Abstract
There is limited knowledge of how philopatry influences the spatial ecology of iteroparous anadromous species during their spawning migrations, but this knowledge is important in understanding population responses to interventions such as river reconnection. Here, acoustic telemetry was applied to twaite shad Alosa fallax and hybrids (n = 184) during their freshwater spawning migration, enabling quantification of philopatry across spawning migrations and assessment of the factors affecting space use. Tagged fish moved a median of 7 km day−1. Their migration routes were tortuous (median ratio of total distance moved/upstream extent = 2.8), and included multiple upstream/downstream direction changes (median = 27) over a median freshwater movement distance of 247 km. Females occupied larger core areas than males, but previous spawning experience, body length, tagging status, and introgression with A. alosa did not predict core area size. Seventy-one fish returned a year after tagging, with a median freshwater residency of 33 days. Between years, intra-individual similarity in space use was significantly greater than inter-individual similarity, providing strong evidence of philopatry. These results provide insights into how spawning philopatry and phenotype influence riverine space use in a threatened anadromous species, and have implications for river reconnection efforts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Iteroparity is a bet-hedging strategy that aims to maximise fitness in relatively unstable environments (Glebe and Leggett 1981; Stearns 1998). In anadromous fish species, iteroparity reduces the relative importance of individual spawning migrations by prioritising adult survival to ensure future reproduction. Iteroparous individuals may therefore be relatively risk-averse in their migratory behaviours (Warner 2005). Natal philopatry is also evident in many anadromous species, especially those in the Salmonidae family (Dittman and Quinn 1996; Thorstad et al. 2021). This brings the advantage of local genetic adaptation, with patterns of local population structuring usually evident within salmonid species (Beacham and Withler 2017; Sylvester et al. 2018). In iteroparous species, spawning philopatry—the return of adults to a previous spawning site—can also occur (Stepien et al. 2009).
Anadromous members of the herring family Clupeidae are distributed throughout the Northern Hemisphere and comprise both iteroparous and semelparous species (Leggett and Carscadden 1978). Some widely distributed anadromous clupeid species utilise both strategies, with iteroparity prevailing in environmentally variable northern rivers and semelparity prevailing in more stable southern rivers (Leggett and Carscadden 1978; Aprahamian et al. 2003a, b). In iteroparous clupeids, individuals can generally survive several annual spawning events, with natal philopatry suggested by strong patterns of population structuring; genetic isolation by distance is usually evident between different spawning populations (Hasselman et al. 2010; Jolly et al. 2012; Sabatino et al. 2022). Distinct sub-populations are also often present in specific river tributaries (Carscadden and Leggett 1975; Jolly et al. 2012). In addition, recent telemetry work has indicated that repeat-spawning adults primarily return to the same catchment, despite sharing marine habitats with individuals from other spawning populations (Davies et al. 2020).
Natal philopatry is likely to increase the likelihood that returning adults will find mates, and favourable spawning and nursery habitats (Hendry et al. 2003). However, whether natal philopatry by virgin spawners leads to spawning philopatry in subsequent migrations is uncertain, especially in rivers where migration routes are potentially impeded by engineered structures. While there is evidence that migration timing can be ‘fine-tuned’ throughout the life cycle of iteroparous fish (Tibblin et al. 2016), there is high uncertainty on the inter-annual differences in, and the biological and environmental drivers of, riverine space use by individual fish, such as body size and sex, despite being important for impact assessments of spawning movements and behaviours (Pess et al. 2014).
While natal philopatry is a highly adaptive strategy, straying can buffer anadromous populations from environmental perturbations and enable colonisation of new habitats and recolonisation after local extinctions (Keefer and Caudill 2014). Increasingly, conservation interventions in impounded rivers, including barrier removals and fish pass installation, are being used to restore aspects of longitudinal connectivity for migratory fishes and provide access once more to former reproductive areas (Nunn and Cowx 2012; Bubb et al. 2021). Reconnection potentially represents a special form of environmental instability where population responses can be influenced by their degree of philopatry to natal sites in the pre-reconnection period (Pess et al. 2014). There are, however, considerable knowledge gaps in how iteroparous, non-salmonid fishes react to river reconnection, including how natal philopatry influences the extent to which individuals alter their selection of spawning areas.
An anadromous and iteroparous clupeid that is becoming increasingly threatened across its range is the twaite shad Alosa fallax, which is distributed across the north-eastern Atlantic and Mediterranean (Aprahamian et al. 2003a). River fragmentation and overexploitation have driven substantial declines in their populations (de Groot 1990; Aprahamian et al. 2003b), resulting in international conservation designations (e.g. listing on Annexes II and V of the European Union Habitats Directive; Council of the European Communities 1992). In the northern part of their range, previously spawned fish often represent over 50% of the spawning run (Aprahamian et al. 2003b). They also readily hybridise with sympatric allis shad Alosa alosa, with higher rates of genetic introgression being associated with the anthropogenic fragmentation of their spawning rivers, which results in overlapping spawning ranges (Jolly et al. 2011; Taillebois et al. 2020; Antognazza et al. 2021; Sabatino et al. 2022). High rates of hybridisation could be important to note given that hybrids may use migration routes and spawning destinations that are intermediate between the parental routes and have higher variability in their migration patterns (Alvarado et al. 2014; Delmore and Irwin 2014).
Advances in passive telemetry have facilitated the identification of the spawning migration routes of anadromous fish (e.g. Kirk and Caudill 2017; Lennox et al. 2019; Mack et al. 2021) and individual variability in their movements and habitat choice (e.g. Moore et al. 2017; Davies et al. 2022). For twaite shad, refinements to tagging protocols have enabled internal implantation of acoustic transmitters to track their spawning migrations and marine habitat use (Bolland et al. 2019; Davies et al. 2020), and enabled individuals to be tracked over multiple spawning migrations. This approach has already revealed the negative effect of anthropogenic barriers on twaite shad upstream migrations in the highly engineered River Severn basin, western Britain. In this basin, a series of navigation and flow regulation weirs have already been demonstrated to prevent and delay the upstream movements of returning spawning shad that were tracked over successive spawning migrations (Davies et al. 2023). Here, the focus is on how philopatry among individuals tracked during at least two successive spawning migrations influenced their riverine spatial habitat use, with the influence of sex, body length, genetic introgression, tagging and migratory experience also considered. The objectives were to (i) quantify the movements, extent of freshwater residency and catchment-scale space use of twaite shad and their hybrids during their spawning migrations; (ii) test the individual and genetic factors affecting riverine space use during their spawning migrations (‘core area size’); and (iii) assess the extent of philopatry during their spawning migration in the River Severn basin by returning fish.
