely increase, affecting population health (i.e., survival, growth, and reproduction) and possibly resulting in extirpation from systems (Sharma et al. 2007; Schulte 2015). To successfully manage cold-water fisheries for ecological resiliency, insight into thermal habitat use and the metabolic costs of movement will be vital (Hansen et al. 2022). Future studies should continue to focus on how multiple factors affect Lake Trout metabolic costs (and those of other fishes) because this information will further facilitate the sustainable management of Lake Trout fisheries.
Data availability
Data is available at the following GitHub repository.
References
Amat-Trigo F, Andreou D, Gillingham PK, Britton JR (2023) Behavioural thermoregulation in cold-water freshwater fish: Innate resilience to climate warming? Fish Fish 24:187–195. https://doi.org/10.1111/faf.12720
Auer SK, Salin K, Rudolf AM et al (2015a) Flexibility in metabolic rate confers a growth advantage under changing food availability. J Anim Ecol 84:1405–1411. https://doi.org/10.1111/1365-2656.12384
Auer SK, Salin K, Rudolf AM et al (2015b) The optimal combination of standard metabolic rate and aerobic scope for somatic growth depends on food availability. Funct Ecol 29:479–486. https://doi.org/10.1111/1365-2435.12396
Auer SK, Salin K, Anderson GJ, Metcalfe NB (2016) Flexibility in metabolic rate and activity level determines individual variation in overwinter performance. Oecologia 182:703–712. https://doi.org/10.1007/s00442-016-3697-z
Beitinger TL, Fitzpatrick LC (1979) Physiological and ecological correlates of preferred temperature in fish. Integr Comp Biol 19:319–329. https://doi.org/10.1093/icb/19.1.319
Bergstedt RA, Argyle RL, Seelye JG et al (2003) In situ determination of the annual thermal habitat use by lake trout (Salvelinus namaycush) in Lake Huron. J Great Lakes Res 29:347–361. https://doi.org/10.1016/S0380-1330(03)70499-7
Bergstedt RA, Argyle RL, Krueger CC, Taylor WW (2012) Bathythermal habitat use by strains of great lakes-and finger lakes-origin lake trout in Lake Huron after a change in prey fish abundance and composition. Trans Am Fish Soc 141:263–274. https://doi.org/10.1080/00028487.2011.651069
Blanchfield PJ, Tate LS, Plumb JM et al (2009) Seasonal habitat selection by lake trout (Salvelinus namaycush) in a small Canadian shield lake: Constraints imposed by winter conditions. Aquat Ecol 43:777–787. https://doi.org/10.1007/s10452-009-9266-3
Brett JR (1971) Energetic responses of salmon to temperature. A study of some thermal relations in the physiology and freshwater ecology of sockeye salmon (Oncorhynchus nerkd). Integr Comp Biol 11:99–113. https://doi.org/10.1093/icb/11.1.99
Brownscombe JW, Gutowsky LFG, Danylchuk AJ, Cooke SJ (2014) Foraging behaviour and activity of a marine benthivorous fish estimated using tri-axial accelerometer biologgers. Mar Ecol Prog Ser 505:241–251. https://doi.org/10.3354/meps10786
Brownscombe JW, Griffin LP, Chapman JM et al (2020) A practical method to account for variation in detection range in acoustic telemetry arrays to accurately quantify the spatial ecology of aquatic animals. Methods Ecol Evol 11:82–94. https://doi.org/10.1111/2041-210X.13322
Brownscombe JW, Raby GD, Murchie KJ et al (2022) An energetics–performance framework for wild fishes. J Fish Biol 101:4–12. https://doi.org/10.1111/jfb.15066
Byström P, Andersson J, Kiessling A, Eriksson LO (2006) Size and temperature dependent foraging capacities and metabolism: Consequences for winter starvation mortality in fish. Oikos 115:43–52. https://doi.org/10.1111/j.2006.0030-1299.15014.x
Chipps SR, Wahl DH (2008) Bioenergetics modeling in the 21st Century: Reviewing new insights and revisiting old constraints. Trans Am Fish Soc 137:298–313. https://doi.org/10.1577/t05-236.1
Christie CC, Regier H, a, (1988) Measures of optimal thermal habitat and their relationship yeilds for four commercial fish species. Can J Fish Aquat Sci 45:301–314
Cruz-Font L, Shuter BJ, Blanchfield PJ (2016) Energetic costs of activity in wild lake trout: A calibration study using acceleration transmitters and positional telemetry. Can J Fish Aquat Sci 73:1237–1250. https://doi.org/10.1139/cjfas-2015-0323
Cruz-Font L, Shuter BJ, Blanchfield PJ et al (2019) Life at the top: Lake ecotype influences the foraging pattern, metabolic costs and life history of an apex fish predator. J Anim Ecol 88:702–716. https://doi.org/10.1111/1365-2656.12956
Dillon PJ, Clark BJ, Molot LA, Evans HE (2003) Predicting the location of optimal habitat boundaries for lake trout (Salvelinus namaycush) in Canadian shield lakes. Can J Fish Aquat Sci 60:959–970. https://doi.org/10.1139/f03-082
Elrod JH, O’Gorman R, Schneider CP (1996) Bathythermal distribution, maturity, and growth of lake trout strains stocked in U.S. waters of Lake Ontario, 1978–1993. J Great Lakes Res 22:722–743. https://doi.org/10.1016/S0380-1330(96)70992-9
Enders EC, Boisclair D (2016) Effects of environmental fluctuations on fish metabolism: Atlantic salmon Salmo salar as a case study. J Fish Biol 88:344–358. https://doi.org/10.1111/jfb.12786
Evans DO (2007) Effects of hypoxia on scope-for-activity and power capacity of lake trout (Salvelinus namaycush). Can J Fish Aquat Sci 64:345–361. https://doi.org/10.1139/f07-007
Fry FEJ (1947) Effects of the environment on animal activity. Publications of the Ontario Fisheries Research Laboratory 55:1–62
Fry FEJ (1971) The effect of environmental factors on the physiology of fish. In: Fish Physiology. Academic press
Gunn JM, Louste-Fillion J (2022) Adaptability of lake charr (Salvelinus namaycush) in a changing world: Newly recolonized landscapes reveal the significance of traits shaped during the Pleistocene. Environmental Biology of Fishes 21. https://doi.org/10.1007/s10641-022-01311-y
Guzzo MM, Blanchfield PJ (2017) Climate change alters the quantity and phenology of habitat for lake trout (Salvelinus namaycush) in small boreal shield lakes. Can J Fish Aquat Sci 74:871–884. https://doi.org/10.1139/cjfas-2016-0190
Guzzo MM, Blanchfield PJ, Chapelsky AJ, Cott PA (2016) Resource partitioning among top-level piscivores in a sub-Arctic lake during thermal stratification. J Great Lakes Res 42:276–285. https://doi.org/10.1016/j.jglr.2015.05.014
Guzzo MM, Blanchfield PJ, Rennie MD (2017) Behavioral responses to annual temperature variation alter the dominant energy pathway, growth, and condition of a cold-water predator. Proc Natl Acad Sci 114:201702584. https://doi.org/10.1073/pnas.1702584114
Guzzo MM, Mochnacz NJ, Durhack T et al (2019) Effects of repeated daily acute heat challenge on the growth and metabolism of a cold water stenothermal fish. J Exp Biol 222:1–12. https://doi.org/10.1242/jeb.198143
Hansen GJA (2021) Novel thermal habitat in lakes. Nat Clim Chang 11:470–471. https://doi.org/10.1038/s41558-021-01067-w
Hansen MJ, Boisclair D, Brandt SB et al (1993) Applications of bioenergetics models to fish ecology and management: Where do we go from here? Trans Am Fish Soc 122:1019–1030. https://doi.org/10.1577/1548-8659(1993)122%3c1019:aobmtf%3e2.3.co;2
Hansen GJA, Wehrly KE, Vitense K, et al (2022) Quantifying the resilience of coldwater lake habitat to climate and land use change to prioritize watershed conservation. Ecosphere 1–18. https://doi.org/10.1002/ecs2.4172
Harvey CJ, Schram ST, Kitchell JF (2003) Trophic relationships among lean and siscowet lake trout in Lake Superior. Trans Am Fish Soc 132:219–228. https://doi.org/10.1577/1548-8659(2003)132%3c0219:TRALAS%3e2.0.CO;2