Methods
Study duration and area
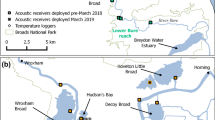
The study was conducted on the spawning migrations of twaite shad (including their hybrids) in the River Severn in 2018, 2019 and 2020. These migrations tend to commence in late April and are concluded by late June (Antognazza et al. 2019). The study area was the lower river catchment that includes confluences with two major tributaries, the River Teme and River Avon, and eight major weirs (four on the main river channel, and two on each of the lower reaches of the River Teme and River Avon) that result in high fragmentation (Fig. 1). The normal tidal limit is at Maisemore (hereafter Weir S1a) and Llanthony Weirs (S1b) on the western and eastern branches of the river, respectively (Fig. 1), although large spring tides can penetrate the river up to Upper Lode Weir (hereafter Weir S2). Further description of the study area, including detailed description of anthropogenic structures and fish passage infrastructure, is provided in Davies et al. (2023).
The River Severn catchment study area, including locations of release of acoustic-tagged twaite shad Alosa fallax (black stars), weirs (bars) and acoustic receivers (black dots within white circles) in the rivers Severn, Teme and Avon, UK. The black arrows denote the direction of the flow. For full description of weirs and characteristics, see Davies et al. (2023)
Twaite shad capture and tagging
At the commencement of their migration season in May 2018 and 2019, upstream-migrating twaite shad were captured by angling in the weir pools of S1a and S2. In addition, twaite shad were captured at S2 using an aluminium trap featuring a manually operated sliding trapdoor positioned at the upstream exit of the notch fish pass. Fish were captured individually as they exited the pass and immediately removed from the trap via hand net. Following their anaesthesia (ethyl 3-aminobenzoate methanesulfonate; MS-222), all fish were weighed (to the nearest 10 g), measured (fork length, to the nearest millimetre) and sexed (see subsection ‘Assignment of sex and hybrid classes’), and approximately three scales were removed for analysis of spawning history. These scales were analysed to determine their number of spawning-marks (i.e. their previous spawning history) on a projecting microscope (×48 magnification) (Baglinière et al. 2001). In addition, a tissue sample from a pelvic fin biopsy was taken from each individual for subsequent genetic analysis.
The fish were tagged with 69 kHz Vemco V9 acoustic transmitters (29 mm long × 9 mm diameter, 4.7 g weight in air) (innovasea.com), following the protocol of Bolland et al. (2019), under UK Home Office project licence PD6C17B56. All captured fish were tagged, apart from those (< 1%) displaying injury due to possible attack by predators. Following tagging, fish were immediately transferred for recovery in the river by being gently held facing upstream, and released when they were able to swim upstream. A total of 184 twaite shad were tagged with acoustic transmitters over the 2 years (Table 1). Most transmitters (n = 173) were programmed to transmit randomly between 30 and 90 s in the period April–June inclusive to encompass the freshwater migration period and then every 10 min July–March inclusive to encompass their marine phase. This programming increased the battery life of the transmitters to approximately 3 years, potentially enabling three consecutive spawning migrations to be tracked. The remaining transmitters (n = 11) had a battery life extending to one spawning season only (random burst interval 30–90 s), with the decision to tag some fish with these shorter-life transmitters based on tag availability. All fish captured at S1a were released upstream of this weir in order to study their migration and the impacts of barriers further upstream. Eighteen fish captured at S2 were released downstream in 2018 (Table 1) as part of wider investigations into the factors affecting barrier passage (Davies et al. 2023).
Acoustic array
An array of Vemco acoustic receivers (VR2-W and VR2-Tx, www.innovasea.com) was installed in the study area (Fig. 1), prior to the commencement of the spawning migration period in each study year. Receivers were deployed upstream and downstream of each navigation weir on the main channel of the River Severn and the flow-regulation weirs on the rivers Teme and Avon, with additional receivers deployed between weirs (Fig. 1). The furthest downstream receiver in the array (51.8347 N, −2.2901 W; Fig. 1) was located in the estuary, 8 km downstream of the tidal limit, and slightly upstream of approximate limit of saltwater incursion into the river (Bassindale 1943). Although no tagging occurred in 2020, the receiver array was installed to track returning fish tagged in previous years. Receivers were anchored on steel fencing pins driven into the river bed. In the River Teme, which has sections of fast-flowing riffle, receivers were deployed in slower-flowing pools to maximise detection distance. In each tracking year, data were downloaded from receivers every 2 weeks until no further movements were detected. Basic range tests were conducted on one day in 2019 during normal flow conditions by suspending a range test tag (V9, fixed-burst interval 10 s) at 1 m depth in the river and moving sequentially away from the receiver. The tag was suspended in the water column for 2 min in increments of 20 m, up to a maximum distance from the receiver of 200 m. These tests revealed that 100% of test tag transmissions were detected a minimum of 100 m away from the receivers in the River Severn, and a minimum of 50 m away from the receivers in the River Teme. Individual receiver detection efficiency in each year was calculated using the actel R package (Flávio and Baktoft 2021) and ranged from 43 to 100%. The two receivers with the lowest detection efficiencies (annual efficiencies = 53 to 93% and 43 to 91%) were in narrow channels and/or high turbidity tidal areas. The detection efficiency of receivers in non-tidal areas of the River Severn was generally high (median: 98.9%).
Assignment of sex and hybrid classes
Of the 184 individuals, 77 (42%) were sexed by positive identification of gonads, eggs or milt during tagging by visual inspection of the body cavity through the tagging incision, or by the presence of milt. Since twaite shad display marked sexual polymorphism (Aprahamian et al. 2003a, b), sex was estimated for any unsexed individuals based on the length/weight ratio-at-age distributions of individuals of known sex. Individuals of a given age were conservatively assigned a sex if they fell outside the size range of individuals determined to be of the other sex. After this process, 155 (84%) individuals had a known or estimated sex classification while the remainder fell within the overlapping size range for length/weight ratio-at-age of individuals of known sex, and so remained unsexed. Assignment of hybrid classes between twaite shad and allis shad was conducted using the protocol of Taillebois et al. (2020) on the fin tissues of a subset of 95 individuals, selected on the basis of their recording across successive spawning migrations. In short, 75 species-specific single-nucleotide polymorphisms (SNPs) were genotyped using the MassARRAY system as described in Taillebois et al. (2020). Individual genetic assignment to one of the 15 purebred or hybrid classes up to three generations were performed using NewHybrids (Anderson and Thompson, 2002 [https://doi.org/https://doi.org/10.1093/genetics/160.3.1217]) jointly with the data from Taillebois et al. (2020) [https://doi.org/https://doi.org/10.5061/dryad.ht76hdr9t] to allow for a good representation of allele frequencies from the two species. NewHybrids was run using uniform priors and a burn of 5000 iterations followed by a record of 10,000 iterations. Each multilocus genotype was assigned to the class that showed more than 50% of membership probability or the most likely hybrid class.
Data processing
All data processing, summaries and analyses were conducted using R statistical software (version 4.0.2, R Core Team 2020), making particular use of the dplyr package for data processing (Wickham et al. 2019). Data were initially processed to identify and remove false detections using the actel R package (Flávio and Baktoft 2021). For individuals that did not emigrate from the river in any given year, detections occurring after the first detection at their final detection location were removed to avoid spatial bias during space use analysis.
Quantifying survival, duration of freshwater residency and movement distances
To estimate the survival rates of newly tagged and returning fish, individuals were classed as surviving or non-surviving depending on whether they emigrated from the river. Fish were classed as having emigrated from the river if their final detection location was the most downstream receiver in the array. Fish that failed to emigrate were assumed to have died within the river (e.g. due to predation (Nolan et al. 2019) or failure to recover from spawning activities).
To provide a general characterisation and comparison of the movements of twaite shad during their spawning migration, summary metrics were calculated for each emigrating individual in each year. First, to estimate the duration of freshwater residency, the time at large was calculated as the time between the first and last detections. Then, to estimate the minimum distance moved while in fresh water, total movement distance was calculated as the cumulative distance moved between receivers. Daily movement rate was calculated as the total movement distance standardised by the time at large. Tortuosity was calculated as the ratio of total movement distance to the theoretical minimum river distance of an individual’s journey to its upstream extent, followed by emigration. To further explore the broadscale movement characteristics of shad in fresh water, and assess the extent to which changes in migration direction were made in response to weirs, we calculated the number of changes of direction occurring in obstructed versus obstructed reaches by tagged fish. Obstructed changes of direction were those occurring immediately downstream of a weir, while unobstructed changes of direction occurred elsewhere.
Movement metrics (time at large, total movement distance, daily movement rate, tortuosity) were summarised for newly tagged and returning individuals in each year as the median, with the range around the median given using the lower (25th) and upper (75th) quartiles (LQ-UQ). Tracking periods lasting less than 10 days were excluded from calculations of distance metrics, to avoid including fish that might have been tagged post-spawning, or ‘fallback’ individuals that emigrated soon after tagging. These individuals were also excluded from quantification of space use (see next section) to avoid potential spatial biases associated with short tracking periods.
Quantifying riverine space use
The space use of individuals during their spawning migration was quantified as a utilisation distribution (UD), a metric representing the relative probability distribution of a tagged fish within the river (Keating and Cherry 2009). Detection histories were converted to UDs by applying a dynamic Brownian bridge movement model (Horne et al. 2007), which incorporated the time and location of each detection, as well as estimated positions of tagged individuals within the river during movements between receivers. UDs for each tagged individual in each year were generated using the dynBBMM function in the RSP R package (Niella et al. 2020).
To visualise and understand the overall distribution of tagged individuals in the study area, a mean UD was generated by calculating mean values of each cell in the UD across all individuals. Mean UDs were calculated separately for (i) individuals tracked upstream of Weir S2 (i.e. individuals released upstream of S2 and individuals that passed S2) and (ii) individuals that did not pass Weir S2 (i.e. individuals released upstream S1 or downstream S2 that did not approach and/or pass S2).
Modelling factors affecting core space use area
The 50% kernel utilisation distribution (KUD50) was estimated for each individual in each year. KUD50 is a widely used metric to quantify the core area or ‘home range’ of an animal (Campbell et al. 2013; Barry et al. 2020), which represents the spatial area or ‘kernel’ in which 50% of an animal’s time is spent. Here, it was applied to assess the core space use area (CSUA) of fish during their freshwater spawning migration. First, the CSUA (km2) was calculated from the UD, in the raster R package (Hijmans, 2020). The individual factors affecting CSUA were then tested using linear mixed-effects models (LMMs) in the lme4 R package (Bates et al. 2015). Individual covariates were body length, sex, spawning history (virgin versus previously spawned) and tagging status (newly tagged versus returning individuals). To account for repeated measures occurring from the same individuals, a random effect of individual ID was included in the models. Data exploration was conducted to assess collinearity between covariates. Since sex and spawning history were both collinear with body length, these factors were not included together within the same models. Then, models containing all possible combinations of covariates (body length, sex, spawning history, tagging status) without interactions were tested and ranked according to the Akaike’s Information Criterion with a correction for small sample size (AICc); models within 2 AICc of the top-ranked model were considered to have strong support (Burnham and Anderson 2002), unless they were a more complex version of a nested model with lower AICc (Richards, Whittingham and Stephens 2011). We considered the risk of obtaining spurious results due to an ‘all possible models’ approach as low, due to the low number of covariates tested (< 6); indeed, including all covariates counters the risks of confirmation bias and minimises the risk of excluding unanticipated results (Alcott et al. 2021). In addition, a univariate LMM containing hybrid class as the sole covariate was fitted on a reduced dataset, to assess the effect of hybridisation on CSUA.
Assessing individual similarity in space use
The degree of similarity in space use by returning individuals between their first and second year of tracking was examined by calculating the intra-individual volume of intersection (VI) of individual UDs in consecutive years. VI is calculated as the cumulative sum of the minimum volume of intersection for corresponding cells between two UDs, and is a widely used metric that represents the degree of similarity in space use between UDs (Fieberg and Kochanny 2005; Dwyer et al. 2020). It is considered superior to area-based indices of overlap between space use polygons or kernels (Millspaugh et al. 2004). To avoid potential biases, two selection criteria were applied to UDs prior to calculation of VI. Firstly, since weirs had the potential to impede the upstream distribution of twaite shad in the river, only individuals that were tracked upstream of Weir S2 in both years, or that were tracked upstream of S2 in neither year, were considered appropriate for calculation of VI. This was to avoid biases in VI calculations that would result in these individuals having low overlap values that did not necessarily reflect a lack of fidelity. This decision was justified based on results presented in Davies et al., (2023), which clearly demonstrated that successful passage of weir S2 in an individual’s first year significantly increased passage likelihood upon return. It was thus assumed that unsuccessful individuals in their return migration were likely to be motivated to pass the weir had they done so previously. Secondly, for individuals that passed Weir S2 in a given year, detections that occurred downstream of this barrier were removed prior calculation of UD, to reduce biases imposed by barrier passage delays on their overall space use distributions.
Inter-individual VI was calculated between the UD of each individual in a given year with the UDs of 10 randomly selected returning fish in the following year. Finally, to test whether intra-individual space use was more similar than inter-individual space use (indicating that individuals displayed fidelity to previously occupied areas), differences in intra-individual VI versus inter-individual VI were assessed using ANOVA.
Modelling factors affecting riverine space use (spawning philopatry)
Factors affecting variation in riverine space use (intra-individual VI) were then tested using binomial generalised linear models (GLMs). Covariates were body length and spawning history at the time of tagging, and sex (with sex and body length excluded from the same models). Model selection was conducted as per the LMMs. In addition, a univariate GLM on the reduced dataset (n = 95) containing individuals with a known hybrid class (pure A. fallax versus A. fallax × alosa backcrosses) was fitted to assess the effect of hybridisation on spawning philopatry.