Hugie DM, Dill LM (1994) Fish and game: A game theoretic approach to habitat selection by predators and prey. J Fish Biol 45:151–169
Jane SF, Hansen GJA, Kraemer BM et al (2021) Widespread deoxygenation of temperate lakes. Nature 594:66–70. https://doi.org/10.1038/s41586-021-03550-y
Kao Y-C, Madenjian CP, Bunnell DB et al (2015) Temperature effects induced by climate change on the growth and consumption by salmonines in Lakes Michigan and Huron. Environ Biol Fish 98:1089–1104. https://doi.org/10.1007/s10641-014-0352-6
Kepler MV, Wagner T, Sweka JA (2014) Comparative bioenergetics modeling of two lake trout morphotypes. Trans Am Fish Soc 143:1592–1604. https://doi.org/10.1080/00028487.2014.954051
Kitchell JF (1983) Energetics. In: Webb PW, Weihs D (eds) Fish Biomechanics. Praeger Publishers, pp 321–338
Kitchell JF, Stewart DJ, Weininger D (1977) Applications of a bioenergetics model to yellow perch (Perca flavescens) and walleye (Stizostedion vitreum vitreum). Journal Fisheries Research Board of Canada 34:1922–1935
Kraemer BM, Anneville O, Chandra S et al (2015) Morphometry and average temperature affect lake stratification responses to climate change. Geophys Res Lett 42:4981–4988. https://doi.org/10.1002/2015GL064097.Received
Kraemer BM, Pilla RM, Woolway RI et al (2021) Climate change drives widespread shifts in lake thermal habitat. Nat Clim Chang 11:521–529. https://doi.org/10.1038/s41558-021-01060-3
Mackenzie-Grieve JL, Post JR (2006) Thermal habitat use by lake trout in two contrasting Yukon territory lakes. Trans Am Fish Soc 135:727–738. https://doi.org/10.1577/T05-138.1
Madenjian CP, O’Connor DV (1999) Laboratory evaluation of a lake trout bioenergetics model. Trans Am Fish Soc 128:802–814. https://doi.org/10.1577/1548-8659(1999)128%3c0802:leoalt%3e2.0.co;2
Madenjian CP, Connor DVO, Nortrup DA (2000) A new approach toward evaluation of fish bioenergetics models1. Can J Fish Aquat Sci 57:1025–1032
Madenjian CP, Keir MJ, Whittle DM, Noguchi GE (2010) Sexual difference in PCB concentrations of lake trout (Salvelinus namaycush) from Lake Ontario. Sci Total Environ 408:1725–1730. https://doi.org/10.1016/j.scitotenv.2009.12.024
Madenjian CP, Pothoven SA, Kao Y (2013) Reevaluation of lake trout and lake whitefish bioenergetics models. J Great Lakes Res 39:358–364. https://doi.org/10.1016/j.jglr.2013.03.011
Magnuson JJ, Meisner JD, Hill DK (1990) Potential changes in the thermal habitat of Great Lakes fish after global climate warming. Trans Am Fish Soc 119:254–264. https://doi.org/10.1577/1548-8659(1990)119%3c0254:pcitth%3e2.3.co;2
Martin NV, Olver CH (1980) The lake charr, Salvelinus namaycush. In: Balon EK (ed) Charrs: salmonid fishes of the genus Salvelinus. Kluwer Boston
McCauley RW, Tait JS (1970) Preferred temperature of yearling lake trout, Salvelinus namaycush. Journal Fisheries Research Board of Canada 27:1729–1733
McDonald ME, Hershey AE, Miller MC (1996) Global warming impacts on lake trout in arctic lakes. Limnol Oceanogr 41:1102–1108. https://doi.org/10.4319/lo.1996.41.5.1102
McMeans BC, McCann KS, Tunney TD et al (2016) The adaptive capacity of lake food webs: From individuals to ecosystems. Ecol Monogr 86:4–19. https://doi.org/10.1890/15-0288.1
Metcalfe NB, Van Leeuwen TE, Killen SS (2016) Does individual variation in metabolic phenotype predict fish behaviour and performance? J Fish Biol 88:298–321. https://doi.org/10.1111/jfb.12699
Morbey YE, Addison P, Shuter BJ, Vascotto K (2006) Within-population heterogeneity of habitat use by lake trout Salvelinus namaycush. J Fish Biol 69:1675–1696. https://doi.org/10.1111/j.1095-8649.2006.01236.x
Morris DW (2003) Toward an ecological synthesis: A case for habitat selection. Oecologia 136:1–13. https://doi.org/10.1007/s00442-003-1241-4