Results
Hybridisation rates and summary of movement metrics
Of the 95 twaite shad analysed, 75 (79%) were pure A. fallax and 20 (21%) were third generation (or greater) backcrosses (A. fallax × A. fallax backcross hybrids), indicating historical hybridisation with A. alosa. Of the 184 twaite shad tagged with acoustic transmitters, 133 (72%) emigrated from the River Severn after concluding their spawning migration. Of those with programmed transmitters allowing the potential for further detection (n = 125) 71 (57%) returned the following year, of which 53 (75%) emigrated, and seven returned for a third successive spawning migration (representing 10% of the 73 fish tagged in 2018 with long-life transmitters, and 29% of those that emigrated in 2019 (n = 24) (Table 2).
In general, the time at large of newly tagged individuals (median (LQ-UQ) = 24 (18–30) days) was less than for individuals returning the subsequent year (33 (27–38) days) (Table 2). During their freshwater migration, the median minimum distance moved between the first and last detections by newly tagged individuals was 156 km (113–223 km), and for returning individuals it was 247 km (188–304 km). Distances moved per day by the two groups of fish were highly similar (newly tagged individuals, median 7 km day−1 (5–8 km day−1), n = 184; individuals returning the subsequent year, 7 km day−1 (6–9 km day−1), n = 71) (Table 2). Fish tended to make multiple upstream and downstream movements; for example, returning individuals in their second year made upstream–downstream changes in direction a median of 27 (18–30) times prior to emigration. Upstream–downstream changes in direction occurred in both obstructed and unobstructed reaches both prior to and following barrier approach (Fig. 2); overall, a median of 60% of downstream reversals occurred in unobstructed reaches. These multiple downstream movements were reflected in migration path tortuosity values of 2.8 (2.1–4.2) for returning individuals.
Movement and space use of four returning acoustic-tagged twaite shad to the River Severn catchment. Left panels show movement in the form of a track, with y-axis values representing river distance (rkm) of detections from the tidal limit by time (x-axis). Horizontal dashed lines represent location of main weirs within the catchment. Right panels show utilisation distributions derived from dynamic Brownian bridge movement model. A Female individual predominantly detected in lower River Teme and confluence with the River Severn. B Male individual predominantly detected at the confluence of the River Teme and River Severn. C Female individual detected widely within the River Severn upstream of Weir S2. D Male individual detected predominantly within the River Teme (color figure online)
Mean utilisation distribution and factors affecting KUD50 area
For individuals that did not pass Weir S2, activity was concentrated in the upstream half of the contiguous reach between Weir S1 and S2, with the highest probability densities occurring at the confluence of the River Severn and River Avon (Fig. 3). For fish tracked upstream of S2, mean UD was characterised by a peak of activity in the upstream half of the contiguous reach between Weir S2 and weirs S3/T1, with the highest probability densities occurring within the lower River Teme and the confluence of the River Teme and River Severn (Fig. 3). Testing of factors influencing the 50% kernel utilisation distribution (KUD50) retained sex as the only significant predictor from 14 best-fitting LMMs (Supplementary Material: Table S1), indicating that females had significantly larger core space use areas than males (Table 3, Fig. 4). A univariate LMM indicated that there was no evidence that hybridisation was a strong predictor of KUD50 (Table 3).
Riverine space use (mean utilisation distribution) of twaite shad in the River Severn catchment. White bars indicate the positions of major weirs. a Utilisation distributions of migrations upstream of Weir S2 but not approaching S3/T1, pooled across years (n = 49 migrations). b Utilisation distribution of migrations upstream of Weir S2 approaching S3 and/or T1 pooled across years (n = 91 migrations). c Utilisation distributions of migrations upstream of Weir S1 but not approaching S2, pooled across years (n = 7 migrations). d Utilisation distributions of migrations upstream of Weir S1 and S2, pooled across years (n = 53 migrations) (color figure online)
Riverine space use by individuals across successive spawning migrations
For the 71 fish tracked across two successive spawning migrations, intra-individual VI (mean ± SD = 55% ± 18) was significantly greater than inter-individual VI (38% ± 21) (one-way ANOVA, F1449 = 26.8, p < 0.01) (Fig. 5), indicating that returning individuals tended to return to areas of river they had used the previous year. There however, no significant predictors of variation in VI, with none of the 28 fitted GLMs tested having better AIC support than the null model (Table S2). A univariate GLM indicated that there was minimal evidence that hybridisation was a strong predictor of intra-individual VI (Table 4).
Boxplot showing inter- and intra-individual volume of intersection of utilisation distributions between first and second year of tracking by twaite shad. Boxplot boundaries represent upper and lower quartiles, central line represents the median. Filled circles represent individual data points (color figure online)
Discussion
Twaite shad movements were highly tortuous during their riverine spawning migrations, undertaking multiple upstream and downstream movements, both in unobstructed reaches and during approach to man-made weirs. Females occupied larger core areas than males, but hybridisation was not a significant predictor of space use. There was evidence of philopatry, with returning fish occupying very similar locations in the river across successive spawning migrations.
Anadromous clupeids are often highly vagile during their spawning migrations. For example, long-distance ‘oscillations’—back and forth movements between spawning grounds and downstream habitats—have been observed in alewife Alosa pseudoharengus (McCartin et al. 2019), and allis shad may exhibit ‘exploratory’ behaviours that have been attributed to the concurrent use of spawning and resting sites (Acolas et al. 2004, 2006). Here, when comparing the distance moved by individuals relative to their upstream extent (i.e., tortuosity), it reflects how their freshwater migration is composed of multiple upstream and downstream movements. As the time-standardised movement rates between the consecutive migration periods were similar (median: 7 km day−1), this suggests that the tagging process did not substantially affect their overall distance moved. The duration of freshwater residency by returning twaite shad (median: 33 days) was commensurate with a recent study on American shad Alosa sapidissima where periods of 35 days were observed in returning individuals (Gahagan and Bailey 2020). Freshwater residency times of between 8 and 33 days have been reported in American shad tagged in the same year (Beasley and Hightower 2000; Aunins and Olney 2009; Aunins et al. 2013; Grote et al. 2014; Raabe and Hightower 2014). Consequently, the duration of spawning periods and vagility and duration of the spawning migration appear to be similar among other members of the Alosa genus.
The mean distribution of twaite shad in this study suggested that specific areas of river were used relatively intensively during the spawning migration, potentially indicating the impact of the weirs inhibiting upstream progression, as well as possible spawning locations. Specifically, the space use of individuals that did not pass weirs S2 and S3 was focused in the 1 to 2 km reaches downstream, and both are known spawning areas (Aprahamian, Lester and Aprahamian 1998). The negative impact of anthropogenic barriers on the upstream migration of twaite shad in the River Severn has already been demonstrated (Davies et al. 2023). Nevertheless, this finding is also consistent with a study that revealed that allis shad spent the majority of their time either in resting areas approximately 1.5 km downstream of spawning grounds which were immediately downstream a major migration barrier (Acolas et al. 2004; Aprahamian, Lester and Aprahamian 1998). The possibility that shad were searching for alternative passage routes or spawning habitats after approaching weirs, as reported for sea lamprey Petromyzon marinus (Davies et al. 2022), also cannot be ruled out. Crucially, fish that did and did not approach S3/T1 both displayed areas of intensive space use in the areas immediately downstream of these barriers; thus it is difficult to disentangle the influence of anthropogenic barriers and biotic influences on space use during twaite shad freshwater spawning migrations. Future efforts to reconnect inaccessible upstream spawning areas through barrier removal and fish pass installation (www.unlockingthesevern.co.uk) may help further this understanding.
Sex has been widely linked to differences in the temporal and spatial characteristics of animal migration (Morbey and Ydenberg 2001; Barnett et al. 2011; Bunnefeld et al. 2011). As a life history strategy, iteroparity should reduce the relative importance of individual spawning events by facilitating survival for future spawning events, with suggestions that the behaviours of females from iteroparous populations are more risk-averse than males (Warner 2005). Here, it was apparent that female twaite shad used significantly larger areas of the river than males during their spawning migrations, although it could not be ascertained whether this was related to reducing, for example, their risk of predation. Studies in multiple Alosa species have suggested that females are batch-spawners, with spawning events separated by a period of days (Olney et al. 2001; Harris et al. 2007; Mouchlianitis et al. 2020). Moreover, batch-spawning by female allis shad has been linked to observed movement differences between sexes (Acolas et al. 2004, 2006), with males more likely to be on spawning grounds on consecutive nights and engaging in more spawning acts, while females spent periods consistent with egg maturation away from spawning grounds. While it was beyond the scope of this paper to explore movement strategies of males and females underlying differences in space use, the male and female movement tracks presented in Fig. 2 provide potential examples of how strategies may differ between sexes, whereby males spend more time in the upper reaches of rivers while females are more prone to long distance movements during their freshwater residency period. Consequently, the greater space use by females versus males here could just reflect movements by females from spawning to resting areas during the egg maturation period. This possibility is also supported by a study on American shad where males used upstream habitats more than females (Raabe and Hightower 2014), potentially reflecting a stronger association with spawning grounds by males. Further studies should seek to understand the sex-based differences in movement strategy underlying space use patterns, and how these may interact with pressures such as anthropogenic barriers.
Philopatry is a widespread feature of animal migration, with natal philopatry being a strategy that increases the likelihood that migrants will encounter mates and suitable reproductive habitat (Greenwood 1980; Dittman and Quinn 1996). In anadromous fishes, the process of natal philopatry (or ‘homing’) is believed to be driven by olfactory imprinting, whereby juveniles form associations with the geochemical signature of water from their natal river (or tributary) during development (Dittman and Quinn 1996; Keefer and Caudill 2014). Although our understanding of this process has mainly been derived from salmonid fishes, natal homing to specific tributaries by American shad has been demonstrated, with marked hatchery-reared individuals returning to the same tributary that they emigrated from as juveniles (Hendricks et al. 2002). Spatial segregation of returning individuals across the river was also noted, suggesting that olfactory-mediated location of natal tributaries may occur. While fidelity to previous spawning rivers by adults has also been observed in iteroparous shad species (Melvin et al. 1986; Davies et al. 2020), how this homing interacts with spawning philopatry (i.e. the return of adults to specific spawning areas) has been unclear. Here, while we could not assess natal philopatry directly, the results revealed that twaite shad did return to similar areas of the river during consecutive spawning migrations, as evidenced by intra-individual VI being significantly greater than inter-individual VI. However, it is acknowledged that how the extent of this repeatability in riverine space use between migrations relates to spawning philopatry is unclear, as the method used (passive acoustic telemetry) was unable to provide information on the actual spawning locations of the fish. Complementary approaches, such as surveys of spawning activity (Langkau et al. 2016; Paumier et al. 2020) and high-resolution telemetry, will therefore be needed if information on the actual spawning areas of twaite shad is required.
Hybridisation between different species or subpopulations with divergent migratory strategies can result in offspring that display intermediate or increased variability in movement patterns; this phenomenon has been best described in migratory birds (Alvarado et al. 2014; Delmore and Irwin 2014). Where allis shad and twaite shad co-occur, the former generally migrate further upstream, with river fragmentation posited as driving their hybridisation through the sharing of spawning areas in more downstream areas (Aprahamian et al. 2003b; Taillebois et al. 2020). Here, 21% of sampled twaite shad were introgressed with allis shad (third generation (or greater) backcrosses with a theoretical 12.5% of the genome from A. alosa), indicating infrequent historical hybridisation with A. alosa, but there was no evidence that this influenced their core area size. Significant knowledge gaps remain on the status of A. alosa in the River Severn, including the extent to which this historical introgression with A. fallax derives from individual A. alosa straying from spawning populations in other rivers, or from a spawning population in the River Severn.
In summary, the twaite shad that were tracked during successive spawning migrations provided important insights into their spatial ecology in a highly regulated river catchment. The duration of their freshwater residency was similar to another Alosa species, differences in space use were greater in females than males, and spawning philopatry to areas within the river basin was apparent in returning individuals. These results should thus provide resource managers with considerable insights into this and similar anadromous and iteroparous species in regulated rivers that can be used to inform management measures, conservation strategies and further research.
Data availability
Data from this study will be made available upon reasonable request.
References
Acolas M, Begountras M, Véron V, Jourdan H, Sabatié M, Baglinière JL (2004) An assessment of the upstream migration and reproductive behaviour of allis shad (L.) using acoustic tracking. ICES J Marine Sci 61(8):1291–1304. https://doi.org/10.1016/j.icesjms.2004.07.023
Acolas ML, Véron V, Jourdan H, Bégout ML, Sabatié MR, Baglinière JL (2006) Upstream migration and reproductive patterns of a population of allis shad in a small river (L’Aulne, Brittany, France). ICES J Marine Sci 63(3):476–484. https://doi.org/10.1016/j.icesjms.2005.05.022
Alcott, D.J., Goerig, E., Rillahan, C., He, P. and Castro-Santos, T. (2021). Tide gates form physical and ecological obstacles to river herring (Alosa spp.) spawning migrations. Canadian Journal of Fisheries and Aquatic Sciences. https://doi.org/10.1139/cjfas-2020-0347.