Muir AM, Hansen MJ, Bronte CR, Krueger CC (2016) If Arctic charr Salvelinus alpinus is ‘the most diverse vertebrate’, what is the lake charr Salvelinus namaycush? Fish Fish 17:1194–1207. https://doi.org/10.1111/faf.12114
Muir AM, Krueger CC, Hansen MJ, Riley SC (eds) (2021) The Lake Charr Salvelinus namaycush: Biology, Ecology, Distribution, and Management
Nathan R, Getz WM, Revilla E et al (2008) A movement ecology paradigm for unifying organismal movement research. Proc Natl Acad Sci 105:19052–19059. https://doi.org/10.1073/pnas.0800375105
Norin T, Malte H (2011) Repeatability of standard metabolic rate, active metabolic rate and aerobic scope in young brown trout during a period of moderate food availability. J Exp Biol 214:1668–1675. https://doi.org/10.1242/jeb.054205
Norin T, Metcalfe NB (2019) Ecological and evolutionary consequences of metabolic rate plasticity in response to environmental change. Philosophical Transactions of the Royal Society B: Biological Sciences 374:20180180. https://doi.org/10.1098/rstb.2018.0180
Papastamatiou YP, Friedlander AM, Caselle JE, Lowe CG (2010) Long-term movement patterns and trophic ecology of blacktip reef sharks (Carcharhinus melanopterus) at Palmyra Atoll. J Exp Mar Biol Ecol 386:94–102. https://doi.org/10.1016/j.jembe.2010.02.009
Pazzia I, Trudel M, Ridgway M, Rasmussen JB (2002) Influence of food web structure on the growth and bioenergetics of lake trout (Salvelinus namaycush). Can J Fish Aquat Sci 59:1593–1605. https://doi.org/10.1139/f02-128
Piccolo JJ, Hubert WA, Whaley RA (1993) Standard weight equation for lake trout. North Am J Fish Manag 13:401–404. https://doi.org/10.1577/1548-8675(1993)013%3c0401:SWEFLT%3e2.3.CO;2
Pincock DG (2012) False detections: What they are and how to remove them from detection data. Amirix Document DOC-004691 Version 03 Available: http://vemco.com/wp-content/uploads/201. https://doi.org/10.1109/TNSRE.2002.802877
Plumb JM, Blanchfield PJ (2009) Performance of temperature and dissolved oxygen criteria to predict habitat use by lake trout (Salvelinus namaycush)1. Can J Fish Aquat Sci 66:2011–2023. https://doi.org/10.1139/F09-129
Plumb JM, Blanchfield PJ, Abrahams MV (2014) A dynamic-bioenergetics model to assess depth selection and reproductive growth by lake trout (Salvelinus namaycush). Oecologia 175:549–563. https://doi.org/10.1007/s00442-014-2934-6
Raby GD, Johnson TB, Kessel ST, et al (2019) Pop-off data storage tags reveal niche partitioning between native and non-native predators in a novel ecosystem. Journal of Applied Ecology 1–11. https://doi.org/10.1111/1365-2664.13522
Reid AJ, Carlson AK, Creed IF et al (2019) Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol Rev 94:849–873. https://doi.org/10.1111/brv.12480
Ridgway MS, Bell AH, Lacombe NA, et al (2022) Thermal niche and habitat use by co-occurring lake trout (Salvelinus namaycush) and brook trout (S. fontinalis) in stratified lakes. Environ Biol Fish. https://doi.org/10.1007/s10641-022-01368-9
Rodrigues TH, Chapelsky AJ, Hrenchuk LE et al (2022) Behavioural responses of a cold-water benthivore to loss of oxythermal habitat. Environ Biol Fish. https://doi.org/10.1007/s10641-022-01335-4
Rottiers DV (1993) Energy budget for yearling lake trout, Salvelinus namaycush. J Freshw Ecol 8:319–327. https://doi.org/10.1080/02705060.1993.9664871
Schindler DW, Curtis PJ, Parker BR, Stainton MP (1996) Consequences of climate warming and lake acidification for UV-B penetration in North American boreal lakes. Nature 379:705–708. https://doi.org/10.1038/379705a0
Schulte PM (2015) The effects of temperature on aerobic metabolism: Towards a mechanistic understanding of the responses of ectotherms to a changing environment. J Exp Biol 218:1856–1866. https://doi.org/10.1242/jeb.118851