Alvarado AH, Fuller TL, Smith TB (2014) Integrative tracking methods elucidate the evolutionary dynamics of a migratory divide. Ecol Evol 4(17):3456–3469. https://doi.org/10.1002/ece3.1205
Antognazza CM, Britton JR, Potter C, Franklin E, Hardouin EA, Gutmann Roberts C et al (2019) Environmental DNA as a non-invasive sampling tool to detect the spawning distribution of European anadromous shads (Alosa spp). Aquatic Conserv: Marine Freshw Ecosyst 29(1):148–152. https://doi.org/10.1002/aqc.3010
Antognazza CM, Sabatino SJ, Britton RJ, Hillman RJ, Aprahamian M, Hardouin EA et al (2021) Hybridization and genetic population structure of Alosa population in the United Kingdom. J Fish Biol. https://doi.org/10.1111/JFB.14917
Aprahamian M, Baglinière J-L, Sabatie M, Alexandrino P, Thiel R, Aprahamian C (2003a) Biology, status and conservation of the anadromous twaite shad Alosa fallax fallax (Lacépède, 1803). Am Fish Soc Symp 35:103–124
Aprahamian MW, Lester SM, Aprahamian CD (1998). Shad Conservation in England and Wales: R & D Technical Report W110. Bristol, UK. https://assets.publishing.service.gov.uk/media/5a7cda3fe5274a2ae6eeb2ab/str-w110-ee.pdf
Aprahamian MW, Aprahamian CD, Baglinière JL, Sabatié R, Alexandrino P (2003b) Alosa Alosa and Alosa fallax spp. Literature Review and Bibliography: R&D Technical Report W1–014/TR. Bristol, UK. https://assets.publishing.service.gov.uk/media/5a7c543840f0b62dffde1617/sw1-014-tr-e-e.pdf
Aunins A, Olney JE (2009) Migration and spawning of American Shad in the James River, Virginia. Trans Am Fis Soc 138(6):1392–1404. https://doi.org/10.1577/T08-160.1
Aunins AW, Brown BL, Balazik M, Garman GC (2013) Migratory movements of American shad in the James River fall zone, Virginia. North Am J Fish Manag 33(3):569–575. https://doi.org/10.1080/02755947.2013.768564
Baglinière JL, Sabatié MR, Aprahamian MW, Alexandrino P, Aprahamian CD, Assis CA et al (2001) Guide pour l’interprétation des écailles et l’estimation de l’âge chez les aloses (Alosa spp.) De la façade Atlantique-est et de la Méditerranée-ouest. Bull Français De La Pêche Et De La Piscic 357–360:485–531. https://doi.org/10.1051/kmae:2001001
Barnett A, Abrantes KG, Stevens JD, Semmens JM (2011) Site fidelity and sex-specific migration in a mobile apex predator: Implications for conservation and ecosystem dynamics. Animal Behav 81(5):1039–1048. https://doi.org/10.1016/j.anbehav.2011.02.011
Barry J, Mcloone P, Fitzgerald CJ, King JJ (2020) The spatial ecology of brown trout (Salmo trutta) and dace (Leuciscus leuciscus) in an artificially impounded riverine habitat: results from an acoustic telemetry study. Aquat Sci 82:63. https://doi.org/10.1007/s00027-020-00737-9
Bassindale R (1943) A Comparison of the Varying Salinity Conditions of the Tees and Severn Estuaries. J Animal Ecol 12(1):1. https://doi.org/10.2307/1407
Bates D, Mächler M, Bolker BM, Walker SC (2015) Fitting linear mixedeffects models using lme. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Beacham TD, Withler RE (2017) Population structure of sea-type and lake-type sockeye salmon and kokanee in the Fraser River and Columbia River drainages. PLoS ONE 12(9):e0183713. https://doi.org/10.1371/JOURNAL.PONE.0183713
Beasley CA, Hightower JE (2000) Effects of a low-head dam on the distribution and characteristics of spawning habitat used by striped bass and American shad. Trans Am Fish Soc 129(6):1316–1330. https://doi.org/10.1577/1548-8659(2000)129<1316:EOALHD>2.0.CO;2
Bolland JD, Nunn AD, Angelopoulos NV, Dodd JR, Davies P, Gutmann Roberts C et al (2019) Refinement of acoustic-tagging protocol for twaite shad Alosa fallax (Lacépède), a species sensitive to handling and sedation. Fish Res 212:183–187. https://doi.org/10.1016/j.fishres.2018.12.006
Bubb DH, Birnie-Gauvin K, Tummers JS, Aarestrup K, Jepsen N, Lucas MC (2021) Short-term effects of low-head barrier removals on fish communities and habitats. Front Ecol Evol 9:697106. https://doi.org/10.3389/FEVO.2021.697106/BIBTEX
Bunnefeld N, Börger L, Moorter BV, Rolandsen CM, Dettki H, Solberg EJ et al (2011) A model-driven approach to quantify migration patterns: Individual, regional and yearly differences. J Animal Ecol 80(2):466–476. https://doi.org/10.1111/j.1365-2656.2010.01776.x
Campbell HA, Dwyer RG, Irwin TR, Franklin CE (2013) Home Range Utilisation and Long-Range Movement of Estuarine Crocodiles during the Breeding and Nesting Season. PLoS ONE. https://doi.org/10.1371/journal.pone.0062127
Carscadden JE, Leggett WC (1975) Meristic Differences in Spawning Populations of American Shad, Alosa sapidissima : Evidence for Homing to Tributaries in the St. John River, New Brunswick. J Fish Res Board Canada 32(5):653–660. https://doi.org/10.1139/f75-084
Council of the European Communities (1992). Council Directive 92/43/EEC of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora. L206, 7–50.
Davies P, Britton RJ, Nunn AD, Dodd JR, Crundwell C, Velterop R et al (2020) Novel insights into the marine phase and river fidelity of anadromous twaite shad Alosa fallax in the UK and Ireland. Aquat Conserv: Marine Freshw Ecosyst 30(7):1291–1298. https://doi.org/10.1002/aqc.3343
Davies P, Britton RJ, Nunn AD, Dodd JR, Crundwell C, Velterop R et al (2022) Individual movement variation in upstream-migrating sea lamprey Petromyzon marinus in a highly fragmented river. Freshw Biol 67:643–656. https://doi.org/10.1111/fwb.13869
Davies P, Britton JR, Castro-Santos T, Crundwell C, Dodd JR, Nunn AD, Velterop R, Bolland JD (2023) Tracking anadromous fish over successive freshwater migrations reveals the influence of tagging effect, previous success, and abiotic factors on upstream passage over barriers. Can J Fish Aquat Sci 80(7):1110–1125. https://doi.org/10.1139/cjfas-2022-0196
de Groot SJ (1990) The former allis and twaite shad fisheries of the lower Rhine, The Netherlands. J Appl Ichthyol 6(4):252–256. https://doi.org/10.1111/j.1439-0426.1990.tb00587.x
Delmore KE, Irwin DE (2014) Hybrid songbirds employ intermediate routes in a migratory divide. Ecol Lett 17(10):1211–1218. https://doi.org/10.1111/ele.12326
Dittman A, Quinn T (1996) Homing in Pacific salmon: mechanisms and ecological basis. J Exp Biol 199(1):83–91
Dwyer RG, Campbell HA, Cramp RL, Burke CL, Micheli-Campbell MA, Pillans RD et al (2020) Niche partitioning between river shark species is driven by seasonal fluctuations in environmental salinity. Funct Ecol 1365–2435:13626. https://doi.org/10.1111/1365-2435.13626
Fieberg J, Kochanny CO (2005) Quantifying home-range overlap: The importance of the utilization distribution. J Wildlife Manag 69(4):1346–1359
Flávio H, Baktoft H (2021). actel: Standardised analysis of acoustic telemetry data from animals moving through receiver arrays. E Codling (ed), Methods in Ecology and Evolution, 12(1):196–203. https://doi.org/10.1111/2041-210X.13503.