Sharma S, Jackson DA, Minns CK, Shuter BJ (2007) Will northern fish populations be in hot water because of climate change? Glob Change Biol 13:2052–2064. https://doi.org/10.1111/j.1365-2486.2007.01426.x
Sherwood GD, Pazzia I, Moeser A et al (2002) Shifting gears: Enzymatic evidence for the energetic advantage of switching diet in wild-living fish. Can J Fish Aquat Sci 59:229–241. https://doi.org/10.1139/f02-001
Shuter BJ, Jones ML, Korver RM, Lester NP (1998) A general, life history based model for regional management of fish stocks: The inland lake trout (Salvelinus namaycush) fisheries of Ontario. Can J Fish Aquat Sci 55:2161–2177. https://doi.org/10.1139/cjfas-55-9-2161
Stewart DJ, Weininger D, Rottiers DV, Edsall TA (1983) An energetics model for lake trout, Salvelinus namaycush : Application to the Lake Michigan population. Can J Fish Aquat Sci 40:681–698. https://doi.org/10.1139/f83-091
Thackeray SJ, Sparks TH, Frederiksen M et al (2010) Trophic level asynchrony in rates of phenological change for marine, freshwater and terrestrial environments. Glob Change Biol 16:3304–3313. https://doi.org/10.1111/j.1365-2486.2010.02165.x
Tunney TD, McCann KS, Lester NP, Shuter BJ (2012) Food web expansion and contraction in response to changing environmental conditions. Nat Commun 3:1105. https://doi.org/10.1038/ncomms2098
Tunney TD, McCann KS, Lester NP, Shuter BJ (2014) Effects of differential habitat warming on complex communities. Proc Natl Acad Sci USA 111:8077–8082. https://doi.org/10.1073/pnas.1319618111
Tytler P, Calow P (eds) (1985) Fish Energetics: New Perspectives. Croom Helm Ltd., London, UK
Vandergoot CS, Murchie KJ, Cooke SJ et al (2011) Evaluation of two forms of electroanesthesia and carbon dioxide for short-term anesthesia in walleye. North Am J Fish Manag 31:914–922. https://doi.org/10.1080/02755947.2011.629717
Winter J (1996) Advances in Underwater Biotelemetry. In: Murphy BR, Willis DW (eds) Fisheries Techniques, 2nd edn. pp 555–590
Zimmerman MS, Krueger CC (2009) An ecosystem perspective on re-establishing native deepwater fishes in the Laurentian Great Lakes. North Am J Fish Manag 29:1352–1371. https://doi.org/10.1577/M08-194.1
Acknowledgements
We thank Dominic Monaco, Douglas Goosen, and Patrick Johnson of Kenauk Nature for their invaluable information about Lake Papineau. We also thank Jessica Reid, Matthew Futia, Auston Chhor, Jennifer Cooke, Brooke Etherington, Shannon Clarke, Mickey Phillip, Josée Gagnon, Peter Holder, Jill Brooks, Jacqueline Chapman, Taylor Ward, Alice Abrams, Jessica Taylor, Jinming Wu, and various Kenauk Interns for assistance in the field, and Connor Reeve for assistance in understanding the relationship between swim-speed, acceleration, and metabolism. Dr. Dirk Algera, Michael O’Brien, and Dr. Robert Lennox provided insight on the statistical analyses used in the manuscript. We also thank several anonymous referees.
Funding
This project was Supported by NSERC-CRD grant Cooke CRD 2017 and Kenauk Nature.
Author information
Authors and Affiliations
Contributions
All authors contributed to the design of the study. Field work and sampling were conducted by all authors except M.P. Data analyses and figures were done by B.L.H. The manuscript was written by B.L.H with all authors contributing to revisions. Study design was conceived by B.L.H., J.E.C., D.P.P, J.E.M., M.P., and S.J.C.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hlina, B.L., Glassman, D.M., Lédée, E.J.I. et al. Habitat-dependent metabolic costs for a wild cold-water fish. Aquat Sci 86, 36 (2024). https://doi.org/10.1007/s00027-024-01052-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-024-01052-3