Gahagan BI, Bailey MM (2020) Surgical Implantation of Acoustic Tags in American Shad to Resolve Riverine and Marine Restoration Challenges. Marine Coast Fish 12(5):272–289. https://doi.org/10.1002/mcf2.10108
Glebe BD, Leggett WC (1981) Latitudinal Differences in Energy Allocation and Use During the Freshwater Migrations of American Shad (Alosa sapidissima) and Their Life History Consequences. Can J Fish Aquat Sci 38:806–820. https://doi.org/10.1139/f81-109
Greenwood PJ (1980) Mating systems, philopatry and dispersal in birds and mammals. Animal Behav 28(4):1140–1162. https://doi.org/10.1016/S0003-3472(80)80103-5
Grote AB, Bailey MM, Zydlewski JD (2014) Movements and demography of spawning American shad in the Penobscot River, Maine, prior to dam removal. Trans Am Fish Soc 143(2):552–563. https://doi.org/10.1080/00028487.2013.864705
Harris JE, McBride RS, Williams RO (2007) Life History of Hickory Shad in the St. Johns River Florida. Trans Am Fish Soc 136(6):1463–1471. https://doi.org/10.1577/t06-187.1
Hasselman DJ, Bentzen P, Bradford RG (2010) Taking stock: Defining populations of American shad (Alosa sapidissima) in Canada using neutral genetic markers. Can J Fish Aquat Sci 67(6):1021–1039. https://doi.org/10.1139/F10-031
Hendry AP, Castric V, Kinnison MT, Quinn TP (2003) The evolution of philopatry and dispersal homing versus straying in salmonids. In: Quinn TP, Hendry AP (eds) Evolution illuminated: salmon and their relatives. Oxford University Press, USA, pp 52–91. https://doi.org/10.1093/oso/9780195143850.003.0003
Hendricks ML, Hoopes RL, Arnold DA, Kaufmann ML (2002) Homing of Hatchery-Reared American Shad to the Lehigh River, a Tributary to the Delaware River. North Am J Fish Manag 22(1):243–248. https://doi.org/10.1577/1548-8675(2002)022%3c0243:hohras%3e2.0.co;2
Horne JS, Garton EO, Krone SM, Lewis JS (2007) Analyzing animal movements using Brownian bridges. Ecology 88(9):2354–2363. https://doi.org/10.1890/06-0957.1
Jolly MT, Maitland PS, Genner MJ (2011) Genetic monitoring of two decades of hybridization between allis shad (Alosa Alosa) and twaite shad (Alosa fallax). Conserv Gene 12(4):1087–1100. https://doi.org/10.1007/s10592-011-0211-3
Jolly MT, Aprahamian MW, Hawkins SJ, Henderson PA, Hillman R, O’Maoiléidigh N et al (2012) Population genetic structure of protected allis shad (Alosa Alosa) and twaite shad (Alosa fallax). Marine Biol 159(3):675–687. https://doi.org/10.1007/s00227-011-1845-x
Keating KA, Cherry S (2009) Modeling utilization distributions in space and time. Ecology 90(7):1971–1980. https://doi.org/10.1890/08-1131.1
Keefer ML, Caudill CC (2014) Homing and straying by anadromous salmonids: A review of mechanisms and rates. Rev Fish Biol Fish 24(1):333–368. https://doi.org/10.1007/s11160-013-9334-6
Kirk MA, Caudill CC (2017) Network analyses reveal intra- and interspecific differences in behaviour when passing a complex migration obstacle. J Appl Ecol 54(3):836–845. https://doi.org/10.1111/1365-2664.12786
Langkau MC, Clavé D, Schmidt MB, Borcherding J (2016) Spawning behaviour of Allis shad Alosa Alosa : new insights based on imaging sonar data. J Fish Biol 88(6):2263–2274. https://doi.org/10.1111/jfb.12978
Leggett WC, Carscadden JE (1978) Latitudinal variation in reproductive characteristics of American shad (Alosa sapidissima): evidence for population specific life history strategies in fish. J Fish Res Board Can 35(11):1469–1478. https://doi.org/10.1139/f78-230
Lennox RJ, Paukert CP, Aarestrup K, Auger-Méthé M, Baumgartner L, Birnie-Gauvin K, Bøe K, Brink K, Brownscombe JW, Chen Y, Davidsen JG, Eliason EJ, Filous A, Gillanders BM, Helland IP, Horodysky AZ, Januchowski-Hartley SR, Lowerre-Barbieri SK, Lucas MC et al (2019) One hundred pressing questions on the future of global fish migration science, conservation, and policy. Front Ecol Evol. https://doi.org/10.3389/fevo.2019.00286
Mack K, White H, Rohde FC (2021) Use of Acoustic Telemetry to Identify Spawning River and Spawning Migration Patterns of American Shad in the Albemarle Sound, North Carolina. North Am J Fish Manag 41(1):242–251. https://doi.org/10.1002/NAFM.10555
McCartin K, Jordaan A, Sclafani M, Cerrato R, Frisk MG (2019) A New Paradigm in Alewife Migration: Oscillations between Spawning Grounds and Estuarine Habitats. Trans Am Fish Soc 148(3):605–619. https://doi.org/10.1002/tafs.10155
Melvin GD, Dadswell MJ, Martin JD (1986) Fidelity of American shad, Alosa sapidissima ( Clupeidae), to its river of previous spawning. Can J Fish Aquat Sci 43(3):640–646. https://doi.org/10.1139/f86-077
Millspaugh JJ, Gitzen RA, Kernohan BJ, Larson MA, Clay CL (2004) Comparability of three analytical techniques to assess joint space use. Wildlife Soc Bull 32(1):148–157. https://doi.org/10.2193/0091-7648(2004)32[148:COTATT]2.0.CO;2
Moore J, Harris L, Le Luyer J, Sutherland B, Rougemont Q, Tallman R et al (2017) Genomics and telemetry suggest a role for migration harshness in determining overwintering habitat choice, but not gene flow, in anadromous Arctic Char. Mol Ecol 26(24):6784–6800. https://doi.org/10.1111/MEC.14393
Morbey YE, Ydenberg RC (2001) Protandrous arrival timing to breeding areas: A review. Ecol Lett 4(6):663–673. https://doi.org/10.1046/j.1461-0248.2001.00265.x
Mouchlianitis FA, Minos G, Ganias K (2020) Timing of oocyte recruitment within the ovulatory cycle of Macedonian shad, Alosa macedonica, a batch spawning fish with indeterminate fecundity. Theriogenology 146:31–38. https://doi.org/10.1016/j.theriogenology.2020.01.050
Niella Y, Flávio H, Smoothey AF, Aarestrup K, Taylor MD, Peddemors VM, et al. (2020). Refined Shortest Paths (RSP): Incorporation of topography in space use estimation from node‐based telemetry data. E. Codling (ed.), Methods in Ecology and Evolution, 11(12), 1733–1742. https://doi.org/10.1111/2041-210X.13484.
Nolan ET, Gutmann Roberts C, Britton JR (2019) Predicting the contributions of novel marine prey resources from angling and anadromy to the diet of a freshwater apex predator. Freshw Biol 64(8):1542–1554. https://doi.org/10.1111/fwb.13326
Nunn AD, Cowx IG (2012) Restoring river connectivity: prioritizing passage improvements for diadromous fishes and lampreys. Ambio 41(4):402–409. https://doi.org/10.1007/s13280-012-0281-6
Olney JE, Denny SC, Hoenig JM (2001) Criteria for determining maturity stage in female american shad, Alosa sapidissima, and a proposed reproductive cycle. Bull Français De La Pêche Et De La Piscic 362–363:881–901. https://doi.org/10.1051/kmae:2001025
Paumier A, Drouineau H, Boutry S, Sillero N, Lambert P (2020) Assessing the relative importance of temperature, discharge, and day length on the reproduction of an anadromous fish ( Alosa alosa ). Freshw Biol 65(2):253–263. https://doi.org/10.1111/fwb.13418
Pess GR, Quinn TP, Gephard SR, Saunders R (2014) Re-colonization of Atlantic and Pacific rivers by anadromous fishes: linkages between life history and the benefits of barrier removal. Rev Fish Biol Fish 24(3):881–900. https://doi.org/10.1007/s11160-013-9339-1
R Core Team (2020). R: A language and environment for statistical computing.R Foundation for Statistical Computing, Vienna, Austria.
Raabe JK, Hightower JE (2014) American Shad Migratory Behavior, Weight Loss, Survival, and Abundance in a North Carolina River following Dam Removals. Trans Am Fish Soc 143(3):673–688. https://doi.org/10.1080/00028487.2014.882410
Sabatino SJ, Faria R, Alexandrino PB (2022) Genetic structure, diversity, and connectivity in anadromous and freshwater Alosa alosa and A fallax. Marine Biology 169(1). https://doi.org/10.1007/s00227-021-03970-4
Stearns SC (1998) The evolution of life histories. Oxford University Press. https://doi.org/10.1093/oso/9780198577416.001.0001
Stepien CA, Murphy DJ, Lohner RN, Sepulveda-Villet OJ, Haponski AE (2009) Signatures of vicariance, postglacial dispersal and spawning philopatry: population genetics of the walleye Sander vitreus. Mol Ecol 18(16):3411–3428. https://doi.org/10.1111/J.1365-294X.2009.04291.X
Sylvester EVA, Beiko RG, Bentzen P, Paterson I, Horne JB, Watson B et al (2018) Environmental extremes drive population structure at the northern range limit of Atlantic salmon in North America. Mol Ecol 27(20):4026–4040. https://doi.org/10.1111/MEC.14849
Taillebois L, Sabatino S, Manicki A, Daverat F, Nachón DJ, Lepais O (2020) Variable outcomes of hybridization between declining Alosa Alosa and Alosa fallax. Evol Appl 13(4):636–651. https://doi.org/10.1111/eva.12889
Thorstad EB, Bliss D, Breau C, Damon-Randall K, Sundt-Hansen LE, Hatfield EMC et al (2021) Atlantic salmon in a rapidly changing environment—Facing the challenges of reduced marine survival and climate change. Aquat Cons: Marine Freshw Ecosyst 31(9):2654–2665. https://doi.org/10.1002/AQC.3624
Tibblin P, Forsman A, Borger T, Larsson P (2016) Causes and consequences of repeatability, flexibility and individual fine-tuning of migratory timing in pike. J Animal Ecol 85(1):136–145. https://doi.org/10.1111/1365-2656.12439
Warner RR (2005) The role of extreme iteroparity and risk avoidance in the evolution of mating systems. J Fish Biol 53:82–93. https://doi.org/10.1111/J.1095-8649.1998.TB01019.X
Wickham H, Averick M, Bryan J, Chang W, McGowan L, François R et al (2019) Welcome to the Tidyverse. J Open Sour Softw 4(43):1686. https://doi.org/10.21105/joss.01686
Acknowledgements
The authors acknowledge funding for the purchase of acoustic tags from the UK Department for Environment, Food and Rural Affairs (DEFRA). PD was supported by a match-funded PhD grant from the ‘Unlocking the Severn’ project (Heritage Lottery Fund, Grant/Award Number: HG/15/04573, LIFE Nature Programme Grant/Award Number: LIFE15/NAT/UK/000219), and Bournemouth University. In addition, we acknowledge the expertise of Natalie Angelopolous during the tagging process, as well as the planning and logistical support from staff of the Environment Agency, Natural England, Severn Rivers Trust, and Canal and Rivers Trust, and permission from the landowner at Maisemore Weir. The SNP genotyping was performed at the PGTB (https://doi.org/10.15454/1.5572396583599417E12) with the help of Laure Dubois and Erwan Guichoux.
Author information
Authors and Affiliations
Contributions
Conceived and designed the field experiments: JDB, ADN, JRD, CC, RV, JRB, and PD. Conducted fieldwork: JDB, ADN, JRB, CC, JRD, and PD. Conducted genetic extraction and analysis: OL, SS, PD, DA. Conducted telemetry analysis: PD. Wrote the article: PD. Edited the article: JDB, DA, JRB, ADN, JRD, CC, OL, SS and RV.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Davies, P., Britton, J.R., Andreou, D. et al. Tracking repeat spawning anadromous fish migrations over multiple years in a fragmented river suggests philopatry and sex-linked variation in space use. Aquat Sci 86, 34 (2024). https://doi.org/10.1007/s00027-024-01048-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-024-01048-z